Abstract
The leucine-rich pentatricopeptide repeat-containing (LRPPRC) protein, a member of the pentatricopeptide repeat (PPR) family, is a mitochondria-associated protein that regulates various biological processes, including cell cycle progression and mitochondrial gene translation. LRPPRC has also been identified as an important causative gene in several mitochondrial diseases. N6-methyladenosine (m6A) is the most prevalent and extensive modification of mRNA in eukaryotes, playing a significant role in cellular proliferation, differentiation, and oncogenesis. As an m6A regulator, LRPPRC has been shown to play an important role in the development of various human metabolic diseases and malignant tumors. This review mainly focuses on summarizing the biological roles of LRPPRC in a variety of human malignant tumors, emphasizing the molecular mechanisms LRPPRC is involved in and its potential impact on tumor prognosis.
Keywords
Introduction
RNA modification is an important component of epigenetics and contributes significantly to the regulation of cellular functions, along with gene and protein modifications. 1 Among eukaryotes, N6-methyladenosine (m6A) is the most widespread and abundant modification observed in mRNA, playing a crucial role in the translation, processing, splicing, stabilization, and degradation of target genes. 2 The methylation of RNA m6A is a highly dynamic and reversible process, governed by three key types of proteins: m6A methyltransferases (writers), m6A demethylases (erasers), and m6A-binding proteins (readers). M6A is installed by methyltransferases, including METTL3,3-5 METTL14, 6 WTAP,7-9 METTL16,10-12 FTO13-15 and ALKBH5,16-18 as examples of demethylases, remove the methyl group from the chain. Additionally, m6A RNA modification can interact with m6A-binding proteins to perform different functions, such as YTH domain family proteins (YTHDF1,19-21 YTHDF2,22-24YTHDF3, 25 YTHDC1,26-28 and YTHDC229-31), insulin-like growth factor 2 mRNA-binding proteins (IGF2BP1,32-34IGF2BP2,35-37 and IGF2BP338-40), LRPPRC have also been identified as m6A readers. 41 As methylation regulators of m6A RNA, these proteins regulate mRNA transcription, maturation, translation, degradation, and stability.42-44 Indeed, studies increasingly indicate that regulators of m6A RNA methylation are linked to the occurrence and development of various malignant cancers.45-49
LRPPRC, a gene spanning 4.8 kb and encoding a protein of 130 kDa, was first identified in HepG2 cells by Hou J in 1994. 50 LRPPRC is a member of a protein family characterized by the PPR motif, and the PPR motif, which consists of a repeated sequence of 35 amino acids, is found in tandem arrays ranging from 2 to 50 iterations within pentatricopeptide repeat proteins. 51 PPR proteins bind to specific RNA sequences and are mainly involved in post-transcriptional events, such as RNA editing, translation, stability, and splicing. 52 It has been shown that LRPPRC is a multifunctional protein found in the inner and outer nuclear membranes as well as in the cytoskeleton, endoplasmic reticulum, nucleoplasm, and mitochondria, regulating multiple biological processes, including energy metabolism. 53
Several studies have shown that LRPPRC is aberrantly expressed as an m6A regulator in various diseases, such as intracranial aneurysms, atherosclerosis, and malignant tumors, and is negatively correlated with most immune cells.54,55 In addition, LRPPRC significantly contributes to anti-apoptotic mechanisms and enhances the invasive potential of tumor cells, leading to increased drug resistance. 56 Therefore, the overexpression of LRPPRC is strongly linked to the progression of disease and the poor prognosis, suggesting that LRPPRC may be used as a diagnostic tool and a new therapeutic target in malignant tumors. Furthermore, mutations in the LRPPRC gene can induce Leigh syndrome French Canadian (LSFC), a neurodegenerative disorder marked by a deficiency in cytochrome c oxidase activity. This condition is characterized by developmental delay, hypotonia, ataxia, mild facial dysmorphism, and growth retardation. 57 And studies have found that patients with Alzheimer’s disease and Parkinson’s disease have significantly reduced LRPPRC gene expression in their brain tissue. 58 Thus, LRPPRC has been identified as a pivotal gene involved in the etiology of early-onset, multisystem, and neurological mitochondrial diseases. 59
In this review, the PubMed (Medline), Embase, ISI Web of Knowledge, Springer, the Cochrane Library, Scopus, BioMed Central, and ScienceDirect databases were used to conduct a comprehensive search for studies. The search strategy included the following keywords, variably combined: ‘LRPPRC and cancer,’ ‘LRPPRC and tumor,’ and ‘LRPPRC and carcinoma’, published between inception and March 2025. After excluding duplicate articles, the current role of LRPPRC in malignant tumors and its potential impact on tumor prognosis are summarized.
