Abstract
Plain Language Summary
This study looked at how well Asian Americans followed through with at-home mt-sDNA tests for colon cancer screening. Researchers used data from a large database of patient, focusing on those who were using the test for the first time. Overall, about 71% of patients completed the test; however, those with Medicaid had the lowest adherence rates (61%). In addition, disparities in adherence persisted across different payer types, sociodemographic groups, and geographic regions. Targeted intervention strategies are warranted to address disparities in colon cancer screening and ensure adherence to mt-sDNA testing, as they are crucial for the early detection and treatment of colon cancer.
Introduction
Asian Americans are the fastest-growing minority group in the US, nearly doubling in number between 2000 and 2019. 1 In 2022, the US had an estimated 24.6 million Asian residents, 1 and projections indicate this population will surpass 46 million by 2060.1,2 Among Asian Americans, cancer is the leading cause of death, and CRC is the second most common cancer in men and woman combined.3,4 Mortality rate for Asian Americans is lowest mortality among racial and ethnic groups at 8.9 per 100 000. 5 Overall, CRC rates in Asian Americans are lower than other ethnic groups but the incidence of distant metastasis at the time of diagnosis is higher than other ethnic groups. 6 In addition, National Cancer Institute SEER 17 data suggests that over half of Asian Americans and Pacific Islanders were diagnosed with regional or distant CRC. 7 Asian Americans have significantly lower CRC screening rates compared with White individuals and have the lowest screening rates among all minority groups which may explain the lower rates of diagnosis but the more advanced stage at diagnosis.6,8-14 For example, in 2021, only 50% of Asian American individuals aged 45 years and older were current with CRC screenings, compared to 61% of both White and Black individuals. 8 This lower screening rate among Asian Americans is especially problematic given that the mortality rate of CRC significantly declines with regular screening and subsequent treatment. 15 Taken together, these findings suggest that the burden of CRC may be demonstrably reduced in the Asian American community through improved screening.
The multitarget stool DNA (mt-sDNA) test is one of the non-invasive, stool-based CRC screening tools included in several guidelines, including those from the American Cancer Society, the Joint Asian Pacific Association of Gastroenterology, and the American College of Gastroenterology.16-18 Although adherence (defined as the return of a valid stool specimen by the patient for testing) to many stool-based screenings is generally suboptimal, mt-sDNA screening has achieved relatively high adherence rates.19-22 Across studies, adherence to mt-sDNA orders for CRC screening ranged from 51% of Medicaid patients to 72% of traditional Medicare patients.20-22
The current investigation builds on previous research by examining the adherence rates of first-time mt-sDNA testing among individuals of Asian American descent across various healthcare payers in the US. This analysis also explores the sociodemographic factors associated with patient adherence to mt-sDNA testing, which can inform strategies to enhance CRC screening uptake and reduce the burden of CRC in the rapidly growing Asian American population.
Method
Data Source
The current study linked two data sources. The first was the Komodo Research Data (KRD) + MapEnhance Komodo Lab database, a comprehensive and representative administrative claims database that includes closed claims data from more than 165 million lives across more than 150 payer sources. The second was the Exact Sciences Laboratories LLC (ESL) database, which provides complete information on mt-sDNA orders and includes data on laboratory test results, patient outreach, health systems, payers, and basic demographic information. Linking KRD and ESL data created the KRD-ESL dataset, which offers complete information on mt-sDNA testing and results, along with comprehensive demographic and health information from the KRD data. All data were de-identified and compliant with the Health Insurance Portability and Accountability Act (HIPAA); this study qualified as exempt research.
Study Design and Sample Selection
This study included individuals self-identified as Asian American who were first-time users of mt-sDNA testing and were shipped a collection kit between 2017 and 2023. Patients were greater than 45 years old and were covered by a commercial payer, managed care organization, Medicare, Medicaid, or Medicare Advantage insurance. We defined the index date as the date of the first prescription order for the CRC screening test/procedure (mt-sDNA) received within an index year (2017 to 2023). Eligible patients were continuously enrolled in a health plan for a minimum of two years, including one year before (baseline period) and one year after (follow-up period) the index date. During the baseline and follow-up periods, eligible patients were required to be of average risk for CRC, defined as having no prior diagnosis of CRC and no high-risk conditions, including inflammatory bowel disease, familial polyposis syndromes, colonic adenomas (based on their existing claims), in addition to no documented family history of CRC or prior colectomy.
