Abstract
Ovarian cancer (OC) is the most lethal gynecological tumor, characterized by its insidious and frequently recurring metastatic progression. Owing to limited early screening methods, over 70% of OC cases are diagnosed at advanced stages, typically stage III or IV. Recently, N6-methyladenosine (m6A) modification has emerged as a hotspot of epigenetic research, representing a significant endogenous RNA modification in higher eukaryotes. Numerous studies have reported that m6A-related regulatory factors play pivotal roles in tumor development through diverse mechanisms. Moreover, recent studies have indicated the aberrant expression of multiple regulatory factors in OC. Therefore, this paper comprehensively reviews research advancements concerning m6A in OC, aiming to elucidate the regulatory mechanism of m6A-associated regulators on pivotal aspects, such as proliferation, invasion, metastasis, and drug resistance, in OC. Furthermore, it discusses the potential of m6A-associated regulators as early diagnostic markers and therapeutic targets, thus contributing to the diagnosis and treatment of OC.
Plain Language Summary
Ovarian cancer (OC) presents a formidable challenge in the medical field, often detected at advanced stages, necessitating urgent exploration of diagnostic and therapeutic avenues. This review delves into the intricate role of N6-methyladenosine (m6A) RNA modification in OC, a dynamic epigenetic process increasingly recognized for its regulatory role in cancer biology. Highlighting recent advancements, the review sheds light on how m6A-related factors influence crucial aspects of OC progression, including tumor growth, metastasis, and resistance to treatment. Specifically, m6A methyltransferases, binding proteins, and demethylases exert multifaceted effects on OC progression, influencing the expression of pivotal oncogenes and tumor suppressors. While promising, translating these insights into effective therapies requires further investigation. By comprehensively understanding the influence of m6A on OC, there lies hope for developing improved diagnostic techniques and novel treatment strategies to combat this complex disease.
Keywords
Introduction
Ovarian cancer (OC) develops insidiously, with inconspicuous early symptoms, limited early screening methods, and a high propensity for recurrence and metastasis. Alarmingly, over 70% of OC cases remain undetected until they have progressed to advanced stages, generally stage III or IV. 1 The latest statistics indicate a 5-year survival rate of only 49.7% patients with OC. 2 Therefore, it is imperative to explore potential diagnostics, therapeutic targets, and biomarkers specific to OC by comprehensively studying its tumorigenic mechanisms, which may significantly improve the clinical outcomes of patients with OC.
Recent advancements in RNA epitranscriptomics have spearheaded the search for novel biomarkers, with numerous studies highlighting the significance of epigenetic dysregulation across various diseases, especially tumors. Among RNA modifications, N6-methyladenosine (m6A) stands out as one of the most prominent RNA modifications in the field of epitranscriptomics. While the initial descriptions of m6A date back to the 1970s,3,4 the specific functions and underlying mechanisms have only been elucidated in recent years.5,6 M6A is the most common endogenous methylation modification in eukaryotic RNAs, 7 preferentially occurring on the adenosine nucleotide at the nitrogen-6 position in the consensus motif ‘RRm6ACH’ (where R = G or A; H = A, C, or U).8-10 Since its initial discovery, m6A has been implicated in an extensive array of RNA metabolic processes, including RNA splicing, structural remodeling, nuclear export, translation, stability, and degradation. 11 These biological functions, as well as the dynamic reversible processes of m6A modification, are regulated by 3 proteins: “writers” (methyltransferases), “erasers” (demethylases), and “readers” (m6A-binding proteins).7,12 Owing to its profound impact on RNA metabolism, m6A is involved in various physiological and pathological processes, including immunity, 13 viral infections, 14 inflammation, 15 metabolism, 16 embryogenesis, 17 and cancer. 18 Currently, numerous studies have underscored the close association between m6A and the development and prognosis of malignant tumors. In this paper, we review the latest findings from m6A research in OC, as well as the mechanisms through which m6A regulates the proliferation, migration, invasion, and drug resistance of OC cells, while also summarizing its impact on the development and prognosis of OC.
Ovarian Cancer and “Writers”
Roles of Methyltransferase in Ovarian Cancer (OC).


Summary diagram illustrating m6A methyltransferase regulation in ovarian cancer (OC) development. The methyltransferase complex mediates m6A modification, with METTL3, METTL14, WTAP, and VIRMA independently influence the tumorigenesis, metastasis, and drug resistance of OC. METTL3 promotes OC progression by upregulating the expression of AXL, EIF3C, CSF-1, FZD-10, AKT, BIRC5, VGLL, RIPK4, pri-miR-1264, pri-miR-126-5p, lncRHPN1-AS1, and circPLPP4, while inhibiting the expression of IFFO1 and lncMEG3. WTAP stimulates the malignant biological behavior of OC by upregulating the expression of AKT, ERK, JNK, p38, FAM76A, HBS1L, and miR-200 and inhibiting EGR3 expression. METTL14 and VIRMA respectively act on TROAP and ENO1, exerting inhibitory and promoting effects on OC.
