Abstract
Background
Epidermal growth factor receptor exon 20 insertion (EGFR ex20ins), an uncommon mutation in non-small cell lung cancer (NSCLC), can induce poor patient response to EGFR tyrosine kinase inhibitors (EGFR-TKI). However, the clinical features and prognosis of patients with EGFR ex20ins are not clearly understood. This study investigated the clinical characteristics and prognosis of advanced NSCLC patients with EGFR ex20ins.
Methods
Advanced NSCLC patients treated at Fujian Cancer Hospital were consecutively recruited from June 1, 2014 to December 20, 2021 and retrospectively examined. EGFR ex20ins was identified by polymerase chain reaction (PCR) or next-generation sequencing (NGS). The clinical characteristics, treatment methods, and patient outcomes were retrieved from the hospital database. The progression-free survival (PFS) and overall survival (OS) were assessed by Kaplan-Meier analysis.
Results
Fourteen mutation subtypes of EGFR ex20ins were identified in the 24 enrolled patients, with EGFR ex20ins mutation more prevalent in non-smoking women. A763_Y764insFQEA and A767_V769dup (12.5% for both) were the most common mutation subtypes. Notably, no significant differences in PFS and OS were found between the first-line targeted therapy group [PFS: 257 days, 95% confidence interval (CI): 116-397 days; OS: not reached] and chemotherapy-based combination therapy group (PFS: 182 days, 95% CI: 156-207 days; OS: 998 days, 95% CI: 674-1321 days). TP53 mutation was the commonest concomitant mutation (62%), followed by EGFR amplification (25%). Chemotherapy combined with immunotherapy improved the prognosis of patients with high PD-L1 expression.
Conclusion
For NSCLC patients with EGFR ex20ins, limited therapeutic benefits can be gleaned from either EGFR-TKIs or chemotherapy-based combination therapy.
Plain Language Summary
EGFR-TKIs have limited efficacy in NSCLC patients with EGFR ex20ins. Combining chemotherapy with immunotherapy may represent a promising treatment approach for individuals with positive ex20ins and high PD-L1 expression.
Keywords
Introduction
Lung cancer, one of the most commonly diagnosed cancers (11.6%), is the leading cause of cancer-related deaths worldwide, 1 with non-small cell lung cancer (NSCLC) accounting for approximately 80%-85% of all lung cancer cases. 2 Among the most common mutations in NSCLC, mutations or deletions in the epidermal growth factor receptor gene (EGFR) are detected in 15%-20% of NSCLC cases in the Caucasian population and up to 50% in the Asian population,3-6 in which deletion in exon 19 and the point mutation L858R in exon 21 account for about 85% of EGFR mutations. 7 EGFR 20 insertion (EGFR ex20ins) constitutes about 1%-2% of all NSCLC and is classified as a rare mutation. To date, the clinical characteristics and prognosis of these patients with EGFR ex20ins are poorly understood and await further exploration.
Despite the similarity of a high incidence in non-smoking females,8,9 compared with patients with common EGFR mutations, NSCLC individuals with EGFR ex20ins are diagnosed at a younger age. 10 Unlike traditional EGFR mutations, EGFR ex20ins does not affect the ATP binding pocket that determines the kinase activity. Instead, it forms a wedge at the end of the C helix, which enhances active kinase conformation but does not increase the affinity for EGFR tyrosine kinase inhibitors (TKI). 11 The decreased drug affinity may arise from steric hindrance, which causes displacement of the C-helix and the phosphate-binding loop of EGFR within the drug-binding pocket. 12 The majority of EGFR ex20ins mutations can increase resistance to both the first-generation EGFR-TKIs like gefitinib and erlotinib, with an objective response rate (ORR) of approximately 5%, and the second-generation EGFR-TKIs, including afatinib and dacotinib. 13 In the latter, regardless of the high efficacy of afatinib against some uncommon mutations (G719X, L861Q, and S768I), a post hoc analysis of clinical trials with LUX-Lung 2, LUX-Lung 3, and LUX-Lung 6 reveals poor ORR and median progressive free survival (PFS) in NSCLC patients with EGFR ex20ins (ORR: 8.7% and PFS: 2.7 months). 14 Nonetheless, the third-generation TKI, osimertinib, demonstrates some efficacy against EGFR ex20ins in vitro, 15 yielding an ORR of approximately 5% and a median PFS of roughly 3.6 months. 16 However, despite the observed responses of EGFR ex20ins to these inhibitors, these inhibitors have not undergone phase III clinical trials. Recently, the U.S. Food and Drug Administration (FDA) has approved several drugs with high selectivity and effectiveness against EGFR ex20ins for the treatment of metastatic NSCLC patients with EGFR ex20ins following platinum-based therapies, including the EGFR-MET bispecific monoclonal antibody, amivantamab (JNJ-61186372), and mobocertinib (TAK-788).17,18 In addition, some studies have documented a strong activity from poziotinib, CLN-081 (TAS6417) and DZD9008, against EGFR ex20ins.19-21 However, the efficacy of these targeted drugs against EGFR ex20ins (with an median PFS of 7-10 months) is still far worse than that of the third-generation EGFR-TKIs against EGFR classical mutations.