The Biological Functions of LRPPRC in Tumor Cells
In various studies, LRPPRC has been implicated in the progression and pathogenesis of malignant tumors, particularly in relation to mitochondrial oxidative phosphorylation60,61 and antophagy.73,74
Various perspectives exist regarding the role of LRPPRC within mitochondria. Many studies have provided compelling genetic evidence that LRPPRC is involved in the stability, polyadenylation, and coordination of mitochondrial mRNA translation.62-64 Nonetheless, some reports suggest that LRPPRC acts as a transcriptional activator by forming a complex with POLRMT,65,66 but Harmel et al. found that LRPPRC does not directly regulate mtDNA transcription, but rather acts as a posttranscriptional regulator of mammalian mtDNA expression. 67 Polyadenylation of mRNA is crucial for its stability and translation in mitochondria, 68 and it has been shown that LRPPRC is essential for the proper polyadenylation of mitochondrial mRNA; without the expression of LRPPRC gene, mRNA polyadenylation is limited to the addition of oligo (A) residues, resulting in the subsequent termination of transcription and the deficiency of translation to proceed. 63 In mammalian cells, LRPPRC plays various roles by interacting directly or indirectly with other proteins. Specifically, LRPPRC forms a complex with the SRA stem-loop interacting RNA binding protein (SLIRP) through the RNA recognition motif-PPR protein interface. The LRPPRC-SLIRP complex interacts with mitochondrial mRNA, stabilizing its structure. 69 This stabilization facilitates the exposure of critical sites needed for polyadenylation and translation, achieved through the regulation of mitochondrial poly(A) polymerase activity. 64
LRPPRC proteins play a critical role in the maturation process of nuclear mRNAs and their subsequent export to the cytoplasm. LRPPRC binds to the ribonucleoprotein complex and shuttles mature mRNAs from the nucleus to the cytoplasm. 70 Additionally, LRPPRC can act as a cofactor for eukaryotic translation initiation factor eIF4E, assisting this factor in regulating nuclear gene expression, which involves managing the export of specific mRNAs from the nucleus to the cytoplasm. 71
As essential organelles in eukaryotic cells, mitochondria are crucial for energy metabolism, sustaining redox equilibrium, and modulating apoptotic processes.72,73 Mitochondria primarily facilitate aerobic respiration via oxidative phosphorylation, generating ATP through the mitochondrial electron transport chain to meet the cell’s energy demands for survival. 74 In cancer cells, mitochondria exhibit distinct characteristics compared to normal cells, notably producing excessive amounts of reactive oxygen species (ROS). This heightened ROS generation leads to telomere erosion and the formation of DNA double-strand breaks, as well as the disruption of mitotic checkpoints, and contributes to cancer progression by inducing genome instability, modulating gene expression, and engaging signaling pathways.75,76 The “vicious circle” between mitochondria, ROS, and genomic instability is perpetuated by mutations in mitochondrial and nuclear DNA caused by oxidative damage. 77 It has been demonstrated that LRPPRC regulates the expression of all mitochondrial DNA-encoded mRNAs except for NADH-dehydrogenase 6 (ND6) mRNA. 78 Pham et al. points to a mechanism of post-transcriptional regulation in muscle mitochondria through the stabilization of mitochondrial mRNA, providing insights into how exercise improves mitoribosome capacity and mitochondrial quality control to correct defects. 79 A deficiency in LRPPRC leads to instability in polyadenylated mitochondrial mRNAs, disruption of mitochondrial ultrastructure, and significant assembly defects in the respiratory chain’s complex IV (CIV) and ATP synthase (CV). 80
Autophagy is a conserved self-degradation system that sequesters misfolded proteins, defective or senescent organelles, and aberrant proteins within double-membraned structures termed autophagosomes, which ultimately merge with lysosomes for degradation. 81 Autophagy exhibits dynamic oncogenic or pro-oncogenic phenomena during the different stages of tumor development. 82 Microtubule-associated protein 1S (MAP1S) is a linker that connects mitochondria to microtubules for transport and links the autophagy machinery to microtubules and mitochondria. MAP1S influences autophagosome biogenesis and degradation, and it interacts with autophagosome markers LC3 and LRPPRC. 83 Bcl-2 proteins associated with follicular lymphoma are crucial for controlling apoptosis and promoting cell survival in response to a variety of apoptotic stimuli, 84 Beclin 1 is the first mammalian autophagy protein identified as a protein interacting with Bcl-2, 85 LRPPRC forms a ternary complex with Beclin 1 and Bcl-2, which is crucial for maintaining the stability of Bcl-2. LRPPRC also inhibits the Beclin 1-PI3KCIII complex, which is involved in the regulation of autophagy, to activate autophagy and mitochondrial autophagy. 86 Wang et al showed that lack of oxidative phosphorylation increased apoptosis and induced autophagy,87,88 suggesting that LRPPRC may play a role in inhibiting autophagy.
