Abstract
Introduction
Platinum-based chemotherapy is important for the treatment for recurrent or metastatic nasopharyngeal carcinomas (RM-NPCs), as recommended by current clinical guidelines. However, the prognosis of RM-NPC patients remains poor. This study aimed to evaluate the efficacy and safety of vascular-targeting agents apatinib or anlotinib plus chemotherapy for RM-NPC treatment.
Methods
This retrospective multicenter study, conducted at 4 hospitals in Guangxi Province, China, between January 2017 and December 2021, included 198 patients. Propensity score matching (PSM) was used to minimize potential selection bias between groups. After PSM, a total of 150 patients were included in the study. Chemotherapy was administered with and without vascular-targeting agents in 58 and 92 patients, respectively. The primary endpoint was progression-free survival (PFS), assessed by a blinded independent review committee according to RECIST v.1.1. The secondary end points were overall survival (OS) and safety.
Results
After PSM, median PFS was significantly higher in the combination group (8.0 months [95% CI, 5.7-8.3 months]) than in the chemotherapy alone group (5.0 months [95% CI, 4.1-5.9 months], P = 0.014), with a hazard ratio of 0.65 (95% CI, 0.46-0.92). The combination group exhibited a higher median OS compared to the chemotherapy alone group (45 months [95% CI, 21.3-52.7] vs 18 months [95% CI, 13.0-27.0 months], P = 0.019), representing a hazard ratio of 0.58 (95% CI, 0.37-0.92). In the combination therapy group, the most common adverse event was hand-foot syndrome (n = 16/58 [27.6%]).
Conclusion
The addition of vascular-targeting agents to chemotherapy for RM-NPC patients provided superior efficacy compared to chemotherapy alone, along with a manageable safety profile.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is a head and neck cancer with an unbalanced geographical distribution; it is particularly prevalent in Southern China, East and Southeast Asia, and North Africa. Globally, approximately 129 000 new cases and 73 000 deaths attributed to NPCs are reported annually.1,2 The incidence of synchronous metastatic disease is about 5% in newly diagnosed patients. Additionally, approximately 20% of the non-metastatic NPC patients develop distant failure despite definitive treatments, accounting for the majority of deaths in these patients.3,4 Guideline-recommended platinum-based chemotherapy is an important treatment for metastatic NPC patients, but its efficacy in terms of median progression-free survival (PFS) and overall survival (OS) remains low. 5 Consequently, the treatment of recurrent or metastatic NPC (RM-NPC) patients remains a major challenge.
Angiogenesis is an important step in the development of several malignant tumors, and vascular-targeting agents, including anlotinib and apatinib, are globally considered an important treatment for cancer. Apatinib is a novel vascular endothelial growth factor receptor-2 (VEGFR-2) tyrosine kinase inhibitor (TKI) that selectively targets intracellular ATP-binding sites and has shown efficacy against various solid tumors.6-8 Anlotinib (AL3818), an oral small-molecular TKI, exerts significant anti-angiogenesis and anti-tumor growth effects by inactivating VEGFR and other factor receptors.9-11 It is an important vascular-targeting agent and has been approved as the third line treatment for locally advanced or metastatic non-small cell lung cancer. Vascular endothelial growth factor (VEGF) is highly expressed in NPCs and correlates with their survival, making them amenable to antiangiogenic therapy. A previous study reported VEGF overexpression in more than 60% of the clinical biopsy samples of NPC. Co-expression of tumor VEGF and hypoxia-related growth factors in NPC is associated with poor prognosis, indicating the effectiveness of apatinib against NPC relapse and recurrences. 12 In a phase II study of RM-NPCs by Ruan et al, the observed objective response rate (ORR) was 36.4%, the median OS was 16 months, and the median PFS was 5.0 months in 33 patients. 13 Vascular-targeting agents combined with chemotherapy are effective in the treatment of lung and colorectal cancers. To our knowledge, the efficacy of vascular-targeting agents apatinib or anlotinib combined with chemotherapy has not previously been investigated in RM-NPC patients. This study aimed to explore the efficacy of vascular-targeting agents apatinib or anlotinib combined with chemotherapy vs chemotherapy alone in RM-NPC patients, along with their potential synergistic effects.
