Abstract
Purpose
To assess the value of pretreatment neutrophil-to-lymphocyte ratio (NLR) and systemic immunoinflammatory index (SII) in the prognosis of nasopharyngeal carcinoma (NPC) patients.
Methods
This retrospective study analyzed a total of 185 NPC patients who visited the clinic from June 2015 to December 2018 and were selected as study subjects. The NLR and SII were calculated based on the collection of demographic information, clinical characteristics, and pre-treatment lymphocyte counts, neutrophil counts, and platelet counts. Predictive efficacy was evaluated using the receiver operating characteristic (ROC) curve, and survival analysis was performed through life table methods and Cox risk-proportional regression.
Results
Using the X-tile software, significant differences were found in clinical factors among NPC patients based on NLR (>2.91) and SII (>535.47). Age, TNM staging, SII, and NLR were identified as independent prognostic factors in a Cox regression analysis. SII had the highest area under the curve (AUC) for predicting 1-year survival, TNM staging had the highest AUC for predicting 3-year survival, and NLR had the highest AUC for predicting 5-year survival. The combined model showed superior predictive accuracy across all time points.
Conclusion
NLR and SII, as biomarkers of inflammation and immune status, have significant clinical applications in the prognostic assessment of NPC. The integrated prediction model combining age, TNM staging, SII, and NLR significantly improved the accuracy of survival prediction and provided a reliable basis for individualised treatment of NPC.
Plain Language Summary
This study looked at two blood markers, the neutrophil-to-lymphocyte ratio (NLR) and the systemic immunoinflammatory index (SII), to see if they can help predict how patients with nasopharyngeal carcinoma (NPC) will do over time. NPC is a type of cancer that occurs in the upper part of the throat behind the nose. We analyzed the medical records of 185 patients who visited a clinic from 2015 to 2018, checking their NLR and SII levels before treatment. We found that higher levels of these markers are linked with a more advanced disease and could influence the survival chances of the patients. Patients with high levels had a worse prognosis, suggesting that these markers could help doctors figure out a patient’s condition more accurately and possibly tailor treatments more specifically to improve outcomes. This research indicates that looking at inflammation and immune status through these markers could be a valuable tool in managing and treating NPC, aiming to enhance the precision of survival predictions and guide treatment decisions.
Introduction
Nasopharyngeal carcinoma (NPC), one malignancy of the head and neck that originates from the mucosal epithelium of the nasopharynx, has significant geographical distribution and epidemiological features. 1 Per the findings of the International Agency for Research on Cancer (IARC), 133 000 new cases of NPC are expected to occur in 185 countries around the world in 2020, of which about 80 000 will die from the disease, with about 70 per cent of new cases concentrated in eastern and southeastern Asia, with a high incidence in China. 2 As the primary tumour focus is often located at the top or lateral wall of the nasopharynx, it is not easy to remove it completely by surgery, making radiotherapy the main treatment modality. 3 The pathological type of NPC is predominantly non-keratinising undifferentiated carcinoma, which is extremely sensitive to radiotherapy. 4 Clinically, the signs and symptoms of NPC lack specificity, and patients often present with enlarged cervical lymph nodes as the first symptom; therefore, the diagnosis of locally advanced disease is reported to occur in 60%–70% of patients, significantly impacting the therapeutic efficacy and the survival rate of patients. 5
Although recent advances in radiotherapy techniques, particularly the introduction of intensity-modulated radiotherapy (IMRT) as well as chemotherapy regimens have brought notable improvement in local control and overall survival in NPC patients, treatment challenges remain. 6 Currently, NPC patients have a recurrence rate and distant metastasis rate of 5%–15% and 15%–30%, respectively, which leads to an increased risk of treatment failure. 7 Existing Tumor Node Metastasis stage (TNM) staging systems are mainly based on anatomical features, but they do not fully reflect the patient’s biological heterogeneity, causing notable differences in the outcomes of patients at the same stage. Such limitations have prompted investigators to explore new prognostic markers to compensate for the shortcomings of the TNM staging system and thereby improve the clinical management and treatment outcomes of NPC. 8