Ovarian cancer and METTL3
METTL3, the most critical component of the m6A methyltransferase complex, is a highly conserved S-adenosylmethionine (SAM)-binding protein. 20 Several studies have underscored the significance of METTL3 in the development and progression of various cancers. 21 In studies related to hepatocellular carcinoma and lung cancer, METTL3 has been identified as an oncogene that promotes tumor proliferation, migration, and colony-forming ability, thereby correlating with poor prognosis in cancer patients.22,23
The expression of METTL3 in OC tissues has been reported to be significantly higher than that in normal ovarian tissues. 24 Additionally, METTL3 has been identified as a regulator of m6A methylation in epithelial ovarian cancer (EOC), independent of METTL14 and WTAP. 25 Regarding the specific mechanism of METTL3 in OC development, various studies have proposed different explanations. For instance, Bi et al demonstrated that METTL3 could recognize the m6A modification site of pri-miR-1246, leading to the upregulation of miR-1246 expression and the inhibition of cyclin G2 (CCNG2), which accelerated the proliferation, migration, and invasion of OC cells while inhibiting their apoptosis, ultimately promoting OC progression. 26 Cui et al demonstrated that in cisplatin-resistant OC cells, METTL3 could enhance the stability of RHPN1-AS1 (lncRNA RHPN1 antisense RNA 1) through m6A modification, 27 which in turn promoted the expression of RHPN1-AS1, activated the PI3K/AKT signaling pathway, and subsequently enhanced cisplatin resistance in OC cells. Additionally, Wenfeng Hua et al reported that METTL3 could indirectly regulate AXL receptor tyrosine kinase (AXL), which plays a pro-oncogenic role in various cancers, including OC. 24 Likewise, Liang et al proposed that METTL3 promotes the malignant progression of OC by activating the AKT-related signaling pathway and thereby stimulating a series of downstream effectors, including p70S6K and Cyclin D1. 28 Moreover, Zhang et al experimentally validated that the low-level expression of oncogene intermediate filament family orphan 1 (IFFO1) in OC was modulated by METTL3/YTHDF2 participation. METTL3, when highly expressed, reduced the post-transcriptional stability of IFFO1, which in turn was degraded by YTHDF2. Consequently, the mechanisms of anti-tumor metastasis and IFFO1-mediated drug resistance reversal were inhibited. 29 Bi et al, conversely, inhibited the development of OC by knocking down METTL3 to suppress the upregulation of phosphatase and tensin homolog (PTEN) mediated by miR-126-5p (a key biomarker for several cancers), thereby preventing the activation of the PI3K/Akt/mTOR pathway. 30 Additionally, Fan et al reported an upregulation of baculoviral IAP repeat containing 5 (BIRC5), an inhibitor of the apoptosis (IAP) family protein, in cisplatin-resistant OC cell lines. They further showed that BIRC5 was upregulated and stabilized by METTL3-mediated m6A modification and IGF2BP1 binding, which in turn promoted platinum resistance in OC. 31 Furthermore, Yin et al demonstrated that the upregulation of receptor-interacting protein kinase 4 (RIPK4), which acts as an oncogene in OC, was facilitated by METTL3-mediated m6A modification. This process was accompanied by concurrent enhancement of RIPK4 mRNA stability mediated by YTHDF1, leading to NF-κB activation and subsequent tumor growth and cisplatin resistance. 32 Moreover, CircPLPP4 was found to be remarkably overexpressed in chemoresistant OC tissues and cells, with its increased levels regulated by METTL3/IGF2BP1 through modulation of its stability. 33 Similarly, vestigial-like 1 (VGLL1) was also found to be upregulated by the combined action of METTL3 and IGF2BP2 in an m6A-dependent manner, which in turn promoted OC proliferation and metastasis. 34 Additionally, a mechanistic study revealed that METTL3/YTHDF2-mediated methylation induces the degradation of the tumor suppressor lncRNA MEG3, which in turn releases the tumor suppression effect and subsequently promotes OC proliferation and metastasis through the miR-885-5p/VASH1 axis. 35 These findings collectively affirm that METTL3 is dysregulated in OC and affects multiple signaling pathways in an m6A-dependent manner, thus playing a pro-cancer role in OC.
Ovarian cancer and METTL14
METTL14 acts as a key component of the m6A methyltransferase complex and influences oncogene expression in tumors by modulating RNA stability and degradation.36-38 Interestingly, in breast cancer cells, METTL14 has been implicated in a pro-tumorigenic role, where it regulates the m6A methylation level of key mRNAs involved in the epithelial-to-mesenchymal transition (EMT). However, its expression is downregulated in various tumors, including hepatocellular carcinoma, bladder cancer, endometrial carcinoma, and glioblastoma, where it functions as a tumor suppressor.39,40 Moreover, in digestive malignant tumors, METTL14 has been observed to play a contradictory role in the regulation of digestive system tumors, 41 with similar findings reported in the studies related to OC. Trophinin-associated protein (TROAP, also known as TASTIN) has been implicated in the proliferation, invasion, and migration processes of various cancers. 42 Li et al demonstrated that METTL14 negatively regulates the expression of TROAP in an m6A-dependent manner, thereby inhibiting OC cell proliferation. 43 However, in another retrospective study, Wei et al proposed a different hypothesis suggesting that high METTL14 expression elevates the level of m6A modification in OC cells, thereby promoting their proliferation, invasion, and migration. 44 Presently, there are limited studies on the association of METTL14 with OC; therefore, further exploration is needed to elucidate the multiple molecular mechanisms underlying the effect of METTL14 on the malignant biological behaviors of OC and the potential theoretical rationale for targeted therapies.
