Abstract
Introduction
This study aimed to evaluate the therapeutic effect of osimertinib and further to compare the results of osimertinib plus brain radiation vs. osimertinib monotherapy in advanced EGFR-mutant non-small cell lung cancer (NSCLC) patients with brain metastases (BMs).
Methods
A retrospective study was conducted involving 62 advanced EGFR-mutant NSCLC patients with BMs who were treated with first-line osimertinib at the Vietnam National Cancer Hospital between April 2019 and December 2023. Patients were categorised in two treatment groups: (1) osimertinib alone (33 patients) and (2) osimertinib combined with locoregional therapy, including stereotactic radiosurgery or whole-brain radiotherapy (29 patients). Endpoints included objective response rate (ORR), central nervous system response rate (CNS-ORR), progression-free survival (PFS), overall survival (OS).
Results
The systemic ORR was 91.9% and the disease-control rate (DCR) was 96.8%. The CNS-ORR was 91.9% and the CNS-DCR was 100%. The median PFS and median OS achieved were 24.5 and 35.2 months, respectively. There was no significant difference in outcomes between patients in either treatment group with respect to CNS-ORR ( P = 1.0), mean best percentage change from baseline in CNS target lesion size (P = .376), median PFS (P = .656), intracranial progression-free survival (iPFS) (P = .706), or OS (P = .734). The occurrence of any-grade adverse events (AEs) did not differ significantly between the two treatment groups (P = .762). However, in the osimertinib plus brain radiation cohort, 3/29 (10.3%) patients experienced radiotherapy-related AEs (2 cases of brain necrosis, 1 case of leukoencephalopathy), which consisted of one case of grade 3 brain radiation necrosis.
Conclusion
Osimertinib shows favorable real-world outcomes in improving PFS, OS, and CNS-ORR in advanced EGFR-mutant NSCLC Vietnamese patients with BMs, with no clear additional benefit from combining with brain radiotherapy.
Introduction
Central nervous system (CNS) metastasis is a serious and common complication in patients with non-small cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) mutations. At the time of diagnosis, approximately 24.4% of these patients present with brain metastases, and this figure rises to 46.7% within three years. 1 Given the detrimental impact of brain metastases on quality of life and overall survival, 2 optimizing treatment strategies remains a critical challenge.
EGFR tyrosine kinase inhibitors (EGFR-TKIs) have become the cornerstone of therapy for advanced NSCLC with EGFR mutations. 3 Among these, osimertinib — a third-generation EGFR-TKI — is currently the preferred first-line agent, offering improved progression-free survival (PFS) and enhanced CNS activity compared to first-generation EGFR-TKIs.4,5 However, access to osimertinib remains limited in many developing countries, including Vietnam, primarily due to its high cost and lack of reimbursement under the Health Insurance Fund. Consequently, real-world data on the efficacy of osimertinib in Vietnamese patients are still scarce.
In addition, local treatments such as surgery and radiotherapy now play an important role in controlling brain metastases. However, with the advent of systemic therapies that exhibit strong intracranial efficacy, the management paradigm for brain metastases in lung cancer has evolved to incorporate both systemic and local approaches. In the specific context of EGFR-mutant NSCLC, osimertinib has demonstrated notable blood-brain barrier penetration and excellent intracranial activity.5,6 Furthermore, local therapies such as radiotherapy carry a risk of treatment-related complications.7,8 The role of upfront cranial radiation in patients initiating osimertinib therapy remains an area of ongoing investigation.
In this study, we conducted a retrospective analysis at the Vietnam National Cancer Hospital to evaluate the therapeutic effectiveness of osimertinib in a real-world clinical setting. Furthermore, we aimed to compare clinical outcomes between patients received osimertinib monotherapy and those receiving osimertinib combined with cranial irradiation among NSCLC patients harboring EGFR-activating mutations and brain metastases.
Materials and Methods
Patients
We conducted a retrospective review of consecutive patients with EGFR-mutated advanced NSCLC who were initially diagnosed with brain metastases (BMs) and received first-line osimertinib treatment at the Vietnam National Cancer Hospital (Hanoi, Vietnam) between April 2019 and December 2023. This study followed the STROBE reporting guidelines. 9 The inclusion criteria were: (1) histologically confirmed diagnosis of NSCLC; (2) presence of EGFR exon 19 deletion (Ex19del), exon 21 Leu858Arg (Ex21 L858R) mutations, or other uncommon EGFR alterations; (3) brain metastases confirmed by magnetic resonance imaging (MRI) or computed tomography (CT), or cytological/radiographic evidence of leptomeningeal metastases (LMs); (4) initiation of osimertinib as first-line systemic therapy for metastatic disease. The cutoff date for data collection was December 2024.