Methods
Design of the Study and its Participants
This was a multi-center, retrospective study conducted at 4 Chinese hospitals between January 2017 and December 2021, including 198 consecutive RM-NPC patients. Propensity score matching was used to minimize potential selection bias between groups. After PSM, a total of 150 patients were included in the study. The study flowchart is shown in Figure 1. The primary outcome measure was PFS. The secondary outcome measures were OS and adverse events (AEs). The data used in this study were extracted from a database constructed by combining information from multiple data sources, including the Hospital Information System and Electronic Medical Record. Patient data used in this study were anonymized, and all participating centers' institutional review committees gave their approval for the trial program, which was carried out in compliance with the Declaration of Helsinki. We have de-identified all patient details. The ethics approval number are: 2022YJSLL-26 (Medical Ethics Committee of The Affiliated Hospital of Guilin Medical University); 2022045 (Medical Ethics Committee of Lingshan County People’s Hospital); LL-2025-062-01 (Ethics Committee of Wuzhou Red Cross Hospital); and IRB-2022-047 (Ethics Committee of Nanxishan Hospital of Guangxi Zhuang Autonomous Region). We have obtained written informed consent from the study participants. Patients have consented to access to their medical records and follow-up information. The reporting of this study conforms to STROBE guidelines.
14
Recruitment and Inclusion of Patients With RM-NPC for Participation in the Study
The inclusion criteria were: (a) age of 18-75 years; (b) histological or cytological diagnosis of RM-NPC; (c) at least 1 cycle of vascular-targeting agents anlotinib or apatinib plus chemotherapy or chemotherapy alone following recurrence or metastasis; (d) Eastern Cooperative Oncology Group (ECOG) performance status (PS) of 0 or 1; (e) at least 1 measurable lesion per Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1; (f) expected survival time > 3 months; (g) hemoglobin ≥90 g/L, absolute neutrophil count ≥1.5 × 109/L, platelet count ≥80 × 109/L, total bilirubin ≤ 1.5 times the upper limit of normal (ULN), alanine aminotransferase (ALT) and aspartate aminotransferase (AST) ≤ 2.5 ULN (≤5 ULN with liver metastasis), and serum creatinine ≤ 1.5 ULN. Exclusion criteria included: (a) previous vascular-targeting therapy; (b) difficulty in oral drug administration or absorption, including dysphagia, gastrointestinal resection, chronic diarrhea, and intestinal obstruction; (c) radiographic evidence of tumor invading tissues surrounding vital blood vessels; (d) pregnancy or breastfeeding; and (e) other simultaneous malignant tumors.
Procedures and Assessments
In this retrospective study, patients in the combination therapy group had received conventional chemotherapy combined with vascular-targeting agents, including apatinib (250 mg orally once daily) and anlotinib (12 mg orally once daily), with cycles of 2 weeks on-treatment followed by 1 week off-treatment. The chemotherapy-alone group had received conventional chemotherapy regimens, including platinum, taxanes, fluoropyrimidine, and gemcitabine. Gemcitabine (1 g/m2) was administered intravenously on days 1 and 8, with cisplatin (80 mg/m2) on day 1. Paclitaxel (135 mg/m2) and docetaxel (80 mg/m2) were administered every 21 days. S-1 dosage (40-60 mg twice daily) was adjusted by body surface area and given for 14 days, followed by a 7-day break, repeated every 3 weeks. Fluorouracil (4 g/m2) was administered via continuous intravenous infusion over 96 h starting from day 1.Baseline evaluation included a physical examination, ECOG status assessment, blood pressure measurement, complete blood count, blood chemistry panel, coagulation function test, routine urinalysis, routine stool test (occult blood testing), and baseline lesion evaluation through contrast-enhanced computed tomography (CT) and magnetic resonance imaging (MRI). Treatment efficacy was assessed after every 2 cycles using contrast-enhanced CT or MRI. Follow-up after the last medication administration aimed to evaluate clinical outcomes, including toxicity, efficacy, and survival, until the patient’s death or data cutoff date.