LRPPRC and Cancers
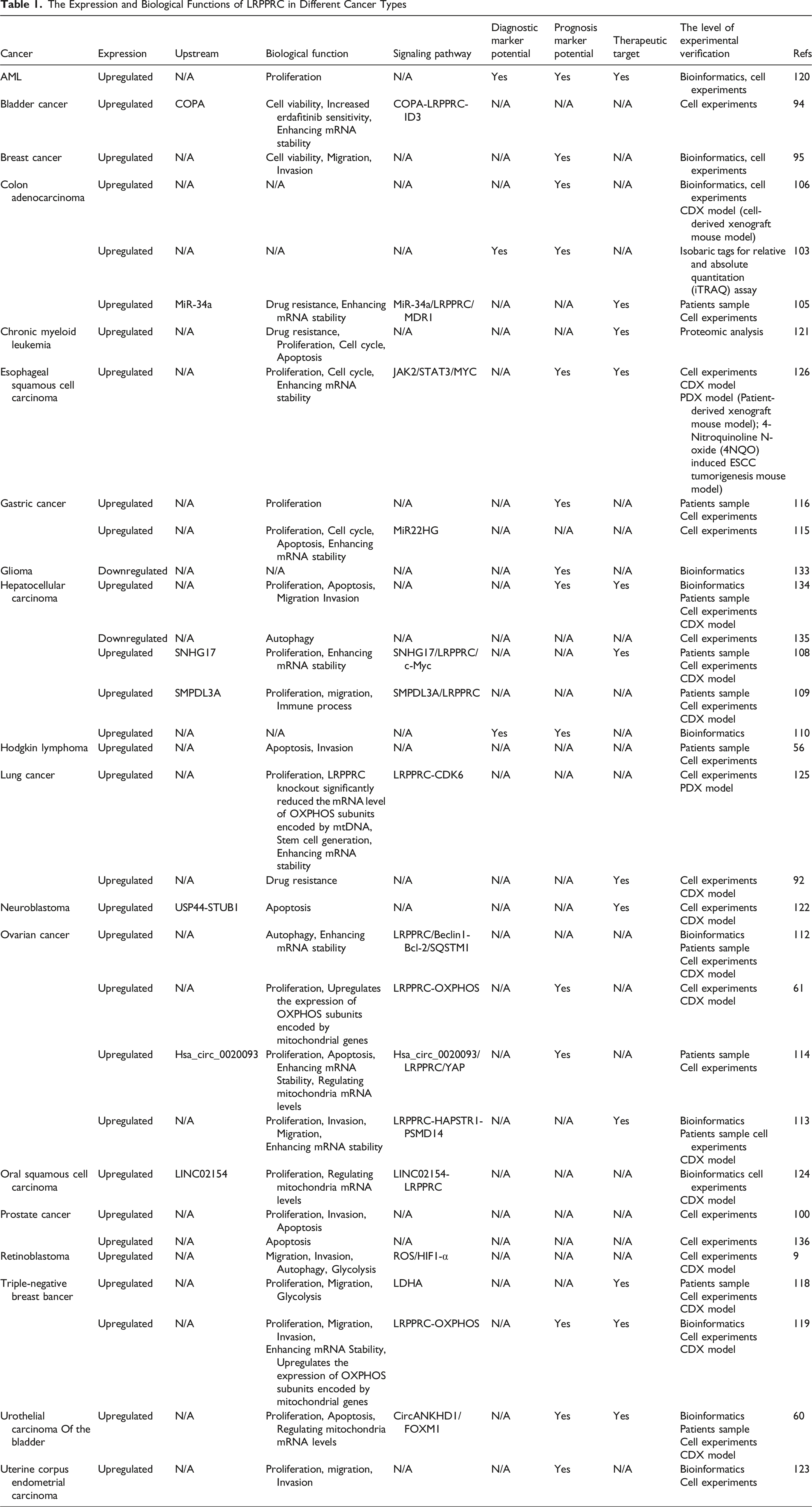
Numerous studies have demonstrated that LRPPRC is upregulated in various human cancers (Figure 1), where it contributes to cancer progression by modulating biological processes, including cell migration and proliferation (Figures 2 and 3), and involving in many signal pathways (Figure 2). We provide an overview of the distinct functions of LRPPRC across various cancer types (Table 1). Expression of LRPPRC Across Various Cancer Types The Role of LRPPRC in Regulating Multiple Human Cancers and Many Signal Pathways The Role of LRPPRC in Regulating Multiple Human Cancers in Radar Chart The Expression and Biological Functions of LRPPRC in Different Cancer Types
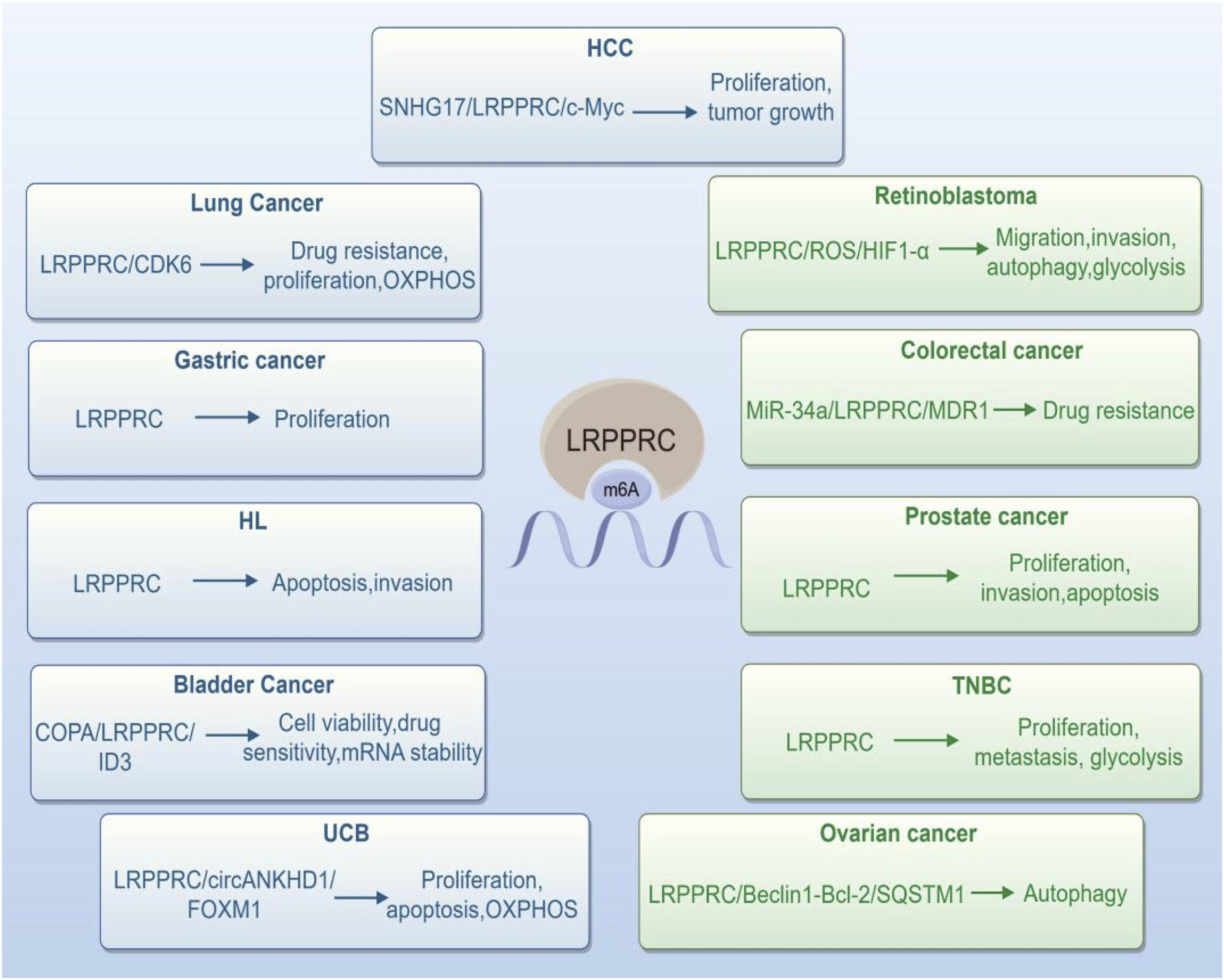

LRPPRC and Lung Cancer
Lung adenocarcinoma (LUAD) represents the most prevalent histological type of lung cancer, comprising roughly 40% of all lung cancers. 89 Yin et al demonstrated increased expression of LRPPRC in malignant cells at the primary site and in metastatic lymph nodes compared with normal lung epithelial cells. It was discovered that the majority of the infiltrating immune cell activities and immune responses had a negative correlation with LRPPRC. And overexpression of LRPPRC were associated with markedly poorer prognosis compared to those with low LRPPRC expression in the anti-PD-L1 treatment cohort. 