Abstract
Introduction
Early-onset colorectal cancer (EOCRC) patients are more likely to have advanced diagnoses across different race/ethnicity or age at diagnosis groups. Thus, we examined the relationship of racial/ethnic disparities with cancer diagnoses (tumor features and histologic types) and 5-year causes-specific survival for EOCRC across three age-at-diagnosis groups.
Methods
We conducted a retrospective cohort analysis using the 2006-2020 Surveillance, Epidemiology, and End Results (SEER) Program, using multivariable logistic and Cox proportional hazards models to examine the association of race/ethnicity and age with survival.
Results
Among 46,956 EOCRC patients, racial minorities were diagnosed with EOCRC at age of 20-29 years while non-Hispanic White patients were more commonly diagnosed at age 40-49 years (P-value <0.001). In adjusted analysis, non-Hispanic Black (NHB) (AOR, 1.65; 95% CI, 1.24-2.18), Asian/Pacific Islander (AOR, 2.22; 95% CI, 1.60-3.08), and Hispanic (AOR, 1.56; 95% CI, 1.27-1.93) patients aged 20-29 years were more likely to be diagnosed at a late stage. These groups were also more likely to have mucinous adenocarcinoma or signet ring cell subtypes (NHB: AOR,1.46; 95% CI, 1.00-2.13; Asian/PI: AOR, 1.89; 95% CI, 1.28-2.77; Hispanic: AOR, 1.95: 95% CI, 1.48-2.59). Regardless of age groups, NHB patients were more likely to die from CRC by 34%-59% (20-29 years: AHR, 1.40; 95% CI, 1.11-1.76; 30-39 years: AHR, 1.59; 95% CI, 1.42-1.77; 40-49 years: AHR, 1.34; 95% CI, 1.26-1.41).
Conclusion
Prioritizing prevention strategies and enhancing access to screening for racial minorities and the 20-29 age group is warranted.
Introduction
The incidence of early-onset CRC (EOCRC, defined as diagnosed with CRC at age 18-49 years) has been increasing rapidly in the United States. 1 EOCRC incidence among adults aged 20-34 years is predicted to increase by 90% by 2030. 2 EOCRC mortality rates have also increased by 1.3% per year from 2008 to 2017. 3 This is particularly concerning because young individuals are currently below the recommended age for screening and may experience limited access to preventive care. Although the contributing factors to these trends of EOCRC are still unclear, prior research has identified different clinical presentations and sociodemographic characteristics between EOCRC and average-onset CRC (defined as diagnosed with CRC at age 50+). For example, EOCRC patients are more likely to present with poorly differentiated tumors and be diagnosed at a later stage compared to older patients (50+ years). 4 This is largely attributed to delayed diagnosis and a lack of awareness or underestimation of risk by healthcare providers. 5 There is also higher prevalence of signet ring carcinomas (SC) and mucinous adenocarcinomas (MAC) among young populations compared to older groups.6,7 Some racial minorities (eg, American Indian/Alaskan Native, Black, or Asian) are also more likely than Whites to have greater risk of CRC mortality or the mentioned advanced diagnoses including right-sided CRC.8,9
Further, younger patients under 40 years, particularly those aged 20-29 years, have higher rates of high pathological grading, late-stage diagnosis, and a greater prevalence of SC and MAC. 4 However, these analyses have not been jointly examined clinical features and survival by race/ethnicity and age. Research on EOCRC survival across age groups is also limited. Such integrated information is important for guiding preventive strategies aimed at screening efficiency and treatment approaches for EOCRC. Therefore, we examined the relationship of racial/ethnic differences with tumor features and histologic types as well as 5-year causes-specific survival for EOCRC while considering the role of age at diagnosis. We hypothesize that racial/ethnic differences in EOCRC are linked to variations in tumor features, histologic types, and 5-year survival, with age at diagnosis as a moderating role.
Methods
We conducted a retrospective cohort analysis using the 2006-2020 Incidence Data with Census Tract Attributes from Surveillance, Epidemiology, and End Results (SEER) Program (22 registries, Nov 2022 submission, https://seer.cancer.gov/) (n = 835,907). We excluded 11,338 patients due to duplicate records and 730,531 patients diagnosed with CRC at age 50 or older. To ensure a more homogeneous sample, we further excluded 47,082 EOCRC patients based on several criteria, including patients diagnosed after 2015 (n = 34,530) due to limited follow-up time, and those with missing data on rurality (n = 5), persistent poverty (n = 28), survival time (n = 183), vital status (n = 1), race (n = 407), surgery (n = 684), stage (n = 2537), pathological grading (n = 8702), or primary site (n = 5). After these exclusions, 46,956 eligible EOCRC patients remained for our analysis.
