Abstract
Introduction
Oropharyngeal Squamous Cell Carcinoma (OPSCC) etiology involves environmental and genetic factors, with Human Papillomavirus (HPV) being a key ecological driver. This study evaluated the relative importance of environmental factors, particularly HPV status, and the genetic polymorphism NKG2D rs1049174 in OPSCC among a Vietnamese population.
Methods
A retrospective case-control study was conducted with 279 OPSCC patients and 250 healthy controls. HPV DNA was screened and typed using PCR and Sanger sequencing. Genotyping of NKG2D rs1049174 was performed using TaqMan assays. Logistic regression, Kaplan-Meier survival analysis, Cox proportional hazards regression, and Random Forest analysis were employed to assess associations between risk factors, genotype, clinical stage, survival, and HPV status.
Results
HPV status strongly predicted overall survival (OS), with HPV-positive patients exhibiting significantly longer survival (adjusted Hazard Ratio (aHR) = 0.32, P < 0.001). Univariate Kaplan-Meier analysis further suggested differential survival among specific HPV types, with HPV16 infection trending toward prolonging OS. In predicting advanced-stage OPSCC, Random Forest analysis identified HPV status as the most critical predictor (Mean Decrease Accuracy = 5.47). Males constituted the vast majority of OPSCC patients in both HPV-negative (97.4%) and HPV-positive (84.6%) subgroups. A statistically significant difference in alcohol consumption patterns was also observed between HPV-positive and HPV-negative patients. Furthermore, the NKG2D rs1049174 polymorphism was significantly associated with OPSCC risk, though not with advanced-stage disease or OS.
Conclusion
HPV infection plays a critical role in OPSCC in this Vietnamese cohort, influencing patient characteristics, clinical presentation, and survival outcomes. The NKG2D rs1049174 polymorphism was identified as a significant factor in OPSCC risk; however, it did not appear to be a significant factor in disease progression or survival in this population. Further research is needed to explore the complex interplay of environmental and genetic factors in OPSCC etiology in Vietnam.
Keywords
Introduction
Oropharyngeal Squamous Cell Carcinoma (OPSCC), a significant subset of head and neck cancers, arises from the squamous epithelium lining the oropharynx, including the tonsils, base of tongue, soft palate, and pharyngeal walls. 1 Globally, head and neck cancers represent a significant health burden, with hundreds of thousands of new cases and deaths annually. 2 The etiology of OPSCC is multifactorial, and recent studies highlight the importance of environmental and genetic factors in its development. High-risk Human Papillomavirus (HPV) infection is increasingly recognized as a strong driver in a growing proportion of Oropharyngeal Squamous Cell Carcinoma (OPSCC) cases, particularly in certain regions.3,4 However, a significant subset of OPSCC remains linked to traditional carcinogens, such as tobacco smoking and alcohol consumption,5,6 with the etiology of OPSCC increasingly understood through distinct pathogenic pathways.
The host immune system, particularly innate immunity, plays a critical role in cancer immunosurveillance. Natural Killer (NK) cells are key effector lymphocytes that can recognize and eliminate transformed cells without prior sensitization. 7 Their function is regulated by a balance of signals from activating and inhibitory receptors on their surface. The NK Group 2, member D (NKG2D) receptor is a potent activating receptor expressed on NK cells, CD8 + T cells, and γδ T cells. By identifying stress-induced ligands (such MICA/B and ULBPs) that are commonly increased on virus-infected or tumor cells, NKG2D triggers cytotoxic reactions and the generation of cytokines, which greatly enhances anti-tumor immunity. 8
Genetic variations within immune-related genes can influence individual susceptibility to cancer. The NKG2D gene (also known as KLRK1) harbors several polymorphisms, including the single-nucleotide polymorphism (SNP) rs1049174 (G/C), located in the 3′-untranslated region (3′-UTR). 9 This SNP defines two functional haplotypes: LNK (associated with the C allele and linked to lower NK cell cytotoxicity) and HNK (associated with the G allele and linked to higher NK cell cytotoxicity). 10 Mechanistically, the G allele has been suggested to impair binding of microRNA-1245 to the NKG2D mRNA, potentially leading to increased NKG2D protein expression and enhanced NK cell function in vitro. 11 Consequently, rs1049174 polymorphism is hypothesized to modulate the efficacy of immune surveillance against developing tumors. Given the significant role of HPV in driving a subset of OPSCC, understanding how this genetic variation in NKG2D might influence the immune response to HPV-infected and transformed cells in the oropharynx is of particular interest.
While the association between rs1049174 and risk has been investigated for various malignancies, including nasopharyngeal carcinoma in Vietnam, 12 its specific role in OPSCC within the Vietnamese population remains largely unexplored. However, the established crucial role of environmental factors, particularly high-risk HPV infection and tobacco use, in OPSCC development in this region suggests a potentially more dominant influence compared to specific genetic polymorphisms. Given the distinct etiologies of OPSCC, where HPV-driven and non-HPV-driven pathways are increasingly recognized, and the potential interplay between genetic susceptibility and these strong environmental exposures, understanding the predominant role of HPV and other environmental factors is essential in elucidating the mechanisms of OPSCC pathogenesis in Vietnam, especially in comparison to the contribution of specific genetic markers like rs1049174.
This study aimed to analyze the roles of HPV infection and NKG2D rs1049174 polymorphism in Vietnamese patients with OPSCC. We hypothesized that while both environmental factors (HPV status, smoking, and alcohol consumption) and the NKG2D rs1049174 polymorphism contribute to OPSCC development, environmental factors would demonstrate a more pronounced or dominant influence on disease risk and progression in this population, providing insights into the main risk factors for OPSCC and contributing to understanding its etiology in Vietnam.
