Abstract
Introduction
The objective of the current study was to examine the impact of Spanish-language patient outreach and navigation services on adherence to initial colorectal cancer (CRC) screening with multitarget stool DNA (mt-sDNA) testing in a predominantly Spanish-speaking patient population receiving care at Federally Qualified Health Centers (FQHCs).
Method
This study included patients aged 45 years or older who identified as Hispanic from FQHCs in a California Health System who were new to mt-sDNA testing and shipped a Cologuard® collection kit between 10-1-2022, and 1-1-2024. Patient outreach was provided only in English prior to 1-22-2023 (pre-intervention period). From 1-23-2023, onward, patient outreach was offered in either English or Spanish based on the patients’ preferred language selection (post-intervention period). Patients were classified into two subgroups: Spanish language preference (SLP) or non-Spanish language preference (NSLP). It was hypothesized that adherence would be greater in SLP patients when patient outreach was provided in Spanish compared to the NSLP.
Results
The final sample comprised 20 341 Hispanic patients who met the study criteria, comprising 15 702 patients with SLP and 4639 with NSLP, stratified across pre- and post-intervention periods. Overall, adherence to initial mt-sDNA testing within 180 days following the index date was 51.4% for SLP patients and 41.3% for NSLP patients, with a significant post-intervention improvement observed after the intervention for SLP patients (47.1% to 52.7%, P < .001), compared to a non-significant improvement (40.7% to 41.4%, P = .713) among NSLP patients.
Discussion
Following the introduction of Spanish-language patient outreach, adherence to mt-sDNA testing improved significantly among SLP patients. The preference of Hispanic individuals for stool-based tests, combined with the non-invasive nature of mt-sDNA testing, supports its suitability as a CRC screening option for this population.
Introduction
The Hispanic population is one of the fastest growing minority groups in the United States (US), rising from 9.6 million people in 1970 to 65.2 million in 2023 1 and is projected to reach 90.5 million by 2050 2 and 111 million by 2060. 3 One particular health concern in this growing population is colorectal cancer (CRC), which, in individuals of Hispanic descent, is the third most commonly diagnosed cancer and second leading cause of cancer death overall. 4 It is estimated that there were 17 100 new cases of CRC and 5300 deaths due to CRC among the U.S. Hispanic population in 2024. 4
Routine screening for CRC can help detect cancer early, which can in turn facilitate treatment and increase chances of survival.5-7 Unfortunately, the CRC screening rates for Hispanic individuals has been found to be significantly lower than for non-Hispanic White individuals, with some studies suggesting that less than half of eligible Hispanic adults receive CRC screening8-12 compared to 59%-72% of the eligible US population.13-15 Consequently, CRC in Hispanic individuals is often diagnosed at a more advanced stage, leading to increased healthcare burden, higher mortality rates, and exacerbating health disparities.8,16-18
Previous work to identify the antecedents of lower CRC screening in Hispanics have pointed to a number of potential factors including distrust in the healthcare system 19 and cultural beliefs about masculinity, 20 as well as limited knowledge of CRC and CRC screening.21,22 However, one of the most salient barriers to CRC screening in Hispanic individuals in the US is a lack of English proficiency,23-28 given that an estimated 28% of Hispanics have limited English proficiency (LEP) and 27% are Spanish dominant.29,30 One factor contributing to CRC knowledge barriers and lower screening completion rates among Spanish speakers is suggested by a recent study that found instructions for stool-based CRC testing were typically not provided in Spanish nor were language translation services offered when scheduling a colonoscopy. 23
Research has shown that the modality of CRC testing is critical to increasing screening rates overall. 15 Furthermore, Hispanic patients have been found to be up to 82% more likely to undergo stool-based CRC screening than non-Hispanic Whites. 31 One such stool-based testing option for CRC screening in individuals at average risk is the multi-target stool DNA (mt-sDNA; eg, Cologuard) test, a non-invasive, at-home screening tool recommended in several guidelines, including those of the American Cancer Society and the American College of Gastroenterology.32,33 Of the available at-home, stool-based testing options for CRC screening, the mt-sDNA test has the advantage of being recommended once every three years when compared to annually for the fecal immunochemical test (FIT) or the high-sensitivity guaiac-based fecal occult blood test (gFOBT).
