Abstract
Background
Combination therapy with multiple tyrosine kinase inhibitors (multi-TKIs) and immune checkpoint inhibitors (ICIs) has been increasingly tested in clinical studies. This study aimed to investigate the effect of the addition of ICI to multi-TKIs on the profile of treatment-related adverse events.
Methods
An electronic database search was performed using PubMed and Web of Science to identify published clinical studies on multi-TKI monotherapy and multi-TKI plus ICI combination therapy from July 20, 2005 to July 1, 2023. The incidence rate of common adverse events caused by multi-TKI monotherapy and multi-TKI plus ICI combination therapy was obtained and compared from the viewpoints of (1) relative risk for the combination therapy vs sunitinib, (2) adverse event incidence rate by clinical trial, and (3) pooled incidence rate. The quality of the evidence was assessed with the Cochrane risk of bias tool. Meta-analysis used random effects models.
Results
This systematic review identified 83 clinical studies involving 7951 patients. The combination therapy of multi-TKI and ICI was associated with an increased risk of diarrhea (relative risk [RR]: 1.24, 95% confidence interval [CI]: 1.15-1.33, P < .001), hypothyroidism (RR: 1.44, 95% CI: 1.11-1.87, P = .0064) and rash (RR: 1.71, 95% CI: 1.18-2.47, P = .0045) compared with multi-TKI monotherapy. The addition of ICI was suggested to decrease the risk of adverse events related to performance status.
Conclusion
Our study identified an increased risk of treatment-related adverse events associated with multi-TKI plus ICI combination therapy. This would help optimize the management of toxicities caused by multi-TKI plus ICI combination therapy.
Keywords
Introduction
In recent years, new anti-cancer drugs, molecular-targeted agents, and antibody drugs have been developed as replacements of conventional cytotoxic compounds. Since the approval of trastuzumab as the first molecular-targeted agent for the treatment of metastatic breast cancer in the US in 1998, 1 a variety of molecular-targeted agents for specific populations have been developed. Multiple tyrosine kinase inhibitors (multi-TKIs) targeting multiple molecules have been shown to be effective in several cancers, and several multi-TKIs, such as sorafenib, sunitinib, axitinib, pazopanib, and cabozantinib have been used in clinical practice.2,3 It has also been reported that multi-TKIs cause pathognomonic toxicities, such as diarrhea, fatigue, nausea, rash, anorexia, vomiting, hand-foot syndrome, hypertension, and proteinuria, which are typical toxicities for multi-TKIs.4,5
Recently, various immunotherapies have been actively developed. In 2011, ipilimumab, the first immune checkpoint inhibitor (ICI), was approved for treating melanoma in the US. 6 Among the various types of ICIs, programmed death receptor-1 (PD-1)/programmed death ligand-1 (PD-L1) inhibitors have been actively developed and indications for various types of cancer have been obtained. Programmed death receptor-1 is overexpressed in dendritic cells and T cells in the tumor environment and binds to PD-L1/PD-L2, which is expressed in cancer cells, to suppress the activity of immune cells, such as T cells and inhibit immune responses.7-9 Blockade of the PD-1/PD-L1 pathway can activate immune responses and enhance anti-tumor effects. Several clinical studies have been conducted on PD-1/PD-L1 inhibitors, and sufficient safety information has been accumulated to understand their safety profile. Safety analyses have been comprehensively performed, and it was shown that these drugs cause immune-related toxicity in various parts of the body. 10
Several clinical studies have been conducted on the use of PD-1/PD-L1 inhibitors in combination with other medications, including multi-TKIs for treating various cancers. Non-clinical data suggest that multi-TKIs produce higher T-cell activation and macrophage polarization, indicating immunomodulatory effects that enhance synergistic anti-tumor efficacy. 11 In recent years, an increasing number of studies using multi-TKIs plus ICIs have been conducted, and several ICIs (avelumab, pembrolizumab, and nivolumab) in combination with multi-TKIs (axitinib, lenvatinib, and cabozantinib) have been approved for the treatment of renal cell cancer. 12 It is assumed that the combination therapy of multi-TKIs and ICIs will be developed for other types of cancer. Therefore, in addition to efficacy, understanding the safety profile of this combination therapy is crucial for toxicity management. Few large-scale studies have investigated multi-TKI plus ICI combination therapy, which makes a comprehensive safety analysis difficult. Moreover, safety assessment of multi-TKI plus ICI combination therapies based on large-scale studies is considered insufficient because the types of multi-TKIs used in the experimental arm and the control arm are usually different.