Endpoints
Efficacy evaluation includes median progression free survival and median overall survival, while safety evaluation includes patient hematologic toxicity and non-hematologic toxicity. The above information is sourced from medical records. The primary endpoint was PFS, defined as the duration from the initiation of vascular-targeting agents plus chemotherapy treatment to either disease progression or death from any cause. Secondary endpoints included OS, defined as the duration from the initiation of vascular-targeting agents plus chemotherapy treatment to death from any cause.15,16 Tumor response was assessed through imaging using RECIST 1.1 by an independent committee every 2 cycles until disease progression or death. AEs were evaluated and graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (version 4.0). 17
Statistical Analysis
Propensity score matching (PSM) was used to balance measurable confounders between combination therapy group and chemotherapy alone group. The propensity score was calculated by logistic regression model with the following covariates age, gender, Chemotherapy drugs, immunotherapy (yes or no), Line of chemotherapy therapy, ECOG PS, metastasis site and initial TNM stage. The matching was performed using a 1:2 nearest-neighbor matching protocol with caliper width 0.2. Continuous variables were expressed as medians and interquartile range (IQR), and were compared using the t test. Categorical variables were presented as frequencies and percentages, and compared using the χ2 test or Fisher’s exact test. Survival rates were estimated using the Kaplan-Meier method. Survival