Abstract
Background
The proportion and impact of minimal pleural effusion (PE) on prognosis remain blurred in operable non-small cell lung cancer (NSCLC) patients who reported minimal PE on imaging.
Methods
Clinical and prognostic data of operable NSCLC patients who presented no distant metastasis, no direct pleural invasion, but minimal PE on preoperative imaging were retrospectively analyzed. The patients were divided into surgical (81 cases) and non-surgical (10 cases) cohorts. Potential or suspected malignant PE or pleural metastases were confirmed by surgery. The overall survival (OS) was analyzed by Kaplan-Meier curve and prognostic factors by multivariate Cox regression.
Results
The surgical cohort reported no pleural invasion on preoperative imaging and no pleural metastases by postoperative pathology, with an overall median disease-free survival of 36.7 (28.6, 44.7) months and a median OS of 59.8 (45.7, 73.2) months, with the latter significantly longer in the surgical cohort than in the non-surgical cohort (59.8 months vs 20.37 months, P < 0.001). Multivariate analysis indicated surgical treatment as an independent prognostic factor for OS.
Conclusion
Malignant PE is rare in operable NSCLC patients who report the presence of minimal PE but no distant metastasis or direct pleural invasion on preoperative imaging. Surgery may be the preferred treatment for these patients.
Plain Language Summary
Subject overview
The proportion and impact of minimal pleural effusion (PE) on prognosis remain blurred in operable non-small cell lung cancer (NSCLC) patients who reported minimal PE on imaging.
Methods
Clinical and prognostic data of operable NSCLC patients who presented no distant metastasis, no direct pleural invasion, but minimal PE on preoperative imaging were retrospectively analyzed. The patients were divided into surgical (81 cases) and non-surgical (10 cases) cohorts. Potential or suspected malignant PE or pleural metastases were confirmed by surgery. The overall survival (OS) was analyzed by Kaplan-Meier curve and prognostic factors by multivariate Cox regression.
Results summary
We found that the surgical cohort reported no pleural invasion on preoperative imaging and no pleural metastases by postoperative pathology, with an overall median disease-free survival of 36.7 (28.6, 44.7) months and a median OS of 59.8 (45.7, 73.2) months, with the latter significantly longer in the surgical cohort than in the non-surgical cohort (59.8 months vs. 20.37 months, p < 0.001). Multivariate analysis indicated surgical treatment as an independent prognostic factor for OS.
Key takeaways
We remain that malignant PE is rare in operable NSCLC patients who report the presence of minimal PE but no distant metastasis or direct pleural invasion on preoperative imaging. Surgery may be the preferred treatment for these patients.
Introduction
Lung cancer, 1 of the most commonly diagnosed cancers (11.6%), is the leading cause of cancer-related deaths worldwide, 1 with non-small cell lung cancer (NSCLC) accounting for approximately 80%-85% of all lung cancer cases. 2 Pleural effusion (PE) is a frequent complication of lung cancer, which has wreaked havoc on the health care system worldwide due to its high mortality rate. 1 The most common causes of malignant PE include lung cancer (37.5%), breast cancer (16.8%), and lymphoma (11.5%). 3 Pleural metastases of non-small cell lung cancer (NSCLC), regardless of the extent of PE and pleural nodules, are designated as an M1a descriptor (stage IVA) in the latest (eighth) edition of the tumor-node-metastasis (TNM) classification and are usually deemed unsuitable for surgical resection, 4 as malignant PE typically indicates an advanced disease progression and a poor prognosis,5,6 and requires local perfusion therapy.7-9 Therefore, an early determination of the nature of PE seems critical and significant for a timely intervention.
The diagnosis of malignant PE requires the presence of tumor cells in PE or pleural biopsy. 10 The amount of effusion and pleural thickening can be assessed by computed tomography (CT), 11 with a specificity of 78% to 100%. However, the sensitivity of CT in PE diagnosis is far from satisfactory, varying from 36% to 68% in practical reports. 12 Alternatively, thoracentesis 13 is often performed for PE diagnosis, which is considered as an appropriate first step. However, approximately 1 in 3 PE patients is prone to missed diagnoses with a single thoracentesis. 14 Currently, it remains a challenge to properly distinguish benign from malignant PE merely by imaging means. 15 Therefore, surgical intervention seems to be a crucial option for a correct diagnosis of PE.