Therefore, to examine the effect of adding ICI to multi-TKI on the safety profile, we conducted a systematic review based on the clinical studies of multi-TKI monotherapy and combination therapy of multi-TKIs and ICIs.
Materials and Methods
Study Selection Strategy
A systematic literature search was conducted to identify published clinical studies on multi-TKI monotherapy and those on multi-TKI plus ICI combination therapy that reported treatment-related adverse events (incidence ≥10%). First, to identify ICIs to be examined in the present study, the search was conducted in PubMed and Web of Science using the terms PD-1, PD-L1, and inhibitor, and then the list was narrowed down using clinical trials. Second, to identify multi-TKIs that are used in combination with ICIs, the search was conducted using the generic names of the ICIs identified above and the term tyrosine kinase and the list was narrowed down using clinical trials. Finally, we searched for clinical studies on multi-TKI monotherapy and combination therapy of multi-TKI and ICI using the generic names of the multi-TKIs identified above and narrowed down the list using clinical trials. The search was conducted for clinical studies reported from July 20, 2005 to July 1, 2023.
In this study, the applicable clinical studies were included in the analysis regardless of whether they were single-arm or randomized clinical studies. Clinical studies in healthy volunteers, pediatric patients, and patients with hepatic disorders were excluded to minimize the impact of differences in the study populations.
Data Extraction and Quality of Evidence
Two independent reviewers (TS and MN) screened the names and designs of the clinical trials for the records derived from PubMed and Web of Science, followed by assessment of eligibility based on the full texts. Disagreements about eligibility were resolved through discussion.
For each of the selected clinical studies, the following information was extracted and tabulated: generic name(s) of the multi-TKIs and ICIs, number of subjects, and study phase. Phase 1/2 was regarded as phase 2.
The following common adverse events were selected for this study with reference to the package insert of multi-TKIs: anorexia, constipation, weight loss, diarrhea, fatigue, hand-foot syndrome (including palmar-plantar erythrodysesthesia syndrome), hypertension, hypothyroidism, nausea, proteinuria, rash, and vomiting. For each adverse event, the number of events was extracted from the clinical studies on multi-TKI monotherapy and multi-TKI plus ICI combination therapy. Incidence rates were calculated by dividing the number of events by the number of subjects in each study. The review protocol was registered with the International Platform of Registered Systematic Review and Meta-Analysis Protocols [INPLASY] (registration number: INPLASY 202390011).
Statistical Analysis
Relative Risk for Combination Therapy with Multi-TKIs and ICIs (Vs Sunitinib)
Several randomized phase 3 trials on multi-TKI plus ICI combination therapy and on sunitinib as a comparator were identified. For each of the selected adverse events, the relative risk (RR) for the combination therapy of multi-TKIs and ICIs compared with sunitinib was calculated. Then, the pooled RR and 95% confidence interval (CI) were calculated using a random-effects model.
The quality and risk of bias of randomized controlled trials (RCTs) were assessed with the revised Cochrane Collaboration’s risk of bias tool (RoB 2.0). 13 We followed the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines for the purpose of this analysis. 14
Comparison of Adverse Events Between Multi-TKI Monotherapy and Combination Therapy of Multi-TKIs and ICIs (Analysis by Clinical Trial)
For each of the selected adverse events, the numbers of studies with ≥10% incidence and those with <10% incidence were counted separately for multi-TKI monotherapy and multi-TKI plus ICI combination therapy. Fisher’s exact test was used to examine whether there was an imbalance in the number of studies between multi-TKI monotherapy and multi-TKI plus ICI combination therapy.
Pooled Incidence Rate of Adverse Events for Multi-TKI Monotherapy and Combination Therapy of Multi-TKIs and ICIs
The pooled incidence rate and its 95% confidence interval for the selected adverse events were calculated separately for multi-TKI monotherapy and multi-TKI plus ICI combination therapy using a random-effects model.