Ovarian cancer and WTAP
WTAP plays a crucial role in localizing the methyltransferases METTL3–METTL14 to nuclear speckles and engages in post-translational regulation, including binding to the 3′-untranslated region (UTR) to enhance mRNA stability and acting as a spliceosome for selective splicing of precursor mRNAs.45,46 Similar to METTL3, WTAP is highly expressed in high-grade serous OC tissues, plays pro-proliferative, migratory, and anti-apoptotic roles, and is associated with patient prognosis and survival. 47 Wang et al confirmed a significant positive correlation between WTAP expression and the expression of Family with sequence similarity 76 member A (FAM76A) and HBS1-like translational GTPase (HBS1L). They hypothesized that WTAP mediates the m6A modification of FAM76A and HBS1L, thereby synergizing with positive transcripts to promote their stability, 48 which in turn promotes OC progression. Additionally, Lyu et al reported that WTAP drives the malignant progression of OC by promoting the maturation of miR-200, which subsequently affects hexokinase 2 (HK2), a key gene in the glycolytic pathway. 49 Furthermore, Fu et al proposed that highly expressed WTAP promotes aberrant mRNA methylation, inhibiting the transcription and translation of the target gene early growth response 3 (EGR3), and should be considered a potential marker for OC subtype diagnosis. 50
Ovarian cancer and VIRMA
VIRMA, also known as KIAA1429, is a recently identified m6A methyltransferase that is highly amplified or mutated in various cancer types, indicating its potential involvement in cancer development. Studies have shown that VIRMA is transcriptionally activated by SPI1, highly expressed in OC, and enhances the stability of enolase1 (ENO1) mRNA in an m6A-dependent manner, thereby promoting aerobic glycolysis and contributing to OC development. 51
Ovarian Cancer and “Readers”
Roles of RNA-binding Proteins in Ovarian Cancer (OC).

Summary diagram illustrating m6A-binding protein regulation in ovarian cancer (OC) development. M6A-modified RNAs interact with m6A-binding proteins, exerting various biological functions. This review primarily summarizes the research on YTHDF1, YTHDF2, IGF2BP1, IGF2BP2, and HNRNPA2B1 in OC. YTHDF1 enhances EIF3C and TRIM29 translation and stabilizes RIPK4 expression, while YTHDF2 promotes BMF expression and suppresses miR-145 and lncMEG3, facilitating OC progression. IGF2BP1 and IGF2BP2 promote SRF, BIRC5, SNAI1, VGLL1, circPLPP4, and circNFIX expression, promoting the malignant development of OC. However, that the context-dependent regulation of OC by IGF2BP1 is exemplified by its positive modulation of lncUBA6-AS1, which exerts anticancer effects. HNRNPA2B1 regulates CDK19 expression, further promoting OC progression.
Ovarian cancer and YTH domain family
YTHDF1–3 are highly homologous proteins that recognize and bind to m6A modification sites through the C-terminal YTH domain. However, they diverge in their main functions: YTHDF1 interacts with translation initiation factors, inducing target mRNA translation; YTHDF2 affects target mRNA stability; and YTHDF3 modulates translation and decay of m6A-modified mRNAs by interacting with YTHDF1 and YTHDF2. 54 Liu’s team showed that YTHDF1 promoted ovarian carcinogenesis and metastasis by binding to the m6A-modified protein translation initiation factor EIF3C mRNA, thereby enhancing EIF3C translation in an m6A-dependent manner, ultimately affecting total protein translation. 55 Hao et al demonstrated that YTHDF1 is recruited by m6A-modified tripartite motif protein 29 (TRIM29) and promotes TRIM29 translation in cisplatin-resistant OC cells. Knockdown of YTHDF1 in these cells suppresses their cancer stem cell (CSC)-like features, which can be reversed by TRIM29 overexpression, suggesting a dependency on m6A-YTHDF1 for its oncogenic role. 56 Furthermore, Xu et al revealed the clinical significance of a novel regulatory axis involving the tumor suppressor F-box and WD repeat domain-containing 7 (FBW7), which mediates the ubiquitination and subsequent proteasomal degradation of multiple oncogenic proteins, in OC. This regulatory axis is known as the FBW7-YTHDF2-BMF (Bcl-2 modifying factor) axis. FBW7 antagonizes YTHDF2, leading to the inhibition of its destabilizing effect on the m6A-modified pro-apoptotic BMF mRNA, which ensures the stable expression of BMF mRNA, consequently hindering OC growth and progression. 57 Li et al found that YTHDF2 expression was significantly up-regulated in OC, where it disrupted miR-145 via a bidirectional negative feedback loop, promoting the proliferation and migration of OC cells. 58 These findings underscore the multifaceted role of the YTH domain family in driving the malignant progression of OC through multiple mechanisms. However, significant uncharted territory remains in the exploration of the role of YTH domain family in OC.
Ovarian cancer and IGF2BPs
The IGF2BP family exhibits high structural similarity and plays crucial roles in regulating mRNA stability, translation, and localization. 59 Upregulation of IGF2BP proteins has been associated with poor prognosis in malignant tumors such as breast, colorectal, glioma, hepatocellular, and pancreatic cancers. Specifically, IGF2BP1 recognizes and binds to m6A-modified serum response factor (SRF) mRNA, thereby enhancing its transcriptional activation and maintaining the expression of various IGF2BP1-SRF target genes, which in turn contributes to tumor growth and invasion, while also promoting CSC-like tumor cell characteristics. Müller et al observed the role of this mechanism in ovarian, hepatocellular, and lung cancers, correlating with poor prognosis and low overall survival. 60 Wang et al found that an OC-inhibiting lncRNA called ubiquitin-like modifier-activating enzyme 6 antisense RNA 1 (UBA6-AS1) increased the m6A level of its target UBA6 mRNA by recruiting RBM15. They further noted an increase in the stability of UBA6-AS1 upon IGF2BP1 binding, which in turn suppressed the malignant phenotype of OC. 61 Additionally, Sun et al demonstrated that IGF2BP2 recognized and bound to m6A modification sites, promoting mRNA stability, and that FTO inhibited SNAI1 mRNA expression in an m6A-IGF2BP2-dependent manner, thereby attenuating OC growth and metastasis. 62 Furthermore, M6A-circNFIX, which is highly expressed in OC under the coregulation of IGF2BP1/2/3, up-regulates the expression of PD-L1 and activates the JAK1/STAT3 pathway via circNFIX/miR-647/IL-6R, thereby promoting OC proliferation, metastasis, and immune escape. 63 The aforementioned studies suggest that IGF2BPs contribute to OC progression primarily through their roles in promoting the transcriptional activity of target mRNAs and enhancing their stability.
