Abstract
Introduction
The platelet-to-lymphocyte ratio (PLR), neutrophil-to-lymphocyte ratio (NLR), and systemic immune-inflammation index (SII) are emerging biomarkers for predicting outcomes in lung cancer. However, their prognostic value in advanced non-small cell lung cancer (NSCLC) patients receiving anti-PD-1/PD-L1 therapy remains uncertain.
Methods
In this retrospective study of 304 advanced NSCLC patients treated with ICIs, we evaluated PLR, NLR, and SII for associations with progression-free survival (PFS), overall survival (OS), and objective response rate (ORR). Stratified analyses were performed based on patient age, lines of immunotherapy, PD-L1 expression, and baseline inflammatory marker levels.
Results
A total of 304 patients were included in the final analysis, with a median follow-up time of 18.6 months. The area under the curve (AUC) for 6-month PFS prediction was highest for SII (0.84), followed by NLR (0.746) and PLR (0.732). Using the optimal SII cutoff value of 776.415, patients were stratified into high and low SII groups. Multivariate analysis demonstrated that patients in the low SII group had significantly longer PFS (HR = 0.466, 95% CI: 0.319-0.681, P < 0.001) and OS (HR = 0.471, 95% CI: 0.317-0.699, P < 0.001) compared to the high SII group. Additionally, ORR was significantly higher in the low SII group (44.5% vs 29.5%, P = 0.007). In univariate analysis, patients with low PD-L1 expression (<50%) showed significantly shorter PFS (HR = 1.555, 95% CI: 1.059-2.284, P = 0.024) and OS (HR = 1.601, 95% CI: 1.075-2.384, P = 0.021) compared to those with high PD-L1 expression (≥50%).
Conclusion
For advanced NSCLC patients receiving anti-PD-1/PD-L1 therapy, lower SII values consistently may correlate with superior treatment response and survival outcomes. In clinical practice where conventional biomarkers like PD-L1 expression offer limited predictive value, SII emerges as a robust complementary indicator that may enhance immunotherapy strategy development.
Keywords
Introduction
Lung cancer remains a leading cause of cancer-related mortality worldwide, with treatment approaches encompassing surgery, radiotherapy, chemotherapy, and targeted therapies. 1 The advent of immunotherapy has revolutionized cancer treatment, offering unprecedented efficacy and durable responses. 2 For advanced non-small cell lung cancer (NSCLC) patients with inoperable oncogenic mutations, immune checkpoint inhibitors (ICIs) targeting programmed death receptor 1 (PD-1) and its ligand PD-L1 have emerged as the standard of care, either as monotherapy or in combination with platinum-based chemotherapy, demonstrating significant survival benefits.3,4 Within the Chinese NSCLC population, four PD-1 inhibitors—sintilimab, tislelizumab, toripalimab, and camrelizumab—have shown clinically meaningful improvements in progression-free survival (PFS) when combined with pemetrexed and platinum-based chemotherapy.5-8 However, despite these advances, significant challenges remain. A subset of patients experiences early mortality, potentially attributable to tumor hyperprogression or immune-related adverse events. This underscores the critical need for reliable predictive biomarkers to identify patient subgroups most likely to benefit from ICI therapy and to optimize treatment strategies.
PD-L1 expression, quantified as the tumor proportion score (TPS), remains the only clinically validated biomarker for ICI therapy in NSCLC. Current clinical trials typically require ≥1% PD-L1 expression as an inclusion criterion for immunotherapy-chemotherapy combinations in advanced NSCLC. 9 However, PD-L1 predictive utility is constrained by significant spatial and temporal heterogeneity, with discordant expression patterns observed between primary/metastatic lesions and biopsy/resection specimens. 10 Tumor mutation burden (TMB), while demonstrating predictive potential in certain malignancies, faces substantial limitations in NSCLC. The lack of standardized computational methodologies for tissue TMB (tTMB) assessment and inconsistent clinical correlations across studies have prevented its establishment as a reliable predictive marker.11,12 Microsatellite instability-high (MSI-H) status, resulting from mismatch repair deficiency (dMMR), represents a pan-cancer biomarker for ICI response. 13 However, in NSCLC, MSI-H/dMMR occurs in <1% of cases, with only partial clinical benefit observed in this subgroup, rendering it impractical as a general predictive tool. 14 The clinical application of existing biomarkers for ICIs is constrained by two critical limitations: suboptimal predictive performance and reliance on tumor tissue acquisition. These constraints are particularly problematic for patients with inadequate tissue samples. Consequently, there is an urgent need to develop more accurate, accessible biomarkers capable of dynamic monitoring to optimize ICI treatment strategies.
