Abstract
Introduction
Whole-brain radiotherapy simultaneous integrated boost intensity-modulated radiotherapy (WBRT + SIB-IMRT) is a potential treatment approach for brain metastasis (BM) that may result in improved overall survival (OS). However, the safety and efficacy of WBRT + SIB-IMRT combined with anlotinib for BM treatment remain uncertain.
Methods
We retrospectively compared the safety and efficacy of anlotinib + WBRT + SIB-IMRT with those of WBRT + SIB-IMRT in patients with BM from 2019 to 2022. The adverse reaction type and grade, intracranial objective response rate (iORR), intracranial disease control rate (iDCR), OS, and intracranial progression-free survival (iPFS) of anlotinib + WBRT + SIB-IMRT were compared with those of WBRT + SIB-IMRT alone.
Results
In total, 63 patients received either anlotinib + WBRT + SIB-IMRT or WBRT + SIB-IMRT alone (n = 31 and 32, respectively). No significant clinical differences were found between the two groups. The iORR and iDCR were higher in the anlotinib + WBRT + SIB-IMRT group than in the WBRT + SIB-IMRT group. The median iPFS and median OS of the 31 patients who received anlotinib + WBRT + SIB-IMRT were 14.5 and 18.9 months, respectively, whereas the median iPFS and median OS for the 32 patients who received WBRT + SIB-IMRT alone were 11.4 and 14.9 months, respectively. Thus, anlotinib combined with WBRT + SIB-IMRT increased the duration of iPFS, but not OS. iPFS was influenced by the Karnofsky Performance Status (KPS) score, age, extracranial distant metastasis, and addition of anlotinib to treatment, whereas OS correlated with age, extracranial distant metastasis, and KPS score. No treatment-related adverse events of grade 3 or higher occurred in either group.
Conclusions
Anlotinib combined with WBRT + SIB-IMRT is effective for BM and is well tolerated by patients.
Keywords
Introduction
Brain metastases (BM) are the most commonly diagnosed type of central nervous system tumor in the adult population, and their incidence is projected to increase as survival rates for primary malignancies improve. 1 BM treatment primarily consists of local and systemic therapy. Historically, whole-brain radiotherapy (WBRT) has been regarded as standard radiation therapy for BM. However, the radiotherapeutic approach to BM has evolved significantly over the last two decades based on evidence from clinical trial. 2 Currently, management of patients with brain metastases may encompass a combination of the following therapeutic approaches: surgery, WBRT, stereotactic radiosurgery (SRS), sequential intensity modulated radiation therapy (sIMRT), or simultaneous integrated boost intensity modulated radiation therapy (SIB-IMRT). The goals of these treatments are to palliate symptoms effectively, maintain neurological function, curb intracranial disease progression, and improve the quality of life.3,4
IMRT is characterized by a highly conformal dose distribution for the target and a limited dose for organs at risk. 5 Dobi et al. have reported that WBRT + SIB-IMRT improved local control rates, prolonged OS, and reduced the probability of cognitive decline, even in patients presenting with a poor performance status and advanced extracranial disease. 6 Despite the potential risk of neurocognitive functional impairment with WBRT, research has demonstrated that it can substantially decrease the recurrence of intracranial tumors, and that the damage to neurocognitive function is considerably less than the neurological loss or impairment resulting from tumor progression. 7 In our clinical experience, patients with BMs who did not receive WBRT had a higher rate of intracranial recurrence. WBRT is reserved for selected patients.
Radiotherapy, a fundamental component of the treatment of intracranial tumors, can damage blood vessels and open the blood-brain barrier. 8 Enhancing the therapeutic response rates and extending the survival rates of patients with BM are the current focus of treatment. With regard to this focus, tumor growth is known to be intricately linked to the proliferation of new blood vessels, and angiogenesis plays a critical role in the development and progression of various cancers. Additionally, an increasing number of studies have recently demonstrated that antiangiogenic therapies can increase the sensitivity to radiotherapy and enhance the oxygenation of tumor cells by normalizing the tumor vasculature and decreasing the fraction of hypoxic cells. 9
Anlotinib is a novel, orally administered tyrosine kinase inhibitor that directly inhibits multiple targets, including PDGFR, VEGFR, c-KIT, and FGFR, thereby suppressing tumor development and angiogenesis.10,11 Ample radiobiological evidence supports the use of anlotinib owing to its radiosensitizing effects. Anlotinib can directly inhibit cell proliferation and, when used in combination with radiotherapy, synergistically suppress tumor cell growth. 12 In addition, the antiangiogenic efficacy of anlotinib has been reported to be significantly better than that of sorafenib and sunitinib. 13 Anlotinib is widely used to treat various types of cancer, including non-small cell lung cancer, soft tissue sarcoma, and esophageal cancer.14-16 Furthermore, a post hoc analysis of the ALTER0303 study indicated that anlotinib may have high antitumor activity in the management of intracranial lesions. 17 This study aimed to determine whether anlotinib combined with radiotherapy has therapeutic potential in patients with BM.