When PE shows a minimal thickness (<10 mm) on the CT scan, it is defined as minimal PE. To date, the diagnosis and prognosis of minimal PE have received limited attention. A recent retrospective study reports that local anesthesia thoracoscopy (LAT) has a sensitivity and specificity of 92.8% and 100% in the diagnosis of minimal PE, respectively. 16 However, the study also indicates that only a few patients with minimal PE received this inspection, with 4 out of 5 patients with minimal PE undergoing LAT and diagnosed as malignant PE. Another study shows a high proportion of minimal PE in a cohort of 2340 NSCLC patients (272/2,340, 13.2%), with patients with minimal PE reporting a significantly shorter survival time than patients without PE (7.7 months vs 17.7 months), and an increasing risk of death by 40%. 17 So far, it remains blurred regarding the proportion of confirmed malignant PE in patients with minimal PE and no definite pleural invasion by imaging is available, let alone their prognostic outcomes.
In the current study, a retrospective analysis was performed to assess the proportion of malignant PE in NSCLC patients with minimal PE, for whom no clear pleural invasion was indicated by imaging, and to explore the significance of surgery for these patients. Clinical data of 2 patient cohorts were retrieved and compared. We found a low proportion of malignant PE in the patients undergoing surgical intervention and a significant survival difference in this cohort when compared with those bypassing the surgery. The findings may provide some insights into the prognostic value of surgical resection in treating NSCLC patients with minimal PE.
Materials and Methods
Study Design
This retrospective cohort study was designed to consecutively collect and analyze the clinical data and prognostic information of patients with NSCLC at operable T and N stages who reported minimal PE, no distant metastasis, and no preoperative indication of pleural invasion in Fujian Cancer Hospital from January 2004 to December 2022. The patients were divided into surgical and non-surgical cohorts according to their surgical status. The study was approved by the Ethics and Scientific Committees of Fujian Cancer Hospital (NO: K2023-213-01). The informed consent was obtained verbally from patients who were still alive during the follow-up (51 patients) or from their family members if the patients passed away amid the follow-up (40 patients). Privacy-related details of all patients were removed. The reporting of this study conforms to REMARK guidelines. 18
Participants
The inclusion criteria of the enrolled patients were as follows: 1. Age ≥18 years; 2. All patients were confirmed as NSCLC by histology or cytology; 3. According to the cancer staging manual of American Association for Cancer Research (Eighth Edition), the preoperative clinical stages included T and N stages that are operable without distant metastasis; 4. The score of Eastern cancer cooperation cohort (ECOG) fell within the range of 0-2; 5. All chest CT scans performed at the time of diagnosis were examined by 2 independent imaging experts, and the minimal PE was designated as effusion less than 10 mm.
Patients were excluded for the following conditions: clear pleural invasion on imaging, active pulmonary tuberculosis, other serious systemic diseases, other confirmed malignancies, and diseases that cause benign PE, ie, the dysfunction of heart, kidney, liver, and other organs.
Measurement of Variables
Patient-related variables included age, gender, smoking history, carcinoembryonic antigen (CEA), hemoglobin (HB), albumin (ALB), serum calcium levels, and pathological characteristics. Tumor-related variables included PD-L1 expression, genetic testing, T stage, tumor size, and N stage. Treatment-related variables involved operation time, surgical approach, postoperative complications, and the administration of neoadjuvant or adjuvant chemotherapy.
Outcomes
Recurrence was defined as the re-emergence of the same lesion from the original surgical site after the surgical treatment or tumor formation of the same type in another site. Time to recurrence was defined as the time from the time of surgery to the time of the first recurrence. Disease-free survival (DFS) was designated as the time from surgery to recurrence at the follow-up, and overall survival (OS) as the time from diagnosis to death or the end of follow-up.
Statistical Analysis
All statistical analyses were performed using SPSS® Software version 24.0 (IBM Corp., Armonk, NY, USA). The baseline characteristics of the patients were analyzed by descriptive statistics. Categorical data were expressed as percentage and analyzed by chi square test or Fisher’s exact test. Survival was analyzed by the Kaplan-Meier method and the log-rank test. Factors affecting PFS were evaluated by the Cox proportional hazards regression model. Significant differences between cohorts were analyzed by the log-rank test. A two-tailed P-value of less than 0.05 was considered statistically significant.