Treatment Protocol
Patients administered osimertinib at a daily dose of 80 mg as first-line therapy. Management of brain metastases was decided by a tumor board comprising medical oncologists, neurosurgeons, radiotherapists, and radiologists. Treatment options included osimertinib monotherapy or osimertinib combined with locoregional interventions such as stereotactic radiosurgery (SRS), whole-brain radiation therapy (WBRT), or surgery. Notably, gamma knife radiosurgery was employed as the specific technique for SRS in this study. No patients underwent brain metastasis surgery; therefore, we categorized patients into two groups: osimertinib monotherapy (n = 33) and osimertinib combined with brain-directed therapies (SRS or WBRT) (n = 29).
Data Collection
All patient details were de-identified. Baseline characteristics collected included age, sex, smoking status, performance status (PS), number and size of brain metastases, neurologic symptoms, and graded prognostic assessment (GPA) scores. We also recorded the dates of initial cancer diagnosis, diagnosis of brain metastases, start and end of osimertinib treatment, locoregional brain treatment modalities, systemic and CNS progression, last follow-up, and death (if applicable).
Systemic response was evaluated by CT scans every 2-3 months using RECIST version 1.1 criteria. Intracranial response was assessed by contrast-enhanced MRI or CT every 6 weeks to 6 months according to the Response Assessment in Neuro-Oncology Brain Metastases (RANO-BM) and Leptomeningeal Metastases (RANO-LM) criteria. PFS was defined as the interval from osimertinib initiation to systemic progression or death. Intracranial PFS (iPFS) referred to the time from osimertinib initiation to documented intracranial progression or death. Overall survival (OS) was calculated from the start of osimertinib to death or last known follow-up. Adverse events (AEs) were assessed and graded based on the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. Leukoencephalopathy was evaluated using the Fazekas grading system.
Ethical Consideration
The study was approved by the Institutional Ethics Review Board of Hanoi Medical University, Vietnam under decision No. 805/GCN-HĐĐĐ NCYSH-ĐHYHN, dated March 27, 2023. The patient data were maintained with confidentiality, in compliance with the Declaration of Helsinki. Informed consent was not required due to the retrospective nature of the study and the use of anonymized clinical data for the analysis.
Statistical Analysis
Data extracted from medical records were analyzed using SPSS version 22.0 (IBM Corp., Armonk, NY, USA). Quantitative variables were expressed as mean ± standard deviation. The Student’s t-test was applied to compare normally distributed continuous variables, while the Mann-Whitney U test was used for non-normally distributed variables. Categorical variables were compared using the Chi-square test or Fisher’s exact test as appropriate. Survival analyses (PFS, iPFS, and OS) were performed using the Kaplan-Meier method, with the log-rank test applied for group comparisons. Multivariate Cox proportional hazards models were utilized to identify independent predictors of survival outcomes. A P-value of less than .05 was considered statistically significant.
Results
Patient Characteristics
Between April 2019 and December 2023, a total of 225 patients with EGFR-mutated advanced NSCLC treated with osimertinib were initially screened. After exclusions — including patients who were treated with second-line osimertinib (n = 57), abandoned treatment (n = 35), lacked complete medical records (n = 5), or did not have baseline brain metastases (n = 66) — 62 patients met the eligibility criteria and were included in the final analysis. The patients in our study is depicted in Figure 1. Patients’ Selection Flowchart. EGFR - Epidermal Growth Factor Receptor, NSCLC- Non-small Cell Lung Cancer, BM- Brain Metastases.
Patient Characteristics and Treatment Response.