Methods
Patients
We retrospectively reviewed the clinical records of patients with BM treated at our hospital between October 2019 and December 2022. All patient identifying information has been completely removed. This study was approved by the Ethics Committee of Nanjing Pukou People’s Hospital (No. 2023-SR-021) and was conducted in compliance with the Declaration of Helsinki. Our Institutional Review Board waived the requirement for informed consent owing to the retrospective nature of the study. This retrospective study conformed to the STROBE guidelines for observational epidemiological research. 18
The inclusion criteria were as follows: (1) patients diagnosed with histologically confirmed malignancies who exhibited 1-10 intraparenchymal BM as identified using computed tomography (CT) or magnetic resonance imaging (MRI); (2) patients aged between 18 and 80 years and with KPS score ≥60; (3) patients who received WBRT + SIB-IMRT along with anlotinib or were treated with WBRT + SIB-IMRT alone; and (4) patients in whom anlotinib treatment was sustained until they developed either intolerance or disease progression. The exclusion criteria were as follows: (1) patients with prior WBRT, SRT, or any other intracranial irradiation therapy; (2) WBRT + SIB-IMRT or concurrent treatment not completed; and (3) patients with a follow-up period of less than 1 month (Figure 1). The flow diagram of included patients.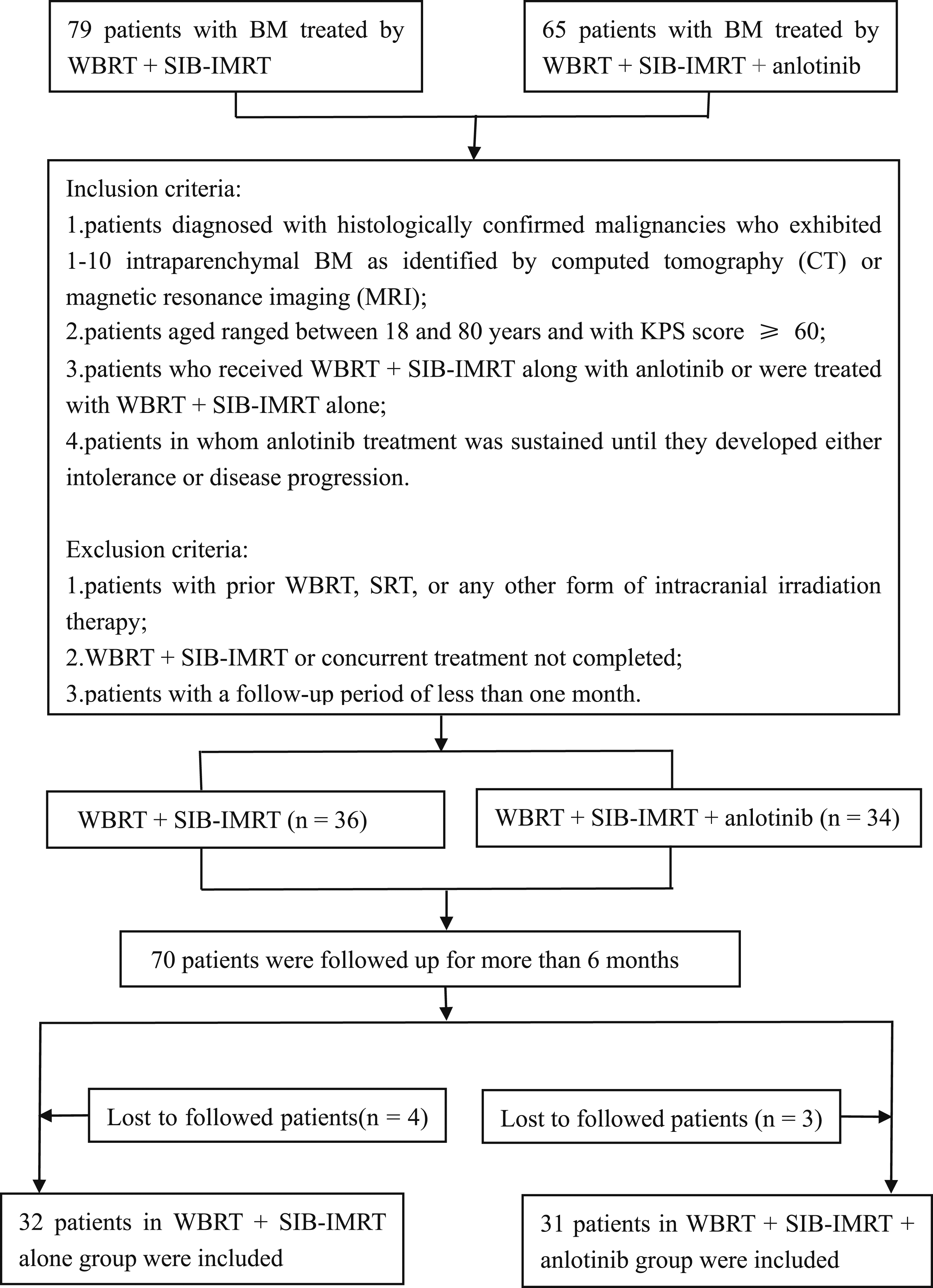
From the clinical records of these patients, we obtained clinical information, treatment processes, imaging dates and clinical outcomes. Clinical information included age, gender, KPS score, primary site, and number of metastases. The tumor diameter and volume were evaluated individually by two radiologists. When these two radiologists disagreed, a third radiologist adjudicated.
Treatment
WBRT + SIB-IMRT was administered at our hospital by using a linear accelerator (Varian, USA). Patients were placed in the supine position and were immobilized with a thermoplastic head mask fixation system (Klarity, Guangzhou, China) to simulate high-resolution thin-slice (3.0 mm thick) Computed Tomography (CT). Target volumes and at-risk organs were contoured using the Eclipse version 13.6 treatment planning system (Varian). The gross tumor volume (GTV) for SIB-IMRT was determined from enhanced areas observed on MRI T1-weighted contrast-enhanced scans. The planned GTV was calculated by adding a 3.0 mm expanded margin around GTV in three dimensions. The prescribed dose for SIB-IMRT was 54 Gy in 20 fractions, whereas that for WBRT prescription dose was 40 Gy in 20 fractions for the entire brain planning target volume. The patients received WBRT + SIB-IMRT on Monday through Friday, and no treatment was administered on weekends.