Heterogeneity among studies was assessed using the Q test and I 2 index, and statistically significant heterogeneity was considered at P < .05 or I 2 >50%. Lastly, Egger’s, and Harbord’s tests were used when necessary to assess publication bias, with significance set at P < .1. All analyses were performed using StatsDirect (Stats-Direct Ltd., Cheshire, UK). A statistically significant association was defined as P < .05.
Results
Search Results and Trial Characteristics
The evaluated trials were identified as described in Figure 1. Our literature search and review of reference lists identified 1971 relevant publications, from which 83 eligible studies involving 7951 patients for the analyses were selected (Supplemental Table 1). Five multi-TKIs that have been used in clinical studies in combination with ICIs were identified. Forty-eight studies were conducted on multi-TKI monotherapy, 34 studies on multi-TKI plus ICI combination therapy, and one study evaluated multi-TKI monotherapy and multi-TKI plus ICI combination therapy. The identified multi-TKI monotherapies included apatinib (n = 9), axitinib (n = 13), cabozantinib (n = 8), lenvatinib (n = 5), and regorafenib (n = 14), with a total of 3879 patients receiving multi-TKIs. PD-1 and PD-L1 inhibitors used included atezolizumab (n = 2), avelumab (n = 7), camrelizumab (n = 12), durvalumab (n = 1), nivolumab (n = 4), and pembrolizumab (n = 9), with a total of 4072 patients receiving ICIs. Phase 2 trial was most common for both multi-TKI monotherapy (n = 36) and multi-TKI plus ICI combination therapy (n = 22) (Table 1). Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow diagram of selecting clinical trials for this analysis. Trial Characteristics. Abbreviations: ICI, immune checkpoint inhibitor; multi-TKI, multiple tyrosine kinase inhibitor.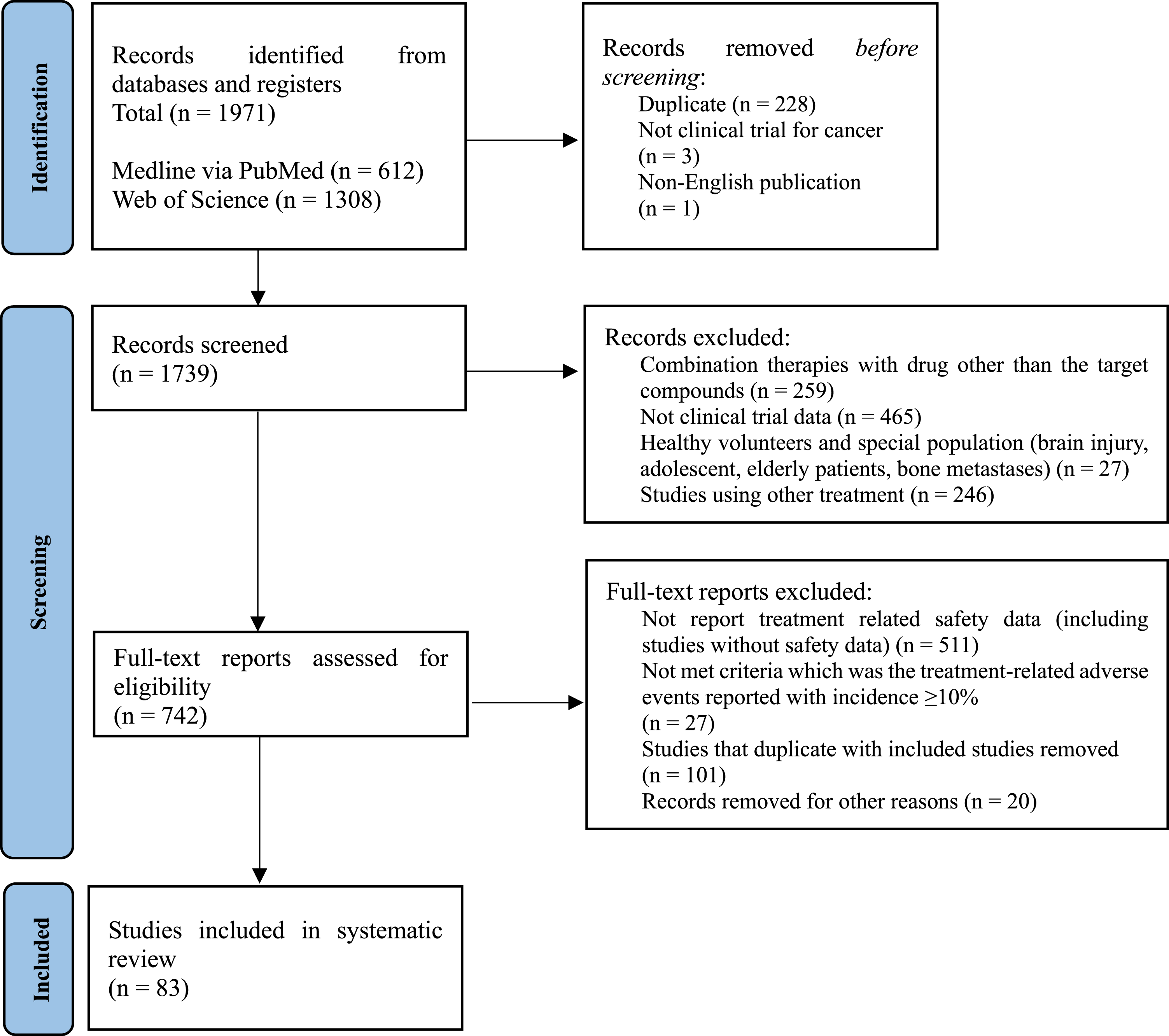

Relative Risk for the Combination Therapy of Multi-TKIs and ICIs (Vs Sunitinib)
Pooled Relative Risk of Adverse Events for Immune Checkpoint Inhibitor Plus Multiple Tyrosine Kinase Inhibitor Compared With Sunitinib Monotherapy.