Materials and Methods
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 13
Study Subjects and Sample Collection
This retrospective case-control study included 279 patients diagnosed with OPSCC recruited from Hanoi Medical University Hospital, the National Hospital of Otorhinolaryngology, and the National Cancer Hospital in Hanoi, Vietnam, between January 2018 and December 2022. During the same period, a total of 260 healthy control subjects were initially considered, of whom 10 declined to participate, resulting in 250 healthy control subjects retrospectively recruited from Hanoi Medical University Hospital. All participants were confirmed to be genetically unrelated and resided in Hanoi or the surrounding regions in northern Vietnam. All patient details were de-identified to ensure the anonymity and privacy of participants. The study size for OPSCC cases was determined by including all available pathologically confirmed cases within the specified recruitment period. While also comprehensively assessing the role of HPV infection and its specific types in the OPSCC patient cohort, the study further investigated the rs1049174 SNP of the NKG2D gene in both patient and control groups to explore its potential contribution to disease susceptibility. The overall study design and participant flow are depicted in Figure 1. The study was approved by the Institutional Review Board (IRB) of Hanoi Medical University, Hanoi, Vietnam (Approval No. 941/GCN-HĐĐĐNCYSH-ĐHYHN). Flow Diagram of Study Participant Recruitment and Analysis
Inclusion Criteria for OPSCC Cases
Patients were included in the OPSCC case collection if they had a pathologically confirmed OPSCC diagnosis and no prior history of malignancies originating from other organs. Furthermore, the availability of formalin-fixed paraffin-embedded (FFPE) tumor samples was required for tissue analysis and subsequent DNA extraction. Cases were consecutively ascertained from the pathology registries and medical records of the participating hospitals. The diagnosis of OPSCC was established based on standard clinical, radiological, and histological evaluations, with histological typing independently confirmed by two pathologists at Hanoi Medical University following the WHO classification system for head and neck tumors. These criteria ensured the inclusion of well-defined primary OPSCC cases representative of the target population for studying the genetic and environmental risk factors.
Control Group
The control group consisted of 250 healthy individuals with no history of cancer, who were frequency-matched to the OPSCC cases based on age and sex distributions to minimize potential confounding factors. Controls were recruited from individuals attending routine health check-ups and non-cancer outpatient clinics at Hanoi Medical University Hospital during the same period as the case recruitment. This control group served as the reference population for analyzing the distribution of the NKG2D rs1049174 polymorphism.
Demographic and Clinical Data
Demographic characteristics and clinical information were extracted from patient medical records. For analysis, age was categorized into three groups: Under 40 years, 40-60 years, and Over 60 years consistent with age stratifications used in previous studies on OPSCC prognosis. 14 Gender was categorized as male or female, and clinical stage was determined according to the American Joint Committee on Cancer (AJCC) TNM staging system for head and neck cancers.
Lifestyle Factors
Information on lifestyle factors, including smoking status and alcohol consumption, was collected from patient records via self-reported questionnaires or direct patient interviews at the time of diagnosis. Participants’ smoking habits were categorized as follows: Heavy Smokers consumed more than 1 pack of cigarettes per day, while Moderate Smokers consumed 1 pack or less per day. Non-smokers were individuals who had never smoked or smoked fewer than 100 cigarettes in their lifetime. Additionally, the categories included Passive Smokers (individuals regularly exposed to secondhand smoke), Traditional Smokers (individuals who smoke “Thuốc lào”, a type of traditional leaf tobacco), and an Unknown category for cases where smoking status could not be determined from records. Regarding alcohol consumption, participants were categorized as Heavy Drinkers (consuming more than one 1-liter bottle of spirits per day), Moderate Drinkers (consuming one 1-liter bottle of spirits or less per day), None-drinkers (individuals who reported no alcohol consumption), or Unknown (alcohol consumption status could not be determined from records).
Molecular Variables
HPV status was determined by PCR and Sanger sequencing as described below and categorized as positive or negative, with high-risk HPV types further identified for specific analyses. The NKG2D rs1049174 polymorphism was genotyped as GG, GC, or CC, and for functional interpretation and specific analyses, these genotypes were categorized into low NK activity (LNK/LNK = GG genotype) and high NK activity (LNK/HNK = GC genotype; HNK/HNK = CC genotype).
Outcome Variables
Regarding outcome variables, OPSCC diagnosis was based on pathologically confirmed OPSCC, as described in the Inclusion Criteria for OPSCC Cases section. Overall survival (OS) was defined as the time from the date of OPSCC diagnosis to the date of death from any cause or the last follow-up.
Sample Preparation and DNA Extraction
Genomic DNA was extracted from FFPE tumor tissue blocks. For control samples, 2 mL of peripheral blood was collected in EDTA anticoagulant tube. The QIAamp DNA FFPE Tissue Kit (Qiagen, Hilden, Germany; Catalog No. 56404) was used for DNA extraction from FFPE tissue, and QIAamp DNA Blood Mini Kit (250) (Qiagen, Hilden, Germany; Cat. No 51106) was applied for blood samples, strictly following the manufacturer’s protocol.
To minimize the risk of cross-contamination, new disposable microtome blades were used for sectioning each tissue block, and laboratory personnel wore fresh gloves throughout the process. Five 5 µm-thick sections were cut and collected in sterile 1.5 mL Eppendorf tubes before DNA isolation for each case. Extracted genomic DNA samples were stored at −20°C until further molecular analyses.