Variables and Outcomes
The primary outcome of this study was adherence rate to mt-sDNA screening, which was defined as the percentage of eligible patients who completed and returned the test kit, with a valid test result obtained by ESL within 365 days of the initial shipment date. Patient demographic characteristics included age group, sex, ethnicity, preferred language, payer type, geographical region, urban/rural classification (coded using the patient’s zip code and was mapped using the ‘2010 Rural-Urban Commuting Area Codes’), 23 ordering provider specialty, and outreach group. We defined four outreach groups by the type(s) of overall communications patients received in addition to the standard outreach (welcomes, reminders, and notifications of test result availability via letters and phone calls). The outreach groups included: (1) fully digital short message service (SMS) plus email, which received both additional SMS and email reminders; (2) digital SMS, which received additional SMS reminders only; (3) digital email, which received additional email reminders only; and (4) no digital communication, which received only the standard outreach with no additional SMS texts or emails. These different outreach groups were included in order to determine what model of communication might be the most effective in promoting adherence.
Statistical Analyses
Baseline by payer type is presented as frequencies and percentages. We reported the primary outcome, adherence rate to mt-sDNA, by payer type and made global comparisons within each payer type chi-square tests. Logistic regression examined factors associated with adherence (yes/no); independent variables included age group (reference group = 45-49 years), sex (reference group = male), payer type (reference group = Commercial insurance), urban/rural classification (reference group = metropolitan), provider specialty (reference group = gastrointestinal), outreach group (reference group = no digital), and preferred language (reference group = English). All statistical analyses were performed using SQL version 8.45.1 and SAS version 9.4. The reporting of this study conforms to STROBE guidelines. 24
Results
Patient Characteristics
Patient Characteristics by Payer Type.
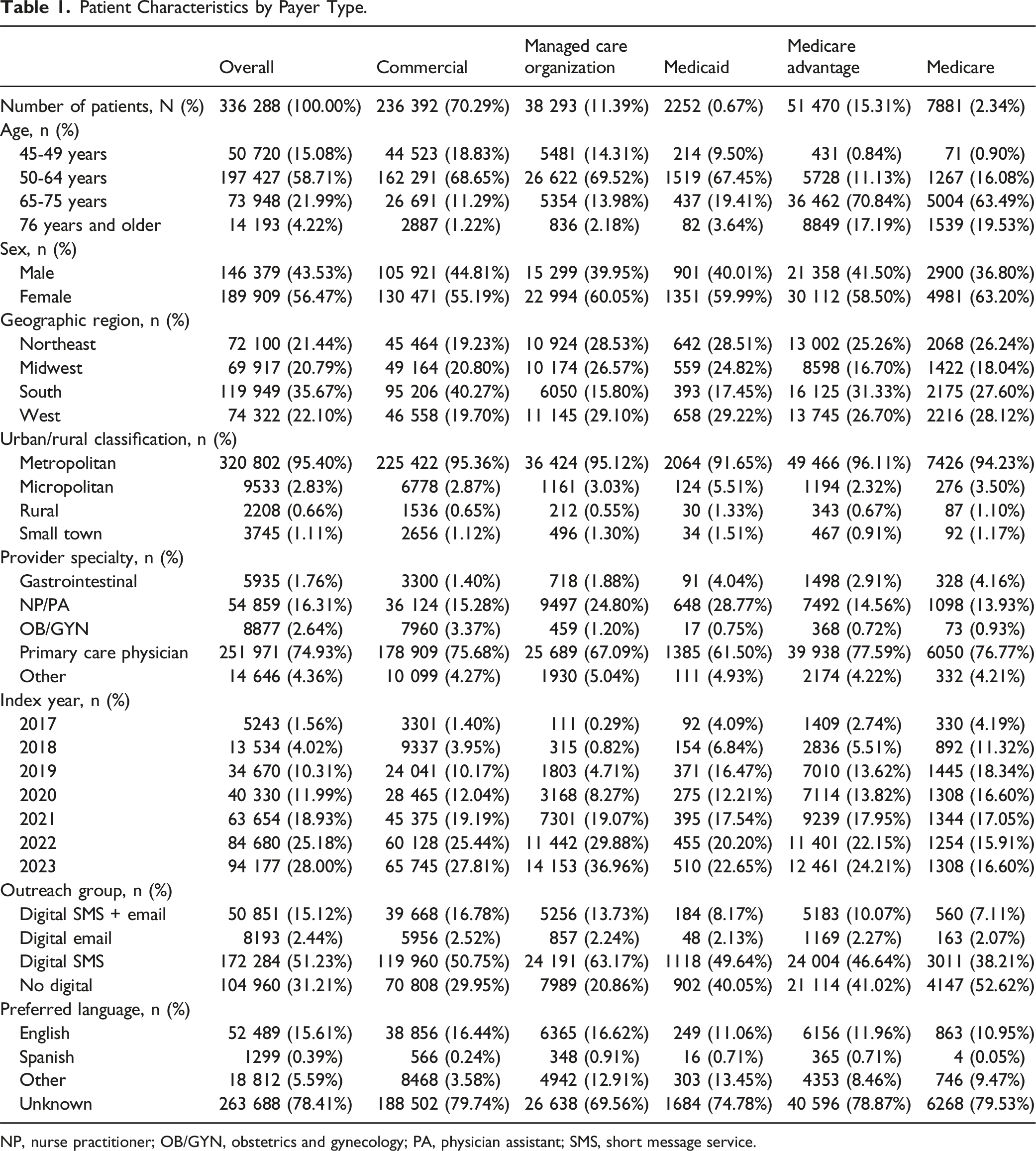
NP, nurse practitioner; OB/GYN, obstetrics and gynecology; PA, physician assistant; SMS, short message service.