Over the past few years, a growing body of research verified the close association between systemic inflammatory changes and tumour cell development, proliferation, metastasis, angiogenesis and progression. 9 Tumour cells evade immune surveillance by avoiding monitoring by immune cells, which leads to the spread and development of cancer cells. 10 Inflammation is considered one of the seven hallmarks of cancer, and the development of about 25 per cent of cancers worldwide is closely linked to inflammation. 11 Increasing research implies that the association between inflammation and cancer can be explained by both endogenous and exogenous pathways, with endogenous inflammatory pathways driven through oncogenic inflammation-associated genes and exogenous inflammatory pathways guided by the inflammatory state. 12 Infiltration of the tumour mesenchyme by leukocyte subtypes is one of the characteristics of NPC, which includes neutrophils, monocyte macrophages and lymphocytes. It has been found that in all types of solid malignant tumours, the more neutrophils, monocytes, macrophages and platelets infiltrate the tumour tissue, the worse the prognosis of the tumour patient.13,14 Contrary to this, patients with a higher degree of lymphocytic infiltration have been shown to have a better prognosis. 15 The use of Neutrophil-to-lymphocyte ratio (NLR) as well as systemic immunoinflammatory index (SII) as potential prognostic biomarkers has been studied through many solid tumours. 16 This has been demonstrated by many clinical studies on cancers like breast, gastric, and liver with NLR and SII found to be high being associated with poor prognosis. 17 It is suggested that the above markers reflecting systemic inflammation and immune status may provide new insights into the prognostic assessment of tumors and become important references for the prognosis evaluation of NPC.
The study aimed at analyzing NLR and SII expressions in NPC patients, and their link to prognosis. We will look into the possibility of NLRs and SIIs being useful in aiding the determination of clinical outcomes of NPC by studying these markers before and after treatment. The findings of this study will result in better prognostic assessment and management for NPC patients, personalized medicine, improved therapeutic response, as well as patient survival. The results of the study will help to improve the prognostic assessment and management of NPC patients, promote individualised treatment, and improve treatment outcomes and patient survival.
Methods and Materials
Patients
Patient Baseline Characteristics.

Note: Body Mass Index (BMI), Epstein-Barr Virus DNA (EBV DNA), Tumor Node Metastasis stage (TNM), Tumor (T) stage, Node (N) stage, Systemic Immune-Inflammation Index (SII), and Neutrophil-to-Lymphocyte Ratio (NLR).
Inclusion and Exclusion Criteria
Inclusion criteria: patients 18 - 80 years old with a pathologically confirmed diagnosis of NPC at our institution who had not yet received any treatment; a physical condition score of Karnofsky performance scores (KPS) ≥70 or an ECOG score of <2; and possession of complete data on the initial clinical case.
Exclusion criteria: patients with immune system-related diseases who have not received previous immunotherapy; the presence of acute or chronic infectious diseases such as active tuberculosis, hepatitis B, or AIDS; patients with active bleeding, severe infections, diffuse intravascular coagulation, history of heparin therapy, or history of connective tissue disease in the past three months; and women who are pregnant; Patients with other systemic diseases (eg, diabetes) that may affect the measurement of inflammatory markers (eg, NLR and SII); patients with other active tumours; patients who have recently used drugs that may affect the immune or inflammatory response (eg, corticosteroids).
Clinical Data Acquisition
Patient information was obtained from electronic medical records, outpatient review records and follow-up data. Clinical data encompassed age, smoking history, gender, pathological classification, Body Mass Index (BMI), T stage, TNM stage, and N stage. Laboratory parameters included Epstein-Barr Virus DNA (EBV DNA), SII, and NLR.
NLR and SII Detection Method
The SYSMEXS haematology analyser XS-500i was used to measure the patient’s blood counts at the time of their first admission to the hospital. nlr = neutrophil count/lymphocyte count, sii = platelet count*(neutrophil count/lymphocyte count).
Follow-Up
Follow-up was conducted on patients every 3-6 months for 2 years and every 6 months to 1 year for 3-5 years after the end of treatment. Follow-up is mainly by telephone.