The platelet-to-lymphocyte ratio (PLR), neutrophil-to-lymphocyte ratio (NLR), and systemic immune-inflammation index (SII; NLR multiplied by platelet count) have emerged as clinically valuable prognostic markers in oncology, offering the dual advantages of cost-effectiveness and routine accessibility.15,16 These hematologic indices not only demonstrate prognostic significance in NSCLC but also show strong correlations with immunotherapy response. Studies have shown that NSCLC patients with lower PLR and NLR exhibit better objective response rates (ORR) and median PFS after two cycles of ICIs treatment compared to those with higher NLR. 17 As the fundamental component of SII, elevated NLR consistently portends poorer clinical outcomes, including reduced PFS and OS.18,19 Liu’s study further revealed that in NSCLC patients treated with nivolumab, pre-treatment SII levels were a decisive factor affecting prognosis, with patients having lower SII showing more favorable PFS and OS. 20 While preliminary investigations have started to uncover the potential prognostic value of the PLR, NLR, and SII in the treatment of NSCLC, there is a pressing need for extensive and systematic retrospective studies focusing on NSCLC patients treated with ICIs. Therefore, this study aims to delve into the predictive potential of PLR, NLR, and SII in NSCLC immunotherapy, in order to provide more precise and personalized treatment guidance for patients.
Materials and Methods
Patient Population
This retrospective cohort study evaluated 304 patients with stage III-IV NSCLC who underwent anti-PD-1/PD-L1 antibody therapy at Weifang Traditional Chinese Medicine Hospital from March 2019 to August 2024. Peripheral blood complete blood counts were obtained within 7 days prior to initial PD-1/PD-L1 inhibitor administration for subsequent inflammatory marker analysis. To ensure data validity, we excluded patients with confounding conditions including active HIV/hepatitis infections, concurrent corticosteroid use, ongoing radiotherapy, autoimmune disorders, chemotherapy-induced myelosuppression, actionable driver mutations (EGFR/ALK/ROS1/RET/MET), or prior targeted therapy exposure. PD-L1 expression was assessed by immunohistochemistry with high expression defined as tumor proportion score (TPS) ≥50% based on membranous staining. All patients provided written informed consent to allow the use of their anonymized medical information for research purposes. All personal information of the patients was de-identified. This study strictly adhered to the principles of the Helsinki Declaration and was formally approved by the Ethics Committee of Weifang Traditional Chinese Medicine Hospital (approval number: 2025YX220). The manuscript of this retrospective study conforms to STROBE guidelines.
21
Patient recruitment and selection criteria, including all exclusion parameters, are systematically presented in the enrollment flowchart (Figure 1). Patient Selection Flowchart for Advanced NSCLC Treated With ICIs. ICI, Immune Checkpoint Inhibitor; NSCLC, Non-Small Cell Lung Cancer
Treatment Protocol
Treatment was administered until disease progression, unacceptable toxicity, patient withdrawal, or death. The immunotherapy regimen consisted of anti-PD-1/PD-L1 monoclonal antibodies, including atezolizumab, camrelizumab, nivolumab, pembrolizumab, sintilimab, tislelizumab, and toripalimab. These agents were administered either as monotherapy or in combination with platinum-based chemotherapy and/or angiogenesis inhibitors (e.g., anlotinib, apatinib, bevacizumab), with treatment selection based on individualized clinical assessment and shared decision-making. All patients participating in the study signed an informed consent form after fully understanding and agreeing, ensuring the ethical compliance of the study.
Assessment Criteria
PFS is defined as the time from the initiation of immunotherapy to the date of documented significant disease progression. OS is measured from the start of anti-PD-1/PD-L1 antibody therapy until the patient’s death from any cause or the last follow-up. All enrolled patients underwent imaging assessments every 4 to 8 weeks following PD-1 antibody treatment, in accordance with the Response Evaluation Criteria in Solid Tumors (RECIST, version 1.1), to rigorously determine treatment efficacy. The data collection cutoff date was set for August 2024. Based on the efficacy assessment results, patient responses were meticulously categorized into four groups: complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD). The overall response rate (ORR) is calculated as the sum of CR and PR, while the disease control rate (DCR) includes CR, PR, and SD.