differences were compared using the log-rank test. Cox proportional hazards regression models were used to calculate the stratified hazard ratios (HRs) and the corresponding 95% confidence intervals (CIs). In subgroup analyses, the widths of the 95% confidence intervals have not been adjusted for multiplicity, and therefore inferences drawn from these intervals may not be reproducible. SPSS 20 (IBM, Armonk, NY, USA) and R (version 4.4.1) were used for statistical analyses. All P values were based on two-sided tests, with results deemed statistically significant at P ≤ 0 .05.
Results
Patient Characteristics
Baseline Characteristics

Abbreviations: ECOG, Eastern cooperative oncology group; FP, fluorouracil plus platinum; GP, gemcitabine plus platinum; TP, paclitaxel plus platinum; PSM, Propensity score matching.
Efficacy
By the data cutoff on August 31, 2023, the median follow-up duration post-treatment was 20 months (IQR, 10-34 months) for both groups. Survival outcomes of the patients with RM-NPC are shown in Figures 2 and 3. Before PSM, in the combination therapy group, 31 of the 66 patients (47.0%) had died, compared to 82 of the 132 patients (62.1%) in the chemotherapy alone group. The median PFS in the combination therapy group was 7.0 months (95% CI, 5.7-8.3 months), significantly greater than that in the chemotherapy alone group (median PFS: 5.0 months [95% CI, 4.1-5.9 months]). The addition of vascular-targeting agents reduced the risk of disease progression by 27% (HR, 0.73 [95% CI, 0.54-0.98]; P = 0.034). Median OS was significantly longer in the combination group (37 months [95% CI, 21.3-52.7 