Abstract
Introduction
We evaluated the clinical efficacy of primary Human Papillomavirus (HPV) testing and liquid-based cytology (LBC) for cervical screening in women below 30 years old.
Methods
This was a retrospective analysis of data collected prospectively from women 25 years or older who attended hospital-based gynaecology clinics for cervical screening. Simultaneous high-risk HPV testing with partial genotyping and LBC testing was performed. Comparison of clinical efficacy of HPV testing and LBC was performed for routine screening discharge rate, colposcopy referral rate, and number of colposcopies needed for each CIN2+ detected.
Results
Of 6398 women included, 503 women (7.9%) tested positive for HPV DNA. The positivity was higher for 25-to-29 years old than for women ≥30 years old (12.6% vs 7.3%,
Conclusions
Our data indicated that cervical screening for women between 25 and 29 years old could adopt HPV screening as the older women.
Keywords
Introduction
Cervical cancer is a highly preventable cancer with mass vaccination against high-risk human papillomavirus (HPV) and eradication of high-grade cervical intra-epithelial lesions neoplasia (CIN) detected in mass screening using high-performance tests. In 2018, the World Health Organisation announced a global call to action towards the elimination of cervical cancer. One of the three strategic actions is to screen 70% or more of women at risk of cervical cancer. 1
Globally, the entry age of women for mass population screening varies according to the constraints of local settings. In 2004, the Singapore Health Promotion Board launched Cervical Screen Singapore (CSS), a cytology-only-based national cervical cancer screening programme for women aged 25 to 69 years old who have had penetrative sexual intercourse. Women screened negative for intraepithelial lesions and malignancy (NILM) are recommended to undergo regular repeat screening every three years.
Meanwhile, irrefutable evidence established that primary HPV screening is more sensitive and cost-effective than liquid-based cytology (LBC) in detecting high-grade lesions equal to or more severe than CIN grade-2 (CIN2+).2–5 In 2019, the updated Singapore national screening guidelines recommended primary HPV-DNA testing in place of LBC for women aged 30 years and above while LBC continues for women below 30 years old. 6
HPV testing for women 30 years or older is universally accepted as the best high performance cervical screening test. However, the method of screening in younger women below 30 years old remains controversial. 7 The guidelines of Canada, Italy, South Korea, and Spain recommend three-yearly cervical cytology, France recommends two annual cervical cytology tests before performing three-yearly cervical cytology, Germany recommends annual cervical cytology and Japan recommends biennial cervical cytology.7,8 In the United States of America, cervical cancer screening recommendations are split into two camps. The United States Preventative Services Task Force (USPSTF), 9 American Society for Colposcopy and Cervical Pathology (ASCCP) 10 and American College of Obstetricians & Gynecologists (ACOG) 11 recommend three-yearly cervical cytology from 21 to 29 years old whereas the American Cancer Society (ACS) recommends five-yearly hrHPV testing. 12 Five-yearly hrHPV testing is also recommended in the national guidelines of the United Kingdom, 13 Sweden, 14 and Australia. 15
Yet, the need to screen young women is compelling. The mean time for progression from a low grade squamous intraepithelial lesion (LSIL) to a high grade squamous intraepithelial lesion (CIN2+) is approximately 6 to 7 years. 16 In Singapore, while the Age-Standardised Incidence Rate (ASIR) of cervical cancer has fallen drastically from 18.0 to 6.8 per 100 000 population from 1968-1972 to 2018-2022, in women aged 30 to 39 years old cervical cancer remains the fourth most common cancer and is ranked the second most common cause of death from gynaecological cancers, with 173 women aged 30 to 39 years old being diagnosed in Singapore in 2018-2022. 17 Detection and elimination of high-grade lesions in young women is a necessary strategy to control invasive cervical cancer in young women. It seems obvious that young women should be screened with high-performing tests, in line with their older counterparts.
Opinions on the choice between primary HPV testing and LBC screening for women below 30 years old is divided. The prevalence of HPV infection is high and mostly transient in nature in these women.18–22 Secondly, the absolute incidence of invasive cancer in this age group is low, accounting for less than 10% of all cervical cancers in women. The benefit of surgical excisional treatment of CIN must be balanced against the potential risk of anatomical damages to the cervix which may result in an increased risk of either cervical incompetence, preterm delivery and premature preterm rupture of membranes, or cervical stenosis which may jeopardize the fertility potential.23–25 Clearly, robust data is needed to guide the development of policy and guidelines of cervical screening for these young women.