The patients received anlotinib orally once daily for 14 days in a 3-week cycle, continuing until disease progression, intolerable toxicities, or death occurred. The starting dose of anlotinib was 12 mg for patients with a KPS score ≥80 and 10 mg for those with a KPS score <80. If grade 3 or 4 adverse events occurred, the anlotinib dose was adjusted to 10 or 8 mg, respectively. Follow-up assessments included the collection of head imaging data and clinical information. Enhanced MRI or CT scans were scheduled for before treatment, 1 month after IMRT treatment, and every 3 months thereafter until treatment ceased to be effective or until death. Treatment responses were classified according to the Response Evaluation Criteria in Solid Tumors (RECIST version 1.1) as complete response (CR), partial response (PR), stable disease (SD), or progressive disease (PD).
Outcomes
The primary endpoint was intracranial progression-free survival (iPFS), defined as the time from the initial diagnosis of BM to the appearance of the first signs of BM progression according to the RECIST or until death from any cause. The secondary endpoints included the intracranial objective response rate (iORR; percentage of patients achieving intracranial CR and PR), intracranial disease control rate (iDCR; percentage of patients with intracranial CR, PR, and SD), and OS (measured from the initial BM diagnosis to death or last follow-up). Treatment-related adverse events were evaluated using the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE version 5.0).
Statistical Methods
Statistical analyses were conducted using SPSS version 26.0 Statistical significance was set at an alpha level of 0.05 (P < 0.05). A normality test was performed on quantitative data. Normally distributed data are expressed as mean ± standard deviation (x ± s), and were compared between groups by using the t test. Data that did not conform to a normal distribution are presented as median and interquartile range (Q1, Q3) and were compared between groups by using the Mann-Whitney U test. Qualitative data are expressed as frequencies and percentages and analyzed using the χ2 test or Fisher exact test. Survival analysis was constructed by Kaplan-Meier survival curves with 95% confidence interval (CI) , and Cox regression analysis was used to evaluate the association between each factor and the observed indicators OS and iPFS.
