Abstract
When considering treatment-related side effects, cancer therapies often present the most significant severity. Given considerations of safety and efficacy, there is a growing inclination towards biological therapeutic approaches, with Oncolytic Viruses (OVs) emerging as a prominent focus. Initial investigations into OVs commenced with Adenovirus, subsequently leading to the study of other viruses, including Herpes Simplex Virus (HSV), Reovirus, and measles virus, evaluating their potential safety profiles and antitumor efficacy in human subjects. The fundamental principle underlying OV-based cancer therapy lies in harnessing viral infectivity and immunogenicity, which are engineered for selective targeting of tumor cells. Following the US Food and Drug Administration (FDA) approval of the HSV-based therapy (T-VEC) for Melanoma, interest in exploring other viral platforms for the potential treatment of various cancer types has notably expanded. A considerable number of OVs have progressed to various stages of clinical evaluation. This review provides an in-depth examination of OV classification, mechanisms of action, delivery strategies, and crucial genetic modifications, furthermore summarizing data from pivotal trials to underscore the efficacy and advancement of OVs from investigational modalities to clinical implementation. For the scope of this review, clinical trial data spanning the past 11 years (2013 to 2024) were accessed from ClinicalTrials.gov and further categorized according to their ongoing clinical trial phases and their utilization in combination with Immune Checkpoint Inhibitors (ICI) or other established therapies. In summary, this work presents a comprehensive overview of recent developments within the field of cancer therapy, specifically concerning Oncolytic Viruses.
Plain Language Summary
This study explains how specially engineered viruses can safely kill cancer cells and help the immune system fight tumors more effectively 4) Plain Language Summary: - This review explores the role of oncolytic viruses (OVs) in cancer therapy. It discusses how these viruses are engineered to target and destroy tumor cells while stimulating immune responses, with growing clinical success—especially when combined with other treatments like immunotherapy and chemotherapy.
Introduction
Despite significant medical advancements in the 21st century, cancer remains a leading cause of mortality and morbidity globally.1,2 Conventional treatments, primarily surgery, chemotherapy, and radiotherapy, often serve as short-term solutions associated with considerable side effects on the patient’s body. This has prompted a growing emphasis on biological therapies, particularly immunotherapy and biotherapy, leading to a significant focus on oncolytic virotherapy.
The fundamental principle of OV therapy lies in engineering viruses with oncolytic potential to specifically infect and target tumor cells. These modified viruses replicate within cancerous cells and induce lysis, directly destroying the tumor.3,4 Furthermore, the viral infection and subsequent lysis release pathogen-associated molecular patterns (PAMPs), which alert the host immune system to the presence of the tumor, initiating an immune response.5,6 Some OVs are further modified to release human cytokines, directly enhancing this immune-mediated attack. This dual killing mechanism, combining is intended to achieve more effective cancer clearance. 6 Certain OVs can also contribute to tumor eradication by inhibiting angiogenesis and inducing indirect cell death. 7 A crucial advantage of oncolytic therapy, distinguishing it from conventional treatments, is the sparing of healthy, non-cancerous cells.
The idea of viruses assisting tumor clearance is not new; tumor regressions coinciding with natural virus infections have been reported since the mid-1800s.8-10 Clinical trials with non-attenuated wild type viruses began in 1949. 8 Although the concept has existed since long prior, studies on oncolytic virus as a focal point has rapidly increased in the past two decades. And more recently we have come far enough to take the concept out in the real world for actual battle against cancer. Talimogene laherparepvec (T-VEC) or IMYLYGIC® is already approved by the FDA to treat metastatic melanoma.11–14 In 2021 Japan approved Delytact (teserpaturev/G47∆) for treating malignant glioma. 15 OVs approved in other countries include RIGVIR (Enteric cytopathic human orphan virus type 7) for melanoma treatment in Latvia (2004),16,17 and Oncorine (modified type 5 human Adenovirus or, HAdV-C5) for head and neck squamous cell carcinoma in China (2005). 18 Other viruses to be used against several other types of cancer are still in pipeline.
Despite the theoretical promises OVs show, their use as monotherapy can still be problematic. Viral genes being so adaptable to changes has given us a wide array of viruses to work with. While some viruses are engineered to produce cytokines, others are altered to make immune checkpoint inhibitors themselves. For instance, An HSV-1 was armed with single-chain Fragment Variable (scFv) antibodies that binds to PD-1 in mouse models of Glioblastoma. 19 Moreover, in recent years, the spectrum of viruses used in cancer therapy has broadened beyond traditional oncolytic viruses. One such example is PV001-DV, an attenuated strain of the dengue virus that has been developed to treat solid tumors. 20 Some OVs can also induce apoptosis or programmed cell death in cancer cells. For instance, the vaccinia virus can release substances such as ATP and high mobility group box-1 protein that stimulate cell death pathways.21,22 This can help further reduce the tumor size and improve treatment outcomes. Another study showed, Oncolytic Newcastle disease virus can cause p53-independent apoptotic cell death by inducing endoplasmic reticulum stress. 23 Studies to combine different cytokine (TNFs, IL-2, IL-12 etc.) producing genes to viral genes are ongoing for many viruses.
Besides, the development stage can be challenging as ensuring the transgenes to be active without interfering with native genes is quite difficult. On the other hand, deletion of a native gene in order to inhibit virulence may result in turning a vital function off. As a result, the selected virus needs to be adaptable and of somewhat known genetic mapping. Even if everything is alright, the therapy might still fail due to the virus being unable to gain access to the site of action or tumor microenvironment (TME). Therefore, the most difficult part of the oncolytic virotherapy is the selection of the virus and a delivery system fitting to the host and the type of cancer being focused.
Classification and General Features of OVs
Oncolytic viruses that are currently being studied can be classified into 2 broad categories, Wild-type or naturally occurring OVs and Genetically modified OVs. The concept of using naturally occurring oncolytic viruses for cancer treatment existed between the mid-twentieths, when several wild type viruses were used in clinical trials for cancer treatment. 8 One major drawback of using the wild-type viruses was uncontrolled pathogenicity. However, the development of newly found wild-type viruses, non-pathogenic to humans, is keeping the focus on naturally occurring viruses. Wild type OVs have natural tropism for tumor specific tissues, as they have the inherent capacity to infect the permissive cells by binding to specific surface markers. For example, Reovirus, a naturally occurring RNA virus targets the RAS activated tumor cells, replicating inside them causing no harm to normal tissues.24,25 Although the primary mechanism of action of wild-type viruses is to lyse the cells directly, they have a significant role in modulating the host immune response targeting the tumor microenvironment.
