Abstract
Introduction
This study evaluates the clinical outcomes, survival benefits, and toxicities of two moderate-hypofractionated radiotherapy (MHRT) patterns, 60 Gy in 20 fractions (60 Gy/20f) and 70 Gy in 28 fractions (70 Gy/28f), in early-stage prostate cancer patients.
Methods
This retrospective study analyzed data from 187 patients diagnosed between 2014 and 2023, using propensity score matching to ensure efficacy assessment accuracy. The primary endpoints reported were overall survival (OS) and disease-free survival (DFS), calculated using Kaplan-Meier analysis. Toxicity and side effects were evaluated using Criteria for Adverse Events v5.0, focusing on the urinary and gastrointestinal (GI) systems.
Results
After matching, each of the 60 Gy and 70 Gy groups included 73 patients. The median follow-up duration for all patients was 36.0 months. The OS rates for the 60 Gy and 70 Gy groups were 86.3% and 89.0%, respectively, with 3-year OS rates of 92.4% and 89.0% (P = 0.375). The 3-year DFS rates were 91.0% in the 60 Gy group and 81.0% in the 70 Gy group (P = 0.096), indicating no significant differences between the groups. The incidence of acute Grade 2 or higher urinary toxicities was comparable between the two groups (60 Gy group vs 70 Gy group: 9.6% vs 9.6%, P = 1.0), while the 70 Gy group demonstrated an advantage for late Grade 2 or higher toxicities (60 Gy group vs 70 Gy group: 12.3% vs 2.8%, P = .028). For the GI system, the incidence of acute toxicities was higher in the 60 Gy group, albeit not statistically significant (60 Gy group vs 70 Gy group: 11.0% vs 6.8%, P = .383), while late toxicities were equivalent between the groups (60 Gy group vs 70 Gy group: 1.4% vs 1.4%, P = 1.0).
Conclusion
Both MHRT fractionation patterns demonstrate comparable survival outcomes and toxicities in early-stage prostate cancer, suggesting MHRT’s viability as a primary treatment. The 60 Gy/20f pattern marginally favored survival, albeit not with statistical significance.
Keywords
Introduction
Prostate cancer is the second most prevalent cancer (after lung cancer) in men and the fourth most common overall.1,2 In 2020, there were more than 1.4 million new cases of prostate cancer. 2 Currently, the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines) for the treatment of localized prostate cancer primarily base their recommendations on a prognosis estimated through risk stratification. 3 The main treatment options are radical prostatectomy (RP), radiotherapy, and androgen deprivation therapy (ADT). As one of the standard treatment choices, radiotherapy primarily comprises two types: brachytherapy (BT) and external beam radiotherapy (EBRT). 4 Most patients undergo external beam radiation therapy.
Radiotherapy is divided into different fractionation patterns based on the single radiation dose. The conventional fractionation regimen consists of 1.8-2.0 Gy per fraction, whereas large fractionation is further subdivided into moderate-hypofractionated radiotherapy (MHRT) (2.4-3.4 Gy per fraction) and ultra-hypofractionated radiotherapy (UHRT) (≥5 Gy per fraction), according to the American Society for Radiation Oncology (ASTRO), American Society of Clinical Oncology (ASCO), and American Urological Association (AUA) hypofractionation guideline. 5 The α/β ratio reflects the fraction size sensitivity of tumor, with a lower α/β ratio suggesting greater sensitivity. 6 For most tumor tissues, the α/β ratio approximates 10 Gy, while for prostate cancer cells, characterized as late-responding tissues, the ratio is only about 1.5 Gy.7-10 This suggests that large fractionation may improve the therapeutic ratio, particularly in relation to late gastrointestinal (GI) and genitourinary (GU) toxicities. 11 Additionally, larger single radiation dose entails fewer fractions, thus facilitating a faster and more cost-effective EBRT course, and enhancing patient compliance. 12 There have been studies demonstrated that MHRT in prostate cancer could achieve higher biochemical control rates.11,13-15 However, studies comparing different dose fractionation patterns within MHRT in early-stage prostate cancer remain relatively limited.