Results
Clinicopathological Characteristics of Minimal PE in Patients Receiving Surgical Treatment or Non-surgical Treatment
A total of 4455 patients were diagnosed with NSCLC, including 1680 patients who reported no distant metastases and an operable T or N stage. After further imaging screening, 91 cases of minimal PE without clear pleural invasion were enrolled in the study, with a median age of 63 ± 5 years old (Figure 1), of which, 81 cases (Cohort 1) underwent surgical resection and 10 cases (Cohort 2) received non-surgical treatment due to patients’ refusal or intolerance. In the enrolled patients, there were 54 smokers, 27 cases of stage I, 18 cases of stage II, and 46 cases of stage III, with adenocarcinoma as the most common pathological type (n = 45, 49.45%) (Table 1). Flow diagram for the enrollment of NSCLC patients with minimal PE in the study. Clinicopathological Characteristics of Minimal PE Patients Between Surgical Treatment and Non-surgical Treatment Groups. Data are presented as N (%) unless otherwise noted. The stage of all patients was defined according to the eighth edition of the TNM classification.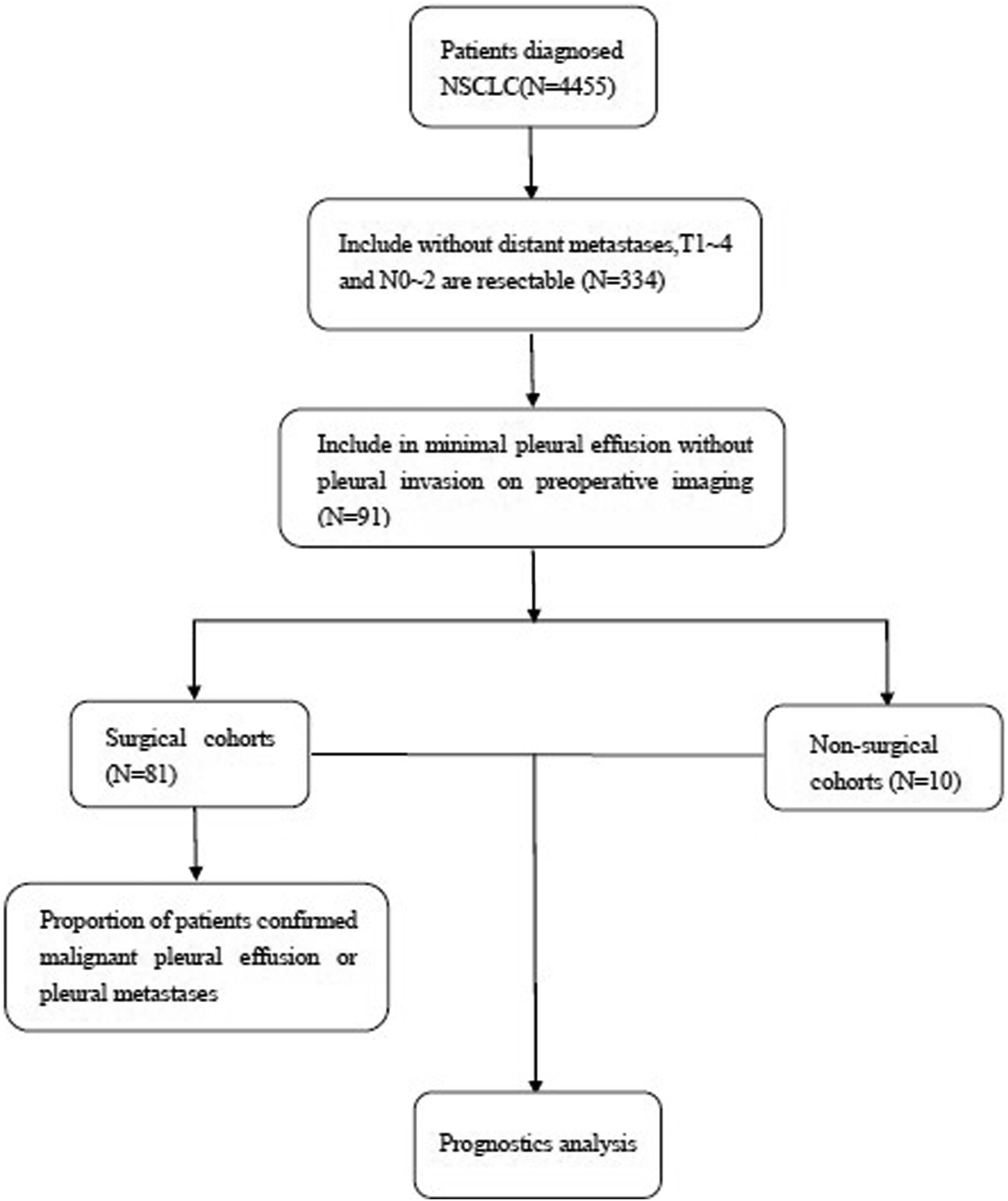

The clinicopathological characteristics were compared between Cohort 1 and Cohort 2, reporting no difference in age, gender, smoking history, CEA, HB, ALB, serum calcium levels, pathological characteristics, or T and N stages (P > 0.05 for all) (Table 1).