Outcome Measurement
(1) X-tile software was adopted for calculating the optimal survival cut-off for NLR and SII. 2. (2) Patients were grouped according to the optimal survival cut-off, and the association of NLR and SII with patients’ pathological data was analysed. (3) Cox regression was performed to analyse the independent prognostic factors affecting patients’ 5-year survival. (4) K-M survival curves were generated for analysing the differences in 5-year survival for indicators with differences in one-way Cox regression. (5) The value of independent prognostic factors for 5-year survival in predicting patients’ 1, 3, and 5-y survival was analysed via time-dependent ROC curves. (6) Construct a Cox regression model based on the beta coefficient of multifactorial Cox regression, and analyse the value of the model in forecasting patients’ 1, 3, and 5-y survival (Figure 1). Sample selection and study protocol Flowchart.

Statistical Analyses
Collected data were analysed using SPSS 26.0 and visualised using Prism 9. The K-S test was adopted for analysing data distribution. Normally distributed data were analysed via the t-test, and the independent sample t-test was adopted for intergroup comparisons, which were expressed as t. The clinical data were partially analysed using count data and the chi-square test, which was expressed as χ2. Cox regression was conducted for screening prognostic factors for the NPC patients’ 5-year survival. The time-dependent ROC curve was generated for analysing the value of independent prognostic factors and risk scores in forecasting NPC patients’ 1-, 3- and 5-y survival. The calibration, time-dependent ROC and DCA curves were drawn using the R software rms, survROC and DCA packages. P < 0.05 implies a conspicuous difference.
Results
Association of NLR Levels with Patient Baseline Data
Relationship of NLR Levels With Pathological Data.

Note: Body Mass Index (BMI), Epstein-Barr Virus DNA (EBV DNA), Tumor Node Metastasis stage (TNM), Tumor (T) stage, Node (N) stage, Systemic Immune-Inflammation Index (SII), and Neutrophil-to-Lymphocyte Ratio (NLR).