As unmodified OVs target the tumor cells intrinsically, leaving the healthy cells unharmed, there is less toxicity in using the wild type viruses. However, their clinical efficacy is still unsatisfactory. Due to the lack of tumor cell specificity and poor clinical results, researchers started to focus on genetically modified viruses, as they gave better results in specificity and more robust immune-mediated tumor destruction. Modified OVs have altered surface receptor genes incorporated into them that are essential for binding into the abnormal cells. One example of this is an oncolytic measles virus, where single-chain (scFv) anti-CD20 antibody genes were fused inside the viral genome to target CD20+ tumor cells. 26 Additionally, modified OVs are better equipped to promote antitumor immunity by inducing pro-inflammatory cytokine production and presenting tumor antigens to the APC’s and killer T cells. 27 Whereas, some tumors give poor response against the wild type viruses due to the intratumoral innate immune responses that inhibit those OVs. To overcome this, a wild type measles virus was engineered to express avoidance proteins to combat the IFN system inside the tumor. 28
Major Types of Oncolytic Viruses
Adenovirus
Adenoviruses are non-enveloped dsDNA viruses containing 28-46 kb genome. With a comparatively complex and large structure, they comprise an icosahedral capsid of diameter of ∼950Å. 29 More than 50 serotypes of adenoviruses, classified into 7 subgroups, are currently identified, out of all the serotypes, AD5 and AD2 are most commonly used in cancer therapy. 30 Enadenotucirev, a chimeric oncolytic adenovirus (serotype Ad3/Ad11p), is also in early phase clinical trials for the treatment of colon cancer. 31 H101 (Oncorin), an oncolytic adenovirus, was the first recombinant oncolytic virus to pass the trial phase and was approved by the Chinese State Food and Drug Administration (CFDA) in 2005 for the treatment of head and neck cancer.18,32
Adenoviruses have a broad tissue tropism, which questions their safety and specificity as an oncolytic agent. 33 Therefore, conditional replication of adenovirus has been introduced to use replication deficient adenovirus in cancer therapy. For example, ONYX-015, with partial E1B gene (blocks p53) deletion, has been used to target tumor cells lacking p53 and cannot replicate inside healthy cells because of the p53 dependent apoptosis. 34 The ability to infect cells independently of their division state and their well understood molecular structure has made adenovirus an ideal candidate for oncolytic therapy.
Herpes Simplex Virus
Herpes simplex virus, a member of a large family of dsDNA viruses called Herpesviridae, is among the few oncolytic viruses that moved beyond the phase trials. 35 Due to their big genetic material (∼150 kb) packed inside a small, complex structure, it makes them an ideal choice as a vector for insertion and deletion of new genes. With two most prevalent serotypes (HSV-1 and HSV-2) around the world, it is responsible for some major diseases like meningitis, encephalitis and genital herpes.36,37
T-VEC, a modified version of Herpes simplex type 1 virus (HSV-1) was approved as treatment against melanoma by the FDA for the first time in 2015.12,13,38 The genetic modification not only deemed cancer cells as its target for destruction, but it also had GM-CSF inserted into it,39,40 an immunomodulatory cytokine that induces production and prolongation of cellular and humoral immunity. Currently, T-VEC remains the only OV approved by FDA. Some other well-known modified variants of HSV type-1 are G207 41 and HSV1716.42,43
Reovirus
Respiratory Enteric Orphan virus (Reovirus) is a non-enveloped, double-stranded RNA (dsRNA) virus with segmented genome. Wild types of the reovirus are non-pathogenic, preferentially replicate in the RAS activated tumor cells, reduce tumor proliferation and induce oncolysis. 44 Out of the 6 species of Orthoreovirus, only mammalian orthoreovirus T3D strain (pelareorep), also known as Reolysin is now manufactured as a therapeutic agent.45,46 This unmodified strain has been tested in over 30 clinical trials of varying types of cancer. 47 Their anti-tumor effects are multifactorial, involving both innate and adaptive immune responses.
However, elementary clinical trials of the oncolytic reovirus highlighted the limited outcomes of using reovirus as monotherapy leading to development of combination therapies for better outcomes. An early Phase I trial combining Reolysin with palliative radiotherapy showed safety and partial responses in advanced malignancies. 48 Present studies use reovirus combined with inhibitor molecules that inhibit the BRAF or MEK genes and enhance ER stress response which triggers apoptosis. 49
Vaccinia Virus
Vaccinia virus, a large dsDNA virus with a genome size of ∼190 kb, has been widely studied for vaccine therapy due to its large genome size and well-known safety profile. Despite the unsatisfactory outcomes in an early study in the 1990s using vaccinia virus against melanoma patients, 50 oncolytic therapies using vaccinia strains are still ongoing. Some strains of Vaccinia virus like Pexa-Vec, GL-ONC1, and TG6002 are extensively studied and have shown excellent safety profiles in HCC, 51 Head and neck cancer 52 and colorectal cancer patients 53 respectively.
Due to the large genome, gene insertion and deletion are quite efficient in vaccinia virus-based therapies. Modified vaccinia viruses have a deletion of TK and VGF genes which increases their safety and selectivity, 54 other than that insertion of several pro-inflammatory cytokines and chemokines have shown an enhanced anti-tumor activity of oncolytic vaccinia viruses in advanced malignancies.55–57 Along with big genome size they have natural tropism for the tumor cells with a very short replication period inside the cell. Besides, a stable antigen presentation ability and unrelatedness to specific receptors for entry into the cells are setting them apart from other OVs in the field.
Mechanism of Action
Viral Entry and Selectivity
Cancer cells differ from normal cells mainly in 6 ways: physiology, growth inhibition signal evasion, metastasis, self-sufficiency in growth signals, unlimited growth potential, sustained angiogenesis etc. 58 These hallmark differences provide a great array for viral selectivity in OV immunotherapy. The tumor specificity and entry of oncolytic viruses can be divided into the following categories: Pro-apoptotic targeting, transduction targeting, transcription targeting and translation targeting. OVs first recognize the tumor cells by the specific receptors or markers like osteocalcin, CD-20, nuclear transcription factor, and cyclo-oxygenase-2 produced by the abnormal cells. 59 Some viruses inactivate apoptosis regulators like p53 and pRb in order to delay cell death and assist their own replication.60–62 For example, Adenoviruses produce protein E1A and E1B to inactivate p53 and pRb respectively. 62
P53 is mutated in >50% of human malignancies, making it an important hallmark for cancer cells.63,64 An adenovirus with deletion in E1 will not be able to replicate in healthy cells where p53 induced apoptosis active, but it will easily infect cancer cells, making itself very specifically tumor targeting via pro-apoptosis.65,66 Tumor cells express high levels of tumor specific receptors (Figure 1), which can be used as oncolytic virus targets for example, overexpression of intra cellular adhesion molecule-1 (ICAM-1) and decay accelerating factor (DAF) being the receptors for coxsackievirus A21(CAV 21).