HPV Screening and Genotyping
The presence of Human Papillomavirus (HPV) DNA in the OPSCC tumor samples was initially screened using a semi-nested Polymerase Chain Reaction (PCR) targeting the L1 gene. A modified set of GP5+/GP6 + primers was employed for this initial screening, which was described in a previous report. 15 First PCR was prepared to reach a total volume of 20 µL containing: 2.5 µL of template DNA, 1X GoTaq® G2 Hot Start Green Master Mix (Promega, Madison, WI, USA; Catalog No. M7428), and 1 µL of each specific primer at a concentration of 5 µM. The thermal cycling conditions for first step PCR were as follows: an initial denaturation step at 95°C for 10 minutes, followed by 40 cycles of denaturation at 95°C for 1 minute, annealing at 45°C for 90 seconds, and extension at 72°C for 1 minute, and next step 72°C for 10 minutes. After that, second PCR was according to reach a total volume of 10 µL contains: 2.5 µL PCR product in first PCR, 1X GoTaq® G2 Hot Start Green Master Mix (Promega, Madison, WI, USA; Catalog No. M7428), and 0.5 µL of each specific primer at a concentration of 5 µM. The thermal cycling conditions for second step PCR were as follows: an initial denaturation step at 95°C for 10 minutes, followed by 40 cycles of denaturation at 95°C for 45 seconds, annealing at 58°C for 45 seconds, and extension at 72°C for 1 minute, and next step 72°C for 10 minutes. PCR products were electrophoresed in a 2% Agarose gel.
Amplification yielding a PCR product of the expected size (approximately 140-150 bp), corresponding to the target L1 gene fragment, was considered HPV DNA positive upon visualization on an ethidium bromide-stained agarose gel. Samples that tested negative with the modified primer set were subsequently re-analyzed using the original GP5+/GP6 + primer set as described in detail in the published report. 15 The PCR product from HPV-positive samples was then purified using a MEGAquick-spinTM plus Fragment DNA Purification Kit (Intron Biotechnology, Korea, Cat No 17290) to remove excess primers and nucleotides. After purification, these samples were sequenced using the Sanger method ABI 3500 Genetic Analyzer (Applied Biosystems, Hitachi). Consensus sequences were extracted from the raw reads using MEGA software version 11.0 16 HPV types were identified using the BLASTN tool on the NCBI website.
NKG2D Genotyping
Genotyping of the NKG2D gene polymorphism rs1049174 was carried out using TaqMan-Allelic Discrimination with a QuantStudio 3 Real-Time PCR system (Applied Biosystems, Foster City, CA, USA) according to previously published methods. 17 To reflect the functional consequences of these genotypes on NK cell activity, the GG genotype was referred to as LNK/LNK (low NK activity), and the GC and CC genotypes as LNK/HNK and HNK/HNK (high NK activity), respectively.