months]) than in the chemotherapy alone group (20 months [95% CI, 13.0-27.0 months]), with a HR of 0.66 (95% CI, 0.45-0.97; P = 0.047). After PSM, in the combination therapy group, 26 of the 58 patients (44.8%) had died, compared to 60 of the 92 patients (65.2%) in the chemotherapy alone group. The median PFS in the combination therapy group was 8.0 months (95% CI, 6.7-9.3 months), significantly greater than that in the chemotherapy alone group (median PFS: 5.0 months [95% CI, 4.0-6.0 months]). The addition of vascular-targeting agents reduced the risk of disease progression by 35% (HR, 0.65 [95% CI, 0.46-0.92]; P = 0.014). Median OS was significantly longer in the combination group (45 months) than in the chemotherapy alone group (18 months [95% CI, 9.1-26.9 months]), with an HR of 0.58 (95% CI, 0.37-0.92; P = 0.019). Median PFS in the chemotherapy plus apatinib and chemotherapy plus anlotinib groups were 8.0 months and 7.0 months, and median OS in the chemotherapy plus apatinib and chemotherapy plus anlotinib groups were 45 months and 31 months. Before Propensity Score Matching, Survival Outcomes of the Patients With RM-NPC. Kaplan-Meier Plots Showing Overall Survival (A) and Progression-Free Survival (B) After Propensity Score Matching, Survival Outcomes of the Patients With RM-NPC. Kaplan-Meier Plots Showing Overall Survival (B) and Progression-Free Survival (A)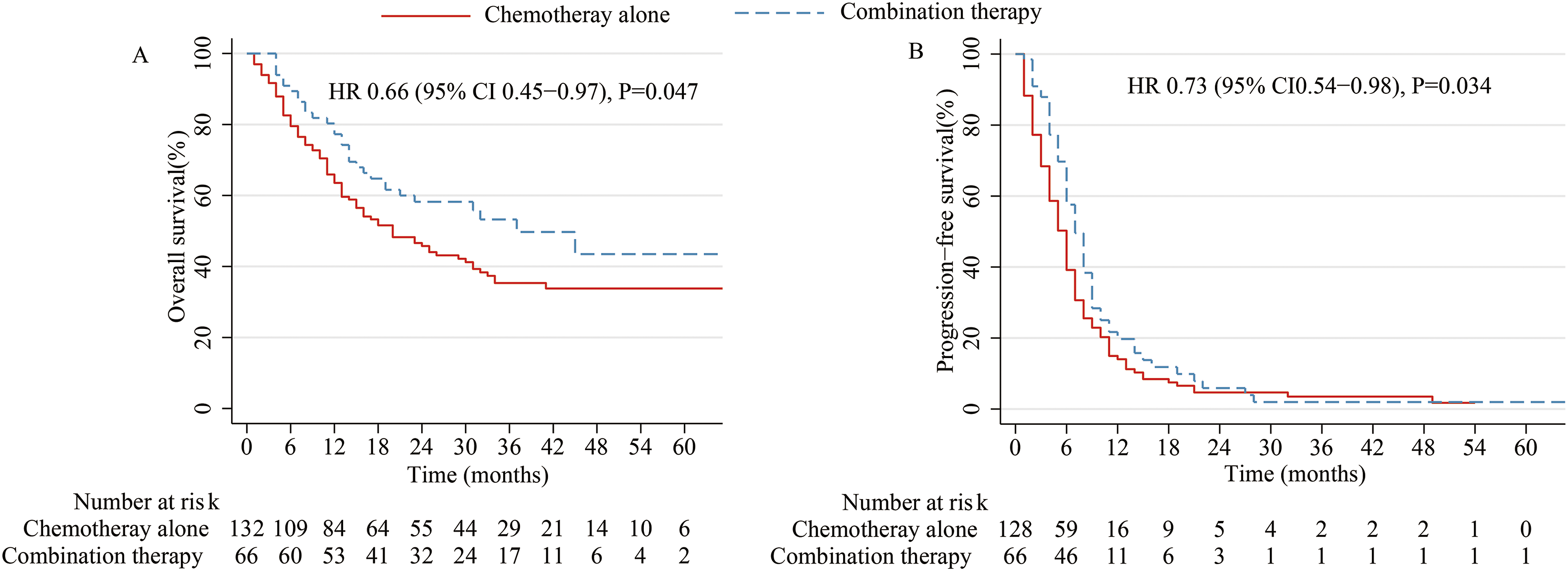

Multivariate Analysis of PFS and OS
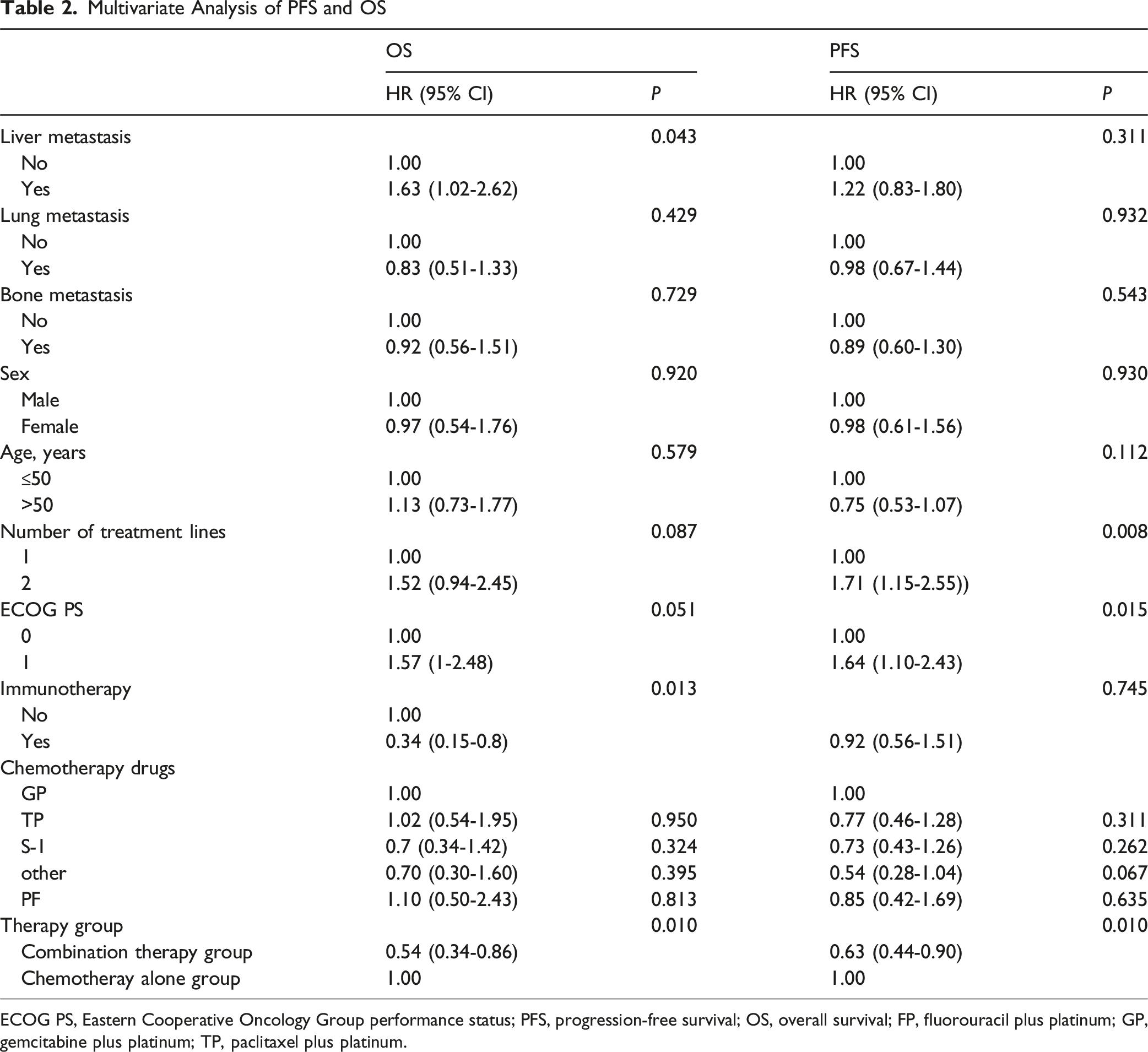
ECOG PS, Eastern Cooperative Oncology Group performance status; PFS, progression-free survival; OS, overall survival; FP, fluorouracil plus platinum; GP, gemcitabine plus platinum; TP, paclitaxel plus platinum.