90 The multidrug resistance protein 1 (MDR1) gene encodes P-glycoprotein (P-gp), which increases drug resistance in cancer cells by reducing anticancer drug concentrations inside the cells , among other effects. 91 Hu et al. also indicated that LRPPRC can further intensify the resistance of lung cancer cells to cisplatin through modulation of the MDR1 transcription process. 92 These findings suggest that LRPPRC could potentially act as a novel regulator of the tumor microenvironment, affect the prognosis of patients receiving immunotherapy, and regulating chemoresistance in lung cancer.
LRPPRC and Bladder Cancer
Urothelial carcinoma of the bladder (UCB) is the most common type of bladder cancer. 93 According to the WEI’s study, LRPPRC regulates mitochondrial mRNA metabolism and orchestrates the circANKHD1/FOXM1 axis to protect UCB cells from oxidative stress. It was found that SLIRP protein directly interacts with LRPPRC to prevent degradation by ubiquitination and proteasome. 60 This research clearly indicates that LRPPRC is indispensable in the regulation of UCB redox homeostasis and tumorigenesis, and suggests that LRPPRC may be further developed as a potential therapeutic target for UCB. Coatomer protein complex subunit α (COPA) is identified as a key target to enhance erdafitinib sensitivity. COPA knockout increases the degradation of LRPPRC protein, leading to reduced inhibitor of DNA binding 3 (ID3) mRNA stability in an m6A-dependent manner. 94 Li et al identify LRPPRC and CXCL11 as oncogenic factors in breast cancer, with LRPPRC partially increasing CXCL11 expression by stabilizing its mRNA. These findings reveal a new mechanism by which m6A modulators promote malignancy and secrete inflammatory factors in BC. 95
LRPPRC and Retinoblastoma
Retinoblastoma (RB) is the most common malignant intraocular childhood tumor. 96 Most retinoblastomas are triggered by biallelic mutations in the developing retinoblastoma tumor suppressor gene, RB1. 97 The expression of LRPPRC is markedly elevated in retinoblastoma tissues compared to normal tissues. Furthermore, LRPPRC can promote tumor migration, invasion, and glycolysis through suppression of autophagy and activation of the ROS/HIF1-α pathway. 98 LRPPRC is expected to be an important prognostic biomarker for retinoblastoma.