67
Another example can be the CD155 receptor, which is abundant in many human cancer cells and is the receptor for binding poliovirus.
68
General Mechanism of Action by Oncolytic Viruses. General Mechanism of Action by Oncolytic Viruses (OVs). Upon Tumor-specific Entry, OVs Replicate Within Cancer Cells, Causing Direct Cell Lysis and the Release of Tumor-Associated Antigens (TAAs), PAMPs, and DAMPs. These Stimulate Innate and Adaptive Immune Responses, Promoting Systemic Antitumor Immunity. Some Genetically Engineered OVs Also Express Immunomodulatory Molecules to Enhance Therapeutic Efficacy and Immune Activation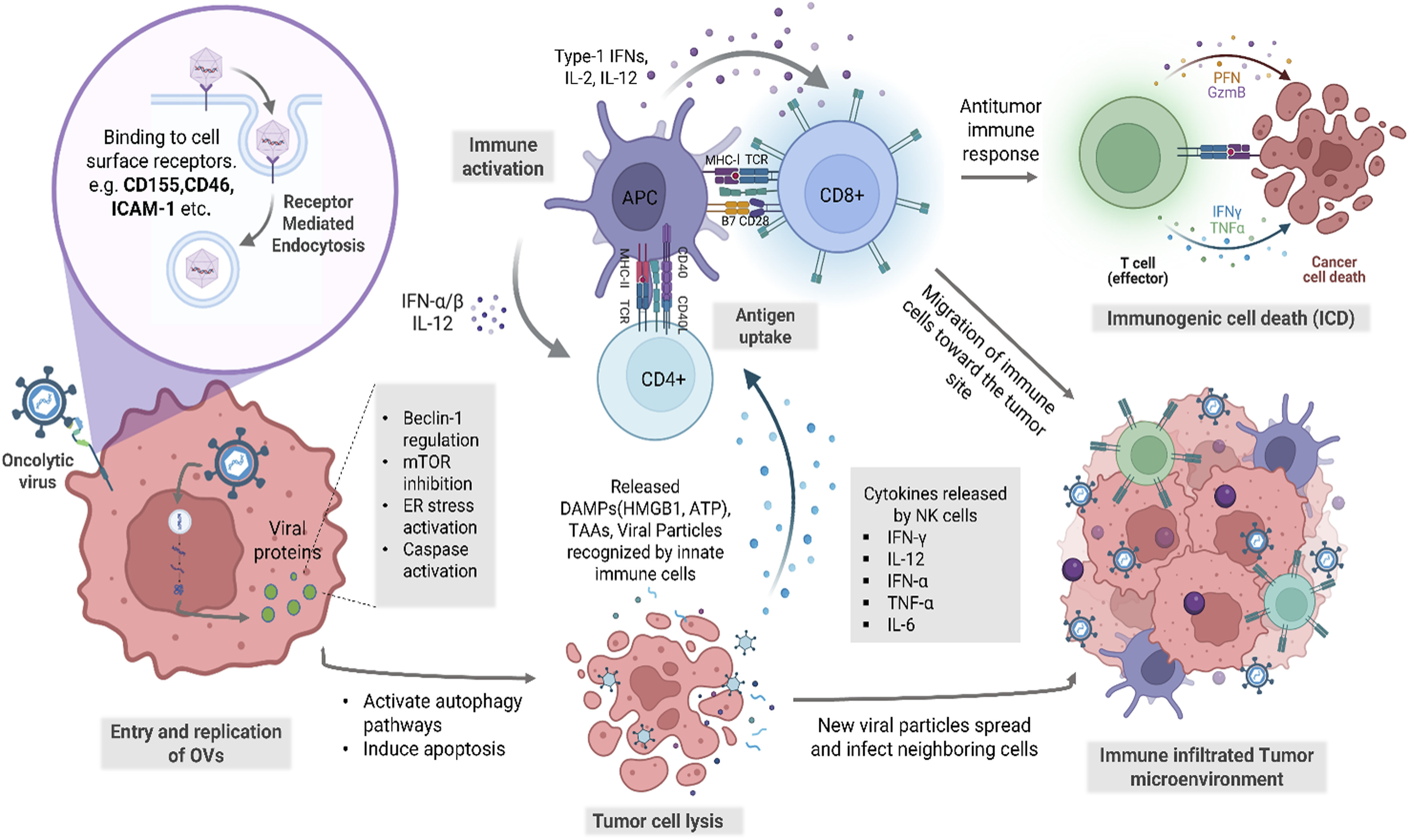
Type 1 interferon or IFN-1 is an important cytokine released in case of viral infection. In some stages of the growth of a cancerous cell, it loses its antiviral mechanism, especially the ability to produce IFN-1. 69 In case of viruses like vesicular stomatitis virus (VSV), which expresses M protein for suppression of genes including the genes responsible for IFN-1 70 and other antiviral mechanism expression, a mutation in M protein producing gene makes them ineffective on normal cells, which are able to produce IFN-1, rendering it selective for only cancer cells. Besides, introducing essential genes that can be regulated by tumor specific promoters into the viral genomes can make them selective for tumors only.
The promoters include human telomerase reverse transcriptase (hTERT) 71 and survivin, 72 which are present in various cancers, and other tumor specific promoters such as tyrosinase for skin, 73 fetoprotein for liver 74 etc.
OBP-301, an attenuated adenovirus was engineered in which the E1 gene expression is controlled by hTERT. 75 Since only cancer cells possess this promoter for telomerase activity, the modified virus could only invade tumor cells.