In this study, we compared two different MHRT dose fractionation patterns, 60 Gy in 20 fractions (60 Gy/20f) and 70 Gy in 28 fractions (70 Gy/28f), among early-stage prostate cancer patients who did not undergo RP. We examined their clinical outcomes, survival benefits, and toxic side effects. Specifically, we performed a propensity score matching analysis 16 to reduce bias in estimating treatment effects when analyzing non-random observational data.
Methods
Patients
This was a retrospective study. We reviewed all patients diagnosed with prostate cancer who received radiotherapy at Peking Union Medical College Hospital (PUMCH), China, from October 2014 to June 2023, totaling 860 cases. The median follow-up period for all patients was 28 months (range: 4-56 months). After excluding those who had undergone surgical local treatment or radiotherapy for metastatic prostate cancer lesions, 187 patients diagnosed with early-stage prostate cancer ultimately met the inclusion criteria: 101 in the 60 Gy group and 86 in the 70 Gy group. All enrolled patients underwent abdominal and pelvic imaging or prostate-specific membrane antigen positron emission tomography (PSMA-PET) scans to confirm the absence of local invasion of adjacent organs, local lymph node metastasis, and distant metastasis. After excluding an additional 25 patients lost to follow-up, 86 patients in the 60 Gy group and 76 in the 70 Gy group were included in the analysis. All patients were diagnosed with pathologically confirmed prostate cancer. Cancer staging utilized the TNM staging system of the American Joint Committee on Cancer (AJCC) and the prostate cancer grading score of the International Society of Urological Pathology (ISUP). Patients were classified as intermediate-risk or high-risk according to the D'Amico risk classification system. 17 The administration of adjunctive ADT before and after radiotherapy was prescribed at the discretion of the attending urologist and recommended for patients at intermediate risk (6 months) and high risk (24-36 months).
The report of this study conforms to the STROBE guidelines. 18 The study protocol was approved by the Ethics Committee of Peking Union Medical College Hospital (PUMCH), China (No. I-25PJ0280). The requirement for obtaining patient consent was waved by the IRB, and we have de-identified all patient details.
Treatment
All patients underwent radiotherapy using intensity-modulated radiotherapy (IMRT), volumetric modulated arc therapy (VMAT), or Tomotherapy (TOMO). For the 60 Gy group, patients primarily followed two treatment plans: the first encompassed the prostate and seminal vesicles within the clinical target volume (CTV) using a dose fractionation pattern of 60 Gy/20f (3 Gy/f); the second comprised CTV1 including the prostate and high-risk seminal vesicles for preventive propose with a dose fractionation pattern of 60 Gy/20f (3 Gy/f), and CTV2 including the low-risk seminal vesicles with a dose fractionation pattern of 52 Gy/20f (2.6 Gy/f). For the 70 Gy group, the CTV included the prostate and seminal vesicles, with a dose fractionation pattern of 70 Gy/28f (2.5 Gy/f). For the delineation of the seminal vesicle target volume, our center referred to the 2018 ESTRO ACROP guideline 19 : For low-risk patients, the seminal vesicles were not delineated. For intermediate-risk patients, the seminal vesicles within 1.4 cm of the prostate on the axial plane were delineated. For localized high-risk patients, the seminal vesicles within 2.2 cm of the prostate on the axial plane were delineated. The planning target volume (PTV) was defined by adding a 5 mm margin around the CTV to avoid the rectum. The principal organs at risk (OARs) included the bladder, rectum, small intestine, and bilateral femoral heads. The regimen and duration of adjunctive ADT before and after radiotherapy were prescribed at the discretion of the attending urologist, tailored to individual patient needs.
All patients underwent bowel preparation and maintained moderate bladder filling before each radiotherapy session. Daily cone beam computed tomography (CBCT) image guidance was performed before each fraction to confirm the target area. For each CBCT, the images were carefully registered with the reference images based on bone and soft tissue to minimize positioning errors. A physician performed quality control on all CBCT images weekly.