LRPPRC and Prostate Cancer
According to Jiang’s research, prostate cancer samples had significantly higher LRPPRC levels than BPH samples, with more than 75% of prostate cancer patients exhibiting high levels of LRPPRC. It was revealed that LRPPRC levels were positively correlated with metastasis, serum prostate-specific antigen levels, tumor grade, while being negatively correlated with sensitivity to hormone therapy at 2 years postoperatively and overall survival. 99 Zhou et al showed that the knockdown of LRPPRC dramatically inhibited invasion and promoted apoptosis in prostate cancer cells. 100 The interaction between mitochondria, microtubule-associated protein MAP1S, and LRPPRC can synergistically regulate the autophagy process. Combining these factors may serve as an important indicator for assessing the prognosis of prostate cancer patients. 101 Additionally, LRPPRC can be used as an independent biomarker in advanced prostate cancer patients with poor prognosis. 102
LRPPRC and Colorectal Cancer
Tomohisa et al used isobaric tags for relative and absolute quantitation (iTRAQ) to identify novel biomarkers for colorectal cancer (CRC), they found that the expression level of LRPPRC was markedly elevated in colorectal cancer tissues compared to its levels in normal colorectal tissues. This finding was also confirmed by immunohistochemistry results, and the expression level correlated with the tumor differentiation, particularly in moderately differentiated colorectal cancer tissues. 103 P53 mutations are an important cause of proliferation, invasion, and metastatic chemoresistance in CRCcells. 104 A study by Yang has demonstrated that LRPPRC is a significant downstream gene and possibly a therapeutic target in CRC cells that are resistant to chemotherapy due to p53 mutations. In non-p53 mutant CRC cells, chemotherapy-induced p53 inhibits the expression of LRPPRC through miR-34a, which subsequently leads to a reduction in the expression of MDR1, resulting in decreased cellular resistance to anticancer drugs. However, when p53 was mutated, it made the chemotherapy-induced miR-34a/LRPPRC/MDR1signaling pathway less active, which resulted in the accumulation of MDR1 and LRPPRC and the enhancement of drug resistance. 105 Lv et al using LASSO logistic regression analysis for selection of optimal the differentially expressed nuclear mitochondrial-related genes (DE-NMRGs) obtained from univariate cox survival analysis and the Kaplan–Meier (KM) survival curve showed high expression levels of LRPPRC had higher overall survival time in Colon Adenocarcinoma. 106 Those finding indicated that LRPPRC can be used as a molecular target for diagnosing or prognosis CRC and provides a corresponding intervention strategy for future precision treatment of CRC.
LRPPRC and Liver Cancer
Globally, liver cancer is the fourth leading cause of cancer-related deaths, continuing to pose a significant threat to global health. Among primary liver cancers, hepatocellular carcinoma (HCC) is the most prevalent type. 107 Liu’s study on human HCC tissues revealed markedly elevated LRPPRC expression in HCC compared to adjacent non-cancerous tissues, correlating high LRPPRC levels with poorer survival outcomes among HCC patients. Moreover, it has been demonstrated that the lncRNA SnoRNA host gene 17 (SNHG17)-LRPPRC-c Myc regulatory axis accelerates the G1/S transition in the cell cycle and promotes cell proliferation, thereby accelerating the growth of tumor cells. 108 LRPPRC is also abnormally overexpressed in HCC, contributing to the unfavorable prognosis of HCC patients, according to Dan’s study. Xu et al have found that METTL3/SMPDL3A/LRPPRC axis promote the tumor genesis of HCC. 109 In HCC, LRPPRC showed a notable increase, and elevated levels of LRPPRC were associated with poorer OS, DSS, and PFI in HCC patients, demonstrating superior diagnostic and prognostic potential. 110 Additionally, the prognosis of HCC and immune microenvironment are investigated using m6A/m5C/m1A-regulated genes. 111
LRPPRC and Ovarian Cancer (OC)