Relationship Between SII Levels and Patient Baseline Data
Relationship Between SII Levels and Pathological Data.

Analysis of Risk Factors Affecting NPC Patients’ 5-Year Survival
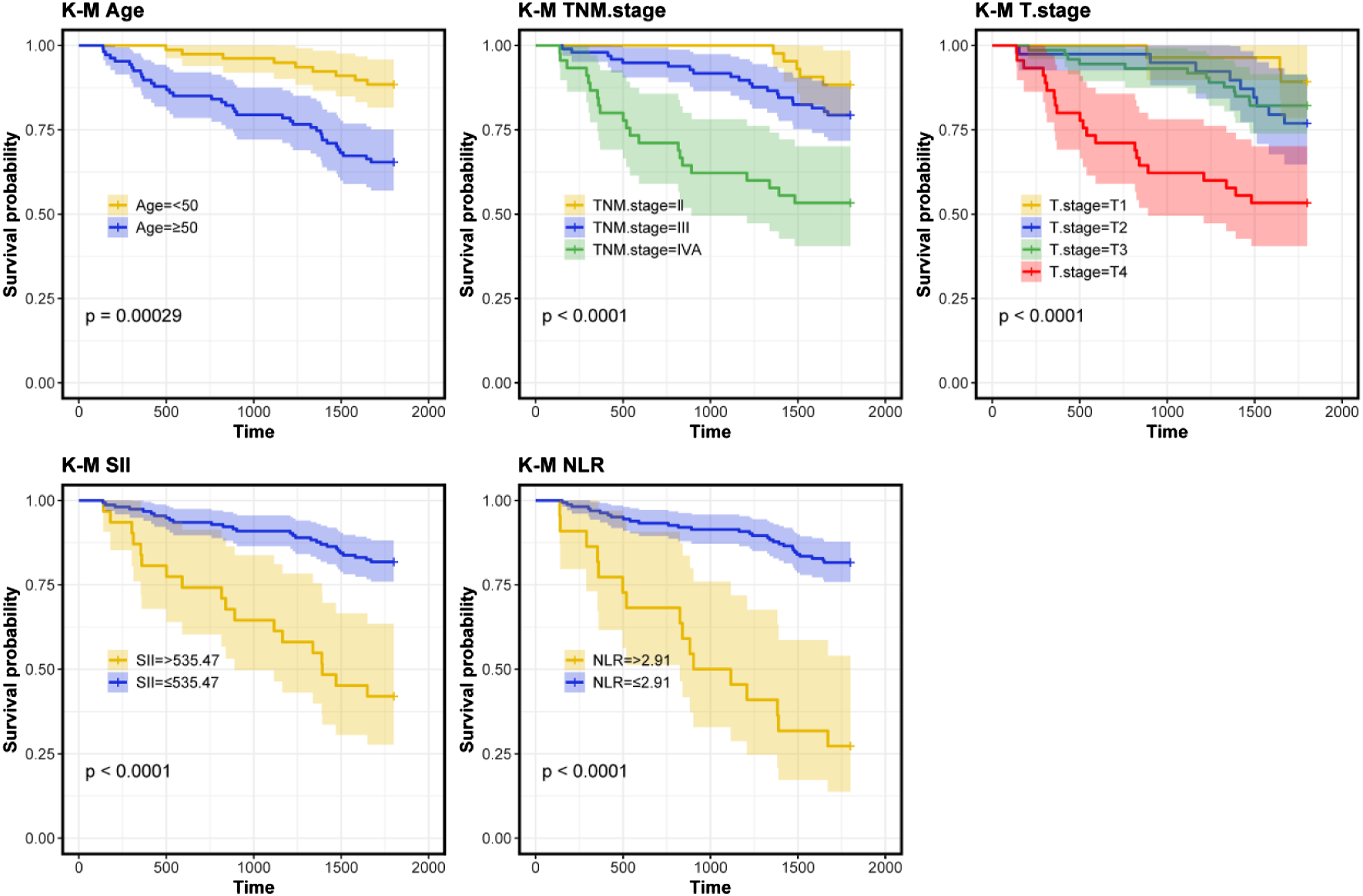
For further understanding of the risk factors impacting NPC patients’ 5-year survival, we analysed them using Cox regression. Univariate Cox regression identified associations of age (P = 0.001), TNM stage (P < 0.001), T stage (P = 0.001), SII (P < 0.001) and NLR (P < 0.001) with 5-year survival (Figure 2). Survival curves for the five factors were generated (Figure 3). Subsequently, a multivariate Cox regression analysis identified age (P = 0.001), TNM stage (P = 0.034), SII (P < 0.001) and NLR (P < 0.001) as independent prognostic factors impacting patients’ 5-year survival (Figure 4). Univariate Cox regression analysis of factors impacting NPC patients’ 5-year survival. K-M survival curves for age, TNM stage, T stage, SII and NLR indicators. Multivariate Cox regression analysis of factors impacting NPC patients’ 5-year survival.

Predictive Value of Independent Prognostic Factors for 1-, 3- and 5-Year Survival
We used time-dependent ROC curves for analysing the predictive value of the four independent prognostic factors for 1-, 3- and 5-year survival. The results showed that SII had the best Area Under the Curve (AUC) (0.847) for predicting 1-year survival, TNM stage had the best AUC (0.795) for predicting 3-year survival, and NLR had the best AUC (0.703) for predicting 5-year survival (Figure 5). ROC curve for independent prognostic factors predicting 1, 3 and 5-year time-dependent NPC A. ROC curve for age as a predictor of 1, 3 and 5-year time-dependent NPC B. ROC curve for forecasting 1, 3 and 5-year time-dependent NPC patients in the light of TNM staging C. ROC curve for SII predicting 1, 3 and 5-year time-dependent NPC D. ROC curve for 1, 3 and 5-year time-dependent ROC curve for NLR in NPC patients.