Ovarian cancer and HNRNPs
The HNRNP family differs from the aforementioned “readers” in that these proteins do not exclusively recognize m6A-modified sites through direct binding. For example, HNRNPC and HNRNPG preferentially bind to m6A-modified RNAs through an “m6A-switch” mechanism. 64 HNRNPA2B1 is highly expressed in OC tissues, garnering attention for its role as an oncogene in OC development. Chen et al identified HNRNPA2B1 as a potential target of miR-30c-5p in OC. They found that miR-30c-5p inhibition led to decreased CDK19 mRNA stability and reduced m6A levels, thereby inhibiting OC progression. 65
Ovarian Cancer and “Erasers”
Roles of Demethylases in Ovarian Cancer (OC).

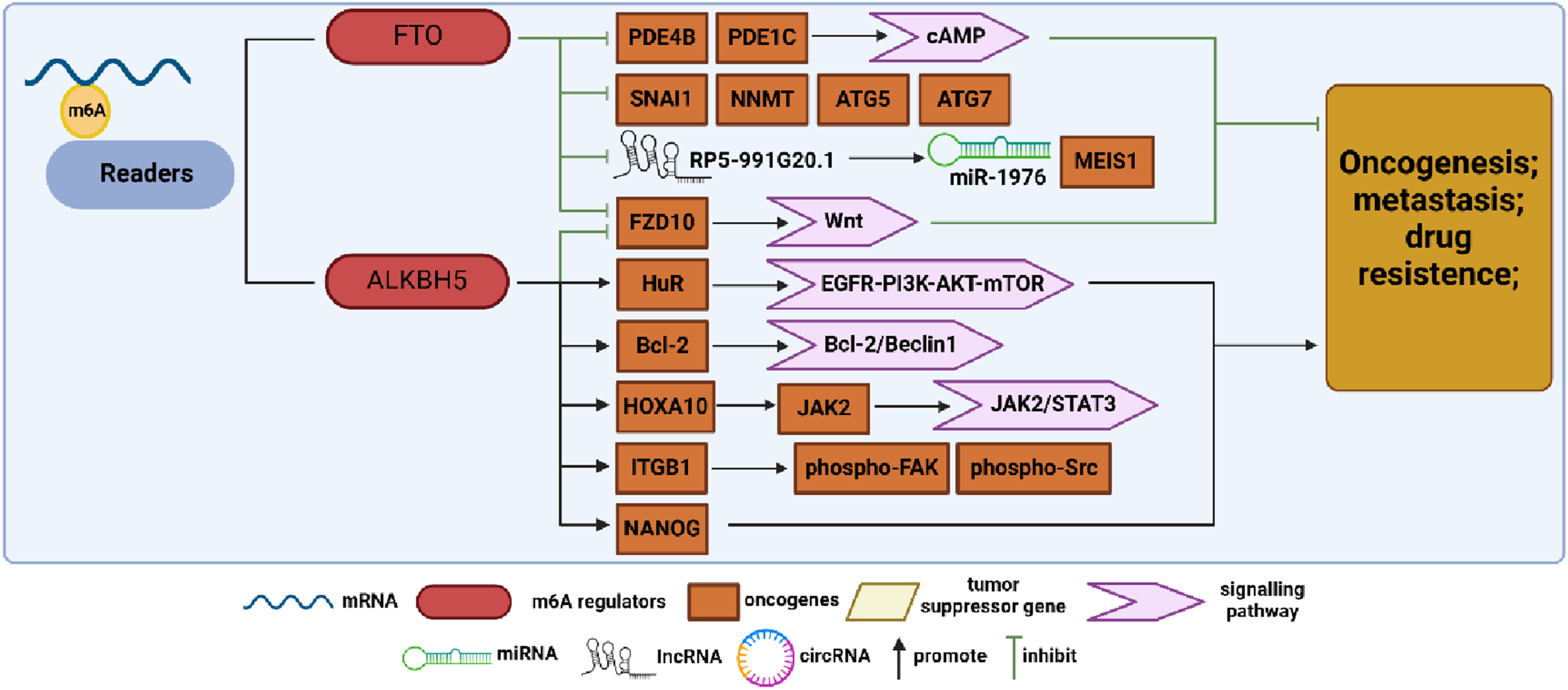
Summary diagram illustrating m6A demethylase regulation in ovarian cancer (OC) development. FTO and ALKBH5 are key demethylases that modulate biological functions by removing m6A modifications. In OC research, FTO acts as a tumor suppressor gene, inhibiting the expression of genes, such as PDE4B, PDE1C, SNAI1, NMMT, ATG5, ATG7, FZD10, and lncRP5-991G20.1, by eliminating m6A modifications, thus suppressing OC progression. Conversely, ALKBH5 exhibits contrasting effects, promoting OC progression by upregulating the expression of HuR, Bcl-2, JAK2, ITGB1, and NANOG.