Outcomes
Biochemical failure followed the Phoenix definition, marked by a nadir plus a ≥2 ng/ml increase in prostate-specific antigen (PSA). Disease-free survival (DFS) was calculated from the end of radiotherapy to the date of biochemical failure, local recurrence, lymph node recurrence, or occurrence of distant metastasis, as diagnosed by imaging. The time from the end of treatment to the date of death from any cause or to the last contact date during follow-up was defined as overall survival (OS). In this retrospective study, the primary endpoints reported were OS and DFS. Follow-up was characterized as the period from the end of radiotherapy to the date of last contact. We systematically assessed toxicity and side effects based on the Common Terminology Criteria for Adverse Events (CTCAE) v4.0 and v5.0 during follow-ups and through review of patient files, specifically targeting GI and GU toxicities. Acute toxicity was defined as that occurring from the start of radiotherapy to three months post-radiotherapy, while events more than three months post-treatment were categorized as late toxicity.
Statistical Analysis
Propensity score generation and 1:1 matching of patients were conducted between the 60 Gy and 70 Gy groups to minimize selection bias in treatment allocation. This process involved sampling without replacement, with the clamp value set to 0.05. Matching relied primarily based on the clinical and pathological baseline characteristics of the patients, including four covariates: age, PSA value at diagnosis, T stage, and