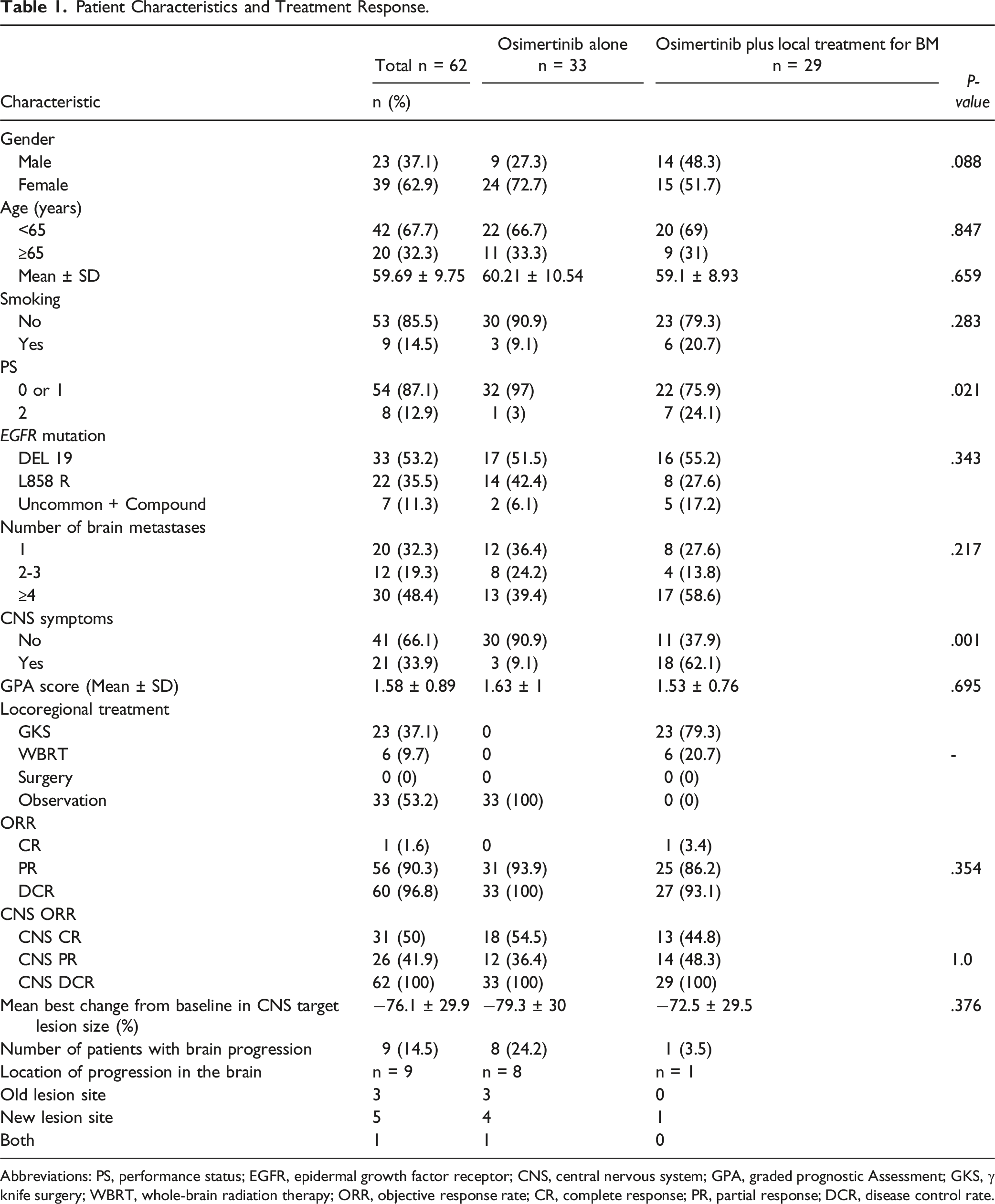
Abbreviations: PS, performance status; EGFR, epidermal growth factor receptor; CNS, central nervous system; GPA, graded prognostic Assessment; GKS, γ knife surgery; WBRT, whole-brain radiation therapy; ORR, objective response rate; CR, complete response; PR, partial response; DCR, disease control rate.
In terms of treatment allocation, 33 patients (53.2%) received osimertinib monotherapy, while 29 patients (46.8%) received osimertinib combined with local brain-directed treatments (23 patients underwent SRS and 6 patients underwent WBRT). No significant differences were observed between the two groups regarding age, sex, smoking status, EGFR mutation type, or number of brain metastases. The osimertinib monotherapy group exhibited better baseline PS (P = .021) and a lower rate of CNS symptoms (P = .001). However, there was no difference in GPA scores between the two groups (P = .695).
Treatment Outcomes
Response evaluation