Replication and Oncolysis
Natural viral infection begins when a host cell is invaded by viral particles; followed by its replication inside the cell. Viruses hijack the cell’s protein synthesis mechanism to fuel itself, while the cell slowly weakens. Eventually, the cell bursts, releasing new viral particles to spread and infect neighboring cells. In oncolytic virotherapy, natural technique for the destruction of tumors is manipulated. Here, OVs are intentionally entered into the host body for specific targeting and destruction of cancer cells. In healthy individuals, upon the onset of a viral infection, multiple signaling pathways interact with each other in order to detect and clear out the infection (Figure 1). IFN and TLRs activated upon recognizing PAMPs on viral particles (DNA, RNA, capsid, protein product etc.) stimulate these pathways. 5 However, in the majority of cancer cells, these signaling pathways are impaired. 76 Hence, most viruses replicate rapidly in tumor cells compared to normal cells. On top of that, some viruses have deletion in genes responsible for producing components that inhibit the transporter associated with antigen presentation, ensuring protection from the host immunity. For example, deletion of α47 gene in T-Vec and G47∆.77,78 Altogether, due to the lack of check from both cellular regulators and immune system, viruses can thrive easily in cancer cells.
In case of replication, oncolytic viruses multiply inside tumor cells much like any other viral infection. Only the selection process of target cells varies due to genetic alteration employed into these viruses. Adenovirus, being the most well studied and arguably most potent oncolytic virus, 79 will be discussed as an example. The receptors on adenovirus bind to the cell surface of the target tumor cell and induce an endosomal encapsulation. Following its entry, endosomal lysis takes place; the viral gene particles are released into the cytosol and transferred to the nucleus for replication. 80 Viral proteins including E1A, E1B and E4 accumulate in the cytoplasm. While E1A and E1B enhance virus directed autophagy, E4 suppresses it.81,82 Using the combination of these proteins, oncolytic adenovirus is capable of modulating autophagy in target cells.82,83 As a response to viral replication inside nuclei, Atg5 is upregulated and the Atg5-Atg12 complex is formed. 84 Following the formation of the complex, components such as p62, LC3-II etc. accumulate in the isolation membrane and cooperatively form autophagosomes. The autophagosome fuses with the lysosome to form autolysosome which has an acidic vesicular organelle (AVO) capable of digesting intracellular organelles including p62. 85 These changes in tumor cells infected with oncolytic virus can be evaluated through the observation of changes in autophagy related biomarkers such as the Atg5 upregulation and p62 downregulation. Additionally, Fas-associated via death domain (FADD)/caspase-8 signaling pathway activation, following autophagy induction has been shown to enhance oncolytic adenovirus-mediated autophagy 8883; which results in autophagic cell death via interaction with Atg5 and FADD. 86
Immune Stimulation and Anti-tumor Response
Oncolytic viruses (OVs) have been shown to have a promising anti-cancer mechanism. When these viruses infect cancer cells, they can selectively replicate within the tumor cells. This process not only kills the cancer cells directly but also stimulates the immune system by exposing the tumor-associated antigens (TAA) that are further taken up by the antigen-presenting cells (Figure 1), which then stimulates anti-tumor immune responses and generate a less immune-tolerant microenvironment by activating danger signals.87,88 Moreover, the danger-associated molecular patterns (DAMPs) like ATP, HMGB1 and pathogen-associated molecular patterns (PAMPs) including viral DNA, RNA and proteins released following the oncolysis of the tumor cells induce immunogenic cell death (ICD) including apoptosis, necrosis, autophagic cell death and pyroptosis. 89 PAMPs and DAMPs then stimulate the production of pro-inflammatory cytokines like type-1 IFNs and TNF-α which then further activates the innate immune responses by recruiting first line immune cells (eg, NK cells, macrophages and DCs) to infiltrate the damaged tumor sites in large numbers. Activated DC and NK cells then recognize PAMPs and act directly to kill the adjacent tumor cells or trigger the long-lasting adaptive immune response.90,91
Multiple studies have highlighted the importance of adaptive immune responses for complete tumor eradication and a better efficacy in oncolytic virotherapy. Mature DCs take up the TAA released from the lysis of the virus infected tumor cells and cross present the antigens in complex with the MHC receptors coupled with some costimulatory molecules on the surface. 92 This cross presentation by the antigen presenting cells subsequently activates the cytotoxic T cells resulting in cytolysis. Moreover, the chemokines and type-1 interferons released in response to viral antigens also facilitate the trafficking of CD8+ T and B cells to the TME.93–95 Additionally, these anti-tumor effects which help in the tumor regression and prevent metastasis, seem to have immune memory too. In brief, induction of the immunogenic cell death primes subsequent innate and adaptive immune responses, which lead to an inflammatory immune response and reverse the immunosuppression in tumor microenvironments.96,97 Also, these TAA, PAMPs and DAMPs can be an invaluable target for several immune-targeted regimens. Besides, OVs can be armed with immunostimulatory cytokines or engineered to insert necessary genes to stimulate a more robust anti-tumor immune response.
Resistance Mechanisms in Tumors and the Tumor Microenvironment (TME)
In spite of the strong antitumor effects observed with OVs, resistance to therapy is a major impediment. Resistance may be generally classified into tumor-intrinsic and microenvironmental. Tumor-intrinsic factors are the re-establishment of protective antiviral pathways that limit OV replication. The ability of viruses to grow despite an impaired interferon signaling in many cancer cells requires that these viruses in rare cancers be able to overcome antiviral defences that have been re-established by upregulation of interferon-stimulated genes (ISGs). 98 Molecules, eg, PKR and MxA, may be overexpressed and suppress the synthesis of viral proteins and prevent productive infection. Also, high autophagy-related proteins activity could stimulate viral particle destruction and slow oncolysis. Furthermore, OVs may selectively kill cancer cells, and the surviving cells can experience clonal evolution towards resistant subpopulations. 99 The TME also compromises the treatment of OV. Heavy stromal obstacles, usually via cancer-associated fibroblasts, make extracellular matrix proteins that limit the spread of viruses physically. Hypoxia, a hallmark of many solid tumors, alters viral replication kinetics and promotes resistance. Immunosuppressive cells within the TME—especially regulatory T cells (Tregs) and myeloid-derived suppressor cells (MDSCs)—release inhibitory cytokines such as IL-10 and TGF-β, which dampen antiviral immunity and reduce OV-driven cytotoxicity. 100 To conquer these impediments new approaches are being worked upon. Genetic engineering has the potential to make OVs that secrete checkpoint inhibitor antibodies (eg, anti-PD-1 scFv) to locally modulate immune response, avoiding systemic toxicity. 101 In other OVs, cytokines such as IL-12 have been encoded to reprogram the TME to an immune-activated state. Viruses can also spread through pharmacologic inhibition of stroma (eg, TGF-B blockers), which may adversely affect the extracellular scaffolding. Combination regimens of OVs with ICIs or stromal-modulating agents are under early-phase clinical investigation, which present potential ways to overcome intrinsic and microenvironment resistance. 102 This twin appreciation of resistance explains the relevance of combining virotherapy as part of a wider immuno-oncology and tumor-microenvironment-focused approach.