After PSM, subgroup analyses are summarized in Figures 4 and 5, the liver metastasis group demonstrated significantly longer OS with combination therapy than with chemotherapy alone. The treatment group after the failure of first-line therapy for RM-NPC patients showed significantly longer OS with combination therapy than with chemotherapy alone. ECOG PS 1 group exhibited significantly longer PFS with combination therapy than with chemotherapy alone. The lung metastasis group demonstrated significantly longer PFS with combination therapy than with chemotherapy alone, these findings are exploratory and require prospective validation. PFS HRs (Combination Therapy Group vs Chemotherapy Group) in Subgroups (Exploratory). The Widths of the 95% Confidence Intervals Have Not Been Adjusted for Multiplicity, and Therefore Inferences Drawn From These Intervals May Not be Reproducible. Abbreviations: ECOG PS, Eastern Cooperative Oncology Group Performance Status; HR, Hazard Ratio; PFS, Progression-free Survival OS HRs (Combination Therapy Group vs Chemotherapy Group) in Subgroups (Exploratory). The Widths of the 95% Confidence Intervals Have Not Been Adjusted for Multiplicity, and Therefore Inferences Drawn From These Intervals May Not be Reproducible. Abbreviations: ECOG PS, Eastern Cooperative Oncology Group Performance Status; HR, Hazard Ratio; OS, Overall Survival; NR, Not Reached

Safety
Cumulative Acute Adverse Events (after PSM)
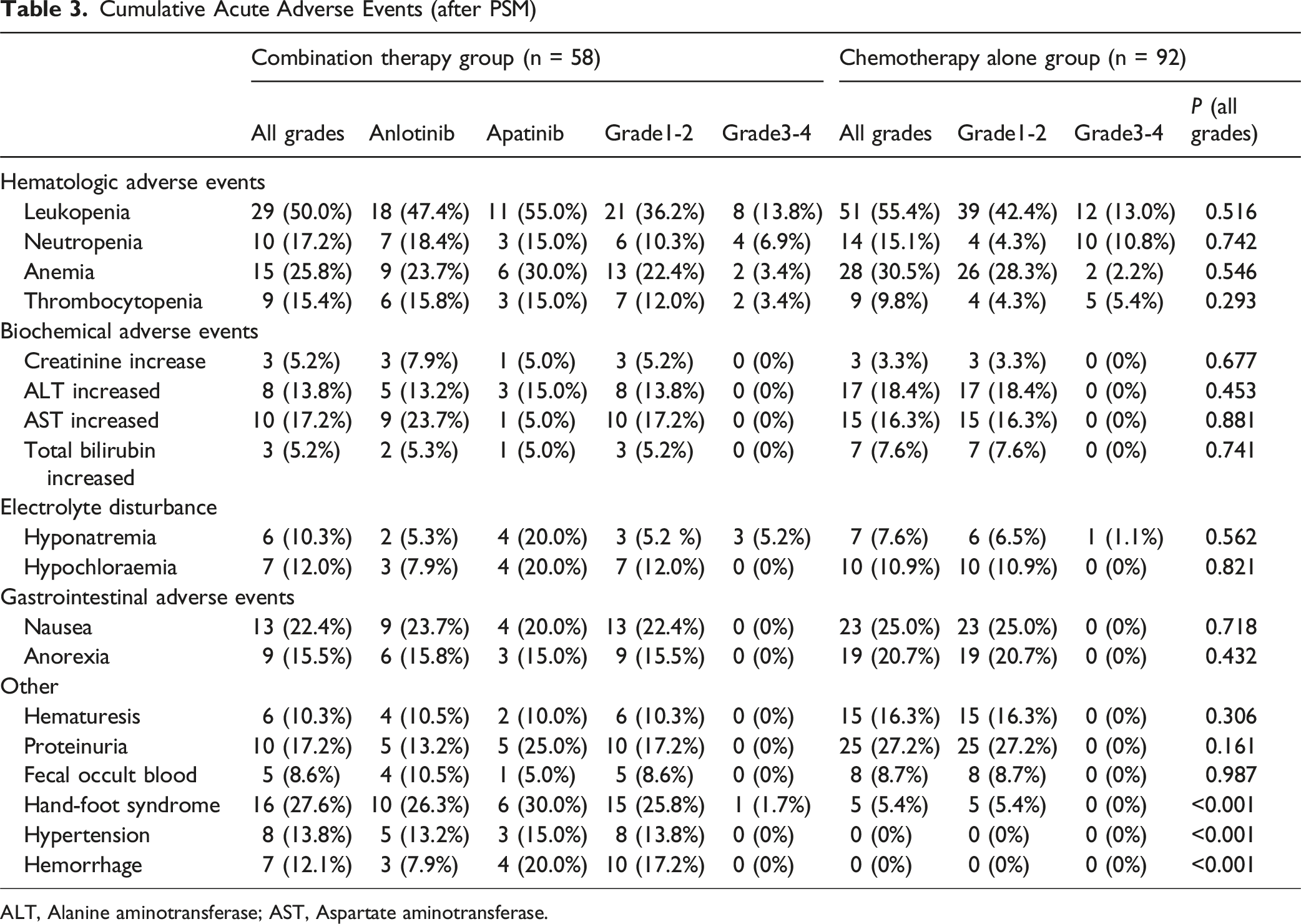
ALT, Alanine aminotransferase; AST, Aspartate aminotransferase.