ISUP grading. OS and DFS calculations were performed using Kaplan-Meier analysis. The log-rank test was utilized to evaluate differences in survival outcomes between the 60 Gy and 70 Gy groups. Continuous and categorical variables were analyzed between the two groups using the t-test and Fisher’s exact test, respectively. All statistical analyses were conducted in the R software environment for statistical computing and graphics (version 4.1). Statistical significance was established at P < .05 for all tests.
Results
Baseline Characteristics for Patients in 60 Gy and 70 Gy Groups Before and After Matching.

Abbreviations: ISUP, International Society of Urological Pathology; PSA, prostate-specific antigen; PSM, propensity score matching; SD, standard deviation; y: year.
Efficacy Comparison Between the 60 Gy Group and the 70 Gy Group After Matching.

Abbreviations: DFS, disease-free survival; OS, overall survival; yr, year.
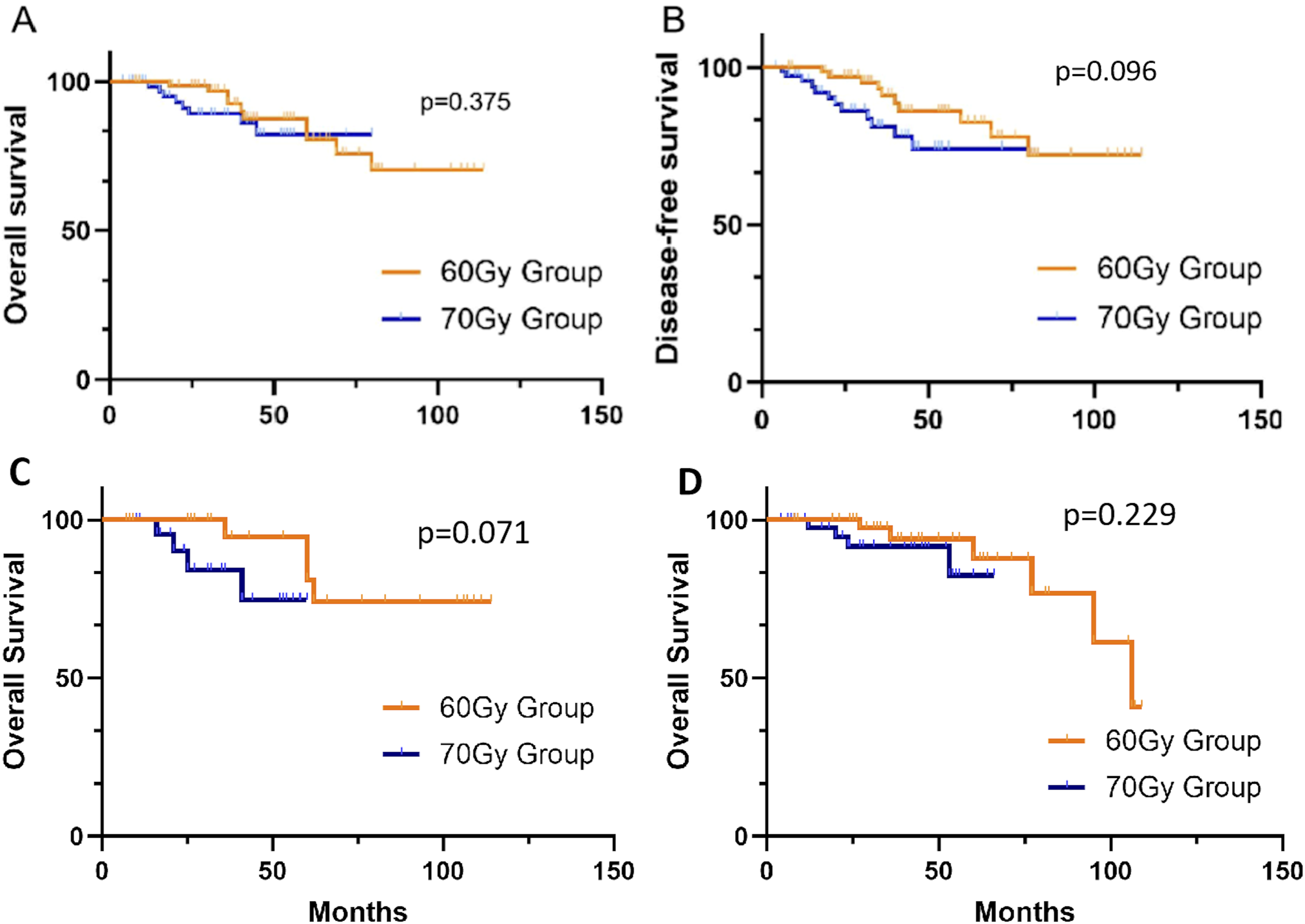
Kaplan-Meier Survival Curves for Patients. (A) Difference in Overall Survival Between the 60 Gy and 70 Gy Groups. (B) Difference in Disease-Free Survival Between the 60 Gy and 70 Gy Groups. (C) Difference in Overall Survival Between the 60 Gy and 70 Gy Groups in the Intermediate-Risk Patient Subgroup. (D) Difference in Overall Survival Between the 60 Gy and 70 Gy Groups in the High-Risk Patient Subgroup.