Administration Routes and Strategies to Overcome Host Defenses
It is necessary for the applied virus to be very specifically targeting tumor cells for better therapeutic efficacy. In order to achieve that, viruses may be modified genetically to recognize only cancer cells. Likewise, entry of oncolytic virus into the patient’s internal systems is a sensitive and delicate process, as suboptimal delivery can lead to treatment failure. The delivery routes can be intratumoral, intravenous or intraperitoneal. Intratumoral route is most commonly used, accounted for 58.6% global OV studies 103 and heavily preferred as it allows control of the virus concentration in the target site and eliminates the possibility of clearance by the host immune system.103,104 However, intratumoral administration cannot eliminate the distantly located tumors, which limits its application to advance metastasis. When it is not feasible to use intratumoral administration, intravenous routes are preferred due to their easy accessibility and widespread application. Other administration routes include intrathecal and subcutaneous. Peritoneal route, commonly used for ovarian cancer, is usually faster than the subcutaneous route, but the latter is easier to operate. 105 When it comes to Central nervous system (CNS) tumors, intrathecal delivery is ideal.106,107
However, the route of administration depends heavily on the treatment objectives. Circulating OVs are required to evade the pre-existing immunity to reduce the chances of neutralization by the immune system. To tackle this, cells with tumor-homing abilities that can be used as vehicles for the oncolytic virus are being developed. Mesenchymal and endothelial cells, renowned for their tumor-homing abilities, were infected by oncolytic virus and tested for their ability to transport the virus directly to the tumor cells. It was shown to have decreased the tumor load in test animals.108,109 Few early studies used transformed cells (Mesenchymal Stem Cells) as carriers to deliver the mutant herpes into the tumor site.109,110 Later, few studies on reovirus showed that immune cell (Dendritic cell, T cell) carriage of the oncolytic reovirus prevents them from immune neutralization and helps them to reach distant tumors. 111 Coating the virus particle with liposomes and cell-derived vesicles could also protect them from immune cells and soluble immune particles.112–114 This outer layer not only protects from neutralizing antibodies but also reduces the production of new antibodies.
Moreover, synthesizing these modified polymers allows the incorporation of specific ligand molecules to target tumor surface receptors. Use of non-human viral strains has been another effective technique to avoid the initial immune responses. For example, a caprine herpes strain showed successful replication cycles in several human cell lines, and triggered apoptosis. 115 Similarly, avian reovirus was able to infect HCC cells and displayed persistence against neutralizing antibodies. 116 Due to the lack of off-target pathogenicity of healthy tissues, these non-human strains hold a safe profile overall and can be an attractive option for future clinical trials. In addition, OVs can be further shielded from the immune system by modifying their surface receptors to halt the interaction with neutralizing antibodies. For instance, altering the structure of glycoprotein D (gD) in HSV has significantly reduced the binding affinity of neutralizing antibodies. 117
A consistent problem with oncolytic virotherapy is the clearance of the viral particles by the host immune system, and this challenge is more significant in patients that have preexisted immunity to the virus due to a previous exposure or a vaccination effect. The presence of neutralizing antibodies and complement protein may destroy the OVs in circulation before they get to the target sites, therefore, minimizing therapeutic efficacy. 6 Numerous strategies have been established on preclinical/early clinic investigational studies in order to circumvent this barrier. Another area of prospective interest is the application of cellular carriers, especially adipose or mesenchymal stem cells (MSCs) and dendritic cells, with their inherent tumor-homing characteristics. 118 The cells can be loaded with the OVs ex vivo and administered back to the patients in order to deliver the virus while protecting it against neutralization by antibodies. MSC carriers in dietary murine glioma or reovirus models have been demonstrated to increase viral persistence and decrease tumor size. In the same vein, OVs can be used as a Trojan horse to deliver themselves to the tumor by using T cells and DCs. 119
The other possible approach is the encapsulation of OVs in liposomes, extracellular vesicles, or nanoparticles, which gives the physical protection against immune recognition. Encapsulation also allows functionalization of surfaces with ligands specific to tumor cells that ensures that these ligands are capable of selectively targeting malignant cells. The preclinical uses that show the benefits of such biomimetic coating to increase circulation half-life and further tumor-uptake of OVs are growing. 120 . There also exists the possibility of exploring non-human strains of the virus. As a further example, caprine herpesvirus as well as avian reovirus effectively infect human cancer cells showing resistance to host-derived neutralizing antibodies in seropositive subjects. These data indicate that in preclinical models, these viruses cause apoptosis and prolonged oncolysis and do not cause toxicity in off-target tissues. Besides, genetic manipulation of viral surface glycoproteins, such as glycoprotein C (gC) in HSV, or gD in herpes simplex virus, has also been demonstrated to decrease antibody binding affinity thus providing enhanced evasion. 121
Advances in Genetic Modification of Oncolytic Viruses
A native virus gains its oncolytic characteristics when it has the ability to attack and kill tumor cells specifically, without the added risk of disease or harming the other body cells. In short, OVs must be engineered to be highly specific and non-virulent for the host in general. Genetic modification of viruses is the foundation of the study of oncolytic virotherapy. In nature, no virus with specific need for tumors for their survival is found. Hence, the beginning of oncolytic viruses is marked by the exploration of the depths of genetic alterations and adjustment of viruses per our need. The focus of genetic modification can vary from reducing viral virulence towards healthy cells and enhancing target specificity to introducing abilities to produce increased amounts of cytokines into viruses. Which specific modification would be instilled and to what extent depends heavily on the specific virus.