Statistical Analysis
Statistical analyses were performed using R software version 4.4.3. One-way Analysis of Variance (ANOVA) was used to compare continuous variables, such as average age, across multiple groups. Differences in genotype and allele frequencies between OPSCC cases and healthy controls were evaluated using chi-square or Fisher’s exact tests, where appropriate. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated to estimate the association between the NKG2D rs1049174 polymorphism and OPSCC risk under different genetic models, including dominant, recessive, and additive. Logistic regression analysis was employed to adjust for potential confounding factors such as age, sex, smoking status, and alcohol consumption. Survival analysis was initially performed using the Kaplan-Meier method to visualize survival probabilities for different groups, including HPV status (HPV type vs negative). Of the 279 initially enrolled OPSCC patients, 223 had complete data for overall survival (OS) and relevant covariates and were included in the multivariate analysis. The remaining 56 patients were excluded due to missing data using listwise deletion. Differences in survival curves were assessed using the log-rank test. To assess the independent prognostic value of HPV status and adjust for potential confounding factors such as age, sex, smoking status, and alcohol consumption, multivariate Cox proportional hazards regression analysis was conducted, with HPV status considered as a binary variable (positive vs negative). A P-value of less than 0.05 was considered statistically significant.
Results
Oropharyngeal Squamous Cell Carcinoma Patient Characteristics by HPV Status
Patient Characteristics by HPV Status and Statistical Results

Notes: - Not applicable or not calculated
Association between NKG2D rs1049174 Polymorphism and OPSCC Risk
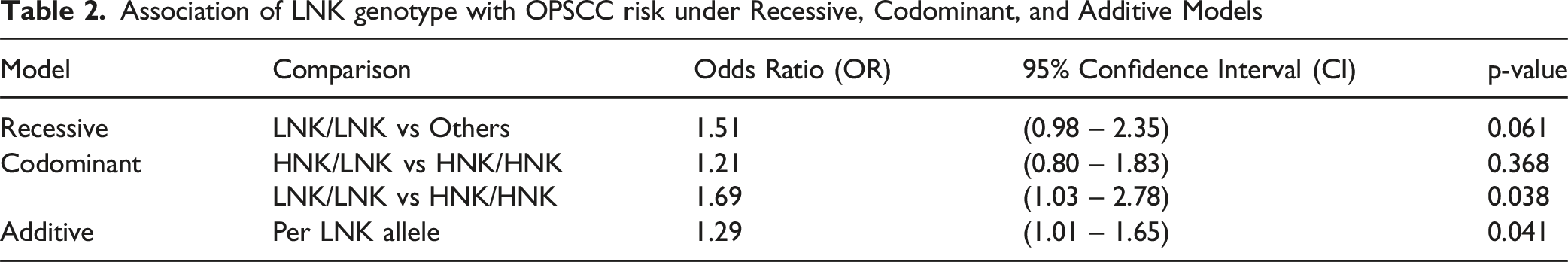
Association of LNK genotype with OPSCC risk under Recessive, Codominant, and Additive Models
Factors Associated with Overall Survival in OPSCC Patients
Univariate Kaplan-Meier Survival Analysis of Factors Associated with Overall Survival in OPSCC Patients

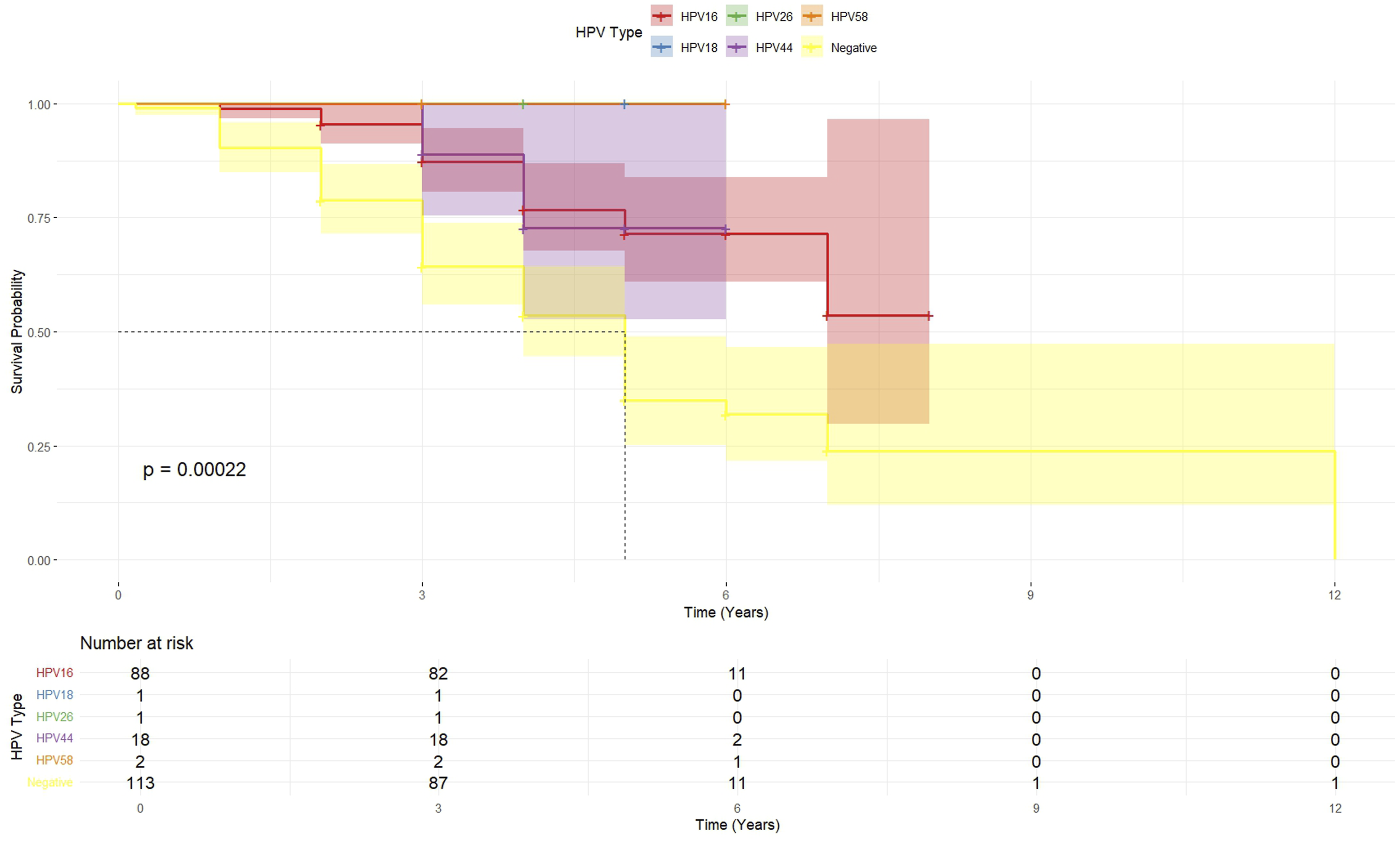
HPV Types Associated With Overall Survival in OPSCC Patients in Vietnam