In both groups, 3 patients in each group did not receive adjuvant or neoadjuvant ADT treatment due to comorbidities or compliance issues. The average duration of continued post-radiotherapy endocrine therapy amounted to 31.7 ± 27.1 months for the 60 Gy group and 23.2 ± 19.7 months for the 70 Gy group. One patient received additional local radiotherapy following local recurrence, whereas all other patients underwent no further treatments beyond endocrine therapy after radiotherapy.
Side events occurred within three months after the end of radiotherapy were designated as acute toxicities, otherwise late toxicities. Among all patients, urinary toxicity was the most prevalent, followed by GI toxicity. A small number of patients experienced acute bone marrow suppression, while very few reported late bone-related radiotherapy side effects. The specific incidence rates of urinary and GI events are detailed in Figure 2. Clinical Manifestations of Toxicities in the Urinary and GI Systems. A1: Urinary Toxicities in the 60 Gy Group; A2: Urinary Toxicities in the 70 Gy Group; B1: GI Toxicities in the 60 Gy Group; B2: GI Toxicities in the 70 Gy group. GI: gastrointestinal.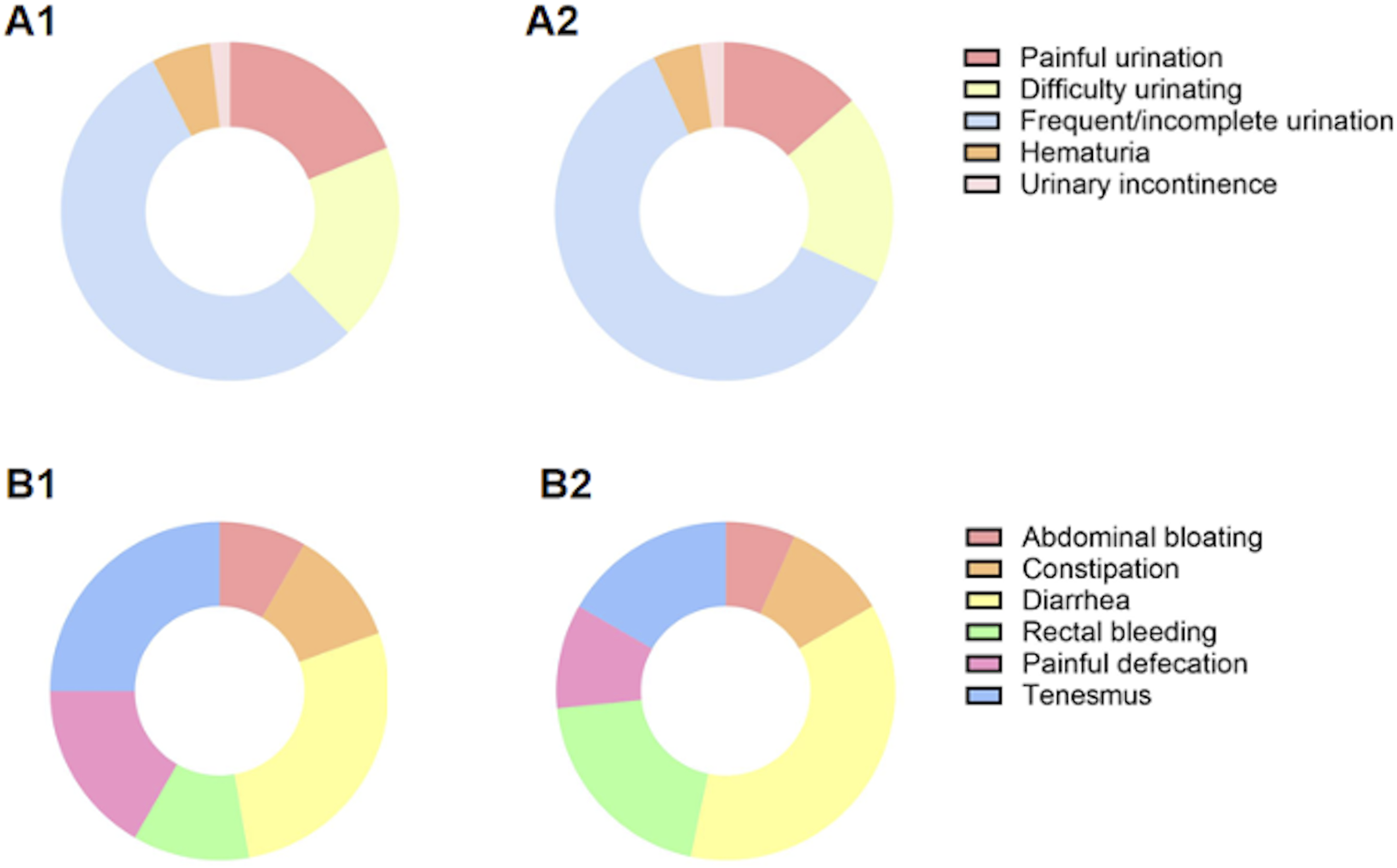
In the 60 Gy group, the occurrence of Grade 2 or higher urinary acute and late toxicities was 9.6% and 12.3%, respectively, while in the 70 Gy group, it was 9.6% and 2.8%, respectively. The incidence of acute Grade 2 or higher urinary toxicities was comparable between the two groups (60 Gy group vs 70 Gy group: 9.6% vs 9.6%, P = 1.0). However, regarding late Grade 2 or higher toxicities, the 70 Gy group demonstrated a significant advantage (60 Gy group vs 70 Gy group: 12.3% vs 2.8%, P = .028).