Adherence
Adherence by Payer Type. a

NP: nurse practitioner; OB/GYN: obstetrics and gynecology; PA: physician assistant; SMS: short message service.
aAdherence: Percentage of eligible patients who completed and returned their test kit, with a valid test result obtained within 180 days of the initial shipment date.
Predictors of Adherence
Logistic Regression Predictors of Adherence. a

NP: nurse practitioner; OB/GYN: obstetrics and gynecology; PA: physician assistant; SMS: short message service.
aAdherence: Percentage of eligible patients who completed and returned their test kit, with a valid test result obtained within 180 days of the initial shipment date.
Discussion
Among Asian American patients aged 45 years and older who were new to mt-sDNA testing, the overall adherence rate was 70.9%, indicating a high level of patient adherence to the mt-sDNA modality of CRC screening. In contrast, the National Health Interview Survey (NHIS) previously reported that across all racial/ethnic groups Asian Americans had the lowest CRC screening rate at 50%, compared with 61% among White individuals. 8 Additionally, only 10% of Asians aged 45 years and older had undergone a fecal occult blood test (FOBT) or fecal immunochemical test (FIT) within the past year, or mt-sDNA test within the past three years. 8 The results of our study suggest that mt-sDNA testing is a viable option for improving CRC screening in the Asian American population.6,8-14 Increasing the rates of screening is particularly important given the higher rate of advanced CRC at the time of diagnosis.
Adherence rates to mt-sDNA testing varied significantly across payer types, with Medicaid patients having the lowest rate of adherence and Medicare patients the highest. This study also found that Medicaid coverage was associated with a 40% lower likelihood of adherence to new mt-sDNA screening compared to commercial insurance. This trend is consistent with previous studies, which have also reported the lowest mt-sDNA adherence among Medicaid patients compared to other payer types regardless of racial group.20-22 Medicaid recipients are primarily low-income, which can contribute to disparities in healthcare access, socioeconomic status, and the availability of resources to support adherence among these populations. 25 However, the number of individuals covered by Medicaid in the current analyses was very small, comprising less than one percent of the overall sample as such these results will need to be further explored in larger cohorts of Medicaid recipients.
Adherence rates also varied by demographic factors, such as age, sex, and urban/rural classification. The logistic regression results suggested that older patients and those living outside metropolitan areas were significantly more likely to return their mt-sDNA tests, while female patients were slightly less likely to return their test. This is consistent with a previous study that found adherence increased with age and was highest among rural patients, with similar adherence rates between males and females, slightly favoring males. 20 The American Cancer Society also reported that CRC screening was the lowest among patients aged 45-49 years (20%) while the screening rate was the highest among patients aged 65-74 years (80%). 6
The current study also found that patients under the care of GI providers had the highest adherence rates at 82% overall and were twice as likely to be adherent compared to those under non-GI providers. This may be due to the expertise of GI providers and their likelihood of offering more targeted patient education and follow-up. Another explanation for this trend is selection bias: patients who receive subspeciality care may be more adherent to all health maintenance recommendations. However, only a small fraction of our study population was seeing a GI provider (<2%); most patients were seeing PCPs, who had the second-highest adherence rate at 72% overall among provider specialties. This suggests that PCPs also play an important role in increasing CRC screening uptake. Our findings are similar to a previous study, which also reported that participants with mt-sDNA tests ordered by GI specialists had the highest adherence rate, at 78.3%, while those ordered by primary care clinicians had an adherence rate of 67%. 20 However, given that Asian American patients have the lowest likelihood of having a PCP among racial/ethnic groups in the US, and the proportion of all Americans with an identified source of primary care is declining, 26 our results also highlight the need to address barriers to accessing primary care. The low rate of Asian American patients having a PCP may help explain the discrepancy between historical CRC screening adherence and the high rate of adherence in our study; Asian American patients may be willing to complete CRC screening but are not offered it due to a lack of a PCP. Consumer-initiated ordering of mts-DNA test facilitated by telehealth provider may help close this gap.