*Constipation, weight loss and proteinuria were not included in the analysis due to insufficient number of events.
Abbreviations: CI, confidence interval.
Treatment-related adverse events with a statistically significant increase in RR for the multi-TKI plus ICI combination therapy compared with sunitinib were diarrhea (RR: 1.24, 95% CI: 1.15-1.33, P < .001), hypothyroidism (RR: 1.44, 95% CI: 1.11-1.87, P = .0064), and rash (RR: 1.71, 95% CI: 1.18-2.47, P = .0045) (Figure 2). Treatment-related adverse events with a statistically significant decrease in RR for multi-TKI plus ICI combination therapy compared with sunitinib were hand-foot syndrome (RR: 0.85, 95% CI: 0.72-1.00, P = .0435) and nausea (RR: 0.82, 95% CI: 0.73-0.93, P = .0016) (Figure 2). Anorexia, fatigue, hypertension and Vomiting were not significantly different between the multi-TKI plus ICI combination therapy and sunitinib (Supplemental Figure 1). Forest plot of relative risk of adverse events (all grade) for (A) diarrhea, (B) hypothyroidism, (C) rash, (D) hand-foot syndrome and (E) nausea for immune checkpoint inhibitors plus multiple tyrosine kinase inhibitor compared with that of sunitinib. Abbreviations: RR, relative risk; CI, confidence interval.
Quality Assessment
The RoB 2.0 results for randomized studies are shown in Supplemental Figure 2, where 4 out of 4 randomized studies were assessed as having some concerns (due to insufficient information of D2 Deviations from the intended interventions and/or D4 measurement of the outcome) for performance. Egger’s, and Harbord’s tests for the treatment-related adverse events revealed no significant difference, indicating no remarkable publication bias in the analysis (P > .1). Meanwhile, the PRISMA checklist for our meta-analysis is given in Supplemental Tables 2-1 and 2-2.
Comparison of Adverse Events Between Multi-TKI Monotherapy and Combination Therapy of multi-TKIs and ICIs (Analysis by Clinical Trial)
Number of Study Arms Classified by Frequency of Adverse Events for the Arms of Multiple Tyrosine Kinase Inhibitor Monotherapy and Immune Checkpoint Inhibitor Plus Multiple Tyrosine Kinase Inhibitor Combination.

