Abstract
Objective
To develop personalized treatment strategies for maintenance therapy in patients with extensive-stage small cell lung cancer (ES-SCLC).
Materials and Methods
We analyzed data from ES-SCLC patients who achieved stable disease (SD) following initial chemotherapy combined with immunotherapy. These patients subsequently received maintenance therapy (MT) with a combination of anlotinib and PD-1/L1 inhibitors. The primary endpoints included progression-free survival (PFS), overall survival (OS), objective response rate (ORR), disease control rate (DCR), and treatment-related adverse events (AEs).
Results
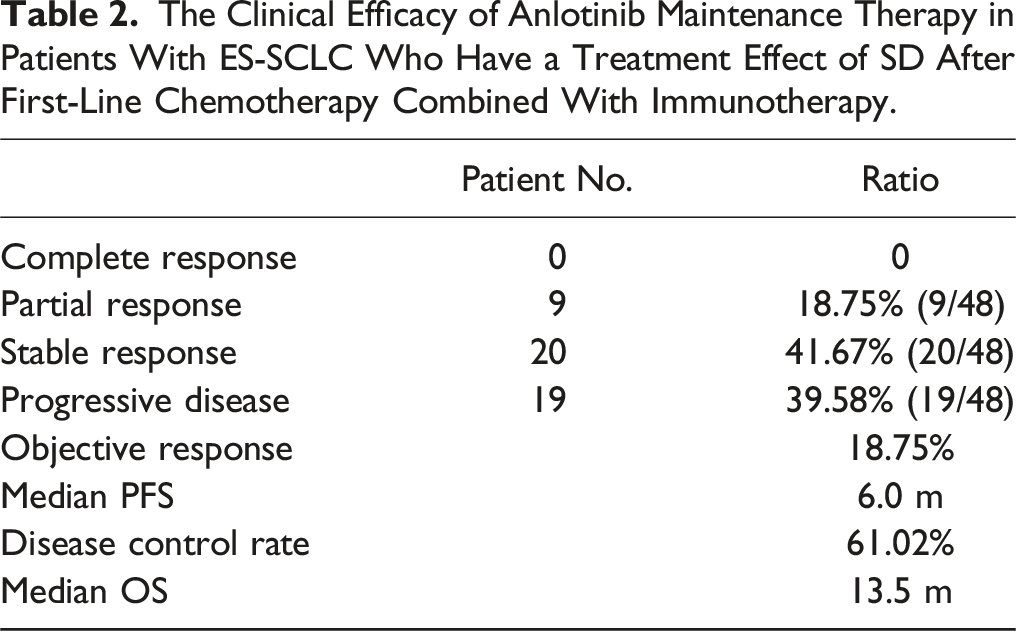
Preliminary findings suggest that this regimen is highly effective, with a median PFS of 6 months and OS of 13.5 months, alongside a DCR exceeding 60%. Subgroup analysis revealed enhanced efficacy in patients with fewer than three metastatic sites and those who experienced hypertension, proteinuria, or hand-foot syndrome during MT. Mechanistic studies showed a notable increase in the proportion of CD8+ T cells in the peripheral blood post-MT, correlating with improved outcomes. These findings imply that the therapeutic effect of MT may be partly due to the direct activation of CD8+ T cells, producing a synergistic anti-tumor response. Despite the prevalence of AEs, AEs were generally manageable, underscoring anlotinib’s potential in this context.
Conclusion
The combination of anlotinib and PD-1/L1 inhibitors offers promising efficacy and manageable AEs in MT, making it a viable option for ES-SCLC patients who achieve SD post-initial therapy. These results justify further prospective studies to validate this approach.
Plain Language Summary
Anlotinib maintenane for SD ES-SCLC: This is the first study to demonstrate that Anlotinib maintenance therapy can provide promising efficacy while the adverse effects are manageable. Further research has identified potential populations that may benefit from this treatment and possible mechanisms of action. This research not only contributes to the understanding of anti-tumor angiogenesis therapies in SCLC but also underscores the importance of personalized treatment strategies based on diease characters and identification of treat-related adverse events.