To evaluate independent predictive markers for OS, a multivariate Cox proportional hazards regression analysis was conducted, concurrently controlling for several covariates (Figure 3). After adjustment, HPV status emerged as the only statistically significant predictor of OS. Patients with HPV-positive tumors exhibited a significantly decreased hazard of death compared to those with HPV-negative tumors (adjusted hazard ratio (aHR) = 0.32, 95% CI = 0.19 - 0.52, P < 0.05). In contrast, the multivariate analysis did not reveal statistically significant associations between OS and other factors such as age (aHR = 1.00 per year increase, 95% CI = 0.98 - 1.03), clinical stage (Stage II vs I: aHR = 0.63, 95% CI = 0.17 - 2.27; Stage III vs I: aHR = 1.18, 95% CI = 0.34 - 4.10; Stage IV vs I: aHR = 1.13, 95% CI = 0.34 - 3.72), gender (Male vs Female: aHR = 1.99, 95% CI = 0.65 - 6.12), family history (Yes vs No: aHR = 1.35, 95% CI = 0.62 - 2.94), smoking status (Moderate vs Non-Smoker: aHR = 1.19, 95% CI = 0.63 - 2.27; Heavy vs Non-Smoker: aHR = 1.59, 95% CI = 0.49 - 5.22; Traditional vs Non-Smoker: aHR = 2.02, 95% CI = 0.69 - 5.96; Unknown vs Non-Smoker: aHR = 2.43, 95% CI = 0.51 - 11.58), and alcohol consumption (Moderate vs Non-Drinker: aHR = 0.66, 95% CI = 0.33 - 3.30; Heavy vs Non-Drinker: aHR = 0.58, 95% CI = 0.06 - 6.13; Unknown vs Non-Drinker: aHR = 0.94, 95% CI = 0.13 - 6.50), as their respective 95% confidence intervals included 1 or the P-values were not statistically significant. The univariate Kaplan-Meier analysis did not show significant differences in OS between the groups for age, alcohol consumption, smoking status, family history, histology, and clinical stage (P > 0.05 for all). Multivariate Cox Proportional Hazards Model for Overall Survival in OPSCC Patients *P < 0.0001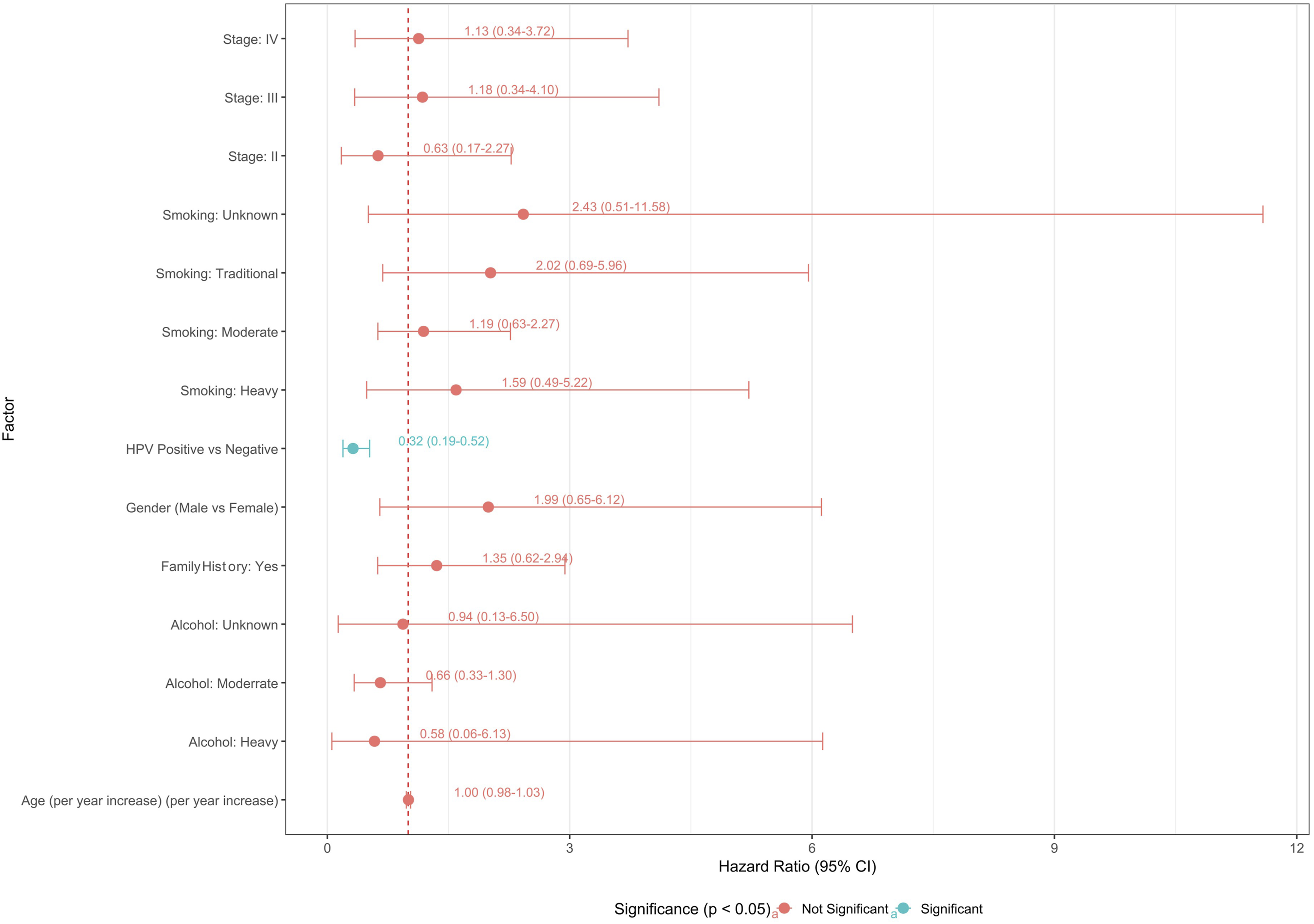
Predicting Advanced-Stage OPSCC
A Random Forest analysis was conducted to predict advanced-stage OPSCC (Stage III/IV vs Stage I/II) to explore further factors associated with tumor presentation. The model utilized HPV Status, Smoking category, patient Age, Alcohol consumption category, presence of the NKG2D LNK allele (LNK allele Presence), and Gender as predictor variables. Variable importance, assessed using the Mean Decrease Accuracy (MDA) metric (which quantifies the reduction in model accuracy when a variable’s values are randomly permuted), is presented in Figure 4. The analysis identified HPV Status as the most critical predictor of advanced stage, yielding the highest MDA value of 5.47. Smoking Category was the second most crucial factor, with an MDA of 4.81. Patient Age also contributed positively to prediction accuracy, albeit to a lesser extent (MDA = 1.08), followed by Alcohol Consumption with minimal positive importance (MDA = 0.22). Notably, both LNK Allele Presence and Gender exhibited negative MDA values (−1.17 and −1.24, respectively), indicating they did not contribute positively to the model’s