Jiang et al indicated that LRPPRC exhibits heightened expression in OC and strongly correlates with adverse outcomes, thereby, fostering the malignant characteristics of OC cells in vitro and in vivo. Zhao et al demonstrated that deubiquitylase PSMD14 expression is upregulated in OC and a direct interaction between PSMD14 and LRPPRC suppresses its ubiquitination, resulting in suppression of autophagy via LRPPRC/Beclin1-Bcl-2/SQSTM1. 112 This indicates that HAPSTR1 prevents LRPPRC degradation via ubiquitination by strengthening the interaction between LRPPRC and PSMD14. Li’s finding revealed that LRPPRC plays a role in HAPSTR1-driven proliferation, invasion, and migration, LRPPRC is an important target for therapy in OC treatment. 113 And it was reported that LRPPRC was negatively correlated with OC prognosis and its depletion impaired the cell viability and enhanced apoptosis of OC induced by cisplatin. 114
LRPPRC and Gastric Cancer (GC)
Ci et al findings indicated that the levels of EEF1A1 and LRPPRC were significantly increased in GC at both RNA and protein level. Silencing LRPPRC inhibited the viability of GC cells and caused cell cycle arrest at the G0/G1 phase, leading to apoptosis in GC cells. 115 Research has shown that LRPPRC expression levels are significantly correlated with tumor infiltration depth, lymph node metastasis (N stage), and distant metastasis (M stage). 56 LRPPRC expression was markedly elevated in GC tissues compared to normal gastric tissues, and a higher level of LRPPRC expression is associated with a lower overall survival rate than a low level of LRPPRC expression. A proliferation assay showed a negative relationship between LRPPRC expression and GC cells growth in vitro. 116 These findings suggest that increased LRPPRC expression in individuals with GC is strongly linked to poor prognosis and decreased overall survival. Therefore, LRPPRC is serve as a prognostic marker for GC patients and may emerge as a therapeutic target for GC.
LRPPRC and Breast Cancer
Breast cancer with triple-negative status (TNBC) is the most aggressive type and has a poor prognosis. 117 Yu et al showed that the overexpression of LRPPRC in TNBC is associated with the identification of the m6A modification site. In vitro and in vivo, LRPPRC promoted proliferation, metastasis, and glycolysis in TNBC cells. Moreover, lactate dehydrogenase A (LDHA) was found to be a novel direct target of LRPPRC, and LRPPRC recognized the m6A site of LDHA mRNA, enhancing its stability and thereby promoting TNBC glycolysis. 118 Furthermore, LRPPRC is serve as a prognostic marker for TNBC patients and may emerge as a therapeutic target for TNBC. 119
LRPPRC and Hematologic Malignancy
It was demonstrated that cells with Hodgkin’s lymphoma and diffuse large B-cell lymphoma expressed significantly higher LRPPRC than mixed inflammatory cells, which served as a control. Hodgkin’s lymphoma cells with low expression of LRPPRC showed reduced resistance to apoptosis, invasion, and colony-forming ability in vitro. 56 Wu et al findings were confirmed through immune correlation analysis and laboratory experiments in acute myeloid leukemia (AML), emphasizing LRPPRC’s potential as a new and dependable diagnostic and prognostic biomarker in AML, emphasizing its role in precision medicine and tumor immunotherapy. 120 LRPPRC was overexpressed in cells that are cross-resistant to MDR/imatinib mesylate (IM) in chronic myeloid leukemia. 121
LRPPRC in Other Cancers
In neuroblastoma (NB), by stabilizing STUB1, USP44 aids in the polyubiquitination and subsequent degradation of LRPPRC, and LRPPRC overexpression diminishes cisplatin sensitivity in both in vitro and in vivo. The USP44-STUB1-LRPPRC pathway is crucial in cisplatin resistance and could include therapeutic targets and prognostic indicators for NB. 122 LRPPRC acts as a new biomarker and enhances the proliferation and invasion capabilities of UCEC and the expression of LRPPRC was identified as an independent prognostic marker through both univariate and multivariate Cox regression analyses. 123 Niinuma et al found that the interaction between LINC02154 and LRPPRC plays a role in regulating mitochondrial genes , and may contribute to the development of innovative treatments for OSCC. 124
Inhibitors of LRPPRC