Tumor features (late stage at diagnosis, high pathological grading, right-sided CRC), histologic types (SC, MAC), and 5-year CRC survival (deaths from other causes censored) were outcomes of interest. We classified high pathological grade as “yes” if tumors were poorly differentiated or undifferentiated, and “no” for all other grades. Stage at diagnosis was defined using SEER summary staging, with regional and distant stages considered late-stage disease (“yes”) and all other stages as “no”. Tumor location was categorized as right-sided (cecum to transverse colon; codes C18.0–C18.4) or left-sided (splenic flexure to rectum; codes C18.5–C18.9, C19.9, C20.9). Histologic types included MAC and SC; these were grouped as MAC/SC (yes or no). Survival time was calculated in months from the date of diagnosis up to 60 months, with follow-up censored on December 31, 2020, or at the time of death from non-CRC causes.
Exposures of interest included age at diagnosis (defined by 10-year ranges within the definition of EOCRC, ie, 20-29, 30-39, 40-49 years) and race/ethnicity (non-Hispanic White [NHW], non-Hispanic Black [NHB], Asian /Pacific Islanders [PI], American Indian/Alaskan Native [AI/AN]). Covariates of interest include sex (male or female), marital status at diagnosis (married or unmarried), census-tract level rurality (yes or no), persistent poverty (yes or no), CRC constitutes first cancer diagnosis in a patient’s lifetime (yes or no), diagnosis year (2006-2010 or 2011-2015), and treatment (surgery, chemotherapy, radiation; yes or no).
Statistical Analysis
Descriptive statistics were used to summarize the distribution of EOCRC patients by race/ethnicity and across three age groups. Frequencies and percentages were reported. Bivariate analyses using chi-square tests were conducted to examine the association between race/ethnicity and age groups. Multivariable logistic regression models were performed to examine racial differences in tumor features and histologic types across three age groups. Further, Cox proportional hazards models were conducted to evaluate the association between race/ethnicity and EOCRC survival by age groups. To address potential bias confounders, logistic regression models were adjusted for all covariates of interest while Cox proportional hazards models were adjusted for all covariates of interest, tumor features, and histologic types. Data were analyzed using SAS version 9.4. Statistical significance was a 2-sided P-value <0.05. Data extracted for this study were publicly available and de-identified and thus considered exempt from the institutional review board at Augusta University. The reporting of this study conforms to RECORD guidelines. 10
Results
Distribution of Race/Ethnicity Across Three Age Groups (n = 46,956)
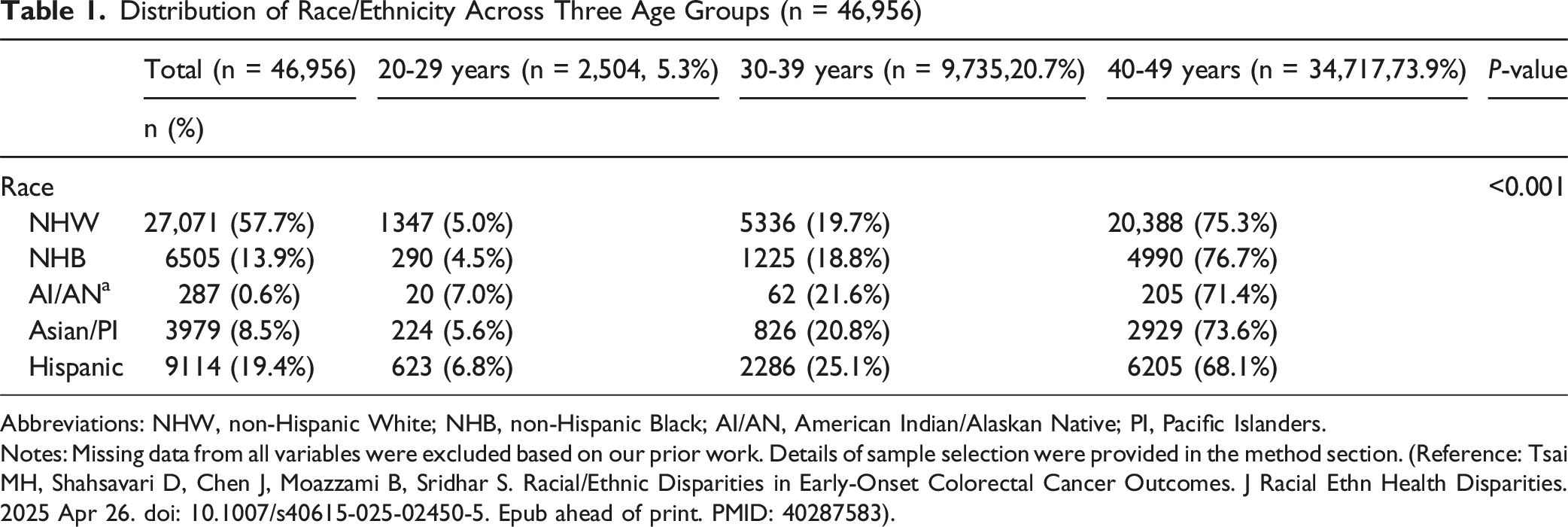
Abbreviations: NHW, non-Hispanic White; NHB, non-Hispanic Black; AI/AN, American Indian/Alaskan Native; PI, Pacific Islanders.