In the combination therapy group, 8 patients (13.8%) required 1 or more interruptions for vascular-targeting agents, and dose reductions were required in 7 patients (12.1%). The most common reasons for dose reduction were hemorrhage (n = 4; 6.9%) and hand-foot syndrome (n = 3; 5.1%). In the chemotherapy group, dose reductions were required in 24 patients (26.1%).
Above, combining vascular-targeting agents with chemotherapy for RM-NPCs demonstrated greater effectiveness than chemotherapy alone, with an acceptable toxicity profile.
Discussion
In this retrospective study, RM-NPC patients who received vascular-targeting agents (apatinib or anlotinib) plus chemotherapy exhibited effective disease control with an acceptable toxicity profile, particularly those with liver metastases. Subgroup analysis identified liver metastasis as a prognostic factor for treatment response. The liver metastasis group showed significantly longer overall survival and progression-free survival with combination therapy compared to chemotherapy alone. These findings support further clinical development of vascular-targeting agents plus chemotherapy for RM-NPCs, associated with dismal survival outcomes and limited treatment options.
To our knowledge, this was the first study to report the efficacy and safety of vascular-targeting agents plus chemotherapy in the treatment of RM-NPCs. Apatinib is a novel VEGFR-2 tyrosine kinase inhibitor (TKI) that selectively targets intracellular ATP-binding sites and has demonstrated efficacy against various solid tumors. Anlotinib (AL3818), an oral small-molecular TKI, significantly inhibits angiogenesis and tumor growth by inactivating VEGFR and other factor receptors. Apatinib and anlotinib, as oral vascular targeting agents for advanced tumors, demonstrated potential in our study. Combination therapy was associated with improved PFS and OS compared to chemotherapy alone, with manageable toxic profiles. Median PFS in the combination therapy and chemotherapy alone groups were 8.0 months and 5.0 months, respectively, representing a 35% reduction in the risk of disease progression. Median OS was significantly longer in the combination therapy group (45 months) than in the chemotherapy alone group (18 months), reflecting a 42% reduction in the risk of mortality.
NPCs are known to be sensitive to platinum-based chemotherapy, with response rates ranging from 40% to 65%. Gemcitabine-cisplatin chemotherapy is an essential treatment for RM-NPCs, with response rates of 67% and a median OS of 29.1 months in a study by Zhang et al.18,19 The extension of full-dose chemotherapy after disease control may be challenging due to cumulative toxic effects and diminishing benefits. However, in the absence of further interventions, most patients with responsive metastatic NPCs experience disease progression soon after the first-line treatment. Compared with full-dose chemotherapy, combination therapy has good tolerability and could be an appealing strategy to further improve outcomes.
There is a lack of standard second-line treatment regimens after the failure of first-line platinum-based chemotherapy for RM-NPC patients. These patients represent those with the most urgent unmet therapeutic needs. Several antiangiogenic multitargeted kinase inhibitors have been investigated in RM-NPCs. Early trials of sorafenib and sunitinib have demonstrated modest efficacy in RM-NPCs, but with a significant risk of fatal hemorrhage.20,21 In another study by Ruan et al, apatinib therapy for RM-NPCs exhibited objective responses in 36% of the 33 patients, achieving a median PFS of 5 months and a median OS of 16 months. 13 In our study, combination therapy outperforms historical controls (study of Ruan et al), median PFS and OS in the chemotherapy plus apatinib group were 8.0 months and 45.0 months. Combination therapy involves the integration of targeted treatment and chemotherapy, which have synergistic effects and enhance drug efficacy. In a phase II clinical study by Zhou et al., the addition of the anti-VEGF antibody, bevacizumab, to paclitaxel plus carboplatin as first-line treatment failed to extend the PFS and OS of RM-NPC patients. 22
Tumor progression is driven in part by hypoxic stress and oncogenic mutations in genes such as K-ras, p53, and HER2/ErbB2, which collectively induce VEGF overexpression in cancer cells. 23 Concurrently, VEGFR-2 is markedly upregulated in tumor-associated vasculature relative to normal vessels.24,25 Previous studies have reported VEGF over-expression in over 60% of clinical NPC biopsy samples. 12 After the binding of VEGF and VEGFR-2, the carboxy-terminal TK domain of the receptor undergoes autophosphorylation, leading to the downstream transduction. Apatinib inhibits VEGFR-2 phosphorylation by competitively binding to its intracellular ATP-binding site, thereby blocking key downstream pathways: the Raf/MEK/ERK pathway (endothelial proliferation), p38-MAPK pathway (endothelial migration), and PI3K/AKT/mTOR pathway (cell survival and vascular permeability). Thus, by targeting VEGFR-2 signaling, apatinib effectively impairs endothelial proliferation, migration, and vascular permeability. 26 In an in vivo murine model of NPC, results demonstrated that combined treatment with apatinib and cisplatin produced significant antitumor effects. 27 Angiogenesis-related molecules can be promising therapeutic targets in NPC. These results provide a rationale for the superior efficacy observed with combination therapy compared to monotherapy in our study.