Results
Patients and Lesion Characteristics
Clinical Baseline Characteristics of Included Patients.

Intracranial Efficacy
ORR and DCR of Intracerebral Lesions in Two Groups.

Analysis of Survival
At the cutoff date of October 30, 2023, of the 31 patients treated with anlotinib + WBRT + SIB-IMRT, 16 patients (51.6%) died due to intracranial progression, and 9 patients (29.0%) died due to extracranial progression. In contrast, among the 32 patients in the WBRT + SIB-IMRT alone group, 20 (62.5%) died as a result of intracranial progression and 7 patients (21.9%) died as a result of extracranial progression. The median iPFS was 14.5 months for those receiving anlotinib + WBRT + SIB-IMRT, compared to 11.4 months for those receiving WBRT + SIB-IMRT alone (HR 0.585, 95% CI 0.351-0.974, P = 0.022) (Figure 2(a)). This suggested that the combination of anlotinib with WBRT + SIB-IMRT significantly prolonged iPFS. Patients in the anlotinib + WBRT + SIB-IMRT group had a longer median OS of 18.9 months compared to 14.9 months in the WBRT + SIB-IMRT alone group, although the difference was not statistically significant (HR 0.832, 95% CI 0.482-1.433, P = 0.500) (Figure 2(b)). Kaplan-Meier survival curves were used to analyze the survival of patients in different treatment groups. (A) iPFS, (B) OS.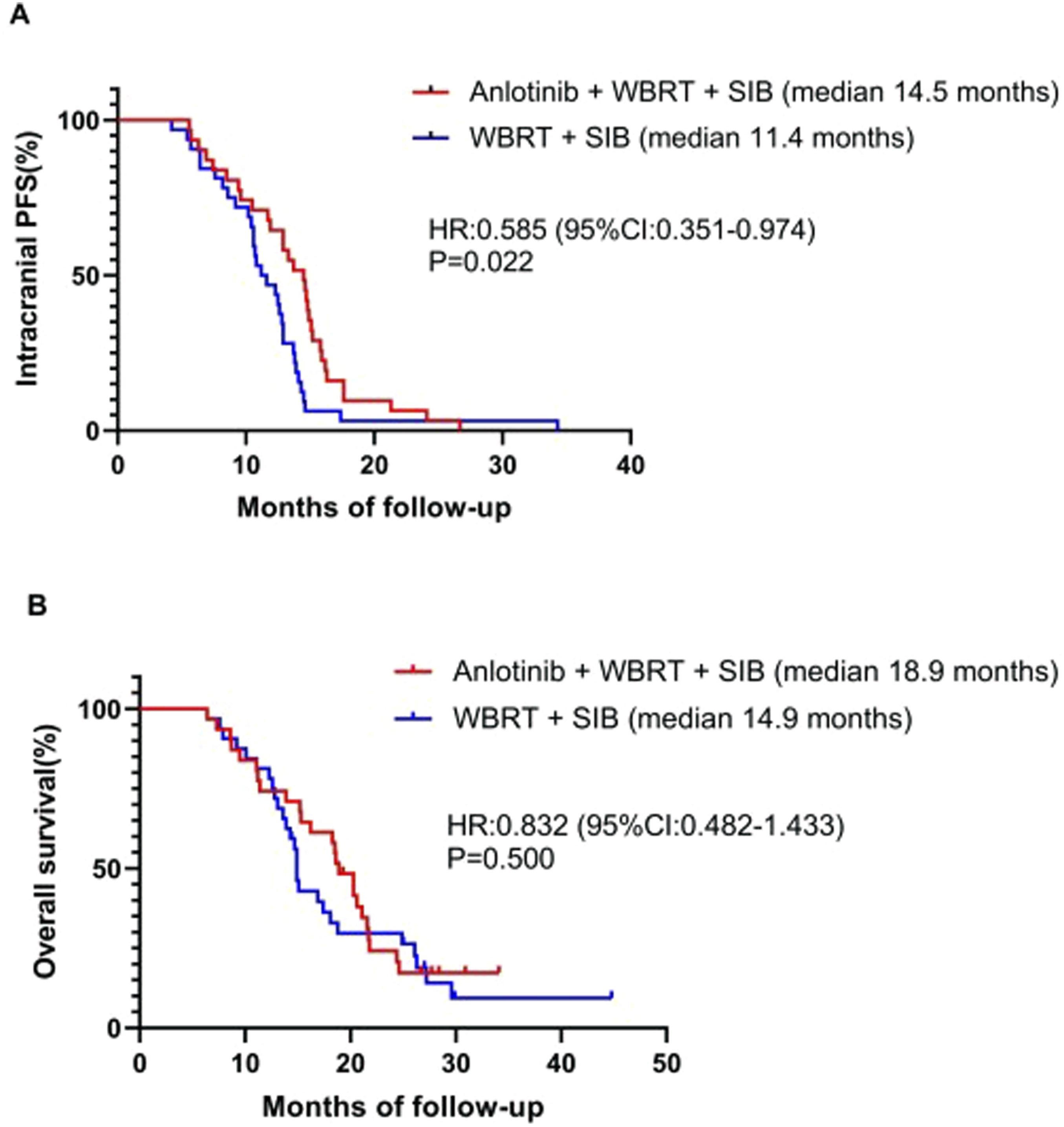
Univariate and Multivariate COX Regression Analysis of iPFS.