Notes: Missing data from all variables were excluded based on our prior work. Details of sample selection were provided in the method section. (Reference: Tsai MH, Shahsavari D, Chen J, Moazzami B, Sridhar S. Racial/Ethnic Disparities in Early-Onset Colorectal Cancer Outcomes. J Racial Ethn Health Disparities. 2025 Apr 26. doi: 10.1007/s40615-025-02450-5. Epub ahead of print. PMID: 40287583).
Association Between Race/Ethnicity and Tumor Features by Three Age Groups (n = 46,956)
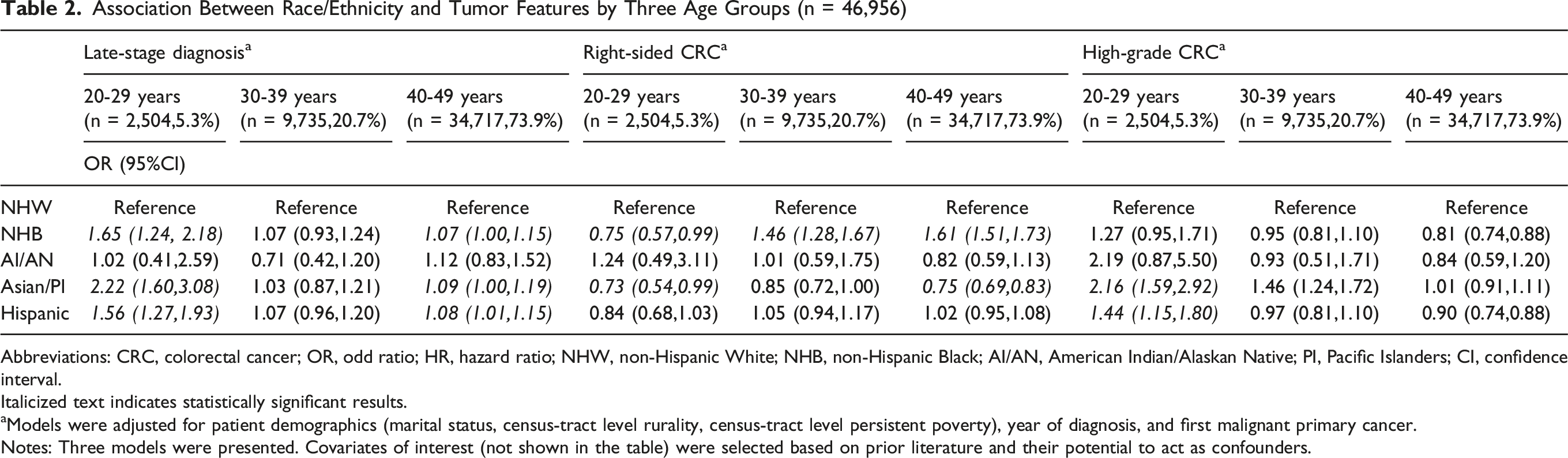
Abbreviations: CRC, colorectal cancer; OR, odd ratio; HR, hazard ratio; NHW, non-Hispanic White; NHB, non-Hispanic Black; AI/AN, American Indian/Alaskan Native; PI, Pacific Islanders; CI, confidence interval.
Italicized text indicates statistically significant results.
aModels were adjusted for patient demographics (marital status, census-tract level rurality, census-tract level persistent poverty), year of diagnosis, and first malignant primary cancer.
Notes: Three models were presented. Covariates of interest (not shown in the table) were selected based on prior literature and their potential to act as confounders.