Virulence Reduction
Ensuring the injected virus does not cause a new disease in a host is a top priority when it comes to using viruses as a treatment option. In Oncolytic HSV, the most common alteration done to HSV-1 genome is the deletion of UL56 and γ34.5 in order to reduce its neuro-invasive capabilities.122,123 The first one associates with host protein KIF1A to facilitate motor invasion in hosts whereas the latter encodes ICP34.5 which assists viral replication in neurons and blocks antiviral responses.124,125
Specificity Enhancement
G207, a second-generation oncolytic virus of HSV-1, has deletions in γ34.5 and an inactivating insertion of LacZ in the ICP6 gene, which encodes the large subunit of the viral ribonucleotide reductase a key enzyme. The combination of both deletions permits its infection in tumor cells while preventing a replication in normal tissue.41,122,126 Due to their wide array tissue tropism, enhanced specificity is the first concern when it comes to using adenoviruses as OVs. The primary receptor for adenovirus found in nature (Coxsackievirus–adenovirus receptor) is generally absent in tumors. 127 In order to counter this, genetic modifications are made into the virus for broadened receptor recognition. An example of this altercation can be the addition of RGD (Arg-Gly-Asp) domain to a protein encoded by a shortened E1A gene (Ad5-Δ24) in recombinant adenovirus. This creates Ad5-Δ24RGD 11128; since RGD is a ligand for αβ integrin receptors abundant in tumor, altogether this addition helps improve specificity of the OV towards malignant cancer cells. 129
Modifications for Immuno-Modulatory Lysis
Along with direct selective lysis of tumor cells, oncolytic viruses are also known for initiating immune-mediated lysis on tumor cells. As they infect the cancer cells, certain genetic modifications allow them to express transgenes that assert immunostimulatory activities; thus, allowing cell destruction by the host immune system. 130
TNFα
Tumor Necrosis factor alpha or TNFα is a cytokine release by several immune cells that induce cell death.131,132 Theoretically, addition of TNFα coding sequences to the genome of oncolytic viruses can induce death by the immune system in tumors. This was done to an oncolytic adenovirus with an Ad5/3 chimeric capsid and a 24bp deletion in the constant region 2 of E1A, allowing it to both produce TNF as well as replicate selectively in cancer cells. The resulting Ad5/3-D24-hTNFa virus was observed to produce TNFα and reduce tumor growth and improve survival when tested on prostate cancer murine models 133 ; it also showed increased infiltration of tumor specific CD8 T cells in metastatic immunocompetent murine model of melanoma.
IL-2
IL-2 is a cytokine that promotes stimulation of effector immune cells which in turn helps in battling cancer. 134 Equipping oncolytic viruses with the ability to produce IL-2 has the goal of mounting a strong immune response from the host through activation of NK cells, T cells, B cells etc. An oncolytic adenovirus engineered to produce TNF had transgenes for IL-2 production added to the E3 region of its genome. 133 The new Ad5/3-E2F-d24-hTNFa-IRES-hIL2 virus was found to increase CD4 and CD8 T cells in cancer microenvironment in the hamster model.33,133
IL-12
Expression of this cytokine in the tumor helps Th1 cell differentiation, promotes T-cell mediated killing and inhibits angiogenesis.135–137 The risk of higher systemic toxicity from direct IL-12 therapies is also reduced when the cytokine is used with OVs. Among the OV-IL12s, Oncolytic HSVs modified for IL-12 production (OHSV-IL12) have come farthest clinically. 138 Murine IL-12 inserted virus G47Δ-mIL12 inhibiting intracranial growth and improving survival in Glioblastoma models, 139 T3855 140 and T3011 141 reducing tumor volume in melanoma are examples. In oncolytic vaccinia virus, subunits for TK (Thymidine Kinase) and RR (Ribonucleotide Reductase) were deleted to ensure tumor specific replication.142,143 Transgene for IL-12 production is inserted into J2R (viral TK) locus. It is controlled by pF17R to limit the production to the cells the virus is actively replicating inside in order to limit systemic toxicity. The resulting AZD4820 demonstrated broad oncolytic activity on cultured human cell lines in vitro, while surrogate virus expressing murine IL-12 anti-tumor activity in M38 and CT26 mouse synergic tumor models. 144
Trail
TNF-related apoptosis-inducing ligand (TRAIL) is a transmembrane protein capable of inducing apoptosis in tumor cells while leaving healthy cells unaffected.145,146 TRAIL transgene introduced into oncolytic adenovirus Ad. IR-E1A/TRAIL showed reduced tumor burden about 2-fold compared to the control group treated with Ad. IR-E1A/AP (without TRAIL), with no toxicity to the liver in mice models. 147 Genetic engineering in oncolytic viruses to exert immunomodulatory effects extends beyond just cytokines. The most successful oncolytic virus till date, T-VEC, is a variation of second generation HSV-1. This variation has deletion in γ34.5 gene as well as in US12, which encodes suppressor of antigen representation, ICP47 protein.122,123 Additionally, T-VEC has human granulocyte macrophage colony-stimulating factor (GM-CSF) cDNA incorporated into its genome for the added ability of recruiting antigen representing cells to tumors and facilitating destruction by immune cells.40,148
Clinical Trials and Efficacy Data
Clinical advance and efficacy of oncolytic viruses (OVs) have shown remarkable improvements in a highly clinical selection process of the viruses in a variety of cancer entities. A multitude of viruses, particularly vaccinia, herpes simplex virus (HSV), and adenoviruses, have been studied broadly, where each exhibit special modifications for increasing specificity, efficacy of treatment, and reducing toxicity.