This study aimed to obtain evidence to fill the information gap in the policy considerations between HPV testing and LBC in cervical screening programs for women below 30 years old. The specific objectives of the study aimed to (1) determine the efficacy of detecting CIN2+ by primary HPV testing and the alternative method by LBC screening among women between 25 and 29 years old in Singapore, and (2) determine the clinical efficacy of the two screening methods between the 25-to-29-year old women, compared to women between 30-to-44-year old, and women in the entire screening age group up to 69 years old.
Methods
Design of Study and Ethical Considerations
This was a retrospective database analysis of women who undertook cervical screening by co-testing in a hospital-based gynaecological clinic in Singapore between 15 November 2013 and 30 April 2019. Relevant clinical data was prospectively updated into the database. Waiver of consent was granted for this study by the hospital’s ethics committee (Centralised Institutional Review Board 2016/2385) and the study was carried out in accordance with the 1964 Declaration of Helsinki ethical standards.
Study Subjects
Women entered this study were consecutive women aged between 25 and 69 years old who had undertaken co-testing with simultaneous HPV DNA testing and LBC in a hospital-based gynaecological clinic in Singapore. The screening was opportunistic cervical screening for women who fulfilled the recommended eligibility criteria for routine screening at 3-year interval. Women who attended the clinic for management and follow up surveillance of cervical neoplasia or prior abnormal cytology were excluded from this study.
Data Collection
The data on cases fulling the study criteria was retrieved. Patients’ demographics, history of cervical screening and diagnosis, attendance and outcome of colposcopy were extracted from the database.
Test Samples and Laboratory Tests
One physician-collected cervical scrape sample was obtained from each woman and kept in 20 mL PreservCyst® Solution (ThinPrep, Hologic USA). The sample was split into two aliquots, one for cytopathology assessment and one to the molecular laboratory for HPV testing. The two laboratories performed the respective tests separately and reported the results independently. HPV DNA testing was performed on Cobas 4800 system (Roche, USA) with partial genotyping. The outcome of HPV testing was reported individually for HPV-16, HPV-18, and the 12 other HPV subtypes (HPV-31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66 and 68) was reported collectively (HPV-12others). LBC was performed on Hologic (USA) liquid-based cytology platform. The outcome of LBC was reported according to the criteria of Bethesda system 2014.
Management of Screen Results
Independent test results on LBC and HPV tests were individually returned to the gynaecology clinic for management according to an existing clinical protocol (Supplemental Figure 1). Criteria for referring women to colposcopy included: cytology abnormality was more severe than ASCUS regardless of HPV status, ASCUS with positive high-risk HPV test, HPV-16 and/or HPV-18 positive regardless of cytology status and hrHPV-12 others positive with abnormal cytology equal to or more severe than ASCUS (≥ASCUS). Repeat screening test at 12 months was done for women who were tested positive on HPV-12 others but negative cytology, and for women whose LBC was unsatisfactory.
All colposcopies were performed by trained gynaecologist colposcopists. Cervical abnormalities identified on colposcopy were biopsied for histopathological examination. The final diagnosis of CIN and invasive cancer was based on the histology of cervical biopsies (cervical punch biopsies or excisional procedures) and hysterectomy specimens where applicable. The results were reviewed by gynaecologic histopathologists. The end points of diagnosis for the current analysis were CIN2+ and CIN3+.
Outcome Measures
Clinical efficacy for the present study was measured by detection rate of abnormal screening results in terms of HPV positivity and/or cytology changes equal to or more severe than ASCUS, discharge rate of screened women to routine program, referral rate to colposcopy, rates of CIN2+ and CIN3+, and number of colposcopies performed for detection of one case of CIN2+ and CIN3+.
Data Analysis
Two analyses were performed. Analysis-1 included women from the entire cohort of women from 25 to 69 years old (Total cohort). The results described the baseline characteristics of the screened population in Singapore as captured in the database. Analysis-2 aimed to determine the impact of the age of women on the efficacy of screening tests. Comparison of clinical efficacy of the two screening tests was made between the 25-to-29 years old and older women who were sub-grouped into 30-to-44 years old and 45-to-69 years old (Supplemental Figure 2).
Statistical Analysis
The characteristics of the clinical outcomes were dichotomous: presence or absence of abnormal screen results, yes or no for discharge to routine screening schedule, yes or no for colposcopy referral, and presence or absence of CIN2+. The data for the two screening methods were obtained from the same group of subjects. The analysis was performed for each of these parameters for women below 30 years old on the null hypothesis that there was no difference in the frequency of individual parameters between the HPV testing and LBC. McNemar’s Chi-square testing for paired variables was conducted on SPSS statistical package.