Ovarian cancer and FTO
Initially identified for its involvement in adipogenesis and obesity, 67 FTO was later found to have demethylase activity. As the first m6A demethylase discovered, FTO is highly expressed and functions as an oncogene in various malignant tumors such as acute myeloid leukemia, pancreatic cancer, hepatocellular carcinoma, and non-small-cell lung cancer (NSCLC).68-70 However, this was not exactly the case in OC. Huang et al demonstrated that FTO acts as an OC suppressor and inhibits the CSC-like features of OC. They further identified 2 phosphodiesterase genes, PDE4B and PDE1C, as downstream FTO targets that regulate the cAMP signaling pathway and play a key role in maintaining ovarian CSC-like features. 71 Based on previous studies showing the involvement of m6A in autophagy, Zhang et al found that CircRAB11FIP1 binds to and upregulates FTO mRNA expression, thereby modulating the m6A methylation levels of ATG5 and ATG7 mRNA through FTO. This mechanism ultimately enhances autophagy and malignant behavior in OC. 72 Additionally, Sun et al demonstrated that decreased FTO expression elevates m6A levels in SNAI1 mRNA, upregulating SNAI1 expression and consequently promoting OC proliferation, invasion, and migration. 62 Furthermore, Huang et al revealed that FTO overexpression enhances OC sensitivity to platinum and induces DNA double-strand breaks, with nicotinamide N-methyltransferase (NNMT) emerging as a novel FTO target regulating OC response to platinum in an m6A-dependent manner. 73 Tumor-associated long non-coding RNAs (lncRNAs) modulated by epigenetic modification switches play pivotal roles in immune evasion and chemoresistance in OC. Chen et al developed a prognostic model integration methylation-associated and immune-associated lncRNAs, revealing that FTO inhibits OC proliferation and drug resistance by regulating the RP5-991G20.1/hsa-miR-1976/MEIS1 signaling pathway. 74 Several studies have confirmed FTO’s role as a suppressor influencing various malignant behaviors in OC, including proliferation, metastasis, autophagy, and drug resistance, through diverse ontogenetic mechanisms.
Ovarian cancer and ALKBH5
ALKBH5, another significant m6A demethylase, modulates various biological processes, such as nuclear RNA export, metabolism, and gene expression, with its involvement in different RNA interactions playing distinct roles across various cancer types. 75 In ovarian cancer, Zhu et al identified ALKBH5 overexpression in tumor tissues, correlating with cancer progression through the inhibition of autophagy in OC cells. 76 In contrast, ALKBH5, in conjunction with FTO, contributes to PARP inhibitor resistance in BRCA1/2 mutant OC cells by regulating frizzled class receptor 10 (FZD10) mRNA expression and mediating the Wnt signaling pathway. 77 Liu et al observed upregulated ALKBH5 expression in cisplatin-resistant OC, leading to enhanced OC cell proliferation and cisplatin resistance both in vivo and in vitro. They identified homeobox A10 (HOXA10) as a transcription factor promoting ALKBH5 transcription, reciprocally regulated by ALKBH5. Further analysis revealed that the sustained upregulation of ALKBH5-HOXA10 promoted OC tumor growth and cisplatin resistance via m6A-dependent activation of the JAK2/STAT3 signaling pathway. 78 Sun et al found that hypoxia could stimulate the expression of hypoxia inducible factor 1 subunit alpha (HIF-1α) and ALKBH5. They observed that ALKBH5 regulated the expression of integrin subunit beta 1 (ITGB1) in an m6A-YTHDF2-dependent manner, which triggered the phosphorylation of focal adhesion kinase (FAK), thus promoting OC lymphangiogenesis and metastasis. 79 In the field of immunology, Jiang et al established an in vitro model by co-culturing M1 and M2 macrophages with OC cells and found that this co-culture significantly increased tumor cell invasiveness. Additionally, they observed that Toll-like receptor 4 (TLR4) upregulated the expression of ALKBH5 in M2 co-cultured group through activation of the NF-κB signaling pathway. ALKBH5, in turn, promoted OC development by demethylating Nanog homeobox (NANOG). 80
M6A Regulators and Ovarian Cancer Prognosis
Since m6A modification is involved in OC tumorigenesis and progression through multiple mechanisms, m6A methylation regulators play a crucial role in tumor prognosis. Fan et al identified IGF2BP1, VIRMA, and ZC3H13 as the most prognostically predictive m6A-associated regulatory proteins, as evidenced by risk model construction. Gene Set Enrichment Analysis (GSEA) further supported the prognostic significance of these proteins by associating them with cancer pathways and the Wnt signaling pathway, thus corroborating their prognostic value. 81 Li et al analyzed the prognostic value of each m6A-modified regulatory factor, identifying VIRMA, IGF2BP1, and HNRNPA2B1 through the Lasso Cox regression analysis and constructed a risk scoring model based on the above 3 factors. Additionally, they constructed a regulatory network of miRNAs, m6A regulators, and m6A target genes and conducted preliminary experimental validation of the miR-196b-5p-IGF2BP1-PTEN pathway within this network, with the results providing some degree of reliability for their constructed regulatory network. 82 Zhang et al analyzed the correlation between each m6A regulator and clinical characteristics of OC (including age, grade, stage, and tumor status). Using univariate and multivariate Cox regression analyses, they identified HNRNPA2B1, KIAA1429, and WTAP as significantly linked to overall survival, leading to the construction of a risk scoring model. Additionally, they observed a correlation between immune infiltration and these 3 m6A regulators. 83 Using the TCGA database, Han et al conducted a bioinformatics analysis, revealing that nearly all OC patients (99.31%) exhibited mutations or copy number variants (CNVs) in at least one m6A-regulated gene, with high WTAP expression correlating significantly with poorer prognosis. 84 Using GEO dataset, Wang et al analyzed the correlation between the altered expression levels of multiple m6A regulators and the prognostic regression of OC, emphasizing the potential involvement of m6A regulators (RBM15 B, ZC3H13, YTHDF1, and IGF2BP1) in immune cell infiltration and immune evasion in OC. 85 Conversely, Tan et al analyzed various OC datasets from TCGA and identified 7 m6A regulators—CBLL1, FTO, HNRNPC, METTL3, METTL14, WTAP, ZC3H13, RBM15B, and YTHDC2—associated with poorer overall survival in OC. They also identified optimal populations that might benefit from immunotherapy. 86 Furthermore, using the TCGA database, Zhu et al found that high expression of KIAA1429 and YTHDC2 correlated with poorer prognosis in OC. They constructed a prognostic model based on 5 m6A RNA methylation regulators in OC (KIAA1429, YTHDC2, ZC3H13, WTAP, and FTO) that are highly expressed in OC. 87
Roles of m6A Regulators in Ovarian Cancer (OC) Prognosis.