Clinical Trial of Vaccinia Virus
Examples of Oncolytic Vaccinia Viruses (VACVs) in Clinical Trials
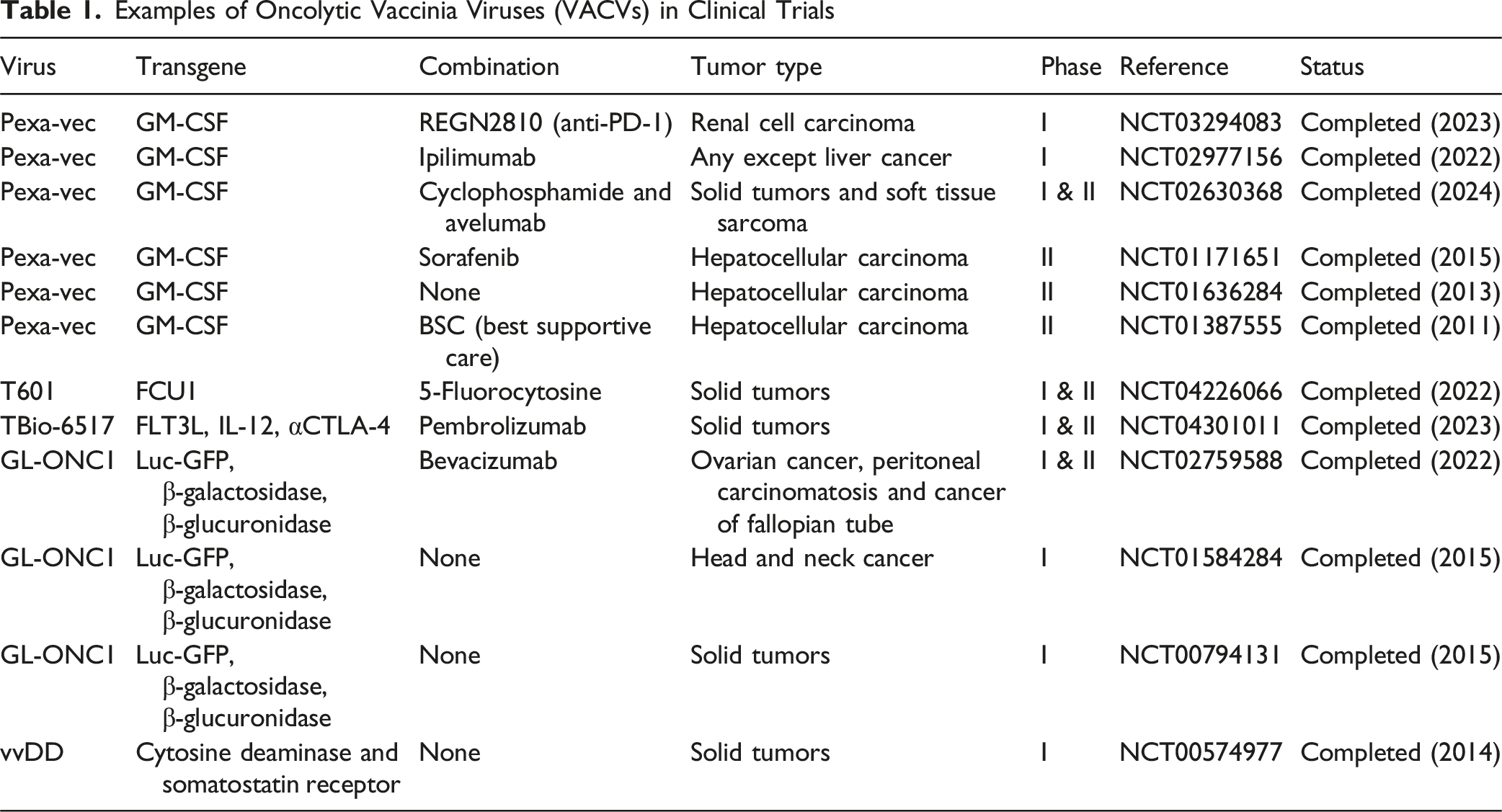
Clinical trials based on vaccinia-based OVs emphasis their multifaceted potential, as the modifications of the transgene like GM-CSF lead to enhanced specificity and better therapeutic results due to intensified anti-tumor immune reactions. In an attempt to extend the therapeutic efficacy of combination strategies, ongoing trials provide exciting routes towards combating complicated malignancies.
Clinical Trial of Herpes Simplex Viruses (HSVs) Virus
Examples of Oncolytic Herpes Simplex Viruses (HSVs) Currently Under Clinical Trials

The HSV-based oncolytic viruses, specifically T-VEC, has created a strong clinical construct with FDA approval for melanoma therapy. Trials repeatedly show their bifunctional potential for direct tumor-lysis as well as a systemic immunological stimulation, which is critical for conquering therapeutic resistance and enhancing clinical outcomes.
Clinical Trial of Adenoviruses
Examples of Clinical Trials With Adenoviruses

In general, these trials serve to emphasize the dramatic potential of OVs as treatments. The synergism between OVs and transgene, as well as combinational therapies, demonstrates increasing successful clinical outcomes of cancer treatment not only because of their direct tumor-lysing capacity but also for the pro-active induction of potent and durable anti-tumor immunity.
Combination of Oncolytic Virus Therapy with Other Approaches
Combining Oncolytic Viruses with Immune Therapies
Targeted immune therapies are designed in a way that they intervene in the progression of the molecules responsible for development of the tumor cells, and this precise targeting ability of this therapy has made it a compelling choice for combining it with virotherapy. While OVs are known for their selective antitumor activities and inducing robust immune responses, host’s pre-existing immunity and anti-viral resistance is limiting the efficacy of the oncolytic viruses as a monotherapy. Also, the expression of the transgenes or genetic modification of the viral vector may reduce the overall fitness and potency of the virus. Considering this, experts are leaning more towards combinational therapies with OVs. Besides, the safety profiles of the OVs and the room for repeated administration make them an attractive option to combine with chemotherapy, targeted therapy, and immunotherapies.
Immune checkpoint inhibitors (ICIs) are one of the most valuable next generation cancer therapies. A major way cancer proceeds spreading throughout the body is by evading surveillance by host immunity using immune checkpoints.149–151 Immune checkpoints on T-cells are homeostatic regulators of immune tolerance. These checkpoints shut the reactivity of immune cells, preventing immune reactions that can be too strong for the host. Cancer cells take advantage of this by bonding with the checkpoint and switching off the T-cell activity.152–154 Immune checkpoint inhibitors are drugs that form bonds with the checkpoints before cancer cells can access it, and thus avoid its deactivation. In 2011 anti-cytotoxic T lymphocyte-associated protein 4 (anti-CTLA-4) for advanced-stage melanoma was approved by FDA. 155 Until 2019, other FDA approved ICIs include, Nivolumab (2014), Pembrolizumab (2014), Atezolizumab (2016), Durvalumab (2017), Avelumab (2017) and Cemiplimab (2019).156,157 Theoretically, Oncolytic Viruses or OVs as foreign agents should induce a strong immune response in the host, while ICIs can ensure the activated cells be effective without disruption.
Summary of Combination of Oncolytic Virus and Immune Checkpoint Inhibitor (ICI) Clinical Trials

Combination of Oncolytic Viruses with Radiotherapy or Chemotherapy
In spite of the fact that radiotherapy is the routine care for local tumors and early-stage disease, there are several drawbacks of this approach, such as non-target effects on normal cells and also emergence of the radio-resistance in the tumor cells, which impair treatment successes.158–160 Moreover, radiotherapy is almost ineffective against metastatic growth.161–163 Likewise, chemotherapeutic agents are usually not selective; they damage normal tissues and cause systemic toxicity and side effects that may have a substantial effect on the quality of patients’ life. 164 Furthermore, tumor cells often mutate thus creating drug-resistant clones that result in diminishing or nullifying the therapeutic effects of chemotherapeutic drugs.