accuracy in predicting advanced stage within this dataset. Thus, the Random Forest model highlighted HPV Status and smoking habits as the predominant factors distinguishing advanced vs early-stage OPSCC presentation. At the same time, the presence of NKG2D LNK alleles and gender were not found to be predictive. Variable Importance in Random Forest Model Predicting Advanced-Stage OPSCC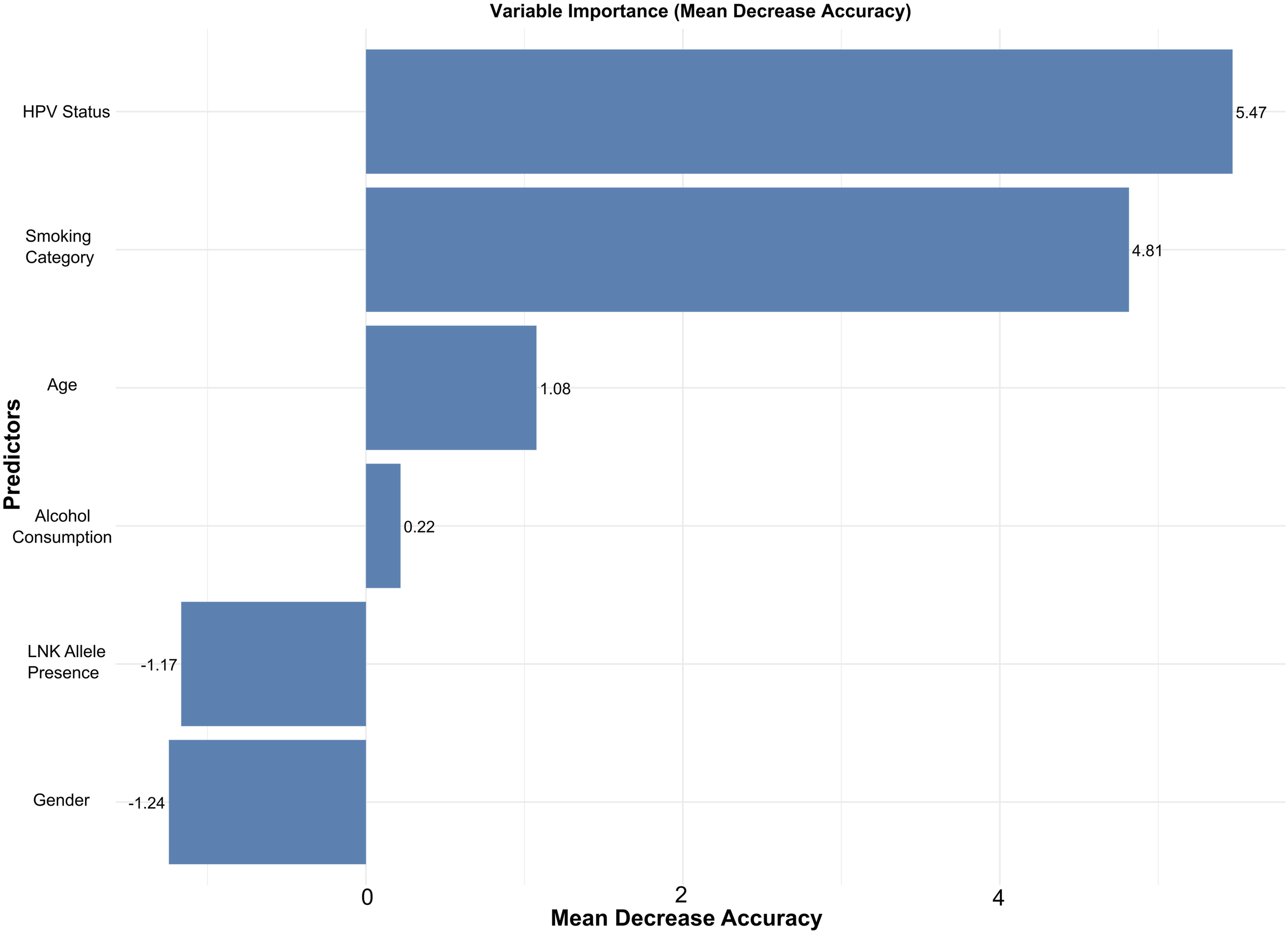
Association Between NKG2D rs1049174 and OPSCC
The association between the NKG2D rs1049174 polymorphism and the likelihood of presenting with advanced-stage (Stage III/IV) vs early-stage (Stage I/II) Oropharyngeal Squamous Carcinoma (OPSCC) was evaluated using multivariate logistic regression under additive, codominant, dominant (user-defined: LNK/LNK vs HNK carriers), and recessive (user-defined: LNK/LNK vs others) genetic models. All models were adjusted for age, HPV status, smoking category, alcohol consumption category, and gender. Figure 5 shows no statistically significant association between the rs1049174 polymorphism and advanced OPSCC stage under any of the tested genetic models after adjustment. Specifically, in the additive model, each additional LNK allele was not associated with the odds of advanced stage (Adjusted Odds Ratio [aOR] = 0.88, 95% CI: 0.60-1.28). Similarly, compared to the HNK/HNK genotype in the codominant model, neither the HNK/LNK (aOR = 0.71, 95% CI: 0.36-1.39) nor the LNK/LNK (aOR = 0.77, 95% CI: 0.35-1.69) genotypes were significantly associated with advanced stage. The dominant (aOR = 0.95, 95% CI: 0.51-1.85) and recessive (aOR = 0.95, 95% CI: 0.51-1.85) models also yielded non-significant results. These findings indicate that the NKG2D rs1049174 genotype is not significantly associated with the clinical stage at presentation in this OPSCC cohort. Association of NKG2D Genotype With Advanced Stage Under Different Genetics Models in OPSCC Patients With Adjusted for Age, HPV Status, Smoking Status, Alcohol Consumption, and Gender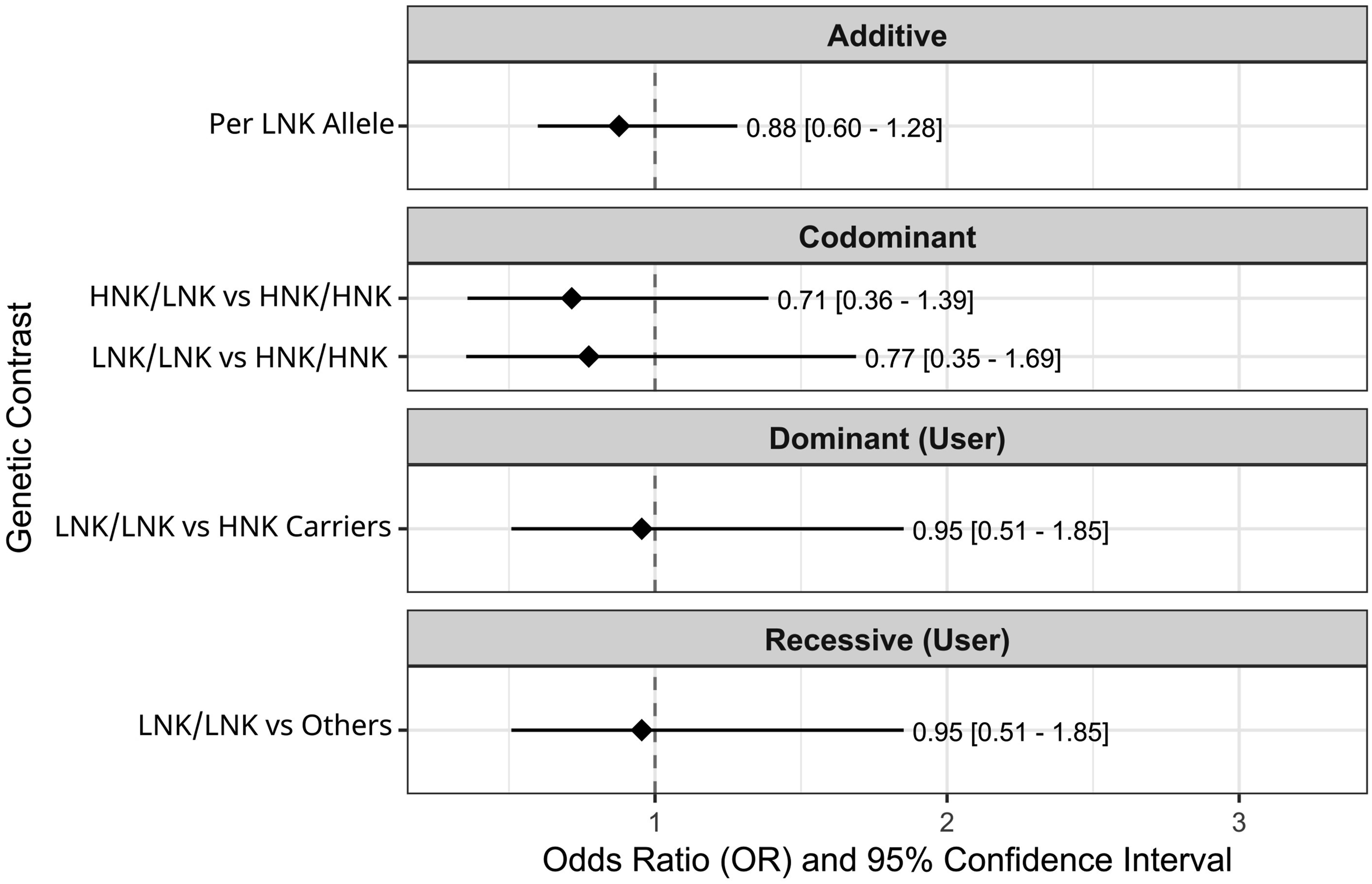
Discussion
This study investigated the interplay between environmental factors and genetic susceptibility in OPSCC among a Vietnamese population. Our findings highlight the significant role of HPV status in OPSCC, regarding patient characteristics and overall survival (OS), and identify the NKG2D rs1049174 polymorphism as a significant factor in OPSCC risk, while not identifying the NKG2D rs1049174 polymorphism as a significant factor in disease presentation or survival in this cohort. The baseline characteristics of our OPSCC patients, analyzed in the whole cohort, revealed a statistically significant difference in gender distribution between HPV-positive and HPV-negative subgroups (P = 0.0004), with a higher proportion of males in both subgroups (97.4% in HPV-negative and 84.6% in HPV-positive patients). This observation aligns with global trends suggesting a stronger association of HPV-positive OPSCC with males. 