Cox Model Constructed for Survival Prediction in NPC patients
The final question of the study was to establish one prediction model through the β coefficient of Cox regression: age*1.314+TNM stage*0.959+SII*1.402+NLR*1.283. Subsequently, the ROC curve analysis found a higher model’s AUC at 1, 3, and 5 years than the best value of the four individual indicators (Figure 6A). Subsequently, the calibration curve verified a certain degree of stability of the model in forecasting the patients’ 1, 3 and 5-years survival (Figure 6B). Finally, the DCA curve revealed a benefit rate of the four prognostic factors and the risk score in forecasting the patients’ 1, 3 and 5-years survival, and the figure showed a higher benefit rate of the risk score than other indicators, indicating that certain clinical value of the model (Figure 6C). Cox model constructed for survival prediction in NPC patients A. Time-dependent ROC curve for evaluating the model’s ability to predict patients’ 1, 3, and 5-years survival B. Calibration curve for assessing the stability of the model in predicting patients’ 1, 3, and 5-years survival C.DCA curve for assessing the clinical value of the model in predicting 1-, 3-, and 5-year survival.
Discussion
The prognosis of NPC is influenced by a multitude of factors, including the disease stage, treatment method, patient’s age as well as general health. 19 Early diagnosis and timely treatment are the key to improving prognosis. However, due to the hidden location of NPC, many patients present with advanced-stage NPC at diagnosis, which makes treatment more difficult and the prognosis worse. 20 Although the advances in IMRT have significantly improved local control rates, some patients still experience recurrence and metastasis, which seriously affects long-term survival rates. 21
NLR is an inflammatory biomarker that has received widespread attention in cancer research in recent years, NLR is a parameter that provides insights into the body’s inflammatory state and immune function by calculating the ratio of neutrophils to lymphocytes in peripheral blood. 22 Some studies have found significantly higher levels of NLR in tumour patients than in the normal population. 23 For example, studies such as Moon et al. 24 have verified increased NLR as one independent prognostic factor for late recurrence and its ability to serve as one reliable, easily accessible and cost-effective test. In addition, Huszno et al. 25 found the ability of NLR before treatment to serve as a prognostic indicator for patients with prostate cancer and an independent prognostic factor. This study found that when the NLR ≤2.91, the number of patients with high TNM stage and age ≥50 years old increased significantly, and the number of patients with SII >535.47 also increased, which was statistically different from that of patients with >2.91. This is due to the fact that high NLR may mean that there is an ongoing inflammation in the body that fuels cancer development and growth.26,27 Inflammation process with a significant role for neutrophils. This triggers the release of various cytokines, chemokines, and enzymes by neutrophils, which stimulate the proliferation of tumor cells, angiogenesis, and metastasis. 28 Moreover, they may also inhibit lymphocyte function thereby suppressing antitumor immune response thus favouring tumour cell expansion. A decrease in lymphocytes may reflect a decrease in the body’s immune function, which cannot effectively identify and eliminate tumour cells. 29 The decrease in lymphocytes and the increase in neutrophils together result in a high NLR value, which implies a poor prognosis.
SII is one comprehensive inflammatory indicator that combines neutrophils, lymphocytes and platelets. 30 Fest et al found that higher levels of SII at baseline in healthy individuals were associated with a significantly increased risk of developing solid cancers. 31 SII not only reflects the patient’s inflammatory state, but also comprehensively considers immune function, and is therefore considered a more comprehensive and sensitive prognostic indicator.32,33 The prognostic value of SII in various cancers has been widely verified. For instance, in a meta-analysis, 34 totally of 30 studies on gastric cancer and SII were analysed, and it was found that patients with gastric cancer and high SII levels before treatment had notably poorer overall survival and disease-free survival, and their prognosis was poor. In addition, Tian et al. 35 found that when SII >750, the overall survival and disease-free survival of cancer patients treated by immune checkpoint inhibitors were significantly shortened. In this study, we found that when SII >535.47, EBV DNA ≥400, TNM high stage, T high stage and NLR >2.91, the number of patients will increase significantly. Patients with high SII values usually have higher tumour burden, stronger invasiveness and lower survival rate. Platelets play a crucial part in the growth and metastasis of tumours. They not only help to form blood vessels for the tumour but also promote the implantation and growth of tumour cells in distant organs by protecting them in circulation. 36 Moreover, the advantage of SII is that it may comprehensively reflect the patient’s inflammatory as well as immune status, which has a higher predictive value than a single NLR or other inflammatory indicators. By monitoring SII, doctors may more accurately assess the patient’s condition and develop more personalised treatment plans, with the aim of improving the patient’s treatment outcomes and long-term survival rate.