Ovarian Cancer and Potential m6A-associated Targeted Drugs
As m6A research continues to advance, there is a burgeoning interest in exploring the therapeutic potential of pharmacological agents targeting m6A-associated regulatory factors for cancer treatment. However, current research into m6A-targeted drugs remains in its infancy, primarily focusing on cancers other than OC.
As the core component of the m6A methyltransferase complex, METTL3 plays a crucial role in tumorigenesis and development, making it a promising therapeutic target for anticancer drugs. Yankova et al reported the first highly potent and selective inhibitor of METTL3, named STM2457, demonstrating its anti-leukemic efficacy in vivo. 88 Furthermore, a newly developed oral small-molecule METTL3 inhibitor named STC-15 has received IND approval for its Phase 1 clinical study in cancer patients, which began in November 2022. 89 In a recent study, orthotopic NAFLD-HCC tumors were treated with STM2457 in vivo, and it was found that STM2457 significantly inhibited tumor growth and synergized with anti-PD-1 therapy, leading to a strong induction of IFN-γ+ and GZMB+ CD8+ T cell infiltration. 90 Adenosine, a SAM-competitive inhibitor of METTL3, has been identified as one of the 2 moieties of SAM. 91 Moroz-Omori et al characterized a nanomolar inhibitor of METTL3 (UZH1a) by screening an adenine-based library and performing a homogenous time-resolved fluorescence (HTRF) enzyme inhibition assay. Further in vitro validation showed a dose‐dependent reduction in m6A methylation levels. 92 In NSCLC, chidamide decreases the stability and translational efficiency of WTAP and METTL3 mRNAs, thereby downregulating c-MET expression and enhancing NSCLC cell sensitivity to crizotinib. 93
Inhibitors targeting m6A “readers” have limited coverage. For example, salvianolic acid C (SAC), a small-molecule inhibitor of YTHDF1, has been reported to reverse fragile X syndrome, 94 but no YTHDF1 inhibitors for cancer therapy have been described yet. Additionally, Wang et al reported that higher levels of IGF2BP2 in patients with AML, particularly in leukemic stem cells/leukemic initiating cells (LSCs/LICs), are associated with poorer prognosis. Subsequently, they identified a compound, CWI1-2, which directly interacts with IGF2BP2 and shows promising anti-leukemic efficacy both in laboratory and animal models. Remarkably, CWI1-2 achieves this therapeutic effect by inhibiting IGF2BP2’s m6A reader activity, with minimal adverse effects. 95 In the field of OC research, the availability of targeted drugs against m6A regulators is limited. Chang Su reported that AE-848, a small molecule targeting IGF2BP3, could be an effective treatment for OC. Through a subcutaneous tumor model, AE-848 has been validated to inhibit tumor growth by reducing the expression of tumor-associated antigens (c-MYC/VEGF/Ki67/CDK2) and enhancing the anti-tumor effect of macrophages. 96
Among the many m6A regulators, FTO stands out as the most promising target for inhibitor development. Over 10 FTO inhibitors have been discovered to date, with their effectiveness validated in various types of tumors. One early-identified FTO inhibitor, meclofenamic acid (MA), exhibits high selectivity by competing for FTO binding sites. 97 Its ethyl ester derivative, MA2, has been shown to suppress glioblastoma stem cell (GSC) growth and self-renewal in vitro, along with inhibiting tumor growth in vivo. Moreover, the combination of MA2 with the chemotherapy drug temozolomide (TMZ) has shown a synergistic effect in suppressing glioma cells. 98 In 2022, Liu et al encapsulated MA into γ-cyclodextrin cavity and developed a photothermal immunotherapy based on gold nanoparticles. This MA-nanodrug specifically targeted prostate tumor cells and selectively accumulated at the tumor site in vivo by increasing CD8+ T cell infiltration while inactivating PD-L1. 99 FB23-2, an improved selective FTO inhibitor synthesized based on MA, exhibits superior anti-proliferation activity and promotes myeloid differentiation and apoptosis in acute myeloid leukemia (AML) by regulating the expression of RARA and ASB2. The patient-derived xeno-transplantation (PDX) AML mouse model further shows its safety and therapeutic efficacy in vivo. 100 Additionally, in a recent study on hepatocellular carcinoma (HCC), FB23-2 was loaded into a nanocarrier for targeted release into tumor-infiltrating dendritic cells (TIDCs) along with tumor-associated antigens (TAA) to investigate its efficacy in HCC. Findings from the in vivo study showed that intratumoral injection of the nanodrug promoted the maturation of dendritic cells (DCs), improved tumor infiltration of effector T cells, and generated immune memory, synergistically inhibiting distant tumor growth and lung metastasis with immune-checkpoint blockade (ICB) therapy. 101 Su et al identified CS1 and CS2 as efficacious FTO inhibitors, exhibiting potent anti-leukemic efficacy by enhancing AML cell sensitivity to T cell cytotoxicity and overcoming immune evasion. 102 Epigallocatechin gallate, a tea flavonoid, exhibits powerful antioxidant, anti-inflammatory, and anticancer effects, likely attributed to the m6A-dependent regulation of cyclin A2 and CDK2 through the downregulation of FTO and upregulation of YTHDF2. 103 FTO-04, which was developed based on the mechanistic structure of FTO, has been found to significantly inhibit patient-derived GSC neurosphere formation without affecting healthy human neural stem cell growth. 104 Dac51, a more potent FTO inhibitor developed by the same group that developed FB23-2, shares a similar mechanism with CS1 and CS2. Dac51 treatment increases CD8+ T cell infiltration and synergizes with anti-PD-L1 blockade. 105 Additionally, Malacrida et al revealed that imidazobenzoxazin-5-thione MV1035, initially synthesized as a sodium channel blocker, exhibits ALKBH5 inhibition and increases m6A levels through off-target interactions and further suppresses U87 glioblastoma cell migration and invasiveness. 106 Another specific inhibitor of ALKBH5, named ALK-04, was found to enhance anti-PD-1 treatment through promotion of immune cell accumulation in the tumor microenvironment. 107