For the comparison of parameter between age groupings, significance of difference was tested with Pearson’s Chi-square statistics using SPSS.
Results
The Analysis-1: Background Information on the Database
The database included 6398 women, mean age 44.2 years old (95% confidence interval [CI] 22.1-66.3 years). Of these, 5733 (89.6%) were women aged 30 years or older.
The initial screening test detected HPV DNA in 503 women (7.9%, 95% CI [7.2-8.6]). The frequency distribution of HPV detection of all the three subtype groupings showed a declining trend throughout the age spectrum of women from 25 years old to 69 years old (Figure 1). Detection Rate of HPV DNA by Age of Women.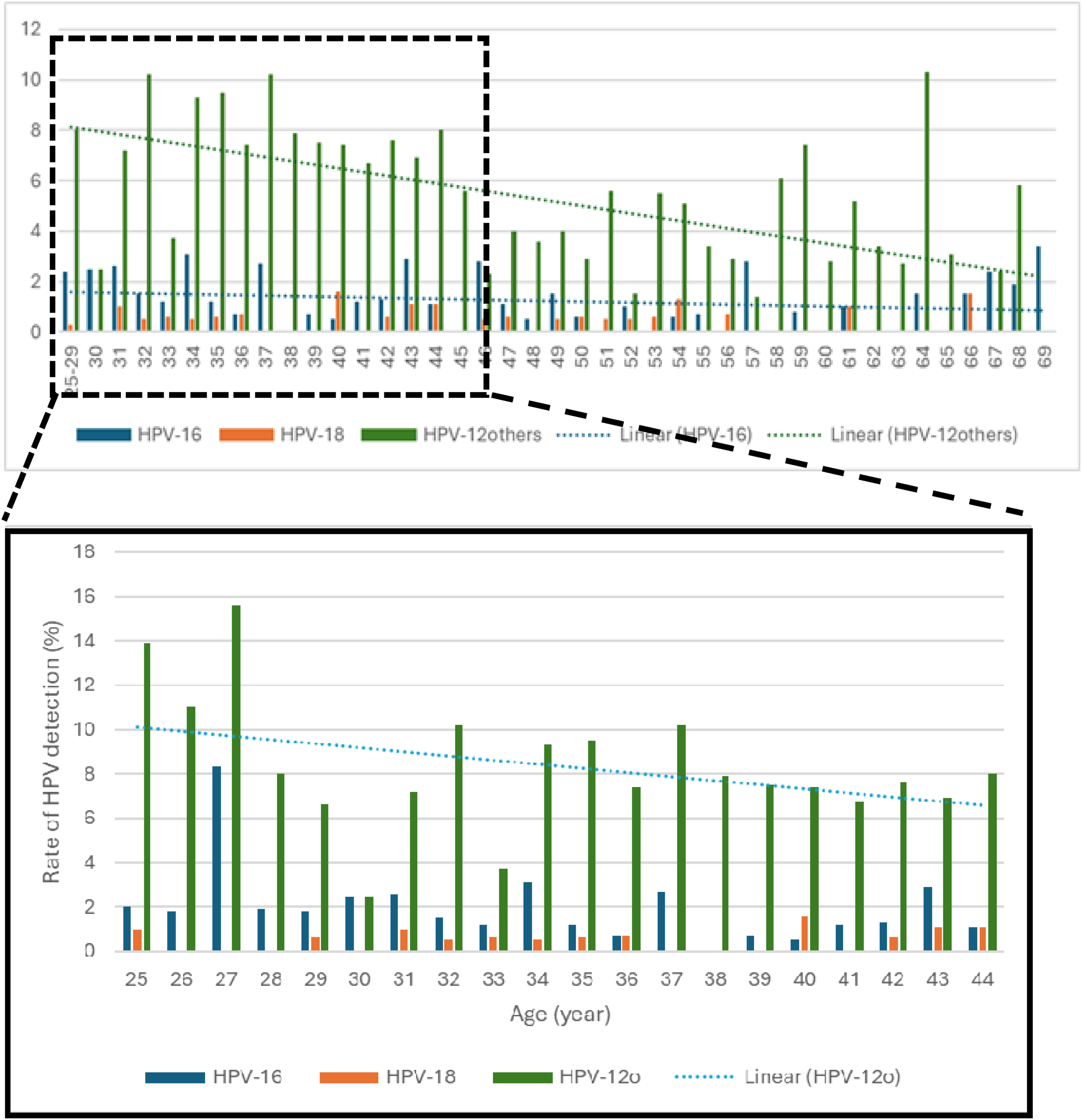
Detection Rate of CIN2 and CIN3+ Lesions of the Cervix Compared by Screening Methods.