The objective of the current study was to examine the impact of Spanish-language patient outreach on adherence to initial CRC screening with mt-sDNA testing in a Hispanic patient population receiving care at Federally Qualified Health Centers (FQHCs). Nationwide, more than 35% of Hispanic individuals seek their healthcare services at FQHCs, 11 where only about 40% of the screen-eligible population treated in these centers are up-to-date with CRC screening overall. 14
Methods
Study Design and Sample Selection
This study included patients of Hispanic or Latino origin or descent aged 45 years or older from FQHCs in a California Health System who did not have a prior claim for mt-sDNA testing and who were shipped a Cologuard® collection kit between October 1, 2022, and January 1, 2024, from Exact Sciences Laboratories LLC (Madison, WI). The index date was the date of the first shipment on the first prescription order (CPT 81528) for the Cologuard test. Patients were required to be continuously enrolled in a health plan for a minimum of one year before and 180 days after the index date. Patients who had multiple orders or were returning patients were excluded. These data were de-identified and compliant with the Health Insurance Portability and Accountability Act (HIPAA); the current study was exempt from Institutional Review Board (IRB) review.
Patient outreach included welcome messages, reminders, and notifications of test result availability via letters and phone calls which all patients received. Patients were also assigned to one of two outreach channels either ‘full digital’ (short message service [SMS] plus email) if the referring health system provided both their phone number and email, or ‘partial digital, SMS only’ if the referring health system only provided the patents’ phone number. Patients who were shipped their mt-sDNA testing kit prior to January 23, 2023, were provided all of the above patient outreach in English (pre-intervention period) regardless of language preference. From January 23, 2023, onward, patient outreach and test navigation services were offered in either English or Spanish based on the patients’ preferred language selection (post-intervention period) determined by their medical records. To reiterate, in the pre-intervention period patients received all patient outreach in English as is normally provided to all patients. In the post-intervention period, the same material was simply provided in Spanish to patients that preferred Spanish.
In both the pre- and post-intervention periods, patients were classified into two subgroups based on their language preference: Spanish language preference (SLP) or non-Spanish language preference (NSLP). Therefore, in the pre-intervention stage both SLP and NSLP patients received patient outreach in English. At the postintervention stage patents in the SLP group received patient outreach in Spanish while NSLP received patient outreach in English.
Variables and Outcomes
The primary outcome of this study was adherence to initial mt-sDNA screening, which was defined as a completed and returned test kit, with a valid test result obtained by Exact Science Laboratories, LLC within 180 days of the shipment date. Specifically changes in adherence in the SLP group from pre- to post-intervention were compared to changes in the NSLP. It was hypothesized that adherence would increase in the SLP patients when they were given patient outreach and test navigation services in Spanish compared to the NSLP patients at both time points
In addition to language preference and outreach channel, patient demographic characteristics included age, gender, ethnicity, payer type, urban/rural classification, Social Vulnerability Index (SVI) score, 34 and median household income by ZIP code.
Statistical Analyses
Study measures and outcomes were summarized using counts and percentages for categorical variables and means with standard deviations (SDs) for continuous variables. Descriptive statistics were used to summarize unadjusted differences in patient characteristics at the index date as well as adherence between the pre-intervention and post-intervention periods separately within the SLP and NSLP subgroups. Logistic regression was used to examined predictors of adherence (yes/no) and tested whether providing patient outreach and instructions in Spanish during the post-intervention period significantly increased adherence in the SLP subgroup after controlling for demographic characteristics. All analyses were performed using R version 4.4.0 (The R Foundation for Statistical Computing, Vienna, Austria). The reporting of this study conforms to STROBE guidelines. 35
Results
Patient Characteristics
Patient Characteristics at Index Date.