The systemic objective response rate (ORR) was 91.9%, and the disease control rate (DCR) was 96.8%. According to RANO criteria, the CNS-ORR was 91.9%, and the CNS-DCR was 100%. No statistically significant difference in intracranial ORR was observed between the two groups: 90.9% in the osimertinib monotherapy group vs 93.1% in the osimertinib combined with cranial radiation group (P = 1.0). The mean best percentage change from baseline in CNS target lesion size was −76.1%. Specifically, the osimertinib-alone group showed a mean reduction of 79.3% (range: 2% to 100%), while the osimertinib plus radiation group had a mean reduction of 72.5% (range: 5% to 100%) (P = .376) (Table 1, Figure 2). Best Percentage Change From Baseline in Central Nervous System Target Lesion Size With (A) Osimertinib Alone and (B) Osimertinib Plus Brain Radiotherapy. TL - Target Lesion, CR - Complete Response, PR- Partial Response. SD- Stable Disease.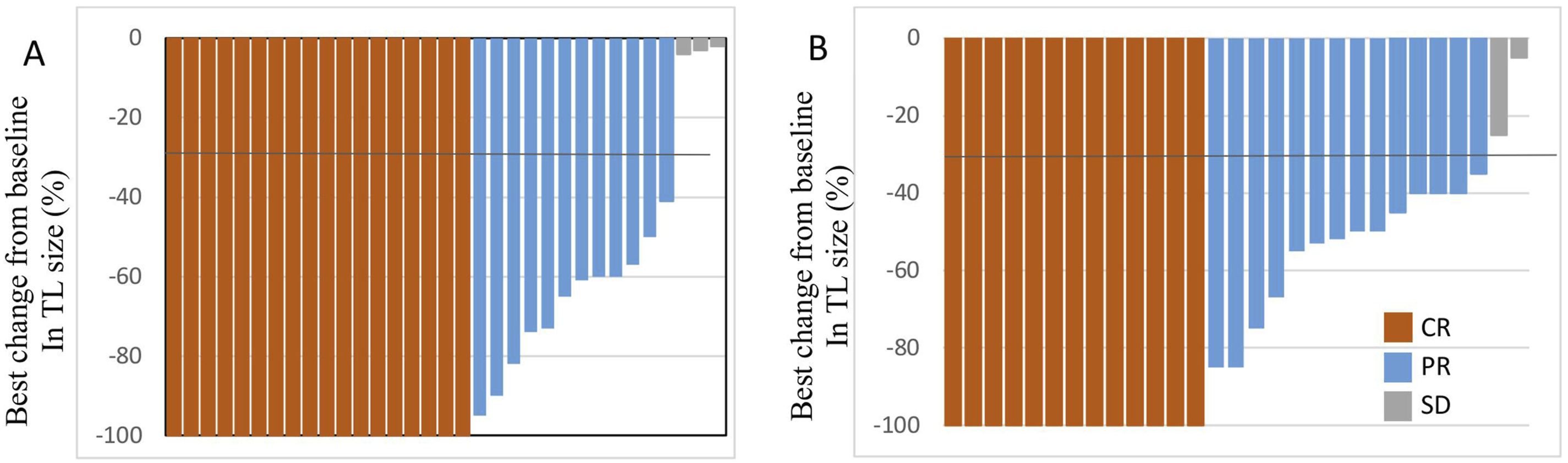
At the data cutoff, brain progression occurred in nine patients: eight patients from the osimertinib-alone group and one patient from the combination therapy group. Details on the location of brain progression in patients showed that, among the eight patients receiving osimertinib monotherapy, three developed progression at the initial