The discharge rate of screened women to routine interval screening was based on the negative screening results. For the all-age group, the discharge rate by primary LBC screening was 92.5% (95% CI [90.00-94.72]), compared to primary HPV screening (92.2%, 95% CI [89.77-94.48]). The difference between the two modes of screening was indiscernible. Similarly, the rate of immediate colposcopy referral did not show any significant difference between primary cytology (4.3%, 95% CI [4.05-5.14]) and primary HPV screening (4.0%, 95% CI [3.54-3.85]). The number of colposcopies needed per case of CIN2+ detected was 6.03 for primary cytology screening and 7.0 for primary HPV screening. The difference was not significant statistically (
The Analysis-2 – Comparison of Women Below and Above 30 Years Old
Comparison of HPV Positivity in Primary Screening Between Women Younger and Older Than 30 Years Old.

aChi-square statistics.
Notable, the stark difference in HPV detection rate between women below 30 years old and women aged 30-to-69-years old was due to a remarkably low HPV detection rate (5.2%). Indeed, the lower overall rate of HPV detection among women aged 30-to-69 years old was due to a lower prevalence of HPV-12 others subgroup of HPV among women above 44 years old. (Supplemental Figure 3)
Rate of Discharge to Routine Interval Screening: Comparison of LBC and HPV Screening Methods.

aComparison between LBC and HPV screening within age group: McNemar’s Chi-square test. **Comparison between age groups by Chi-square statistics for LBC and HPV screening individually.
Comparison of clinical efficacy of primary HPV testing and LBC based screening:
(i) Rate of discharge to routine screening after the initial screening
The discharge rate of women to routine interval screening based on negative initial screening test was summarized in Table 3. For women between 25-to-29 year old, the discharge rate was 89.5% by LBC and 87.4% by HPV testing. McNemar’s test statistics was 2.1125,
(ii) Colposcopy referrals:
Comparison of Rate of Immediate Colposcopy Referral Between Cytology and HPV Screening.

aComparison between LBC and HPV screening within age group: McNemar’s Chi-square test. **Comparison between age groups by Chi-square statistics for LBC and HPV screening individually.
(iii) Detection of CIN2+ and number of colposcopies needed per case of CIN2+ detected:
High-grade lesions of the cervix were more prevalent in younger women between 25-29 years old and 30-44 years old compared to women 45 years or older (Table 1).
Comparison of Number of Colposcopies Per Case of CIN2+ Detected by Method of Screening.

aComparison between cytology and HPV methods of screening with the age group. McNemar’s Chi-square statistics.
Discussion
We presented a real-world experience comparing the effectiveness of primary cytology and primary HPV testing in cervical cancer screening in the younger population. Our data found that the clinical efficacy of primary HPV screening for women of 25-to-29 years old was similar to LBC screening for screening discharge rate (87.4% vs 89.5%) and number of colposcopies needed for detection of a case of CIN2+ (7.0 vs 5.0). Of clinical importance, our data showed that the rate of discharge to routine screening for women below 30 years old was similar for primary cytology-based and primary HPV-based screening protocols. We found that the rate of cytology abnormality and HPV positivity were equally higher among 25-to-29 years old women compared to older women. Consequently, our data showed a higher colposcopy referral rate for both screening tests among the younger women compared to women above 44 years old. The higher number of CIN2+ among the below and above 44 years old lead to a reverse trend of 2-fold higher number of colposcopies per case of CIN2+ among women above 44 years old.
HPV prevalence in our study was higher in younger women aged 25-29 years old compared to women 30 years and older. Our findings concurred with a consistent observation world-wide that HPV prevalence is higher among the young and declines with increasing age.26–28 We would like to emphasize that the prevalence of HPV detection was, in fact, similar between the 25-to-29 years, 30-to-34 years, 35-to-39 years and 40-to-44 years age-groupings. This observation highlights the disproportionate emphasis of higher prevalence of HPV detection in 25-to-29 years old when comparing women below and above 30 years old in general. The lower prevalence of HPV for 30-69-years-old women arose principally from the very low prevalence of HPV for women above 44 years old, a fact not well-recognized and not adequately addressed. This has led to a blanket recommendation of restricting HPV screening to 30 years old or older women.