Univariate and Multivariate COX Regression Analysis of OS.

Safety of Treatment
The Most Common Treatment-Related Adverse Events.

Discussion
In patients with malignant solid tumors, the presence of BMs correlates with a poor prognosis and indicates an advanced of the disease stage. Therefore, treatment strategies should be directed toward maximizing survival time, improving quality of life and reducing neurological deterioration. Previous studies have indicated that effective therapeutic options include, but are not limited to surgical resection and WBRT and SRS/SRT, either alone or in combination. More importantly, some studies have shown that SRS can achieve a similar OS to that of surgical resection, with a higher rate of local control.19-21 The RTOG 9508 study evaluated the efficacy of WBRT with or without an SRS boost in patients with one to three BM and demonstrated that the SRS boost significantly improved local control. 22 However, the SRS requires a standalone treatment planning system, relatively advanced technology, and high medical expenses, making its routine application difficult in many medical institutions. Hence, many clinicians are seeking an alternative to the WBRT + SRS regimen, with SIB-IMRT emerging as the most promising treatment. Several studies have proposed the use of SIB-IMRT for the treatment of BMs using various techniques. These studies were feasibility assessments aimed at achieving dose levels comparable to those of sequential SRS based on a linear quadratic cube model. In recent years, several studies have tentatively confirmed the dosimetric advantages and higher radiotherapy-related safety of WBRT + SIB-IMRT than that of WBRT + SRS.23-25 Rodrigues et al. conducted a retrospective study of 500 patients who received SRS alone or WBRT + SIB-IMRT and found no significant differences in OS; however, SIB-IMRT was correlated with a lower incidence of intracranial failure, which might be ascribed to the WBRT component of the treatment. 26 Therefore, we retrospectively applied the WBRT + SIB-IMRT technique to treat patients with BMs and observed its efficacy when it was combined with anlotinib.
Anlotinib is a novel small-molecule and multi-targeting TKI that exerts antitumor effects mainly through the anti-VEGF pathway. 27 Recent studies have shown that anlotinib possesses intracranial activity and is capable of controlling intracranial tumors.17,28,29 Anlotinib can normalize blood vessels in metastatic tumors, modulate the tumor internal microenvironment, restore normal vascular permeability, and synergize with radiotherapy to increase radiosensitivity and decrease brain edema. Two prospective studies30,31 and one retrospective study 32 suggested that the combination of anlotinib with radiotherapy for lung cancer patients with brain metastasis has satisfactory efficacy. However, there is no evidence to determine whether the therapeutic regimen of anlotinib combined with WBRT + SIB-IMRT is superior to WBRT + SIB-IMRT alone in patients with BM.