Finally, digital outreach with both SMS and email increased the likelihood of screening adherence by 30% compared with no digital outreach. This finding supports recent research indicating that electronic communication significantly contributes to higher screening rates, particularly among Asian patients. 27 The significant impact of digital interventions on improving adherence has also been reported in a systematic review and meta-analysis of randomized controlled trials, which showed that digital interventions were 31% more likely to promote CRC screening uptake compared to no intervention or usual care, highlighting the continued importance of patient connectivity. 28 Future research should focus on developing tailored digital interventions that address the specific needs and preferences of different demographic groups, particularly those with historically low screening rates, such as Asian populations.
Limitations
While the results of this study provide further insight on adherence to mt-sDNA testing in Asian Americans they should be considered along several caveats. First, this study only included new mt-sDNA users who were continuously enrolled in a health plan for a minimum of two years, which may introduce selection bias as repeat mt-sDNA users or patients with unstable insurance status were not considered. Second, this study did not include other factors that may influence adherence rates, such as education level, family income, and comorbidities, and may not fully address the barriers to adherence and the factors that drive patient behavior in CRC screening. Third, the large number of patients for whom preferred language was unknown is limiting; however, even with the reduced sample size a preference for Spanish was a significant predictor of adherence. Patients who preferred Spanish may belong to the growing population of individuals who identify as both Latino and Asian American or Pacific Islander. 29 Census data indicates that an estimated 3% of Asian Americans residing in the United States identify as Asian Latino or Asian Hispanic.29,30 Fourth, the current study examined Asian Americans as a single group and did not account for ethnic and cultural heterogeneity. The incidence of CRC differs among Asians with Japanese Americans and Native Hawaiians having higher rates than white Americans. 6 Additionally, previous studies have found that CRC screening rates vary between Asian American subgroups, with Filipinos having the highest screening rates (55.0%) and Asian Indians (48.6%) the lowest. 31 Fifth, this study benefited from a very large sample which increased the statistical power and therefore caution is suggested when differentiating between statistical and clinical significance. Sixth, the study did not examine whether patients that received a negative test result underwent repeat screening. Seventh, race/ethnicity is missing from approximately 20% of the Komodo database and therefore some Asian American patients may have not been included in the current analysis limiting the generalizability of the results. Finally, the study did not include a comparison group such as patients using fecal immunochemical or guaiac-based fecal occult blood tests.
Conclusion
In this study, Asian American individuals demonstrated high adherence rates to mt-sDNA testing among new users, especially when supported by digital outreach and specialist care. This suggests that mt-sDNA testing, a non-invasive CRC screening tool, is a viable option for Asian American patients who have demonstrated suboptimal adherence rates to CRC screening in previous studies. However, disparities in adherence persist across different payer types, sociodemographic groups, and geographic regions. Targeted intervention strategies are warranted to address disparities in CRC screening and ensure adherence to mt-sDNA testing, as they are crucial for the early detection and treatment of CRC.
Footnotes
Statements and Declarations
Author Contributions
All authors have made substantial contributions to the conception or design of the study, or the acquisition, analysis, or interpretation of data, drafting the manuscript and revising it critically for important intellectual content, and have provided final approval of this version to be published and agree to be accountable for all aspects of the work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Exact Sciences Corporation, Medical writing support was provided by Jason Allaire, PhD of Generativity Health Economics and Outcomes Research and wad funded by Exact Sciences Laboratories LLC.
Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MG, MC, AL, RK,ABO, and PL are employees of and own stock in Exact Science Corporation. MF directs the University of Michigan Center for Value-Based Insurance Design. He reports providing consulting services to AbbVie, CareFirst Blue Cross Blue Shield, Centivo, Clover Health, Community Oncology Association, Covered California, Elektra Health, EmblemHealth, Employee Benefit Research Institute, Exact Sciences, GRAIL, Health[at]Scale Technologies, HealthCorum, MedZed Inc., Merck and Company, Mother Goose Health, Phathom Pharmaceuticals, Sempre Health, Silver Fern Healthcare, Soda Health, US Department of Defense, Virginia Center for Health Innovation, Wellth, Yale-New Haven Health System; holding equity interest in Health[at]Scale Technologies, HealthCorum, Mother Goose Health, Sempre Health, Wellth Inc., and Zansors; receiving research support from the Agency for Healthcare Research and Quality, West Health Policy Center, Arnold Ventures, National Pharmaceutical Council, Patient-Centered Outcomes Research Institute, Pharmaceutical Research and Manufacturers of America, the Robert Wood Johnson Foundation, the state of Michigan, and the Centers for Medicare and Medicaid Services; serving as co-editor for the American Journal of Managed Care; and maintaining a partnership at VBID Health. MD is an Associate Professor of Medicine at Duke University and has no financial interests to disclose.
Data Availability Statement
The data that support the findings of this study are available from Exact Sciences Laboratories LLC. Restrictions apply to the availability of these data, which were used under license for this study. Data are available from the authors with the permission of Exact Sciences Laboratories LLC.