Statistical Analysis
Continuous variables were expressed as mean ± standard deviation, and categorical variables were analyzed using chi-square or Fisher’s exact tests. The optimal SII cutoff for predicting 6-month PFS was determined by Receiver operating characteristic (ROC) analysis (Youden index), stratifying patients into low-SI (below the cutoff value) and high-SII groups (equal to or above the cutoff value). Survival outcomes (PFS/OS) were evaluated via Kaplan-Meier curves with log-rank tests, while univariate and multivariate Cox regression models assessed prognostic factors. Statistical significance was set at P < 0.05 (two-tailed), with 95% CIs reported. Analyses were performed using SPSS for Windows version 29.0 (Inc., Chicago, Illinois).
Results
Patient Characteristics
In this study, a total of 304 patients were included based on the predefined inclusion and exclusion criteria. The cohort consisted of 224 males and 80 females, with ages ranging from 32 to 90 years. Specifically, 119 patients received immunotherapy as first-line treatment, whereas the remaining patients received it as second-line or third-line treatment. Among the participants, 249 underwent immunotherapy in combination with chemotherapy, while 55 patients received immunotherapy alone or in combination with angiogenesis inhibitors. The median number of cycles of anti-PD-1/PD-L1 antibody treatment administered to the patients was 7 (range, 2-34 cycles). The median SII was 775.09 (range, 133.58-3991.52); the 95% CI for mean SII was 829.73-952.87. The median PFS was 8.16 months, and the median OS was 16.48 months, with a median follow-up duration of 18.6 months.
Receiver operating characteristic (ROC) analysis identified an optimal cutoff value of 776.415 for the SII in survival analysis, as determined by the Youden index, with an area under the curve (AUC) of 0.84 (P < 0.001). For comparison, the AUC values for the NLR and PLR were 0.746 and 0.732. Kaplan-Meier analysis revealed significant differences in PFS and OS among patients stratified by SII quartiles (P < 0.001) (Figure 2). ROC Analyses of Inflammatory Biomarkers. (A) SII for 6-Month PFS Prediction, (B) Comparative Performance of SII, NLR and PLR, (C) PFS by SII Quartiles and (D) OS by SII Quartiles. SII, Systemic Immune-Inflammation Index; NLR, Neutrophil-to-Lymphocyte Ratio; PLR, Platelet-to-Lymphocyte Ratio; PFS, Progression-Free Survival; OS, Overall Survival; AUC, Area Under the Curve
Patient Baseline Characteristics

PFS and OS in NSCLC Patients Treated With ICIs
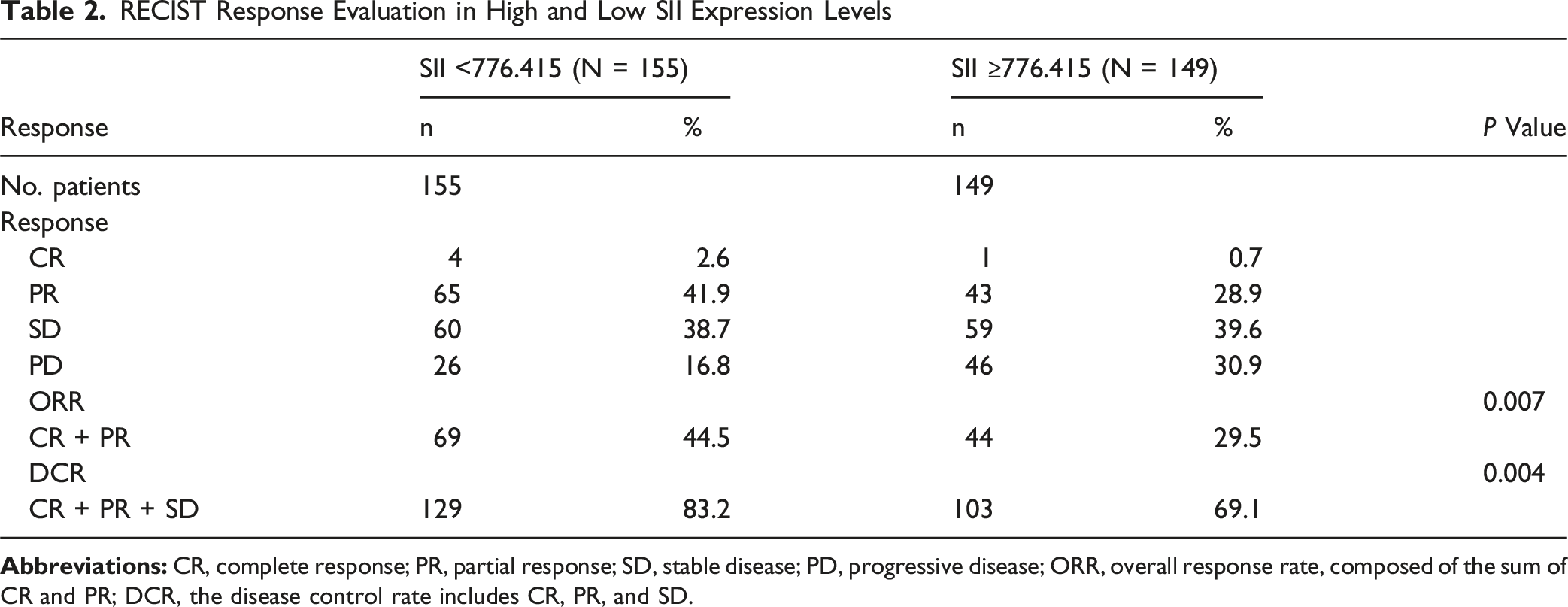
The most frequently used immune drugs included in the analysis were Sintilimab, Tislelizumab, Camrelizumab, Toripalimab, and Pembrolizumab (Figure 3). In the low SII group, 4 out of 155 patients achieved CR, whereas only 1 out of 149 patients in the high SII group achieved CR. The ORR was 44.5% in the low SII group, compared to 29.5% in the high SII group. The DCR was 83.2% in the low SII group and 69.1% in the high SII group. Statistically significant differences were observed in both ORR and DCR between the two groups (Table 2). Utilization Frequency of Immune Checkpoint Inhibitors (ICIs) in the Study Cohort RECIST Response Evaluation in High and Low SII Expression Levels