In our study, anlotinib + WBRT + SIB-IMRT resulted in significantly better median iPFS compared with WBRT + SIB-IMRT alone (14.5 vs 11.4 months, P = 0.022). However, this combination therapy did not result in a statistically significant improvement in OS (P > 0.05), although the median OS in the anlotinib + WBRT + SIB-IMRT group was longer than that in the WBRT + SIB-IMRT alone group. These findings are consistent with the results of the ALTER0303 study, in which anlotinib positively influenced PFS but did not significantly prolong OS in patients with BM. 17 Additionally, we found that iORR was higher in patients who received anlotinib + WBRT + SIB-IMRT than in those who received WBRT + SIB-IMRT, which might be related to the synergistic effect of anlotinib in combination with WBRT + SIB-IMRT on controlling intracranial lesions. Anlotinib has a radiosensitizing effect that can strengthen the lethal impact of radiotherapy on tumor cells and thus enhance local therapeutic efficacy. Univariate and multivariate analyses of the patients’ clinical information and survival data showed that the addition of anlotinib was an independent prognostic factor for improved iPFS (P < 0.05) and that a higher KPS score and younger age were also independent prognostic factors for improvement (P < 0.05). This may be attributed to the fact that a patient’s physical condition can affect treatment efficacy. Concurrently, our study showed that the iPFS and OS of patients with extracranial distant metastasis were significantly shorter than those of patients without extracranial distant metastasis, which is consistent with the conclusions of previous studies.33,34
In the present study, all patients completed toxicity assessments. All patients exhibited good tolerance to the treatment, with no grade ≥3 toxicities observed. Adverse reactions were primarily grades 1-2 and improved after symptomatic treatment. The most common treatment-related adverse events were fatigue, hypertension, dizziness, and appetite loss. In two patients, the dosage of anlotinib was reduced from 10 mg to 8 mg due to hypertension, loss of appetite, and fatigue. No instances of treatment-related discontinuation of anlotinib have been observed.
The findings of this study are important for several reasons. First, by employing consistent SIB-IMRT and WBRT protocols throughout the study, we minimized the variability in treatment-derived factors when analyzing the efficacy of the therapy. Second, SIB-IMRT can simultaneously provide different radiotherapy doses to clinical and focal target areas, and integrate radiotherapy in different target areas into one plan, which has good target conformance and can better protect surrounding normal tissues and shorten hospitalization time. Third, our study was designed as a preliminary evaluation of the therapeutic efficacy and safety of WBRT + SIB-IMRT alone or in combination with anlotinib for BM treatment, and obtained encouraging outcomes.
Although this study yielded several significant findings, its limitations should be acknowledged. First, the conclusions of this study are based on its retrospective design. Despite using relatively strict inclusion and exclusion criteria, the potential for selection bias and patient heterogeneity could not be eliminated, which reflects the inherent limitations of a retrospective study design. Second, during the period of radiotherapy and follow-up, some patients underwent systemic treatment, which could affect their survival rates and intracranial local control. Third, the patient sample size in our study was not determined through an a priori power analysis and was less than ideal. Given these limitations, future studies should use an expanded sample size, employing a multi-institutional randomized prospective clinical trial design to reduce heterogeneity and thereby draw more robust conclusions.
Conclusion
In this study, we evaluated the therapeutic efficacy of anlotinib combined with WBRT + SIB-IMRT in patients with BM. The findings revealed that the combined application of anlotinib + WBRT + SIB-IMRT presented obvious clinical benefits, particularly in terms of improving iORR and extending iPFS in patients with BM. Our research provided important reference values for clinical treatment strategies in patients with BM. To obtain more precise data, prospective studies with larger sample sizes should be conducted.
Supplemental Material
Supplemental Material - Whole-Brain Radiotherapy Simultaneous Integrated Boost Intensity-Modulated Radiotherapy Combined With Anlotinib in the Treatment of Brain Metastases
Supplemental Material for Whole-Brain Radiotherapy Simultaneous Integrated Boost Intensity-Modulated Radiotherapy Combined With Anlotinib in the Treatment of Brain Metastases by Wei Tao, Chunyu Jiang, Jiaqi Xie, Wei Liu, Shuan Wang, Jianyu Zhang, Xue Qiao, Jingyi Yu, Ting Jia and Yuandong Cao in Cancer Control.
Footnotes
Acknowledgments
The authors would like to thank all of the participants in the study.
Author contributions
W.T. conceived, designed, analyzed, interpreted and wrote the paper, C.J.; J.Z.; X.Q.; and J.Y. data collation and analysis, J.X. and W.L. helped to draft the manuscript, S.W. review and edit the manuscript, T.J.and Y.C conceived, designed, analyzed, interpreted and wrote the paper. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Institute of Hospital Administration, NHC. 2023 Medical Quality (Evidence-Based) Management Research project (YLZLXZ23G129); Project of Chinese Hospital Reform and Development Institute, Nanjing University (NDYGN2023034).
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