Background
Small cell lung cancer (SCLC) is a highly aggressive malignancy, accounting for approximately 15% of all lung cancer cases. 1 Extensive-stage small cell lung cancer (ES-SCLC), the most common form at diagnosis, typically presents with a particularly poor prognosis. Patients have a median survival of only 10 to 12 months despite aggressive treatment approaches. 2 The standard first-line treatment for ES-SCLC combines platinum-based chemotherapy and etoposide, with immunotherapies such as atezolizumab 3 or durvalumab 4 supplementing individual treatment plans. Although these immunotherapies have shown promise in improving overall survival and response rates5,6 they provide long-term survival benefits primarily to patients who achieve a partial response (PR) or complete response (CR), which constitute fewer than 20% of all patients. For these patients, CR or PR to initial treatment, single-agent immunotherapy in maintenance therapy proves highly effective, rendering an upgrade in maintenance treatment unnecessary and potentially leading to increased side effects and costs. 7 However, patients exhibiting stable disease (SD) or progressive disease (PD) typically see little to no survival benefit from continued immunotherapy.8,9 Therefore, enhancing efficacy through upgraded treatments in these cases is crucial. As the effectiveness of first-line treatments reaches a plateau, maintaining and upgrading therapy becomes a viable strategy.10,11
In terms of drug selection, small molecule inhibitors primarily targeting VEGF might be suitable for maintenance therapy. While their efficacy is well established in first-line and salvage treatments for advanced lung cancer, including SCLC, their role in maintenance therapy for patients achieving SD after first-line chemotherapy combined with immunotherapy remains to be determined. This retrospective study assesses the efficacy and safety of anlotinib plus PD-1/L1 inhibitors as maintenance therapy in patients with ES-SCLC who achieved SD post-initial treatment. Our aim is to provide a personalized treatment foundation for maintenance therapy and expand the range of treatment options for small cell lung cancer.
Materials and Methods
We conducted a retrospective study by collecting data from patients with ES-SCLC who received MT with anlotinib and PD-1/L1 inhibitors after first-line treatment with chemotherapy and immunotherapy at our centers from 2021 to 2024. This study adheres to the STROBE guidelines,
12
and aims to evaluate the efficacy and safety of anlotinib plus PD-1/L1 inhibitors as maintenance therapy in this patient population (See Figure 1). Flowchart of the patient enrollment process.
Study Population
We reviewed the medical records of patients from January 2021 to January 2024. Inclusion criteria were 1 : histologically confirmed ES-SCLC 2 ; first-line treatment with a platinum-based chemotherapy regimen and PD-1/L1 inhibitors (atezolizumab, durvalumab, sintilimab, tislelizumab, etc.) 3 ; stable disease (SD) as assessed by the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 4 ; an Eastern Cooperative Oncology Group (ECOG) performance status of 0-2 and adequate organ function as determined by laboratory tests 5 ; initiation of anlotinib within four weeks of completing first-line treatment. Exclusion criteria included prior use of VEGFR inhibitors, untreated brain metastases, significant cardiovascular disease, active infections, or any severe or uncontrolled medical conditions that might interfere with the study. The study protocol received approval from the institutional review board (IRB) of Hubei Cancer Hospital Affiliated with Tongji Medical College (Approval No. LLHBCH2024YN-090). As this was a retrospective study, the requirement for obtaining informed consent was waived. The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice (GCP) guidelines.
Lymph Cell Subpopulation Analysis by FCAM
Blood samples (2 mL) treated with ethylenediamine tetraacetic acid (EDTA) were collected from patients with advanced NSCLC before the initial treatment and after subsequent treatment cycles. Samples were processed within 6 hours of collection. CD3+/CD4+/CD8+ T-cell, CD19+ B-cell, and CD16+CD56+ natural killer (NK) cell counts (cells/μL) were quantified using multicolor flow cytometry with human monoclonal anti-CD3-fluorescein isothiocyanate (FITC), anti-CD4-phycoerythrin (PE), anti-CD8-allophycocyanin (APC), anti-CD19-PE, anti-CD16-APC, and anti-CD56-PE antibodies [BD Multitest; Becton, Dickinson, and Co. (BD) Biosciences, Franklin Lakes, NJ, USA] following the manufacturer’s instructions. The cells were analyzed using a BD FACS Canto II flow cytometry system (BD Biosciences).
Treatment Protocol
In our study population, maintenance therapy consisted of anlotinib and PD-1/L1 inhibitors. Anlotinib was administered orally at a dose of 12 mg daily on a 2-week-on, 1-week-off schedule (a 21-day cycle), while PD-1/L1 inhibitors were given according to their respective standard treatment protocols. This regimen continued until disease progression or the occurrence of intolerable toxic side effects. Dosage adjustments were made based on individual tolerance and side effects.