In summary, m6A-targeted drugs show significant feasibility and promise in cancer therapy. Moreover, the aforementioned drug studies indicate that targeting m6A regulatory factors may mediate immune infiltration and potentially synergize with immune checkpoint inhibitor (ICI) immunotherapy. However, current research data are limited, with most targeted drug studies primarily focusing on glioma, AML, and HCC, and notably scarce research on other tumors, including OC. This underscores the need for further exploration in this area as it presents a promising avenue for further exploration.
Discussion and Perspectives
OC ranks third among gynecological tumors but has the highest mortality rate. Owing to the absence of typical early clinical symptoms, most patients (70%) are diagnosed at advanced stages. Currently, the treatment to OC primarily relies on surgery, complemented by radiotherapy, chemotherapy, targeted therapy, and other therapeutic means, yet long-term survival rates remain largely unchanged. 108 Therefore, the urgent focus of current OC research is to identify more accurate early diagnostic markers and therapeutic targets. In recent years, that the significance of m6A modification, one of the most prevalent RNA modifications in eukaryotes, has emerged in post-transcriptional regulation of gene expression. Notably, m6A-related regulators are implicated in the progression and prognosis of various tumors. Current studies have highlighted m6A as a “double-edged sword”, regulating oncogenes or suppressor genes through various methyltransferases, m6A binding proteins, and demethylases, which are involved in the regulation of growth, proliferation, migration, invasion, metastasis, drug resistance, and prognosis of OC.
The majority of m6A studies have focused on its regulatory factors in various aspects of OC, including proliferation, metastasis, and drug resistance. Our review consolidates the specific mechanisms of action of diverse m6A-associated regulatory factors in these areas (Figure 4). Among them, METTL3 and WTAP facilitate m6A modification by promoting the expression of oncogenic mRNAs or non-coding RNAs (ncRNAs), while concurrently suppressing the expression of tumor suppressor genes, ultimately promoting the proliferation and metastasis of OC. However, METTL14 has been observed to impede OC proliferation by reducing the stability of TROAP mRNA. Yet, research on METTL14 remains limited, and the generalizability of its findings warrants further verification. Another less-explored methyltransferase, VIRMA, activated by SPI1, enhances ENO1 mRNA stability in an m6A-dependent manner, thereby contributing to proliferation, migration, and invasion effects in OC. In the “readers” section, we provide an overview of the specific mechanisms of various m6A-binding proteins, such as YTHDF1, YTHDF2, IGF2BP1, IGF2BP2, and HNRNPA2B1, in tumor proliferation and metastasis. We elucidated these mechanisms from perspectives such as mRNA, ncRNAs, and downstream signaling pathways. In “erasers”, the primary subjects of research concerning metastasis and proliferation are FTO and ALKBH5, with both exhibiting contrasting roles in OC development. FTO inhibits OC migration and proliferation by removing m6A modifications, thereby stabilizing the expression of PDE4B and PDE1C. Conversely, ALKBH5 enhances OC proliferation, migration, and invasion through multiple signaling pathways. Although research on drug resistance is limited, the overall roles of these regulatory factors appear to parallel their functions in proliferation and metastasis. METTL3-mediated m6A modification upregulates RIPK4, with YTHDF1 enhancing RIPK4 mRNA stability, subsequently activating the NF-κB signaling pathway to confer OC resistance to cisplatin. Additionally, METTL3 and IGF2BP1 regulate BIRC5 and circPLPP4 expression and stability through m6A modification, thereby promoting OC resistance to cisplatin. Conversely, downregulation of FTO and ALKBH5 increases m6A modifications in FZD10 mRNA, leading to upregulation of the Wnt signaling pathway and reduced resistance to PARPi, ultimately inhibiting PARPi resistance in OC. The synergistic effects between these complexes play a pivotal role in drug resistance. Furthermore, there is a significant scarcity of cross-research between m6A-associated regulatory factors and other phenotypes of OC, such as immune evasion, immune infiltration, autophagy, metastasis, and others. Considering the potential significance of these areas for clinical treatment, we advocate for continued exploration of these areas in future studies. Summary diagram illustrating the effects of m6A-related regulatory factors on various clinical phenotypes of ovarian cancer (OC). This review categorizes and summarizes experimental studies, identifying 4 main clinical phenotypes of OC regulated by m6A modifications: proliferation, metastasis, drug resistance, and others. Most studies indicate the roles of m6A-related regulatory factors in regulating OC proliferation and metastasis. Apart from FTO and METTL14, most factors promote these processes. Several studies have elucidated the regulatory mechanisms of METTL3, IGF2BP1, YTHDF1, FTO, and ALKBH5 in OC drug resistance, with conclusions largely parallel to those regarding proliferation and metastasis. However, research on immune-related phenotypes, metabolism, autophagy, and other aspects remains limited. Only IGF2BP1 has been reported to promote immune evasion by upregulating circNFIX, while circRAB11FIP1 promotes autophagy through FTO-mediated upregulation of ATG5 and ATG7.