The PFS for the low SII group was 10.20 months, compared to 4.53 months for the high SII group (HR = 0.389, 95% CI: 0.297-0.509, P < 0.001). The OS for the low SII group was 20.37 months, compared to 13.90 months for the high SII group (HR = 0.492, 95% CI: 0.374-0.649, P < 0.001) (Figure 4). Kaplan-Meier Survival Analysis. (A) Progression-Free Survival (PFS) and (B) Overall Survival (OS) Stratified by Optimal Systemic Immune-Inflammation Index (SII) Cutoff Value
Cox Regression Analysis in NSCLC Patients Treated With ICIs
Univariate and Multivariate Analysis of PFS

Univariate and Multivariate Analysis of OS

Discussion
The introduction of ICIs has significantly improved the prognosis of patients with advanced NSCLC who do not have driver gene mutations. Since June 2018, nivolumab, pembrolizumab, durvalumab, and atezolizumab have been successively launched in China, becoming important members in the field of NSCLC immunotherapy, and their effectiveness and safety have been widely validated in practical applications.22,23
PD-L1 expression is routinely evaluated using immunohistochemical (IHC) staining of tumor specimens, with quantification methods and interpretation thresholds varying across clinical assays. PD-L1 expression (TPS ≥50%) serves as a key predictive biomarker for improved clinical outcomes with ICI therapy in NSCLC. 24 In the retrospective analysis of 304 patients with NSCLC, data on PD-L1 expression levels were unavailable for 148 patients (48.7%). Further multivariate analysis demonstrated that patients with low PD-L1 expression (1%-49%) had significantly shorter PFS compared to those with high PD-L1 expression (≥50%). Although the difference in OS did not reach statistical significance in the multivariate analysis. PD-L1 testing is simple, rapid, and cost-effective. However, it cannot provide highly accurate predictive value for positive or negative outcomes. Numerous clinical trial results have shown that many patients with high PD-L1 expression in NSCLC do not respond to immune checkpoint inhibitors, even when used in combination with chemotherap.25,26 Conversely, some patients with negative PD-L1 also show improved PFS and OS when treated with chemotherapy combined with pembrolizumab. 27 PD-L1 may be a useful but imperfect predictor of lung cancer response to ICIs, which may be related to tumor heterogeneity and the dynamic changes of the tumor microenvironment. Radiotherapy and chemotherapy may affect the expression of tumor immune factors (TH1, TH2, Treg), reprogram the tumor immune response, and potentially impact the efficacy of immunotherapy.28,29 The principal clinically validated biomarkers for predicting immunotherapy response include PD-L1, MSI/dMMR, and tumor mutational burden (TMB). Nevertheless, these biomarkers present significant clinical limitations: dependence on high-quality tumor tissue samples, and suboptimal predictive accuracy in NSCLC due to methodological variability. Developing more reliable, accessible biomarkers for precision immunotherapy remains an urgent need.