The Proportion of Malignant PE in Cohort 1
We further detected the proportion of malignant PE in all patients. Due to the lack of diagnostic examinations for PE in the non-surgical cohort, only patients in the surgical cohort were analyzed by postoperative pathology for the proportion of malignant PE, which reported no incidence of pleural metastases.
Prognostic Analyses of NSCLC Patients with Minimal PE
The overall median DFS and median OS were 29.9 (95% CI: 20.4, 39.5) months and 56.8 (95% CI: 48.6, 65.1) months, respectively (Figure 2). We further investigated the prognosis of NSCLC patients with minimal PE. On the basis of surgical status, the median DFS was significantly longer in the surgical cohort than in the non-surgical cohort [(36.7 months (95% CI: 28.6-44.8) vs 8 months (95% CI: 1.4-14.6), P < 0.001). The median OS was significantly longer in the surgical cohort than in the non-surgical cohort [59.5 months (95% CI: 45.7-73.2) vs 20.4 months (95% CI: 13.5-27.2), P < 0.001] (Figure 3). Moreover, the multivariate Cox regression analysis showed that surgical treatment was an independent prognostic factor for OS (Table 2). As the control group displayed a high proportion of N2 stage patients, though with no significant statistical difference, we further analyzed the prognosis of patients with N2 nodal stage. The results showed that the surgical cohort reported longer DFS or OS when compared with the non-surgical counterpart [DFS: 19.0 months (95% CI: 14.6-23.4) vs 4 months (0-9.6); OS: 60.8 months (95% CI: 26.7-94.8) vs 18 months (95% CI: 10.6-25.4)]. Despite the limited sample size, the finding in the N2 stage patients was consistent with that of the cohort comparison (Suppl. Figure 1). The prognosis of NSCLC patients with minimal PE. (A) DFS of patients with minimal PE. (B) OS of patients with minimal PE. Kaplan-Meier survival curves for NSCLC patients with minimal PE received surgical intervention or non- surgical intervention. (A) DFS of patients who received surgical intervention or non- surgical intervention. (B) OS of patients who received surgical intervention or non- surgical intervention. Multivariate Analysis for Factors Predictive of Overall Survival.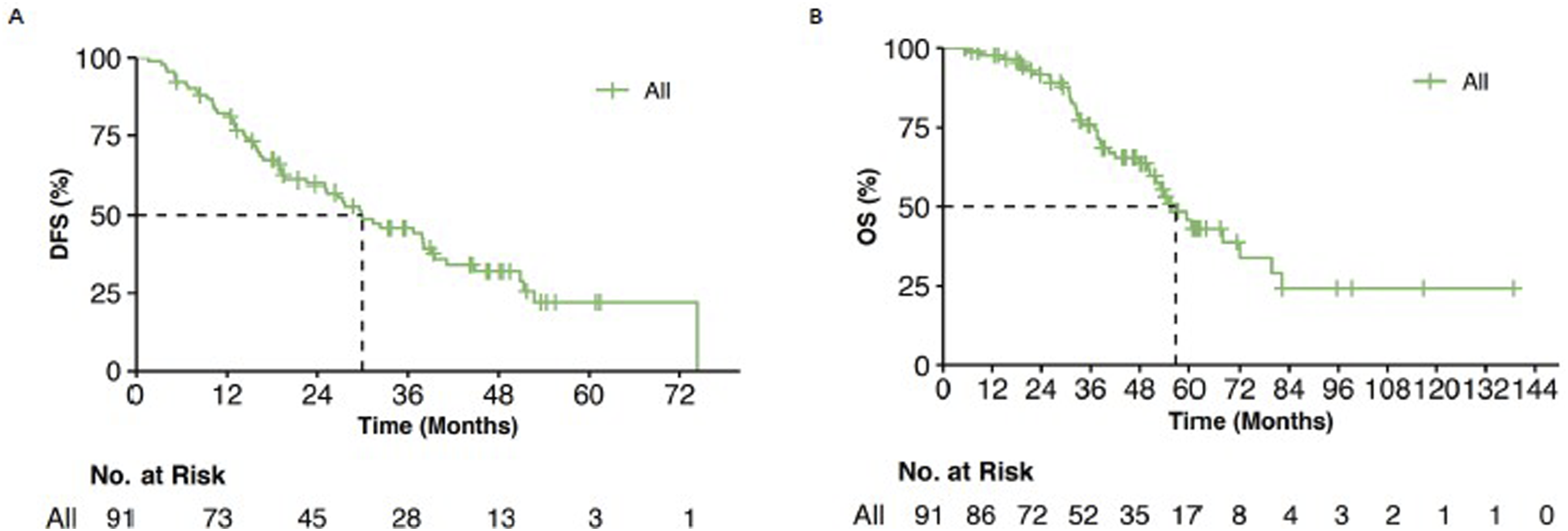


Prognostic Analyses of NSCLC Patients with Minimal PE in Cohort 1
The overall median DFS and median OS in the surgical cohort were 36.7 (95% CI: 28.6, 44.8) months and 59.5 (95% CI: 45.7, 73.2) months, respectively (Figure 4). Further analyses revealed different median DFS and median OS in the Cohort 1 of different stages: The median DFS was 51.3 months in stage I, 29.8 months in stage II, 18.8 months in stage III (P = 0.008); the median OS was 72.0 months in stage I, 53.0 months in stage II, 42.0 months in stage III (P = 0.010) (Figure 5). The prognosis of NSCLC patients with minimal PE in the surgical group patients (cohort 1). (A) DFS of patients who received surgical intervention. (B) OS of patients who received surgical intervention. The overall median DFS and median OS in the surgical group patients between different stages. (A) DFS of patients who received surgical intervention in different stages. (B) OS of patients who received surgical intervention in different stages.