For the GI system, the primary acute toxicity included diarrhea and rectal bleeding, whereas the predominant late toxicity was rectal bleeding. In the 60 Gy group, the occurrence of Grade 2 or higher GI acute and late toxicities was 11.0% and 1.4%, respectively, while in the 70 Gy group, it was 6.8% and 1.4%, respectively. Between the 60 Gy and 70 Gy groups, the incidence of acute toxicities was higher in the 60 Gy group, although not statistically significant (60 Gy group vs 70 Gy group: 11.0% vs 6.8%, P = .383). The incidence of late toxicities was identical between the two groups (60 Gy group vs 70 Gy group: 1.4% vs 1.4%, P = 1.0).
Comparison of Toxicities in Urinary and Gastrointestinal Systems Between 60 Gy and 70 Gy Groups.

Abbreviations: GI: gastrointestinal.
Regarding dose limitations for OARs in radiotherapy, assuming an α/β ratio of 2 for normal tissue, the average biologically equivalent dose (BED) for 50% of the rectum dose in the 70 Gy and 60 Gy groups was 19.9 ± 8.7 Gy and 21.2 ± 11.7 Gy respectively, showing no significant difference (P = .17). The average BED for 50% of the bladder dose was 19.0 ± 18.0 and 22.4 ± 16.0 Gy respectively for 70 Gy and 60 Gy (P = .36). Detailed information is provided in Figure 3. Comparison of BED for Organs at Risk Between the 70 Gy and 60 Gy Groups. From Left to Right: *V50% Rectum, **V50% Bladder. BED: Biologically Equivalent Doses.
Discussion
Completed Studies on Moderate-Hypofractionated Radiotherapy (MHRT) in Prostate Cancer.

Abbreviations: AEs, adverse events; BCDF, biochemical and/or clinical disease failure; BCFF, biochemical or clinical failure free; CFRT, conventionally fractionated radiotherapy; DFS, disease-free survival; FFBF, freedom from biochemical failure; GI, gastrointestinal; GU, genitourinary; MHRT, moderate-hypofractionated radiotherapy; OS, overall survival; PCa, prostate cancer; PSS, prostate cancer-specific survival; QOL, quality of life; SBRT, stereotactic body radiotherapy; xGy/nf, x Gy in n fractions.
However, studies that compare different dose fractionation patterns in MHRT are relatively scarce. In this study, we compared two distinct MHRT dose fractionation patterns, 60 Gy/20f and 70 Gy/28f, among early-stage prostate cancer patients who had not undergone surgery and were undergoing radical radiotherapy. This methodology is relatively novel compared to other studies. Furthermore, we conducted a propensity score matching analysis to mitigate potential biases in the efficacy evaluation, thereby enhancing the reliability of the results.
This study encompassed 162 early-stage prostate cancer patients, and following propensity score matching, each of the 60 Gy and 70 Gy groups included 73 patients. The median follow-up duration for all patients was 36.0 months. From the results, the 60 Gy group shows a slight survival advantage compared to the 70 Gy group, but there is no significant statistical difference in OS and DFS. Regarding side effects, the incidence of grade 2 or higher acute urinary toxicities was similar between the two groups. However, the 70 Gy group exhibited significantly fewer late urinary toxicities, especially concerning long-term side effects such as urinary frequency and urgency. In terms of GI toxicities, the 60 Gy group experienced more noticeable acute GI toxicities, including constipation and diarrhea. Grade 2 or higher late GI toxicities were limited in both groups, and no significant difference was observed between the groups. Overall, apart from late Grade 2 or higher urinary system toxicity where the 70 Gy group held an advantage, there were no significant statistical differences in efficacy and overall urinary and GI system toxicity between the groups.