Efficacy Assessment
Efficacy outcomes included progression-free survival (PFS), overall survival (OS), and the disease control rate (DCR). PFS was defined as the time from the initiation of anlotinib therapy to the earliest sign of disease progression or death from any cause. OS was measured from the start of anlotinib treatment until the date of death from any cause. Responses were assessed via computed tomography (CT) scans every 6 to 8 weeks. DCR was calculated as the proportion of patients achieving a complete response (CR), partial response (PR), or SD at the first evaluation after starting anlotinib.
Safety Evaluation
Adverse events (AEs) were assessed using the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. AEs were monitored throughout the treatment period, and their severity was graded as mild (grade 1), moderate (grade 2), severe (grade 3), or life-threatening (grade 4). The most common AEs, such as hypertension, fatigue, and gastrointestinal disturbances, were analyzed in detail. The overall incidence of AEs, as well as any dose modifications or treatment interruptions due to toxicity, were reported.
Statistical Analysis
Descriptive statistics summarized the patient demographics and clinical characteristics. We performed survival analyses for PFS and OS using the Kaplan-Meier method. Comparisons between groups were made using the log-rank test. Statistical significance was established at P < 0.05. All analyses were conducted using SPSS version 13.0.
Results
Patients’ Characteristics
Baseline Clinical Characteristics of the Study Cohort.

Efficacy
The Clinical Efficacy of Anlotinib Maintenance Therapy in Patients With ES-SCLC Who Have a Treatment Effect of SD After First-Line Chemotherapy Combined With Immunotherapy.

The median PFS and OS for patients with ES-SCLC receiving first-line chemotherapy combined with immunotherapy, showing a treatment effect of SD, who then receive anlotinib maintenance therapy in the general population and subgroups. (A, B) The overall PFS and OS in this study. (C, D) Comparisons of PFS and OS between these patents with less metastasis site (<3) and more metastasis site (≥3) (metastasis site <3 vs metastasis site ≥3). (E, F) Comparisons of PFS and OS between these patents with sAEs (Mainly referring to hypertension, proteinuria, and hand-foot syndrome) and without during the whole treatment (with sAE vs without sAE). (G, H) Comparisons of PFS and OS between these patents with CD8+ ratio elevation and without (CD8+% increase vs CD8+% no increase). mPFS, median progression-free survival; mOS, median overall survival; sAE, specifically refers to any adverse event including hypertension, proteinuria, and hand-foot syndrome.
Safety
The Side Effects of Anlotinib Maintenance Therapy in Patients With ES-SCLC Who Have a Treatment Effect of SD After First-Line Chemotherapy Combined With Immunotherapy.

Mechanism Exploration
We analyzed the proportion of peripheral blood immune cells before and after treatment. Our findings indicated a significant increase in CD8+ cells post-treatment compared to baseline (Paired t test, P < 0.05; see Figure 3). In contrast, the proportions of other immune cells (CD4+, NK+, Treg) showed no significant changes (see Supplementary Figure). Notably, patients with an elevated CD8+ ratio exhibited higher objective response rates (ORR) and DCR as well as better PFS and OS (mPFS 6.0 m vs 5.0 m, P < 0.0001, HR = 0.15: 95% CI:0.06-0.39; mOS 15.0 m vs 13.0 m, P < 0.0001, HR = 0.25:95% CI:0.11-0.57 (Figures 2 and 3). These results suggest that the regimen exerts a synergistic anti-tumor effect through immune system activation, and the upregulation of the CD8+ ratio may serve as a potential molecular biomarker for predicting the efficacy of anlotinib and PD-1/L1 inhibitors. The changes in the proportion of CD8+ cells in peripheral blood before and after anlotinib maintenance therapy in patients with ES-SCLC, who received first-line chemotherapy combined with immunotherapy showing a treatment effect of SD, and a comparison of ORR and DCR among different populations based on changes in CD8+ cell proportions.