Abbreviations: MCO: Manage Care Organization; SMS: Short Message Service; SVI: social vulnerability index.
aPatients who were shipped their testing kit prior to January 22, 2023 (patient outreach and test navigation services only in English).
bPatients who were shipped their testing kit from January 22, 2023, onward (patient outreach and test navigation services in either English or Spanish based on patient language preference).
Adherence Rate
Adherence Rate a Stratified by Language Preference.

Abbreviations: MCO: Managed Care Organization; SMS: Short Message Service; SVI: social vulnerability index.
aAdherence rate was calculated as the percentage of eligible patients who completed and returned the test kit, with a valid test result obtained within 180 days of the initial shipment date.
bPatients who were shipped their testing kit prior to January 23, 2023 (patient outreach and test navigation services only in English).
cPatients who were shipped their testing kit from January 23, 2023, onward (patient outreach and test navigation services in either English or Spanish based on patient language preference).
Significant differences in adherence rates across payer types were observed. Both SLP and NSLP patients covered by commercial health insurance demonstrated significant increases in adherence rates with SLP patients improving from 37.7% to 52.7% (P < .001) and NSLP patients increasing from 30.6% to 44.4% (P < .001). Lastly, among Medicaid-insured patients, SLP patients experienced a significant improvement in adherence from 19.6% to 32.4% (P = .022), while no significant differences were observed for NSLP patients.
Predictors of Adherence
Multivariable Logistic Regression Predicting Adherence a

Abbreviations: MCO: Manage Care Organization; SMS: Short Message Service; SVI: social vulnerability index.
aAdherence rate was calculated as the percentage of eligible patients who completed and returned the test kit, with a valid test result obtained within 180 days of the initial shipment date.
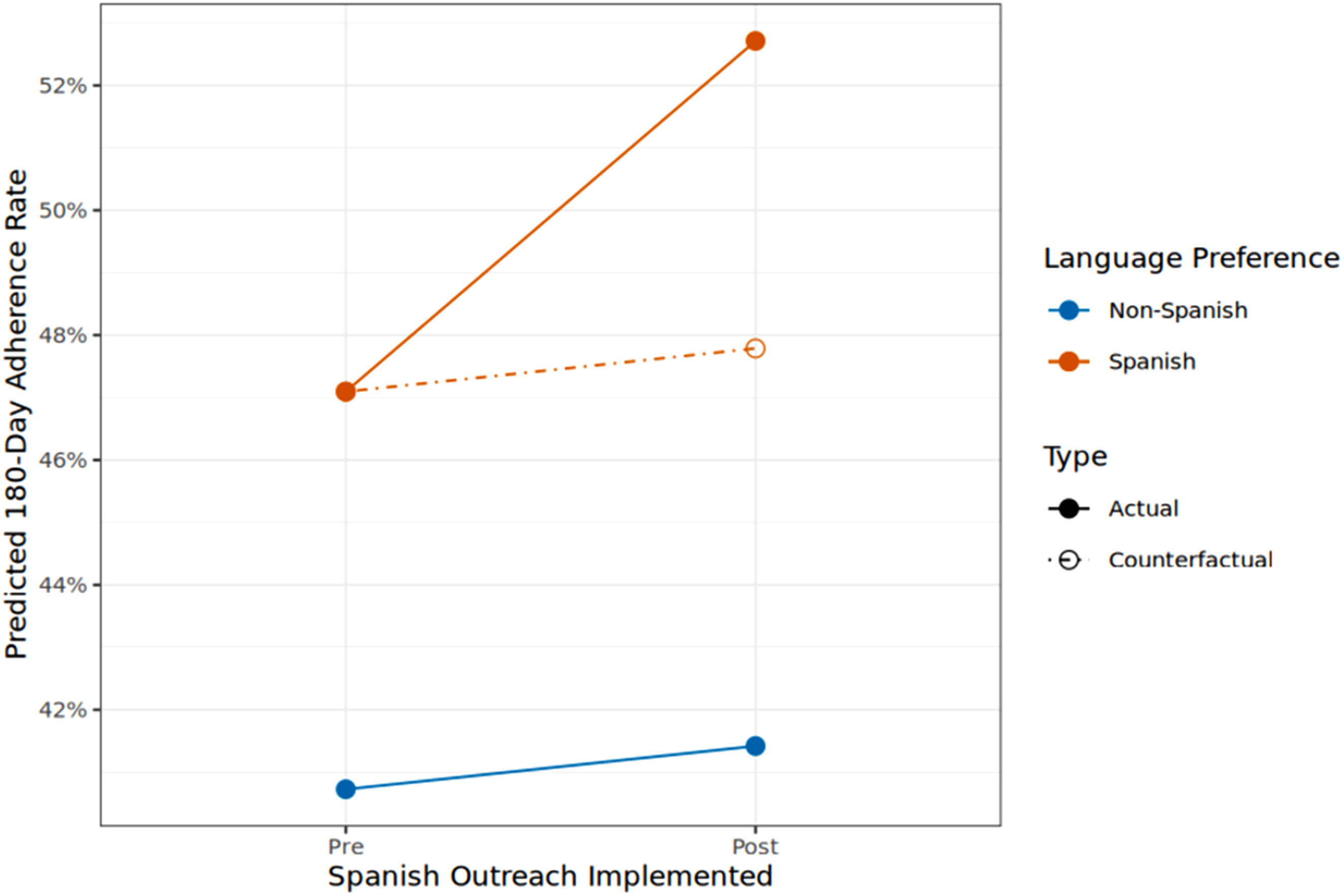
Predicted Adherence Rates Pre- and Post-Implementation of Spanish Outreach.
Discussion
This study evaluated the impact of Spanish-language outreach and multichannel navigation on adherence to CRC screening with mt-sDNA test within a Hispanic patient population receiving care at FQHCs. SLP patients adherence increased from 47% to 53% after patient outreach and test navigation services were provided in Spanish, while NSLP patients adherence remained the same. Coupled together, these findings suggest that the 6% increase in adherence in the SLP patients can be directly attributable to the patient outreach and test navigation services being offered in Spanish to this population of Hispanic patients.