Overall, methyltransferases, including METTL3, WTAP, VIRMA, and m6A-binding proteins YTHDFs consistently play pro-carcinogenic roles in OC, whereas the demethylase FTO is downregulated and plays a cancer-suppressing role. However, the roles of other regulators in OC show pleiotropy and contradiction, potentially attributed to the differences in upstream and downstream signaling pathways involved in, as well as the complex tumor microenvironment. This paper suggests that further exploration is needed to elucidate the mechanisms underlying this paradoxical phenomenon. Furthermore, the complexity and diversity of m6A modification regulators and related mechanisms have rendered many m6A regulators in OC largely unexplored territory. The heterogeneity and complexity of m6A modification in OC, including the potential synergistic or antagonistic effects of different regulators on the carcinogenesis, warrant further investigation. Beyond mechanistic studies, greater emphasis on the clinical translation of m6A by researchers is also essential.
Early diagnosis has always been a challenge in OC management, and there is a pressing need to explore the potential of m6A-related regulators as biomarkers for early OC detection. While several risk scoring models indicate the potential of m6A-related regulators for prognostic assessment, they are primarily based on analyses of public databases, lacking validation through large-scale, multi-center experiments and clinical trials. Addressing these shortcomings will be crucial in future studies. Meanwhile, certain studies have indicated correlations between m6A-related regulators and OC immune cell infiltration, immune evasion, and immune response, which provides feasibility and theoretical support for immunotherapy in OC. We believe that future studies will achieve significant breakthroughs in this field.
Another significant challenge lies in devising effective treatment strategies for OC, where m6A-targeted therapy holds promise. Currently, METTL3 inhibitors stand out as the sole m6A-targeted drugs in clinical trial stages, indicating their potential as the most promising therapeutic targets, with their research progress and efficacy worth anticipating. While research on FTO inhibitors is relatively extensive, with significant anti-cancer effects demonstrated in both in vitro and in vivo experiments uncertainties prevail. As described in the preceding section on FTO in this paper, its anti-cancer effect in OC is ultimately achieved by reducing the m6A RNA levels. Therefore, whether FTO inhibitors might have a contrary effect in OC and whether transitioning to FTO activators is necessary for effective treatment in OC remain unknown and warrant confirmation in future research. Overall, a wide array of m6A regulatory factors play extensive roles in the regulation of gene expression and signaling pathways, influencing various pathological and physiological processes in the human body. However, given that current research findings are predominantly derived from animal experiments, the safety and potential side effects of m6A-targeted therapy in humans remain uncertain and warrant further evaluation. Consequently, future clinical trials must prioritize rigorous testing and measures to mitigate the risk of in vivo side effects associated with m6A-targeted drugs.
Conclusion
Our review delves into the advancements of m6A RNA modification in OC, elucidating the impact of methyltransferases, m6A-binding proteins, and demethylases on tumor proliferation, metastasis, and drug resistance. These insights underscore the potential significance of m6A modification in the diagnosis, prognostic assessment, and treatment of OC. Investigating m6A regulatory factors as potential molecular targets present a novel approach to addressing this complex malignancy. The development of effective targeting RNA regulatory factors may offer novel therapeutic avenues for managing OC and overcoming recurrent drug resistance in the future.
Footnotes
Author Contributions
All authors contributed to study conception and design. YHZ, YFL, YZ and XS were responsible for material preparation, reference collection and interpretation. YHZ and YFL wrote the first draft of the manuscript. FRS, JHZ and YGC read and revised the format. YZG, FY and JW reviewed and revised the manuscript. JW supervised the project. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported by the grants from the Project of Suzhou Science and Technology Development Plan (No. SLJ202006), the Project of Jiangsu Health Development Research Center (No. JSHD2022067), as well as the Fundamental Research Funds for Soochow University (No. H230337).