Discussion
In this retrospective study, we assessed the efficacy and safety of anlotinib and PD-1/L1 inhibitors as maintenance therapy in patients with ES-SCLC who achieved SD following initial treatment with chemotherapy and immunotherapy. While the combination of chemotherapy and immunotherapy has shown promise in improving initial response rates, their effectiveness and safety in maintaining disease control post-treatment remain uncertain.11,13
Our findings indicate that anlotinib combined with PD-1/L1 inhibitors may prolong the period of disease control, particularly in patients who have achieved SD after initial therapy. 14 Notably, patients with fewer metastatic sites and those who experienced specific adverse effects during treatment exhibited better outcomes with this maintenance therapy. This underscores the significance of recognizing clinical features and adverse effects in predicting treatment outcomes and customizing treatment approaches. 15 The safety profile of anlotinib combined with PD-1/L1 inhibitors in our cohort aligns with previous studies. 16 Hypertension, hand-foot syndrome, and proteinuria were the most common adverse events, typical toxicities associated with anti-tumor angiogenesis treatments, especially those targeting VEGF with small molecule inhibitors. No patients discontinued treatment as these adverse effects can be effectively managed with established guidelines and extensive clinical experience. Given its promising efficacy and manageable toxicity, anlotinib could be incorporated into treatment regimens for these patients to enhance quality of life. 17
Our study has several significant implications. Firstly, to the best of our knowledge, this is the first report on precision maintenance therapy for patients with ES-SCLC who achieved SD following first-line chemotherapy combined with immunotherapy. Its efficacy surpasses that reported in previous studies.10,18 Besides, this study included a subset of patients with liver and brain metastases, 19 broadening the potential applicability of this regimen.20-22 In addition to notable efficacy, the combination of anlotinib and immunotherapy for maintenance treatment exhibited relatively mild side effects, most of which were manageable, indicating a high level of tolerance for this regimen. This allows patients to maintain their quality of life during treatment.17,23 Furthermore, the study identified potential biomarkers that may predict therapeutic efficacy, such as the number of metastatic sites and specific side effects like hypertension, hand-foot syndrome, and proteinuria during treatment, which correlate with better outcomes. This provides a basis for selecting advantageous patient populations for the clinical application of this regimen. 24 Additionally, our study suggests that the mechanism of action likely involves immune activation, contributing to a synergistic anti-tumor effect, thereby offering insights and directions for future research.23,25,26
While our study provides valuable insights, it has limitations. The retrospective nature may introduce biases inherent to patient selection and data collection.27,28 Additionally, the small sample size may limit the generalizability of our findings. 21 Therefore, larger, prospective, randomized controlled trials are essential to confirm the efficacy and safety of anlotinib plus PD-1/L1 inhibitors in this setting and to determine optimal treatment durations and combinations with other therapeutic agents. 29 Future research should also explore the biological mechanisms underlying the response to the combination of anlotinib and PD-1/L1 inhibitors. 30 Identifying predictive biomarkers could enhance patient selection for maintenance therapy,21,22 allowing clinicians to tailor treatments based on individual tumor characteristics and molecular profiles.8,26 Furthermore, investigating combination strategies with other agents may further improve outcomes for patients with ES-SCLC.7,27,28,31,32
In conclusion, the combination of anlotinib and PD-1/L1 inhibitors represents a promising maintenance therapy for patients with ES-SCLC who achieve SD following first-line chemotherapy and immunotherapy. Its favorable efficacy and safety profile, coupled with improvements in quality of life, make it a compelling option in the evolving landscape of SCLC management. 15 As the field continues to advance, further studies are warranted to establish definitive treatment guidelines and explore the full potential of targeted therapies like anlotinib in prolonging survival and enhancing the quality of life for patients battling this challenging disease.13,20
Supplemental Material
Supplemental Material - Efficacy and Safety of Anlotinib and PD-1/L1 Inhibitors as Maintenance Therapy for Extensive-Stage Small Cell Lung Cancer Patients who Have Achieved Stable-Disease After First-Line Treatment with Chemotherapy and Immunotherapy: A Retrospective Study
Supplemental Material for Efficacy and Safety of Anlotinib and PD-1/L1 Inhibitors as Maintenance Therapy for Extensive-Stage Small Cell Lung Cancer Patients who Have Achieved Stable-Disease After First-Line Treatment with Chemotherapy and Immunotherapy: A Retrospective Study by Yi Peng, De Wu, Jing Tang and Xiaobing Li in Cancer Control.
Footnotes
Acknowledgement
We would like to thank Xichen Wang (MSD China, Shanghai, China) for providing academic information consulting support.
Author Contributions
Yi Peng: drafting manuscripts. De Wu: Analyzing data. Jing Tang: collecting data. Xiaobing Li: responsible for the implementation of clinical research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Science and Technology Innovation Medical Development Foundation of Beijing (KC2023-JX-0186-PQ008).
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