Regrettably, MHRT has not yet achieved widespread adoption in prostate cancer radiotherapy. Although many studies have demonstrated that MHRT is safe and effective for localized prostate cancer, its efficacy is not significantly superior to that of CFRT, and it may lead to an increase in early adverse reactions.22,33 Furthermore, owing to substantial inter-fraction errors and the steep dose-volume relationship of late toxicity post-treatment, MHRT necessitates advanced radiotherapy technologies and precise image-guidance capabilities.11,34 Nonetheless, MHRT continues to represent a significant future trend in prostate cancer radiotherapy. Compared to CFRT, MHRT utilizes higher single doses, potentially reducing treatment time by 1-2 weeks. Shorter treatment durations entail more efficient courses of treatment and can enhance patient compliance. 12 Higher single doses also elevate the therapeutic gain ratio, which benefits the enhancement of killing power against tumor cells. 11
Due to the numerous advantages of large fraction radiotherapy over CFRT, UHRT has increasing come into focus and commenced usage in clinical trials concerning prostate cancer radiotherapy. In 2019, Marvaso et al 35 involved 421 patients with localized prostate cancer and, after conducting a propensity score matching analysis, compared the efficacy and post-treatment toxicity between UHRT (32.5-35 Gy/5f) and MHRT (70.2 Gy/26f). The study results demonstrated that, compared to MHRT, UHRT presented reduced risks of both acute and late toxicity while providing comparable oncological outcomes. Widmark et al 14 conducted a randomized, non-inferiority Phase 3 trial, HYPO-RT-PC, enrolling 1200 patients with intermediate to high-risk prostate cancer who were randomly assigned to UHRT (42.7 Gy/7f) and CFRT (78 Gy/39f) groups. The results indicated that for intermediate to high-risk prostate cancer, UHRT’s survival without failure was not inferior to that of CFRT, though early side effects were more pronounced. Late toxicity was comparable between the two treatment groups. It is noteworthy that only one patient in each group reported significant sexual dysfunction, which seems to contradict the application of ADT in these patients. This may be due to the already weak baseline sexual function of these elderly patients or their potential to conceal the situation for privacy reasons. Currently, numerous clinical trials investigating prostate cancer UHRT are ongoing. The optimal radiotherapy fractionation regimen for radical prostate radiotherapy, whether it should be UHRT, MHRT, or CFRT, and whether personalized fractionation schemes are recommended for different patients, requires further investigation.
Additionally, most patients in our cohort were in the high-risk group. Stratified survival analysis showed that MHRT seemed to have an advantage in the intermediate-risk group, whereas there was no significant difference in overall survival in the high-risk group. The POP-RT trial, 36 a phase III, single-center, randomized controlled trial comparing survival differences between high-risk prostate cancer patients, found that whole pelvic radiotherapy provided better DFS and biochemical failure-free survival (BFFS) compared to prostate-only radiotherapy. However, there was no significant difference in OS. These findings suggest that for high-risk patients, it may not be enough to explore different dose fractionation schemes alone. Further research is needed to optimize target volume irradiation, and potentially even explore the joint optimization of fractionation schemes and target volumes, to achieve additional survival benefits.
Despite employing propensity score matching analysis, this study, being non-prospective and single-center, still exhibits inherent flaws, including a limited patient cohort, short follow-up time and biases that are challenging to control or eradicate. Moving forward, it would be prudent to design a prospective, randomized controlled trial (RCT) to more thoroughly compare and analyze the differences in efficacy and post-treatment toxicity between the two MHRT dose fractionation patterns.
Conclusions
Our study demonstrates that among early-stage prostate cancer patients who have not undergone surgery and are receiving radical radiotherapy, the two MHRT dose fractionation patterns, 60 Gy/20f and 70 Gy/28f, yield comparable survival and oncological outcomes with no significant statistical differences. In terms of safety, aside from the 70 Gy group displaying an advantage in late Grade 2 or higher urinary system toxicity, there were no significant differences in overall acute or late urinary and GI toxicities between the two groups. This further substantiates the efficacy and safety of MHRT in early-stage prostate cancer patients undergoing radical radiotherapy, underscoring MHRT’s potential as a primary treatment modality for prostate cancer in the future.
Footnotes
Acknowledgements
We would like to extend our gratitude to all our colleagues for their support.
Statements and Declarations
Author Contributions
Y.T. and W.C. contributed equally to this manuscript.
Y.T. and W.C. were responsible for the conceptualization of the study, study design, data analysis and wrote the manuscript draft.
H.Z., J.S., H.G., X.H., K.H., F.Z. and Z.L. were responsible for the data collection and curation.
Z.L. contributed to the design and supervision of the study.
All authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Key R&D Program of China, Ministry of Science and Technology of the People’s Republic of China. (Grant No. 2022YFC2407100, 2022YFC2407102).
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