Discussion
In the current study, a significantly longer median OS and no postoperative pleural metastases were reported in the 81 patients with minimal PE who reported no pleural invasion in preoperative imaging when compared with the non-surgical counterparts. The findings seem to suggest that surgery may be the preferred treatment for potentially operable NSCLC patients with minimal PE on imaging.
Minimal PE has imposed a challenge on clinicians for an accurate clinical diagnosis. However, its incidence has remained low in clinical practice. Retrospective studies have found that minimal PE is uncommon in patients who underwent surgical resection. A study of 9576 surgical NSCLC cases found that only 83 cases (0.9%) met the definition of ‘unexpected’ macroscopic malignant pleural nodules, including 52 cases without visible PE and 31 cases with a minimal volume of PE. 19 Another study reported that minimal PE was present in 125 out of 2061 patients (6.1%), with the proportion of each stage as follows: stage I, 5.2%; stage II, 10.9%; stage IIIA, 13.2%; and stage IIIB, 23.8%. 17 Consistently, the current study found no malignant PE in those undergoing surgical resection. These findings seem to suggest that the proportion of minimal PE in operable patients is extremely low.
Despite the low proportion, minimal PE has been reported to be a poor prognostic factor for either NSCLC or SCLC. Available evidence shows that minimal PE is significantly correlated with a shorter survival time in patients with minimal PE than those with no PE (median OS: 7.7 vs 17.7 months). 17 A review of 2093 consecutive NSCLC patients who underwent thoracotomy from January 2006 to January 2015 reported malignant PE in 110 NSCLC patients (110/2,093, 5.26%), reporting a 5-year estimated survival rate of 28%. 20 Another study of SCLC patients found minimal PE in 74 patients (20.6%) and malignant PE in 83 patients (23.0%), with a significantly different median survival in patients with no, minimal, or malignant PE (median survival: 11.2, 5.93, and 4.83 months, respectively; P < 0.001). 21 Collectively, these findings suggest that minimal PE can serve as a prognostic indicator for patients with lung cancer.
As surgery is not a preferred treatment for patients with malignant PE and PE cannot be clearly indicated in all patients before surgery, malignant PE or pleural nodules may be accidentally detected in some patients during surgery.