The dual role of inflammation in cancer—both activating antitumor immunity and fostering immunosuppressive microenvironments—has been widely recognized. 30 The detection of systemic inflammatory markers is increasingly valued in the field of tumor immune therapy response and prognosis assessment. 31 Systemic inflammatory markers derived from routine complete blood counts (NLR, PLR, SII) have gained prominence for their prognostic value in cancer.32,33 Similarly, their predictive value in tumor immunotherapy has attracted widespread attention.34-36 While elevated NLR has been associated with poorer OS and PFS in ICI-treated patients. 37 Some studies have found that there is no significant difference in survival rates between NSCLC patients with high baseline NLR and PLR levels and those with low baseline PLR levels, which makes the relationship between NLR, PLR, and immunotherapy outcomes still controversial. 36
The current study concentrates on the prognostic value of three blood inflammatory markers, including NLR, PLR, and SII, in NSCLC patients undergoing anti-PD-1/PD-L1 antibody treatment. The findings indicate that SII outperforms NLR and PLR in prognostic assessment. Multivariate analysis further solidifies that SII is an independent prognostic factor, with patients presenting lower SII levels experiencing longer PFS and OS. Moreover, the low SII group exhibits a higher ORR compared to the high SII group. This result aligns with the study by Liu et al, thereby further substantiating that SII holds superior predictive value in contrast to PLR and NLR. 20 Although multiple studies have examined pre-treatment SII association with survival in NSCLC patients undergoing ICI therapy, reported cutoff values and prognostic significance remain inconsistent.38,39 These discrepancies may be due to population heterogeneity, varying study designs, and the differential effects of specific ICIs.
The predictive role of SII in patient survival can be further elucidated by examining the functions of lymphocytes, neutrophils, and platelets in cancer progression and metastasis. Previous studies have demonstrated that a high density of tumor-associated neutrophils (TANs) is strongly associated with unfavorable prognosis. 40 Neutrophils contribute to tumor progression by inducing angiogenesis, facilitating tumor invasion and metastasis, accelerating tumor growth, and evading immune surveillance. 41 Moreover, they can suppress T-cell activation through the release of inhibitory cytokines such as IL-10 and upregulation of PD-L1, thereby impairing anti-tumor immunity. 42 Conversely, tumor-infiltrating lymphocytes (TILs), particularly intra-stromal CD8+ TILs, play a critical role in the efficacy of immune checkpoint inhibitors (ICIs). 43 Immune checkpoint blockers enhance anti-tumor responses by “reprogramming” CD8+ TILs through the targeting of inhibitory receptors.44-46 Additionally, platelets actively participate in the tumor microenvironment of NSCLC. They internalize PD-L1 from tumor cell surfaces via a fibronectin, integrin α5β1mechanism, thereby shielding circulating tumor cells from immune attack and promoting hematogenous metastasis.44,45
In summary, SII demonstrates broad potential as a comprehensive prognostic marker in cancer, given the intricate roles of neutrophils, platelets, and lymphocytes—and their interactions—in tumor growth and metastasis. However, this study has several limitations. As a single-center, retrospective analysis, data collection and processing relied on manual extraction from medical records. Although laboratory results and survival data were rigorously verified, potential patient selection bias cannot be entirely ruled out. Furthermore, the cohort primarily included patients treated with ICIs that have a higher level of evidence in the Chinese NSCLC population; thus, the generalizability of our findings warrants cautious interpretation. While the optimal SII cutoff value was determined using large-sample data, its accuracy and clinical applicability require validation through prospective studies. Consequently, larger prospective trials are urgently needed to confirm our results and establish more robust guidelines for ICI use in clinical practice. Despite these limitations, this study presents a convenient and practical predictive tool, offering valuable insights for assessing treatment efficacy in NSCLC patients receiving ICIs.
Conclusion
SII, as a composite biomarker reflecting both inflammatory and immune status, represents a robust prognostic tool for predicting survival outcomes in NSCLC patients undergoing anti-PD-1/PD-L1 therapy. The integration of SII with other relevant parameters, including NLR and PLR, may offer clinically meaningful insights for optimizing personalized immunotherapy strategies in NSCLC management.
Footnotes
Acknowledgments
We sincerely appreciate all study participants for their valuable contributions to this research.
Ethical Approval
This study was conducted in accordance with the Declaration of Helsinki and received formal approval from the Ethics Committee of Weifang Traditional Chinese Medicine Hospital (approval number: 2025YX220). Written informed consent was obtained from all participating subjects.
Author Contributions
All authors contributed to study design, data collection/analysis, manuscript drafting/revision, and approved the final version. All agreed on journal submission and are accountable for all work aspects.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Shandong Province Natural Science Foundation through its Innovation and Development Joint Fund (Grant No. ZR2023LZY006) and the Shandong Province Natural Science Foundation for Traditional Chinese Medicine Joint Fund Key Project (Grant No. ZR2021LZY015).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