lesion site, four had new brain lesions, and one had both old and new lesion progression. In the combination group, one patient developed a new brain metastasis (Table 1).
Survival Outcomes
At the latest follow-up, there were 14 deaths and 19 progression events recorded. The cohort’s median PFS was 24.5 months, and median OS was 35.2 months. Median iPFS was not reached (Figure 3). In unmatched, unadjusted Kaplan-Meier univariate comparisons, evaluating the association between PFS, OS and factors, no clinical and laboratory characteristics were found to influence OS and PFS (Table 2). Multivariate Cox regression analysis similarly did not identify significant independent predictors (Supplemental Tables 1 and 2). Progression-free Survival (PFS) of all Patients (A), Intracranial PFS (iPFS) of all Patients (B), Overall Survival of all Patients (C). (NR- Not Reached). Progression-free Survival and Overall Survival of all Patients. Abbreviations: PS, performance status; EGFR, epidermal growth factor receptor; GPA, graded prognostic Assessment. PFS, Progression-free survival; OS, overall survival; NR, not reached.

Of a total of 62 patients, 33 patients (53.2%) received osimertinib monotherapy, and 29 patients (46.8%) received osimertinib combined with brain radiotherapy, including 23 patients who received SRS, 6 patients who received WBRT. No significant differences were observed between patients received osimertinib alone and those receiving osimertinib combined with radiation in terms of median time to any progression (NR for osimertinib vs 24 months for RT + osimertinib, P = .656), intracranial progression (NR vs NR, P = .706), or overall survival (36 months vs 35.2 months, P = 0.734) (Figure 4). Progression-free Survival (PFS) (A), Intracranial PFS (iPFS) (B), and Overall Survival (C) of Osimertinib Alone Group and Osimertinib Plus Intracranial RT Group. (RT – Radiotherapy, NR – Not Reached).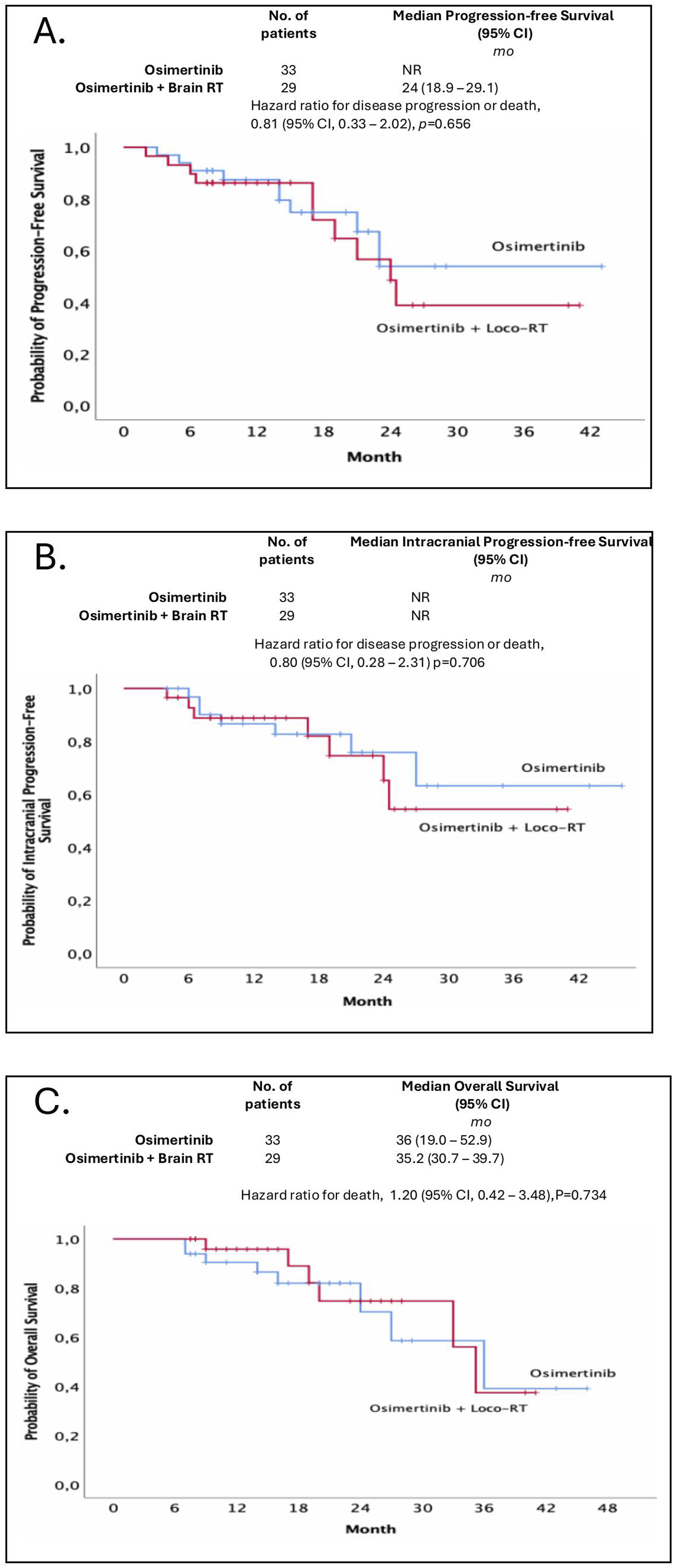
Treatment-related toxicities
Incidence of Adverse Events.
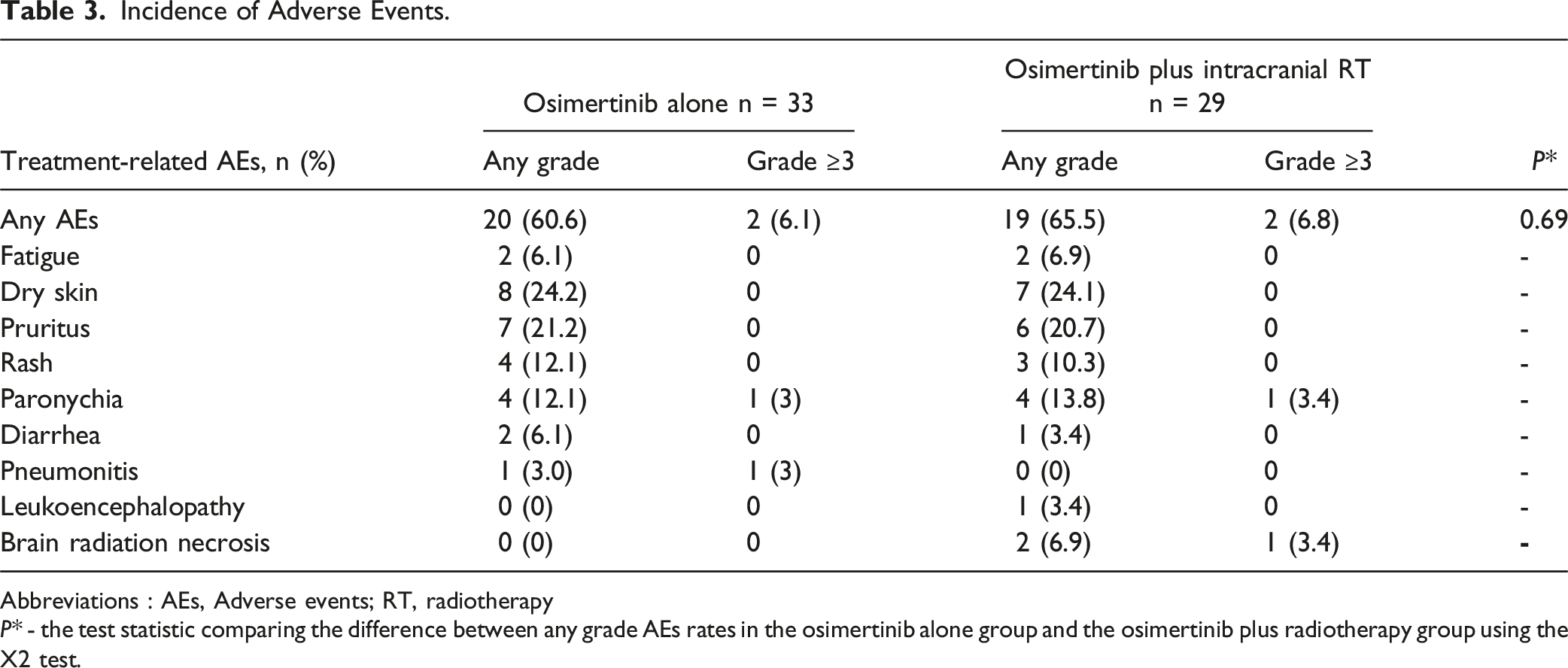
Abbreviations : AEs, Adverse events; RT, radiotherapy
P* - the test statistic comparing the difference between any grade AEs rates in the osimertinib alone group and the osimertinib plus radiotherapy group using the X2 test.