18 Notably, we also found a significant difference in alcohol consumption patterns (P = 0.0002), with the HPV-positive group exhibiting a higher proportion of non-drinking patients compared to the HPV-negative group. This inverse association between HPV positivity and alcohol consumption is consistent with the established understanding that HPV infection is a primary driver in a subset of OPSCC cases, distinct from those driven by traditional risk factors like alcohol and tobacco.19,20 Our survival analysis, conducted on a subset of 223 patients with complete data, underscored the prognostic significance of HPV status in OPSCC. Patients with HPV-positive tumors demonstrated markedly longer OS compared to those with HPV-negative tumors in univariate Kaplan-Meier analysis. This finding is further strengthened by the multivariate Cox regression analysis, where HPV status emerged as the only independent predictor of overall survival, with HPV-positive status associated with a significantly decreased hazard of death (aHR = 0.32, P < 0.0001). This robust association between HPV positivity and improved survival in OPSCC is well-documented in numerous studies worldwide.21-23 Moreover, our stratified Kaplan-Meier analysis by specific HPV types (HPV16, HPV18, HPV26, HPV44, and HPV58) further suggested differential survival probabilities among these types, with HPV16 showing a trend towards better survival than HPV-negative cases. While our sample sizes for some specific HPV types were limited, the overall significant P-value (P = 0.00022) warrants further investigation in larger cohorts to delineate the precise prognostic roles of individual HPV types in OPSCC. Interestingly, while the baseline characteristics in the whole cohort showed significant differences in gender, smoking habits, and alcohol consumption between HPV-positive and HPV-negative groups, the multivariate survival analysis in the 223-patient cohort identified HPV status as the sole independent predictor of OS. This discrepancy might be attributed to the exclusion of 56 patients with missing OS data, potentially introducing bias or reducing the statistical power to detect independent effects of these other factors in the survival analysis. Furthermore, the substantial prognostic impact of HPV status in this cohort might have overshadowed the contributions of these different variables in the multivariate model.