Association Between Race/Ethnicity and Histologic Types and Causes-specific Survival by Three Age Groups (n = 46,956)

Abbreviations: EOCRC, early-onset colorectal cancer; OR, odd ratio; HR, hazard ratio; NHW, non-Hispanic White; NHB, non-Hispanic Black; AI/AN, American Indian/Alaskan Native; PI, Pacific Islanders; MAC/SC, mucinous adenocarcinomas/ signet ring; CI, confidence interval.
Italicized text indicates statistically significant results.
aModels were adjusted for patient demographics (marital status, census-tract level rurality, census-tract level persistent poverty), year of diagnosis, and first malignant primary cancer.
bModels were adjusted for patient demographics (marital status, census-tract level rurality, census-tract level persistent poverty), year of diagnosis, first malignant primary cancer, tumor features (late stage at diagnosis, high pathological grade, primary site), histologic types (adenocarcinoma, mucinous adenocarcinoma/signet-ring cell carcinoma), and treatment initiation (surgery, radiation, chemotherapy).
Notes: Two models were presented. Covariates of interest (not shown in the table) were selected based on prior literature and their potential to act as confounders.
Discussion
This is the first study to examine integrated relationships of race/ethnicity and age at diagnosis with various clinical features and mortality for EOCRC by using a representative US sample. Our findings indicate NHB, Asian/PI, and Hispanic patients aged 20-29 years were more likely to have advanced diagnoses, including late stage at diagnosis, MAC/SC subtypes, and/or high pathological grading. No prior research has examined race/ethnicity and age groups together, making direct comparisons difficult. However, these clinical presentations are generally linked to poor EOCRC prognosis. 4 Our analysis confirms that NHB patients are more likely to die from EOCRC, 9 with the highest estimate observed among those aged 30-39 years. It is possible that younger racial minorities with advanced EOCRC diagnosis or death have lower awareness of CRC or lack access to healthcare that leads to a delayed diagnosis. 5 One modifiable factor that could improve CRC outcomes is CRC screening because it can detect tumors at earlier stages and remove pre-cancerous polyps 11 ; however, younger populations often fall below the recommended screening age and may face additional barriers to accessing care, limiting early detection opportunities.
Our findings highlight disparities in clinical outcomes among adults aged 20-29 and racial minorities (NHB, Asian/Pacific Islander, and Hispanic individuals), underscoring the need for targeted, tailored prevention strategies. Young adults, particularly from racial minority groups, often face barriers such as limited access to care and low cancer risk awareness, contributing to delayed and advanced diagnoses. 5 This is especially relevant for racial minorities, where the complex interplay between race/ethnicity and social determinants of health—such as access to care, socioeconomic status, and structural barriers—may further impact cancer outcomes. 1 Therefore, culturally tailored patient-provider communication about CRC risk is especially important for these racial minorities aged 20-29. Screening recommendations that specifically address this age group and these populations may improve the effectiveness of prevention strategies. 8 Additionally, increased investment in CRC education programs is also essential to improve risk awareness among these young racial minorities. An important limitation is that several important factors (eg, the family history of CRC) associated with advanced diagnoses and mortality are unavailable in cancer registries. Future research should explore additional clinical factors (eg, biomarkers) linked to EOCRC diagnoses in young racial minorities to help reduce outcome disparities.
Conclusion
Racial minorities aged 20-29 are more likely to have advanced diagnoses, and NHB patients faced higher EOCRC mortality across all ages. These findings emphasize the need for earlier CRC risk assessment and improved access to care for young racial minorities, supported by targeted education and navigation programs to raise awareness and enhance preventive care.
Footnotes
Ethical Approval
This research does not involve human and/or animal experimentation by any of the authors. Data extracted for this study was publicly available and de-identified, and thus considered exempt from IRB review at Augusta University.
Author Contributions
Dr Tsai had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: Tsai.Acquisition, analysis, or interpretation of data: Tsai, Cortes, Vega. Drafting of the manuscript: Tsai, Cortes, Vega. Critical revision of the manuscript for important intellectual content: Tsai, Cortes, Vega. Statistical analysis: Tsai. Obtained funding: Tsai. Administrative, technical, or material support: Cortes, Vega. Supervision: Cortes.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Tsai was supported at least in part through the Georgia Cancer Center Paceline funding mechanism at Augusta University (MCGFD01071) and National Cancer Institute (NCI, R21CA301113). The content is solely the responsibility of the authors and does not necessarily represent the official views of Augusta University and NCI.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