Although surgery is not a preferred treatment for patients with malignant PE, existent studies indicate that it can improve the overall survival of patients with PE. A previous study found that the median OS after surgery was significantly longer in the patients without PE than in the PE group (37 vs 22 months, P = 0.005), 19 which also confirms that malignant PE is an indicator of a poor prognosis. A Japanese study of 11,420 registered NSCLC patients showed that macroscopic complete resection was associated with a better survival in 329 patients with pleural carcinomatosis. 22 In the current study, the overall survival of the surgical cohort was significantly longer than that of patients without PE, with a median DFS and median OS of 36.7 (28.6, 44.7) months and 59.8 (45.7, 73.2) months, respectively. Taken together, these pieces of evidence suggest that survival resection seems to be a preferred alternative for the treatment of patients with minimal PE.
Although minimal PE is a factor for poor prognosis, primary tumor resection is still associated with a better survival of patients with minimal PE. A previous study found, after tumor resection, a long-term survival in patients with T1-2 of NSCLC and only a small amount of PE, in which given the T factor of the primary tumor, regardless of the presence or absence of PE or pleural dissemination, patients with T1 (MST: 37 months) or T2 (MST: 15 months) showed a significantly better prognosis than those with T3 (MST: 9.9 months) (P = 0.0004 and 0.011, respectively). 23 Another study reported high CEA level (P = 0.006), advanced N stage (P = 0.005), adenocarcinoma (P = 0.001) and pleural invasion (P = 0.041) as independent risk factors for the incidence of pleural metastasis. 20 In line with previous studies, 4 the current study demonstrated prognostic differences in patients undergoing surgical resection at different stages, showing surgical treatment to be an independent prognostic factor for OS. These findings suggest that the nature of PE should be determined as accurately as possible in these patients and that surgical resection can serve as the first choice for patients with potentially resectable minimal PE but no distant metastases.
Some limitations remain in the current study, however. First, this was a single-center study with a small sample size, which may limit the extent to which our findings can be generalized to other populations. Second, the sample size of the non-surgical cohort was relatively small and the qualitative diagnosis of the small effusions in this cohort was unavailable. Third, as this was a retrospective study, bias in the type and the modality of surgery for the surgical cohort cannot be controlled. Therefore, prospective large-sample studies are needed to further validate the results of this study.
Conclusions
This retrospective study reports an extremely low proportion of malignant PE in patients with minimal PE but no pleural invasion on preoperative imaging. The findings suggest that surgery may be the preferred treatment for potentially operable NSCLC patients with minimal PE on imaging.
Supplemental Material
Supplemental Material - Prognostic Value of Surgical Resection for Non-small-cell Lung Cancer Patients Comorbid With Minimal Pleural Effusion
Supplemental Material for Prognostic Value of Surgical Resection for Non-small-cell Lung Cancer Patients Comorbid With Minimal Pleural Effusion by Haibo Wang, Tao Lu, Xinlong Zheng, Kan Jiang, Longfeng Zhang, Xiaobin Zheng, Qian Miao, Shiwen Wu, Zihua Zou, Shanshan Yang, Yujing Li, Sihui Chen, Yiquan Xu, and Gen Lin in Cancer Control.
Footnotes
Acknowledgments
The authors thank Prof. Hongzhi Huang for English language editing.
Author Contributions
Yiquan Xu and Gen Lin conceived the study and devised the study’s design. Haibo Wang performed this study. Tao Lu Performed data analysis for clinical records. Xinlong Zheng, Longfeng Zhang, Kan Jiang, Xiaobin Zheng, Qian Miao, Shiwen Wu, Zihua Zou, Shanshan Yang, Yujing Li and Sihui Chen collected the clinical records. Haibo Wang wrote the manuscript in consultation with Gen Lin. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Startup Fund for scientific research, Fujian Medical University (No. 2021QH1147), the National Natural Science Foundation of China (Grants No. 82072565 and No. 82372954), the Fujian Provincial Health Systemic Innovation Project (Grant No. 2020CXA010), the Scientific and Technological Innovation Joint Capital Projects of Fujian Province (Grant No. 2020Y9038), the Beijing Xisike Clinical Oncology Research Foundation (Grant No. Y-2019AZZD-0386), the Major Project for Young and Middle-aged Scientists of Fujian Provincial Health Commission (Project No. 2023ZQNZD008), the High-Level Talent Training Project of the Fujian Cancer Hospital Committee (Project No. 2024YNG04), the Joint Funds for the Innovation of Science and Technology, Fujian Province (2021Y9226), the High-level Talent Development Program of Fujian Cancer Hospital (2023YNG14) and the Fujian Cancer Hospital “Outstanding Youth” Project (to Y. Xu).
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