Abbreviations: ICI, immune checkpoint inhibitor; multi-TKI, multiple tyrosine kinase inhibitor.
In multi-TKI monotherapy, all-grade treatment-related adverse events occurred in 2270 of 2393 patients (94.9%) in 25 studies, and grade 3 or higher treatment-related adverse events occurred in 1321 of 2251 patients (58.7%) in 20 studies. In the multi-TKI plus ICI combination therapy, all-grade treatment-related adverse events occurred in 3643 of 3792 patients (96.1%) in 28 studies, and grade 3 or higher treatment-related adverse events occurred in 2383 of 3773 patients (66.5%) in 27 studies. Of the adverse events with a statistically significant difference in incidence between multi-TKI monotherapy and multi-TKI plus ICI combination therapy, hypothyroidism (P = .0003) and rash (P = .0462) were significantly higher in the combination therapy group, and constipation (P = .00175) was higher in the monotherapy group. There were no significant differences in the incidence of anorexia, weight loss, diarrhea, fatigue, hand-foot syndrome, hypertension, nausea, proteinuria, or vomiting.
Pooled Incidence Rate of Adverse Events for Multi-TKI Monotherapy and Combination Therapy of Multi-TKIs and ICIs
Pooled Incidence Rates of Adverse Events for Multiple Tyrosine Kinase Inhibitor Monotherapy and Immune Checkpoint Inhibitor Plus Multiple Tyrosine Kinase Inhibitor.
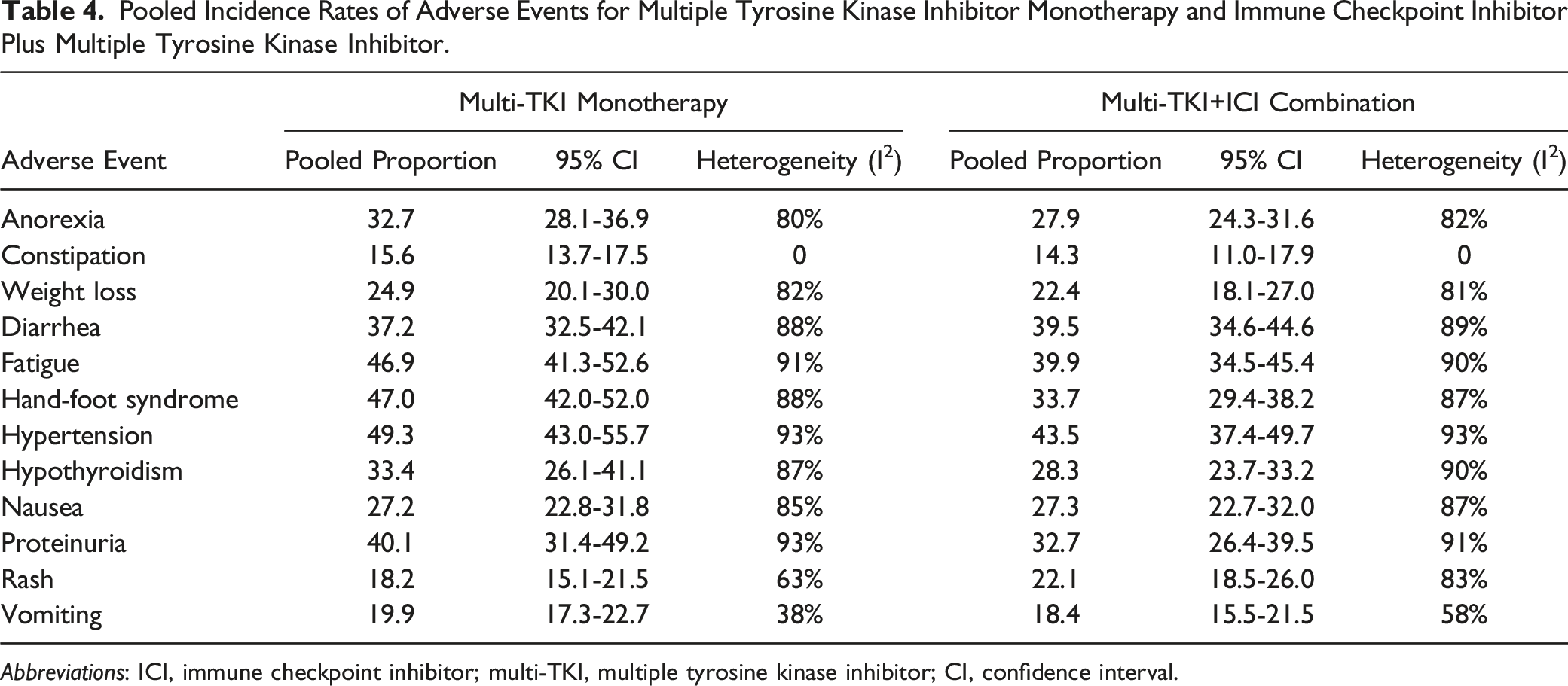
Abbreviations: ICI, immune checkpoint inhibitor; multi-TKI, multiple tyrosine kinase inhibitor; CI, confidence interval.