On the contrary, oncolytic viruses specifically infect and kill tumor cells. Besides, although radiotherapy and chemotherapy are usually localized, the use of oncolytic viruses can target metastatic tumors because oncolytic viruses have an ability to infect tumor cells. Therefore, the integration of oncolytic viruses with chemotherapy or radiotherapy can offer local and systemic disease control. There are many preclinical trials using the combination of oncolytic virus with radiotherapy and chemotherapy. It has been reported that when using the combination of temozolomide or vincristine with oncolytic viruses, one can significantly kill mouse tumors.165,166 Simultaneously, oncolytic viruses were seen to increase the efficacy of mitomycin and hydroxycamptothecin. 167
Clinical Trials of Oncolytic Virus Combined With Radiotherapy and Chemotherapy

Manufacturing and Scale-Up of Oncolytic Viruses
The transition of OVs into the clinic cannot be successful without effectiveness and reproducible manufacturing process. This means that production has to be based on high yield, viral stability and must GMP, according to the highest standards. On a laboratory scale, a permissive cell substrate that facilitates efficient replication of the virus is selected. 168 Vero, HeLa, or HEK293 cells have all been successfully used. A specific multiplicity of infection of the virus is added and growth is ensured by repeated replication. Typical histological harvesting is carried out at peak cytopathic effects with further purification steps involving either a gradient centrifugation, or tangential-flow filtration, or chromatography may be used to remove the cellular debris and contaminants that remain in the sample.
To scale-up to the pilot or industrial scale, bioreactors, which allow regulated culture conditions to propagate large viral volumes, must be used (Figure 2). To optimize on productivity, it is common to use stirred-tank or fixed-bed bioreactors.
169
Nonetheless, scale-up is associated with issues like genetic stability of viral stocks, consistent potency between batches and effective downstream purification. Regulatory agencies such as the FDA and EMA allow only a high level of quality control test of each stage ie, sterility, adventitious agent testing, genome sequencing, and potency assays is allowed.
170
A further degree of difficulty is the viability and long-term storage of OVs because several are temperature sensitive and must be cryopreserved or lyophilized during transport. Cryoprotectants and stabilizing excipients are being enhanced to have a longer shelf-life and better accessibility. Manufacturing Steps of OVs. A Schematic Flow Diagram Illustrating: Cell Substrate Selection → Viral Infection & Amplification → Harvest → Purification (Chromatography/Filtration) → Quality Control (Sterility, Potency, Stability) → Scale-Up in Bioreactors → GMP Distribution
Future Directions
The effectiveness, safety, and clinical applicability of oncolytic virus require further research in an attempt to maximize their therapeutic efficacy. Future directions should focus on optimizing the genetic modifications for a greater specificity of the viruses and reduction in the occurrence of off-target effects. Virus-host interaction strategies of genetic engineering can be further capitalized to enhance selective targeting of tumor microenvironments and systemic toxicity. In addition, the exploration of new range and complexity of viral transgenes, including immune-stimulatory cytokines, checkpoint inhibitors, or apoptosis-inducing molecules, seems promising in increasing anti-tumoral effectiveness. Detailed research works on mechanisms of viral replication in the different types and stages of tumor are critical in determining the best conditions for OV treatment.
Nevertheless, combination therapies, particularly with the use of immune checkpoint inhibitor (ICIs), chemotherapy and radiotherapy, will remain a crucial field of study. With regard to difficult tumor immune evasion mechanisms, the future clinical studies must be heavily focused on developing combinational regimens, especially integrated with simultaneous multiple ICIs or targeted immunotherapies with OV-based strategies. In addition, personalized medicine structures using cutting-edge biomarker analysis and genomic profiling can be useful in determining OV patient populations with the highest likelihood of benefitting from the OV therapeutics, enhancing clinical outcomes.
Evasiveness of host immune responses is still a big hindrance, leading to the need for more innovation in the modes of delivery. Prospective studies should focus on enhanced systemic delivery systems such as engineered cellular carriers, nanoparticle coatings, and altered viral envelopes for the protection of OVs from their host immune neutralization. Studies on the use of advanced biomaterials and bioengineering principles in controlling virtual distribution and existence in tumor tissues in an accurate manner can significantly increase the therapeutic success. In addition, comprehensive studies on tumor resistance to OV-based therapy should be carried out. Knowledge of the fundamental genetic, as well as immunological molecular mechanisms that produce a state of treatment resistance can be used to develop more robust viral constructs. There is also potential for expansion of CRISPR/Cas9-based gene-editing technologies, in the context of improved OV safety profiles, viral tropism modification and site-specific gene knock in tumor cells.
Conclusion
Oncolytic viruses (OVs) are a revolutionary leap in cancer therapies exerting substantial clinical potential because of their unique properties to preferentially infect and destroy cancer cells and at the same time drive potent anti-tumor immunity. Phase I-III clinical trials with various viruses, especially adenovirus, herpes simplex virus, and vaccinia virus, have provided compelling arguments in favor of their efficacy and their capacity for effectiveness, especially in combination with immune checkpoint inhibitors, chemotherapy, or radiotherapy. Additional genetic alterations including the immunostimulatory transgenes such as GM-CSF, IL-12, and TNFα have improved OV therapies more in terms of specificity, efficacy, and profile safety. In spite of significant progress, there are still the challenges of host immune neutralization, delivery efficacy and tumor resistance mechanisms that call for the need for more innovation in design of OVs and combination strategies. A focus on optimization of systemic delivery modalities and fine-tuning the use of personalized medicine strategies as well as patients’ biomarker-driven stratification will be among future directions in OV therapy, along with the use of advanced technologies for gene-editing. Finally, long-term interdisciplinary cooperation and extensive clinical validation are key to the transformation of compelling preclinical results into standardized, widely usable treatments.
Footnotes
Acknowledgements
We extend our heartfelt gratitude to the corresponding author, Dr S. M. Bakhtiar UL Islam, for his invaluable supervision and guidance throughout the development of this manuscript. His deep engagement, insightful feedback, and consistent support were instrumental in shaping the core ideas and refining the research. His active role in overseeing the manuscript’s progress significantly contributed to its overall depth, clarity, and scholarly quality.
Author Contributions
SMBUI and TA conceived the idea and prepared the outline of the review. SMSA, MT and TA performed the literature search and data extraction, analysis of extracted data and manuscript preparation. SMBUI and TA supervised the manuscript preparation and prepared the final draft. The authors read and accepted the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