In contrast to the strong influence of HPV status on survival, our Random Forest analysis, aimed at predicting advanced-stage OPSCC, identified HPV Status as the most important predictor, with HPV-positive cases being less likely to present at an advanced stage. This finding supports the notion that HPV-driven OPSCC often presents with distinct clinical characteristics and potentially earlier detection compared to HPV-negative disease. 24 Smoking category and patient age also showed positive, albeit lesser, contributions to predicting advanced stage. Interestingly, the NKG2D LNK allele presence and gender did not positively contribute to the model’s accuracy. Importantly, our primary analysis revealed a significant association between the NKG2D rs1049174 polymorphism and OPSCC risk. Specifically, the LNK/LNK genotype was significantly associated with an increased odds of OPSCC compared to the HNK/HNK genotype (OR = 1.69, P = 0.038), and each additional LNK allele showed a similar association (OR = 1.29, P = 0.041). This suggests that this genetic variant may indeed contribute to OPSCC susceptibility in this Vietnamese cohort. However, it is notable that despite its association with OPSCC risk, our analysis of the NKG2D rs1049174 polymorphism did not reveal a significant association with the likelihood of presenting with advanced-stage OPSCC under various genetic models after adjusting for key confounders. Similarly, we found no significant difference in the distribution of NKG2D rs1049174 genotypes or allele frequencies between HPV-negative and HPV-positive patient subgroups, nor did genotype influence overall survival in multivariate analysis. While genetic variations in immune-related genes like NKG2D have been implicated in cancer susceptibility in other populations and cancer types,11,25,26 our findings provide evidence for its role in OPSCC risk, while suggesting it may not play a significant role in OPSCC progression or patient survival within this Vietnamese cohort. This could be due to differences in genetic background, environmental exposures, or the specific role of this polymorphism in the context of HPV-related vs HPV-unrelated OPSCC. 27
Our study found a significant link between the NKG2D rs1049174 polymorphism and a higher risk of OPSCC. Previous studies have highlighted NKG2D’s crucial role in immune surveillance. It helps the body fight off virally-infected cells and tumor cells. 28 Polymorphisms in NKG2D can affect how susceptible someone is to various cancers. 11 This includes nasopharyngeal carcinoma (NPC), where its role is often more pronounced, especially with Epstein-Barr virus (EBV) infection. 29 The impact of the NKG2D rs1049174 polymorphism appears to differ in OPSCC compared to NPC. Its relationship with HPV vs EBV may also vary. Several factors could explain these differences. These might include distinct tumor microenvironments that modulate immune responses differently.30,31 Variations in how HPV vs EBV evade the immune system could also play a role.32,33 Finally, unique genetic and environmental interactions within the Vietnamese population may influence the polymorphism’s penetrance and its effects on immune pathways relevant to OPSCC. Further functional studies and larger cohorts are needed. These should explore gene-environment interactions. This will help us fully understand the complex interplay of NKG2D polymorphisms, viral infections, and environmental factors in head and neck cancer development.
Despite providing valuable insights into the role of HPV and NKG2D rs1049174 polymorphism in OPSCC within a Vietnamese population, our study has several limitations that warrant consideration. Our study may still be limited in detecting subtle effects, particularly for less frequent HPV types and specific genetic subgroups. The relatively small number of patients with certain HPV types (HPV18, HPV26, HPV44, HPV58) resulted in wide confidence intervals in the survival analysis, limiting our ability to draw definitive conclusions about their prognostic roles. Secondly, our study population was recruited from hospitals in Hanoi and surrounding areas, which may not fully represent the genetic and environmental diversity of OPSCC patients across Vietnam, potentially limiting the generalizability of our findings to the entire Vietnamese population. Thirdly, while we adjusted for several key confounding factors in our multivariate analyses, other unmeasured or unknown factors could influence OPSCC risk and survival. Crucially, our study did not collect information on sexual behaviors, particularly the history of oral sex, which is a known risk factor for HPV infection in the oropharynx and could potentially confound the observed associations between HPV status and other variables.4,34 Notably, our study did not collect detailed information on the specific treatment modalities received by the patients, such as surgery, radiotherapy, chemotherapy, or combinations thereof. These treatment interventions are known to impact survival outcomes in OPSCC significantly, and the lack of this data limits our ability to fully account for their potential confounding effects on the observed associations. Furthermore, our investigation focused on a single SNP of the NKG2D gene; other genetic variations within NKG2D or other immune-related genes might play a more significant role in OPSCC susceptibility and progression. Finally, while efficient for investigating associations, the case-control design cannot establish causality. Future prospective studies with larger, more diverse cohorts and comprehensive data on treatment regimens and genetic profiling are needed to validate our findings and further elucidate the complex interplay of genetic and environmental factors in the development and progression of OPSCC in Vietnam.
Conclusion
In conclusion, our study underscores the critical role of HPV infection in OPSCC among Vietnamese patients, significantly impacting both clinical presentation and overall survival. While traditional risk factors like smoking and alcohol remain relevant, HPV status appears to be a dominant factor. In contrast to previous understanding, we found a significant association between the NKG2D rs1049174 polymorphism and OPSCC risk in this cohort. However, this polymorphism was not significantly linked to OPSCC progression (advanced stage) or overall survival. Further research with larger sample sizes and exploring other genetic polymorphisms in the context of HPV status and environmental exposures is warranted better to understand the complex etiology of OPSCC in Vietnam.
Footnotes
Acknowledgements
The authors would like to express their sincere gratitude to the staff and patients of Hanoi Medical University Hospital, the National Hospital of Otorhinolaryngology, and the National Cancer Hospital in Hanoi for participating and cooperating in this study. We also thank the two pathologists at Hanoi Medical University for their independent confirmation of the histological diagnoses. The authors gratefully acknowledge the technical assistance provided by the laboratory personnel at the Molecular Pathology Department, Faculty of Medical Technology, Hanoi Medical University, Hanoi, Vietnam, for their work in sample processing, DNA extraction, PCR, Sanger sequencing, and genotyping analyses.
Ethics Statement
This study was approved by the Institutional Review Board (IRB) of Hanoi Medical University, Hanoi, Vietnam (Approval No. 941/GCN-HĐĐĐNCYSH-ĐHYHN, dated 30th June 2023). Written informed consent was obtained from all participants prior to their enrollment in the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