The combination therapy group experienced more AEs than the chemotherapy alone group. In particular, hypertension, hand-foot syndrome, hemorrhage, and proteinuria occurred more frequently in the combination therapy group. As observed in previous studies by Ruan et al and Fang et al, the administration of vascular-targeting agents, such as apatinib and anlotinib, was associated with hypertension and hand-foot syndrome, constituting the most common TRAEs.13,28 Grade 3 or 4 hand-foot syndrome (1.7%) was the only severe AEs in the combination therapy group. However, most of the AEs were manageable through dose modifications, and no treatment-related deaths occurred. The safety profile of combination therapy appeared generally acceptable among patients receiving treatment for RM-NPCs.29,30
In a phase II clinical study of Pu et al, anlotinib plus docetaxel offers a viable therapeutic alternative for patients with advanced NSCLC who failed first-line platinum-based treatments. 31 In another study of Cao et al, Apatinib combined with oral etoposide was feasible in pretreated advanced triple-negative breast cancer. 32 In the treatment of recurrent and metastatic nasopharyngeal carcinoma, immunotherapy also plays a significant role. In a phase II clinical study of Wang et al., 33 toripalimab therapy for RM-NPCs exhibited objective responses in 20.5% of the 190 patients. In another study by Ruan et al., apatinib therapy for RM-NPCs exhibited objective responses in 36% of the 33 patients. The response rate to targeted therapy is higher than immunotherapy. In sequence, we can first use targeted therapy followed by immunotherapy. This study was the first cohort study to compare the efficacy and safety of vascular-targeting agents plus chemotherapy with chemotherapy alone in the treatment of RM-NPCs. However, this study also had some limitations. First, this was a retrospective study and selection bias could not be excluded because of the lack of randomization. Second, the population size for the study was relatively small, the results of subgroup analysis may be limited by the small cohort size, these findings are exploratory and require prospective validation. Thirdly, this was a retrospective study, so the calculation of the sample size selected in this study is lacking. Finally, only patients from 4 hospitals were enrolled in the trial, limiting the generalizability of the results. Future studies should assess the efficacy and safety of vascular-targeting agents plus chemotherapy in NPC patients from diverse ethnicities across different countries.
Conclusions
The addition of vascular-targeting agents to chemotherapy for the treatment of RM-NPCs exhibited superior efficacy compared to chemotherapy alone, along with a manageable safety profile. Further randomized controlled and prospective clinical studies with larger sample sizes are needed to confirm the efficacy of vascular-targeting agents combined with chemotherapy in RM-NPCs.
Footnotes
Ethical Considerations
The ethics approval numbers are: 2022YJSLL-26 (Medical Ethics Committee of The Affiliated Hospital of Guilin Medical University); 2022045 (Medical Ethics Committee of Lingshan County People’s Hospital); LL-2025-062-01 (Ethics Committee of Wuzhou Red Cross Hospital); and IRB-2022-047 (Ethics Committee of Nanxishan Hospital of Guangxi Zhuang Autonomous Region).
Consent to Participate
We have obtained written informed consent from the study participant. Patients have consented to access to their medical records and follow-up information.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Guangxi Medical and health key discipline construction project; The “139” Talent Cultivation Plan Program of Guangxi Medical High-level Backbone, The National Natural Science Foundation of China (No. 82160479); The CSCO Youth Innovative Oncology Research Fund (No. Y-Young2020-0520). The funders of the study had no role in study design, data collection, data analysis, data interpretation, as well as writing the report. The corresponding author had the sole responsibility for choosing to submit the report for publication, and had complete access to all the data generated during the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