The adherence rates observed in this study are consistent with findings from broader studies that have indicated that individuals with LEP, particularly within Hispanic communities often exhibit lower CRC screening rates compared to English-proficient populations.12,28,36,37 Notably, the improvement in adherence rates observed in our study among SLP patients was higher after implementation of the intervention compared with adherence rates reported in prior studies examining CRC screening among Hispanic individuals using FIT/FOBT tests.38,39 For example, a previous study examining FOBT usage among Hispanic adults living in Puerto Rico ranged from 20.5% in 2012 to 45.6% in 2020, while on the US mainland, FOBT usage ranged from 9.9% in 2012 to 16.7% in 2020. 12 This observed difference between may be partially attributable to less frequent testing intervals for mt-sDNA (every three years) compared to FIT/FOBT (annually), as well as the additional impact of tailored Spanish-language outreach in addressing barriers specific to LEP populations. Previous research has highlighted that individuals with LEP are more likely to encounter structural barriers in access to healthcare, including inadequate health insurance coverage 40 and ineffective patient-physician communication. 41 These language barriers can impede comprehension of screening recommendations and access to healthcare information, resulting in significantly lower CRC screening rates compared to English-proficient populations 36
Additionally, adherence rates after the intervention with Spanish language varied significantly by demographics and socioeconomic status, with SLP patients in the most socially vulnerable quartile demonstrating significant adherence improvement post-intervention. This observation is notable as language barriers often intersect with other forms of social disadvantage, creating compounding obstacles to healthcare access and utilization. 42 Age also influenced adherence, with SLP patients aged 50-64 years showing the largest improvement, while older SLP patients (65+ years) exhibited smaller, non-significant improvements. This trend observed may suggest that age-related factors, such as health literacy, mobility, or competing health priorities, may affect adherence differently across age groups. Limited health literacy has been associated with lower screening uptake, and interventions aimed at improving health literacy have been shown to enhance adherence to CRC screening 43 and older individuals often manage multiple health conditions, which may lead to competing health priorities and deprioritization of preventive services like CRC screening. Other characteristics associated with significant improvements in adherence included gender, with a similar trend for females, patients who received ‘partial digital, SMS-only’ for both SLP and NSLP patients, income status ($75K-$100K), as well as patients covered by commercial insurance and Medicaid.