We could not emphasize more that the high prevalence of HPV detection was reflected in our observed higher prevalence of CIN2+ among women aged 25-to-44 years compared to women 45 years or older. A large contribution of case load of CIN2+ from young women have been reported widely.19,29,30 These findings strongly question the validity of the belief that HPV infection in the younger women is a mere transient infection that can be managed in a lesser stringent manner than that in the older aged group of women. Instead, it provides strong evidence for active screening of young women with a test of high-performance.
Considering the high prevalence of high-grade lesions in the young women, our data showed that colposcopy referral and number of colposcopies performed for detection of one case of CIN2+ was in fact similar for the age groupings of 25-to-29 years and 30-to-44 years. In contrast, the comparatively lower prevalence of CIN2+ in women above 44 years old was associated with two-fold higher number of colposcopies needed for one case of CIN2+ compared to the younger women. In fact, our data showed that, compared to cytology-based screening, the numbers of colposcopies required to detect a case of CIN2+ did not significantly increase with our current protocol of primary HPV-based screening regardless of age groups. This dispels the myth that transiting to primary HPV screening in younger women would increase the burden on colposcopic evaluation services. It is also reasonable for one to speculate that there would be no increased rate of surgical intervention among younger women by applying HPV testing.
Some countries have transited to primary HPV-based screening protocols for their younger women. For example, Australia, 15 the United Kingdom 13 and Sweden 14 have implemented primary HPV screening in 2014, 2019 and 2022 respectively, which includes younger women aged 25 to 29 years old in Australia and the United Kingdom, while in Sweden HPV screening begins at 23 years old. In addition, the American Cancer Society who initially recommended primary cytology has updated their recommendations to primary HPV screening as the preferred choice in 2020, 12 after HPV screening received the United States Food and Drug Administration (FDA) approval. Moreover, in 2018, the USPSTF 9 has also included primary HPV alone, primary cytology alone and co-testing as equal options. International guidelines recommend that surgical excisional procedures are only undertaken in women with high-grade lesions. Ablative procedures for high-grade CIN in young women has been shown to be effective with a low risk of recurrence.31,32 Ablative procedures have advantages over excisional procedures by minimizing impact on future pregnancies and easy implementation in the outpatient setting. 32 With high rates of regression of CIN2+, 33 an alternative approach that the authors recommend is a proactive screening, diagnosis, followed by active surveillance of these young women with CIN2+ as these women remain at risk of developing cancer. 34
Strengths and Weaknesses of Study
The strength of the study is that it was based in a single institution which ensured the completeness and accuracy of the clinical information, and consistency of quality of colposcopic evaluation, and of cytopathological and histopathological examination and reporting. Moreover, data was collected prospectively. One limitation of the study is that the study period was prior to the introduction of school-based HPV vaccination in Singapore in 2019. The prevalence of HPV and proportion of the subtypes may differ in the future with mass HPV vaccination and may lead to changes in the performance indices and clinical efficacy of primary HPV screening in the younger population. In addition, the results of the study are applicable to Singapore and regions with similar epidemiological characteristics and HPV prevalence.
Conclusion
The findings of this study confirmed that clinical efficacy of primary HPV testing in women between 25 and 29 years old was comparable to cytology in cervical cancer screening for settings similar to Singapore. Furthermore, the efficacy was comparable to older women between 30 and 44 years old who are recommended primary HPV-based screening. Our data provided strong support for incorporating women aged 25 to 29 years old into primary HPV-based screening program in line with the rest of older eligible women in Singapore. A single screening test for all women at risk of cervical cancer would streamline the execution of mass screening programmes and the longer screening interval in HPV screening is a much-desired benefit of HPV testing over morphology-based LBC screening.
Supplemental Material
Supplemental Material - Comparison of Clinical Efficacy of Liquid-Based Cytology and High-Risk Human Papillomavirus Testing With Partial Genotyping in Cervical Screening of Women Below 30 Years Old
Supplemental Material for Comparison of Clinical Efficacy of Liquid-Based Cytology and High-Risk Human Papillomavirus Testing With Partial Genotyping in Cervical Screening of Women Below 30 Years Old by Joella Xiaohong Ang, and Sun Kuie Tay in Cancer Control
Footnotes
Acknowledgements
The authors would like to thank the doctors, nurses and staff of the Department of Obstetrics and Gynaecology of Singapore General Hospital for their contributions to the study.
Ethical Approval
Waiver of consent was granted for this study by the hospital’s ethics committee (Centralized Institutional Review Board 2016/2385).
Author Contributions
SKT conceived and designed the study and edited and reviewed the manuscript. JXA collected and analysed the data, drafted, edited, reviewed and prepared the manuscript. Both authors have read and approved the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Our data is available upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