Most reported 2 direct small molecule inhibitors of LRPPRC were: gossypol acetate (GAA) and 5,7,4'-trimethoxyflavone (TMF), along with their analogues. GAA as an old medicine in managing gynecology diseases, including menorrhagia, uterine fibroids, and endometriosis. Recently, has shown tumor suppressive effects for lung cancer, CRC, OC, leukemia, and esophageal cancer.61,105,125-127 GAA inhibited OXPHOS by directly binding LRPPRC and induces LRPPRC rapid degradation in a ubiquitin-independent manner.61,119 GAA has been used in clinical trials targeting several cancer types, where it is viewed as a BCL2 inhibitor when combined with chemotherapy drugs for unscreened patients. Nonetheless, the total clinical advantage is still limited.128-130GAA still facing problems such as reproduction toxicity, limited affinity and relatively high effective concentration. To avoid reproduction toxicity, a new LRPPRC inhibitor TMF discovered, which can directly attach to LRPPRC. Liu et al found that TMF was confirmed to attach to LRPPRC, STAT3, and CDK1, disrupting the interactions between LRPPRC-JAK2-STAT3 and JAK2-STAT3-CDK1, which resulted in reduced tumor development in ESCC mouse models induced by 4-Nitroquinoline N-oxide. 126 In order to increase the extraction rate of TMF from Tangerine Peel, structural modifications of TMF have led to 2 series of TMF derivatives, among all compounds, 11g demonstrated its ability to attach to LRPPRC, STAT3, and CDK1, leading to the breakdown of the LRPPRC-JAK2-STAT3 and JAK2-STAT3-CDK1 complexes, thereby inhibiting the JAK2/STAT3 signaling pathway. 131 In vivo and in vitro studies have shown that chemotherapy with GAA and 5-fluorouracil (5FU) can reduce chemo-resistance and improve therapeutic response by effectively degrading LRPPRC protein. 106
Recently, demethylzeylasteral (T96), a novel inhibitor of LRPPRC, which bound to the RNA-binding domain of LRPPRC directly and inhibited the nucleic acid binding ability of LRPPRC, this led to instability in both mt-mRNA and LRPPRC protein. 132
Discussion
LRPPRC functions as a mitochondria-associated protein that regulates the transcriptional and post-transcriptional expression of mitochondrial DNA and influences mitochondrial OXPHOS and cellular autophagy in cancer cells. As a m6A regulator, LRPPRC expression is elevated in a variety of human malignant tumors and negatively correlates with tumor stage, prognosis, and survival (Figure 2). Since its discovery, there has been notable advancement in understanding the structure, function, mechanism, and inhibitors of LRPPRC indifferent types of cancer. However, from TCGA and GTEX database, the expression of LRPPRC is decreased in some cancers, such as Glioblastoma multiforme (GBM)、 Kidney Chromophobe (KICH)、Kidney renal clear cell carcinoma(KIRC) 110 and glioma. 133 Furthermore, the specific signaling pathways through which LRPPRC drives tumorigenesis and development remain unclear and require further investigation, but this necessitates a deeper understanding of LRPPRC’s mechanisms of action.
Due to its RNA binding function, LRPPRC specifically bound to the mRNA of MDR1, increasing MDR1 mRNA stability and protein expression. The effect of LRPPRC on MDR has been studied in many tumor types. Cells resistant to MDR/IM in chronic myeloid leukemia exhibit overexpression of LRPPRC. 121 And LRPPRC silencing increased cisplatin sensitivity in lung cancer cells. 92 However, as the first specific inhibitor, GAA effectively induced degradation of the LRPPRC protein and reduced chemoresistance in CRC cells with P53 mutation, 105 the use of inhibitors can improve tumor drug resistance mediated by LRPPRC.
Nonetheless, LRPPRC still has a significant journey ahead before it can be used as a biomarker and therapeutic target in cancer diagnosis, treatment, and prognosis. Need to focus on unresolved issues, including: exploring the structural foundation of LRPPRC and its interacting partners; examining its function in posttranslational modifications such as phosphorylation, ubiquitination, and acetylation, investigating the interaction between LRPPRC and cancer characteristics (signaling pathways, immune response, metabolites, microbiome); identifying new direct inhibitors of LRPPRC.
Footnotes
Author Contributions
Conceptualization, J.X.T., X.C. and Y.Y.Z.; data curation, J.X.T., S.Y.Q., Y.X. and J.X.L.; writing J.X.T.; writing—review and editing, X.C.; supervision, J.L.; funding acquisition, X.C. and Y.Y.Z. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The APC was funded by the Natural Science Foundation of Shandong Province, ZR2020MH314 (to Xian Chen), and by the Natural Science Foundation of Shandong Province, ZR2021MH311 (to Yunyuan Zhang), and by Qilu Health and Health Outstanding Youth Talent Training Fund (to Xian Chen and Yunyuan Zhang).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