Discussion
Combination therapy with multi-TKIs and ICIs has already been shown to be highly effective for several types of cancers, and it is expected that various combinations of multi-TKIs and ICIs will be actively examined in the future. However, the number of large-scale studies on multi-TKI and ICI combination therapy is limited, and the safety profile has not been fully elucidated. Few clinical studies have used the same multi-TKIs as a background therapy in combination with ICI, which could be one of the factors for not having clarified the impact of adding ICIs to multi-TKIs on safety. Therefore, the present study investigated the effect of adding ICIs to multi-TKIs on the safety profile from various perspectives, especially focusing on the pathognomonic toxicities derived from multi-TKIs.
In phase 3 trials of multi-TKI plus ICI combination therapy compared with sunitinib, increased risk for diarrhea, hypothyroidism, and rash was identified to be associated with the combination therapy. Similarly, in the analysis of clinical trial data from a variety of studies on multi-TKI monotherapy and multi-TKI plus ICI combination therapy, where the impact of adding ICIs to multi-TKIs was evaluated, hypothyroidism and rash were also identified as increased events in the combination therapy. Furthermore, the pooled incidence rates of fatigue, hand-foot syndrome, and proteinuria decreased in the combination group with multi-TKI and ICI. Based on these results, the addition of ICIs to multi-TKI therapy would increase the risk of diarrhea, hypothyroidism, and rash compared with multi-TKI monotherapy. Simultaneously, it was suggested that the addition of ICI would decrease the risk of adverse events related to performance status, presumably because of enhanced efficacy. It was reported that severe treatment-related adverse events seemed to be largely related to multi-TKIs, rather than anti-PD-1 monotherapy. 15 To identify safety profile with multi-TKI plus ICI combination therapy, it is necessary to separately investigate severe and common treatment-related adverse events.
Our systematic review and analysis indicated that the incidence rates of diarrhea, hypothyroidism, and rash were increased by adding ICIs to multi-TKIs, but these events have been reported as common adverse events in ICI monotherapy. As for diarrhea, colitis has been known as an immune-related adverse event (irAE) caused by ICI monotherapy, and diarrhea could be developed as a symptom of colitis.16,17 Forty-five of 49 studies on multi-TKI monotherapy and 31 of 35 studies on multi-TKI plus ICI combination therapy reported diarrhea as a treatment-related adverse event with an incidence rate of more than 10%. According to the results of RR against sunitinib, multi-TKI plus ICI combination therapy increased the risk of diarrhea. Although diarrhea has been recognized as a side effect of multi-TKI monotherapy, appropriate measures should be taken for multi-TKI plus ICI combination therapy because the addition of ICI could further increase the risk of diarrhea.