Results from the multivariable logistic regression analyses in our study revealed important factors influencing adherence to mt-sDNA screening among Hispanic patients in this sample of FQHC patients. Older SLP patients (aged 65 years and above) were less likely to adhere compared to younger patients aged 45-49 years. The issue of cost as a barrier to adherence also played a significant role, as SLP patients covered by MCO, Medicare Advantage, and Medicare insurance tended to have higher adherence rates compared to Commercially insured patients, potentially reflecting better coverage or fewer out-of-pocket costs associated with these programs. In contrast, Medicaid-insured and self-pay patients exhibited lower adherence rates compared to the same reference group, suggesting that financial barriers remain a critical challenge for these groups despite targeted outreach efforts. Outreach methods also influenced adherence rates in our study; at the descriptive level patients engaged through partial digital outreach experienced increases in adherence from pre- to post-intervention and results from the logistic regression suggested that full digital outreach significantly improved adherence controlling for other factors. These findings have important implications for intervention design, highlighting the potential value of communication strategies in improving screening adherence, particularly among diverse populations. Lastly, the odds of adherence were 23% higher for SLP compared to NSLP in the post-intervention period (when SLP received outreach in Spanish) relative to the pre-intervention, reflecting the success of linguistically tailored outreach in addressing barriers specific to limited LEP populations.
Study Limitations
This study has a few limitations. First, the data were obtained from a single health system in California, which may limit the generalizability of the findings to other regions or populations, as it may lack detailed clinical information and potentially underestimate adherence rates. Specifically, we cannot ascertain whether any patients included in this study were experiencing minor symptoms attributed to colorectal cancer that prompted them to undergo mt-sDNA testing. Additionally, we do not know if those who did not complete the mt-sDNA test recently underwent other forms of screening for CRC that would render the mt-sDNA test redundant and unnecessary. Second, while the study relied on self-reported language preference, additional details regarding bilingualism or the frequency of Spanish use were not captured, which could influence the observed effects of Spanish-language outreach. Third, the study’s observational design precludes causal inference regarding the impact of the intervention, as other factors could have contributed to the observed improvements in adherence rates. Finally, though many differences between the two cohorts were controlled for in the logistic regression it is likely that inherent differences between cohorts may have contributed to bias in our results. A random controlled trial would help to further our findings by providing more insight to causality and providing a greater level of control.
Conclusion
Our findings highlight the significant role of Spanish-language outreach in improving adherence to CRC screening with mt-sDNA testing among a predominantly Hispanic population. Following the introduction of Spanish-language patient outreach, adherence to mt-sDNA testing improved significantly among SLP patients. Additional factors beyond the Spanish language intervention influencing adherence included older age, gender, payer type, and full digital outreach (SMS plus email). The preference of Hispanic individuals for stool-based tests, combined with the non-invasive nature of mt-sDNA testing, supports its suitability as a CRC screening option for this population. These findings demonstrate that by addressing language barriers through linguistically tailored communication and navigation services, healthcare systems can foster greater engagement and adherence to screening recommendations.
Footnotes
Acknowledgments
Medical writing support was provided by Jason Allaire, PhD of Generativity Health Economics and Outcomes Research and wad funded by Exact Sciences Laboratories LLC.
Ethical Statement
Author Contributions
All authors have made substantial contributions to the conception or design of the study, or the acquisition, analysis, or interpretation of data, drafting the manuscript and revising it critically for important intellectual content, and have provided final approval of this version to be published and agree to be accountable for all aspects of the work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Exact Sciences Corporation.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MG, TP, ABO, DB, and JK are employees if and own stock in Exact Science Corporation. JVRL and MD have no conflicts to report.
Data Availability Statement
The data that support the findings of this study are available from Exact Sciences Laboratories LLC. Restrictions apply to the availability of these data, which were used under license for this study. Data are available from the authors with the permission of Exact Sciences Laboratories LLC.