In the osimertinib plus radiotherapy group, 3/29 (10.3%) patients experienced radiotherapy-related side effects, which consisted of 2 cases of brain necrosis due to SRS (Table 3) and 1 case of leukoencephalopathy following WBRT. Among them, 1 patient had grade 3 brain radiation necrosis (Figure 5). Image of Brain in a Patient With Brain Radiation Necrosis. The Patient With Stage IV Non-small Cell Lung Cancer (NSCLC), T3N2M1 (Brain Metastasis), Pathology: Adenocarcinoma (AC), EGFR (+) Exon 19 Mutation, Treated With Osimertinib in Combination With Stereotactic Radiosurgery for the Brain Tumor. (A) Image of Brain Tumor before Treatment, Lesion Located in Right Frontal Lobe Measuring ∼ 22 × 15 × 14 mm, Increased Signal, Strong and Homogeneous Enhancement on T1 Gd (+), No Brain Edema. (B) Post-treatment, the Tumor Responded With Tumor Image Measuring ∼ 18 × 15 × 14 mm, Rim-Shaped Enhancement on T1 Gd (+), Mild Brain Edema. (C) 8 months after Radiosurgery, the Patient had Increased Headaches, Severe Nausea, MRI Showed a Rim-Shaped Enhancement Lesion Measuring ∼ 23 × 19 × 17 mm, Irregular Edges, and Extensive Surrounding Brain Edema. The Patient was Diagnosed With Post-radiation Brain Necrosis and Treated With Dexamethasone Followed by Bevacizumab. After Treatment, the Patient’s Condition Stabilized, and Neurological Symptoms Improved. (GSK -Gamma Knife Radiosurgery).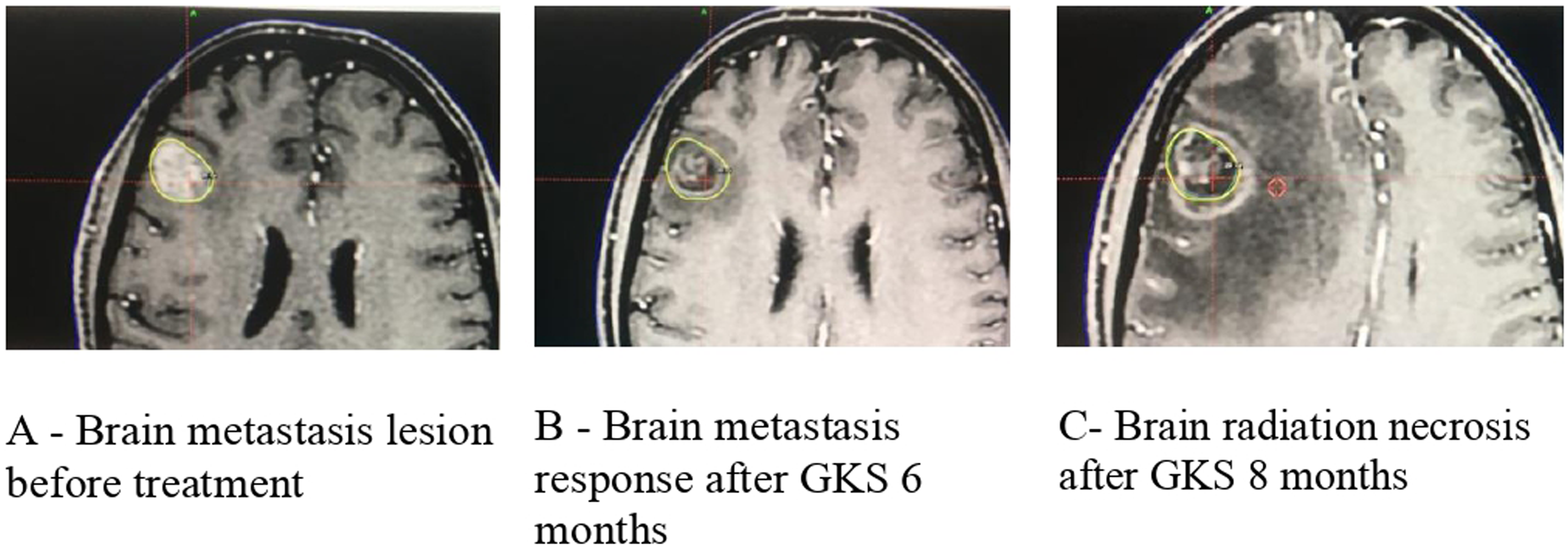
Discussion
To the best of our knowledge, this is the first retrospective study conducted at the Vietnam National Cancer Hospital evaluating the outcomes of first-line osimertinib in patients with NSCLC harboring EGFR mutations and BMs. Data from this analysis showed that osimertinib provides a favorable treatment effect in improving PFS, OS and CNS-ORR for this patients group. Additionally, survival outcome and intracranial response were noninferior to osimertinib alone compared with osimertinib plus brain radiotherapy. These results suggest that, in selected patients, the potent CNS activity of osimertinib may allow for the deferment of local brain-directed therapies. This treatment approach needs additional investigation through prospective trials.
In our study, osimertinib demonstrated a high intracranial response rate, with a significant reduction in CNS target lesion size. This result is similar to some other research data, confirming the excellent intracranial penetration of osimertinib. For instance, the FLAURA study showed a CNS ORR of 91% among patients with measurable brain metastases treated with osimertinib compared to 68% with first-generation EGFR-TKIs, with a median reduction in CNS lesion size of 64%. 5 Similarly, Brandon S. Imber and colleagues observed a 100% lesion-level best objective response in untreated brain metastases among patients receiving osimertinib, with a very low local failure rate at one and two years. 10 These findings reinforce the strong intracranial efficacy of osimertinib.
Our study reported a median PFS of 24.5 months and a median OS of 35.2 months, outcomes that are superior to those observed in the FLAURA subgroup with brain metastases, where median PFS was 15.2 months. 4 Consistent with real-world data, our results align with a study by Y. Zhao et al., which showed that first-line osimertinib achieved significantly longer iPFS, PFS, and OS compared to first-generation EGFR-TKIs, despite patients having more and larger brain metastases. In particular, the osimertinib group reached a PFS of 34.7 months and an overall survival of 40.6 months. 11 Our study does not have a control group to compare the effectiveness with first- and second-generation TKIs. However, based on the results of this study and other studies evaluating the effectiveness of first- and second-generation EGFR-TKIs in Vietnamese patients with EGFR-mutant NSCLC, osimertinib not only improves response rates and controls brain metastases but also provides significant survival benefits. In the Vietnamese population, studies showed that when treating EGFR-mutated advanced NSCLC with BMs by 1st and 2nd generation TKIs, the median PFS and median OS were only 11 months and 25-29.9 months, respectively.12,13
The combination of EGFR-TKIs and radiotherapy has been hypothesized to enhance therapeutic efficacy by increasing radiosensitivity and improving blood-brain barrier penetration.14,15 Several earlier studies demonstrated improved outcomes with upfront cranial radiation plus EGFR-TKIs compared to TKIs alone.16-20 For example, Magnuson et al. reported a significantly longer OS and iPFS with the combination of SRS or WBRT plus erlotinib vs erlotinib monotherapy. 16 A meta-analysis by Du et al. similarly indicated survival benefits when cranial radiation was combined with first- or second-generation EGFR-TKIs. 17 However, these studies did not include osimertinib, which has superior CNS penetration compared to earlier-generation TKIs.