In hypothyroidism, the thyroid is one of the organs associated with the immune system, and some autoimmune diseases can develop because of thyroid abnormalities. Therefore, hypothyroidism is clearly one of the side effects associated with ICIs and has already been reported as a common irAE for ICI monotherapy. 18 In this study, 28 of 35 studies on multi-TKI plus ICI combination therapy presented hypothyroidism as a treatment-related adverse event, with an incidence rate of more than 10%. On the contrary, in the studies on multi-TKI monotherapy, the number of studies with more than 10% incidence rate of treatment-related hypothyroidism differed depending on the multi-TKI compound. Therefore, it should be considered whether the combination therapy of multi-TKI and ICI increases the risk of hypothyroidism additively or synergistically.
Rash is also one of the side effects related to the immune system and is a well-known irAE of ICI monotherapy. 19 It has been reported that irAEs, such as disorders of the skin, endocrine organ, and gastrointestinal tract could be predictors of the efficacy of ICI monotherapy. 20 In the present study, 17 of 49 studies on multi-TKI monotherapy and 25 of 35 studies on multi-TKI plus ICI combination therapy presented rash as a treatment-related adverse event with an incidence rate of more than 10%. Rash was reported most frequently with regorafenib in 7 studies. In addition, some multi-TKI plus ICI combination therapies showed more than 2-fold increase in RR of rash against sunitinib; the RR differed depending on the type of combination of multi-TKIs and ICI. Therefore, it should also be considered whether the combination therapy of multi-TKIs and ICIs causes an increased risk of rash additively or synergistically. Overall, the risk of these adverse events could be increased by adding ICIs to multi-TKI monotherapy. It is important to discuss these adverse events with specialists early and to take appropriate measures, which will improve patient safety. 21
The pooled incidence rates of fatigue, hand-foot syndrome, and proteinuria were decreased by the addition of ICIs to multi-TKI monotherapy. The results of health-related quality of life in previous phase 3 trials indicated improved performance status of multi-TKI plus ICI combination therapy compared with multi-TKI monotherapy.22,23 Although the results of the present study should be interpreted with caution because of limited data, the improvement in adverse events related to quality of life was consistent with the previous reports.
The prediction of adverse events has been a challenge for many years. Several machine learning and data mining techniques are explored to find the treatment-related adverse events. 24 For the safety assessment of multi-TKI plus ICI combination therapy, the use of these techniques may enable more accurate identification of adverse events based on safety information such as real-world data.
Our study had several limitations. First, only treatment-related adverse event data with >10% incidence rates were collected in this study. Second, the type of cancer was not specified in this study, and clinical studies on various types of cancer were included in the analyses. However, data on treatment-related adverse events were collected to minimize the influence of using data for various types of cancer on the analyses. Finally, because several types of multi-TKIs and ICIs were analyzed together, the results of this study are highly heterogeneous.
Conclusion
Our systematic review and analysis identified diarrhea, hypothyroidism, and rash as treatment-related adverse events leading to increased toxicities with multi-TKI plus ICI combination therapy compared with multi-TKI monotherapy. Additionally, some adverse events related to performance status were improved by the addition of ICIs to multi-TKIs. The results of the present study are expected to optimize the management of toxicities caused by multi-TKI plus ICI combination therapy in individual patients, as the number of clinical studies using this combination therapy and opportunities for their use in clinical practice are expected to increase. As this study was conducted based on the results of limited clinical studies, further investigation is needed based on the accumulating results of future clinical studies and real-world data.
Supplemental Material
Supplemental Material - A Systematic Review of Treatment-Related Adverse Events for Combination Therapy of Multiple Tyrosine Kinase Inhibitor and Immune Checkpoint Inhibitor
Supplemental Material for A Systematic Review of Treatment-Related Adverse Events for Combination Therapy of Multiple Tyrosine Kinase Inhibitor and Immune Checkpoint Inhibitor by Takashi Sawada and Mamoru Narukawa in Political Cancer Control
Footnotes
Author’s Contributions
Sawada T (study design, data collection, data analysis, data interpretation, and manuscript writing), Narukawa M (data interpretation and manuscript writing).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Takashi Sawada is an employee of MSD K.K. (a subsidiary of Merck & Co., Inc., Kenilworth, N.J., USA). Mamoru Narukawa declares that he has no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
The authors confirm that all data generated or analysed during this study are included in this published article.
Supplemental Material
Supplemental material for this article is available online
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