In this study, we further analysed the prognostic factors impacting patients’ 5-year survival using Cox regression. The results identified age, TNM stage, SII and NLR as independent prognostic factors affecting the NPC patients’ 5-year survival. Several reports have shown that age and TNM stage affect the prognosis of NPC patients.37,38 Wang et al. 39 reported that it could be acted as one independent predictor in prognosis and survival in NPC patients, with high accuracy of the prediction by SII. According to a study by Liu et al. 40 the SII was one independent prognostic biomarker correlated to poor OS as well as DFS in newly diagnosed metastatic NPC patients. The previous study by Ye et al. 41 confirmed NLR as one predictor of survival in advanced NPC patients. The fact that an increase in SII and NLR results in a worse balance between inflammation and immunity may be an essential indicator of the NPC patients’ prognosis. Through this study, the role of SII and NLR in the prognosis assessment of NPC was further verified. We found that SII performed particularly well in short-term survival prediction, while NLR also had significant value in long-term survival prediction. According to the time-dependent ROC curve analysis results, the AUC of SII in predicting patients’ 1-year survival is 0.847, the AUC of TNM staging in predicting patients’ 3-year survival is 0.795, and the AUC of NLR in predicting patients’ 5-year survival is 0.703. Considering these biomarkers together may more accurately assess survival prediction at different stages and improve the scientific nature of clinical treatment decisions. At the end of the study, we constructed a comprehensive prediction model based on Cox regression models, which further improved the accuracy of survival prediction by combining the risk scores of age, TNM stage, SII and NLR. Previously, Liu et al. 42 developed a forecasting model for the survival of patients who suffered nasopharyngeal squamous cell carcinoma using one line chart. The AUC of their model for forecasting the patients’ 1-, 3-, and 5-year survival was greater than 0.75. The AUC of the model in this study for predicting the 1-, 3-, and 5-year survival of patients was higher than that of any single indicator, and the AUC of the prediction was greater than 0.8. The finding implies that the comprehensive prediction model constructed by the study has higher accuracy and reliability in survival prediction. This result shows that considering multiple prognostic factors may significantly improve the performance of the prediction model. In particular, combining biomarkers of inflammation and immune status (such as SII and NLR) with traditional clinical factors (such as age and TNM stage) may provide more comprehensive information on patient survival.
This study has also some space to improve. The pre- and post-implementation survey and focus group data that were reviewed to answer this question were not randomized, which introduces potential selection bias and information bias, and may not be completely accurate and/or unbiased Second, the sample was drawn from a single hospital and so has not been validated with an independent external dataset, and the findings may not be generalizable. The very limited amount of patients in the sample (185) and the short follow-up time hindered the statistical analysis stability and thus the long-term survival and long-term prognosis could not be wholly estimated. Last but not least, we only had a brief look at the impact of different therapy modalities on prognosis, and it would take a major part in the patients’ survival rate and prognosis. A larger sample size, longer follow-up time, multicenter, the status of different treatment model and external data confirmation are needed in the follow-up research for confirming the reliability and generalization of the conclusions of the study.
Conclusion
NLR and SII are of significant clinical application value regarding the prognosis assessment of NPC as biomarkers of inflammation and immune status. The comprehensive prediction model combining age, TNM stage, SII and NLR significantly improves the accuracy of survival prediction and provides a reliable basis for the individualised treatment of NPC patients.
Supplemental Material
Supplemental Material - High Expression of NLR and SII in patients With Nasopharyngeal Carcinoma as Potential Prognostic Observations
Supplemental Material for High Expression of NLR and SII in patients With Nasopharyngeal Carcinoma as Potential Prognostic Observations by He Lv, Xing Chen, Xudong Chen, Ji Li and Xu Lu in Cancer Control
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research received funding from the Zhejiang Province Medical and Health Project, Project name: IL-39 regulates the role of allergic rhinitis through STAT3 signalling and its diagnostic reagents, Project number: 2024646849.
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