Following the emergence of osimertinib as a first-line treatment for advanced NSCLC harboring EGFR mutations, and osimertinib is known to better penetrate the CNS.4,6 One remaining question is the role of upfront cranial radiation therapy in osimertinib-treated EGFR-mutant NSCLC with BMs. In our study, no significant differences in systemic or intracranial outcomes were observed between patients receiving osimertinib monotherapy vs those receiving osimertinib combined with brain radiotherapy. These findings are consistent with other reports, such as those by Thomas et al., who found no difference in PFS, iPFS, or treatment failure rates between patients receiving CNS-penetrant TKIs alone and those combined with radiation. 21 Similar results were confirmed in a study at the Stanford Cancer Center, or in a real-world Canadian data, both showing no significant differences in median OS and median time-to-treatment failure between patients receiving Osimertinib alone or Osimertinib with radiotherapy.22,23 Furthermore, a study by Xiaoyang Zhai et al. in 61 NSCLC patients showed no significant differences in intracranial or systemic responses, iPFS, or OS between patients treated with osimertinib alone vs osimertinib plus radiotherapy. 24 Notably, in Zhai’s study, a relatively high incidence of leukoencephalopathy was observed among patients receiving combined therapy, emphasizing the potential risks associated with brain irradiation. Our study also recorded 3 patients (10.3%) experienced radiotherapy-related side effects in the osimertinib plus radiotherapy group, which consisted of two cases of brain necrosis due to SRS and one case of leukoencephalopathy following WBRT. Among them, 1 patient had grade 3 brain radiation necrosis. Based on these studies, delaying cranial radiotherapy in patients with EGFR-mutant NSCLC that has spread to the brain and are being treated with more brain-penetrating drugs such as osimertinib does not appear to impact survival outcome or intracranial response and may lower the risk of radiation-induced neurotoxicity. This approach is also supported by ASCO-SNO-ASTRO guidelines, which recommend deferring local therapy until intracranial progression in asymptomatic patients receiving osimertinib. 25
Nevertheless, although toxicity remains a concern, the effectiveness of local therapies in controlling brain disease is undeniable. Our findings also support this, demonstrating similar brain disease control with the combination of osimertinib and local treatment. At the latest follow-up, 9 patients experienced brain progression, with 8 of them receiving osimertinib alone, and 4 of those 8 patients (50%) saw regrowth at the original BM site. In contrast, only 1 patient in the group receiving osimertinib combined with brain radiation developed brain progression, which occurred in a new brain area. Furthermore, several other studies reported results inconsistent with our study. Retrospective study by Takehiro Tozuka et al reported significantly longer OS and CNS-PFS in patients who received upfront local therapy followed by osimertinib compared to osimertinib alone. 26 Another real world data showed upfront cranial local therapy was associated with improved iPFS and PFS, particularly in patients with oligo-metastatic disease with prolonged PFS and OS. 11 So, delaying cranial radiotherapy in EGFR-mutant NSCLC patients with BMs receiving osimertinib is controversial. These discrepancies highlight that the decision to pursue early cranial radiotherapy must be individualized, balancing potential benefits against risks. Ultimately, given the retrospective nature of available data, larger prospective trials are necessary to definitively determine the optimal timing of cranial radiation in EGFR-mutant NSCLC patients treated with osimertinib. Until then, multidisciplinary discussions tailored to individual patient characteristics — including symptoms, brain disease burden, and performance status — remain essential to guide therapeutic decisions.
Our study has several limitations. The most prominent is its retrospective design and the limited sample size from a single center, which may reduce statistical power and hinder the detection of significant differences between the two groups. Additionally, the follow-up duration may not be sufficient to capture long-term outcomes. Moreover, the imbalance in performance status and neurological symptoms between the two groups, despite no significant difference in GPA scores, could have introduced a bias in the comparison of treatment outcomes. Larger prospective studies are needed to prove this hypothesis.
Conclusions
While osimertinib provides excellent treatment responses and CNS activity for advanced EGFR-mutant non-small cell lung cancer (NSCLC) patients with brain metastases, there was no observed survival outcome or intracranial response benefit when combining osimertinib and brain radiotherapy compared to the osimertinib monotherapy. These results offer preliminary suggestion that the intracranial activity of osimertinib may enable local radiation to be deferred in appropriately selected patients. As a small retrospective study, prospective studies are needed to elucidate the longer‐term benefits and risks of this approach.
Supplemental Material
Supplemental Material - Therapeutic Outcomes of Osimertinib in EGFR – Mutant Non-Small Cell Lung Cancer With Brain Metastases: Results From a Retrospective Study at Vietnam National Cancer Hospital
Supplemental Material for Therapeutic Outcomes of Osimertinib in EGFR – Mutant Non-Small Cell Lung Cancer With Brain Metastases: Results From a Retrospective Study at Vietnam National Cancer Hospital by Hoa Nguyen Thi Nhu, Quang Le Van, Phuong Nguyen Thi Bich, Hau Tran Thi, Tai Nguyen Van, The Dao Minh, Lien Nguyen Duc, Duong Phan Thanh, Lam Ngo Le, Chu Nguyen Van, Hoang Nguyen Cong, Duc Nguyen Dinh, and Kien Do Hung in Cancer Control
Footnotes
Ethical Statement
Author Contribution
Conception and design: HNTN, QLV, KDH
Collection and assembly of data: HNTN, QLV, KDH
Data analysis and interpretation: HNTN, QLV, KDH
Manuscript writing: All authors
Final approval of manuscript: All authors
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