Given that these FDA-approved targeted drugs are still not the first-line standard, together with the expensive drug price and limited drug accessibility, this study attempted to retrospectively analyze the clinical characteristics and prognosis of NSCLC patients with EGFR ex20ins in our center in the hope of shedding more lights on the clinical management of this particular patients‘ population.
Materials and methods
Patients and Data Collection
This retrospective study recruited NSCLC patients with EGFR ex20ins treated at Fujian Cancer Hospital from June 1, 2014, to December 20, 2021. The clinical information of patients was obtained from the medical records. In addition, the clinical and pathological characteristics of the patients and their responses to chemotherapy, TKIs, or immune checkpoint inhibitors (ICIs) were analyzed. The patients were divided into 2 groups respectively receiving targeted therapy (the targeted therapy group) and chemotherapy-based combination therapy (the chemotherapy-based combination therapy group) according to the first-line treatment plan. The inclusion criteria of the enrolled patients were as follows: (i) age ≥18 years; and (ii) diagnosis of inoperable locally advanced or metastatic NSCLC; (iii) the presence of EGFR ex20ins detected by polymerase chain reaction (PCR) or next-generation sequencing (NGS). The exclusion criteria were: (i) incomplete data; (ii) patients with other malignant tumors; and (iii) patients with severe failure of other major organs. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and approved by the Ethics and Scientific Committees of Fujian Cancer Hospital (No. K2023-136-01). The informed consent was obtained verbally from patients who were still alive during the follow-up (15 patients) or from their family members if the patients passed away amid the follow-up (9 patients). Privacy-related details of all patients were removed. The reporting of this study conformed to the STROBE guidelines. 22
Testing for EGFR 20ins Mutations
EGFR ex20ins was detected in lung tumors, peripheral blood, pleural effusion, or cerebrospinal fluid samples by PCR or NGS. Given the limitations of PCR in detecting the various EGFR ex20ins mutations, all types of EGFR ex20ins were considered in this study.
Treatment Assessment and Efficacy Evaluation
In all patients, treatment strategies were chosen by the attending physician on the basis of the physical and disease status of the patient. Baseline imaging of the chest and abdomen was conducted by CT, magnetic resonance imaging (MRI), or whole body scanning of measurable target lesions according to the radiologic criteria for solid tumor response (RECIST; version 1.1). Treatment response was evaluated one month after the treatment and followed up every 2 months, which was based on the medical records and categorized as complete response (CR), partial response (PR), stable disease (SD), or disease progression (PD). The objective response rate (ORR) was defined as the proportion of patients showing PR and CR in all the analyzed cases that received a similar treatment. The disease control rate (DCR) was designated as the proportion of patients showing PR, CR, and SD in all cases that received the same treatment. PFS 1 was calculated from the initiation of the first-line treatment to the point of disease progression or death. PFS 2 was calculated from the initiation of the second-line treatment to the point of disease progression or death. Overall survival (OS) was calculated from the time of diagnosis to the end of follow-up or the time of death.
Statistical Analyses
All statistical analyses were performed using SPSS® Software version 24.0 (IBM Corp., Armonk, NY, USA). The baseline characteristics of the patients were analyzed by descriptive statistics. Categorical data were expressed as a percentage and analyzed by chi square test or Fisher’s exact test. Survival was analyzed by the Kaplan-Meier method and the log-rank test. Factors affecting PFS were assessed by the Cox proportional hazards regression model. Significant inter-group differences were evaluated by the log-rank test. The heatmap was depicted with the R software package to visualize gene mutations. A two-tailed P-value of less than .05 was considered statistically significant.
Results
Patient Characteristics
A total of 33 patients were initially included in this study. Among them, 9 were excluded because of incomplete data. Among the included cases, 14 (58.3%) patients were female and 10 (41.7%) were male, with a median age of 62 (51-71) years. Eighteen patients (75.0%) were non-smokers, whereas 6 were smokers. Twenty-one cases (87.5%) were diagnosed at stage IV. The most common distant metastasis was the opposite lung (12, 50.0%), followed by bone metastasis (11, 45.8%), central nervous system metastasis (6, 25.0%), and liver metastasis (1, 4.2%). PD-L1 expression was found in 13 patients, including 8 cases (61.5%) with positive expression (≥1%) and 4 cases (37.5%) with high expression (≥50%). Mutations were mainly detected in the lung tumor tissue (22 cases, 91.7%).
The Baseline Characteristics of the 24 Patients With EGFR ex20ins Mutations.

Abbreviations: CNS, central nervous system; PD-L1, Programmed cell death 1 ligand 1.
EGFR ex20ins Mutation Subtypes and Concomitant Mutations
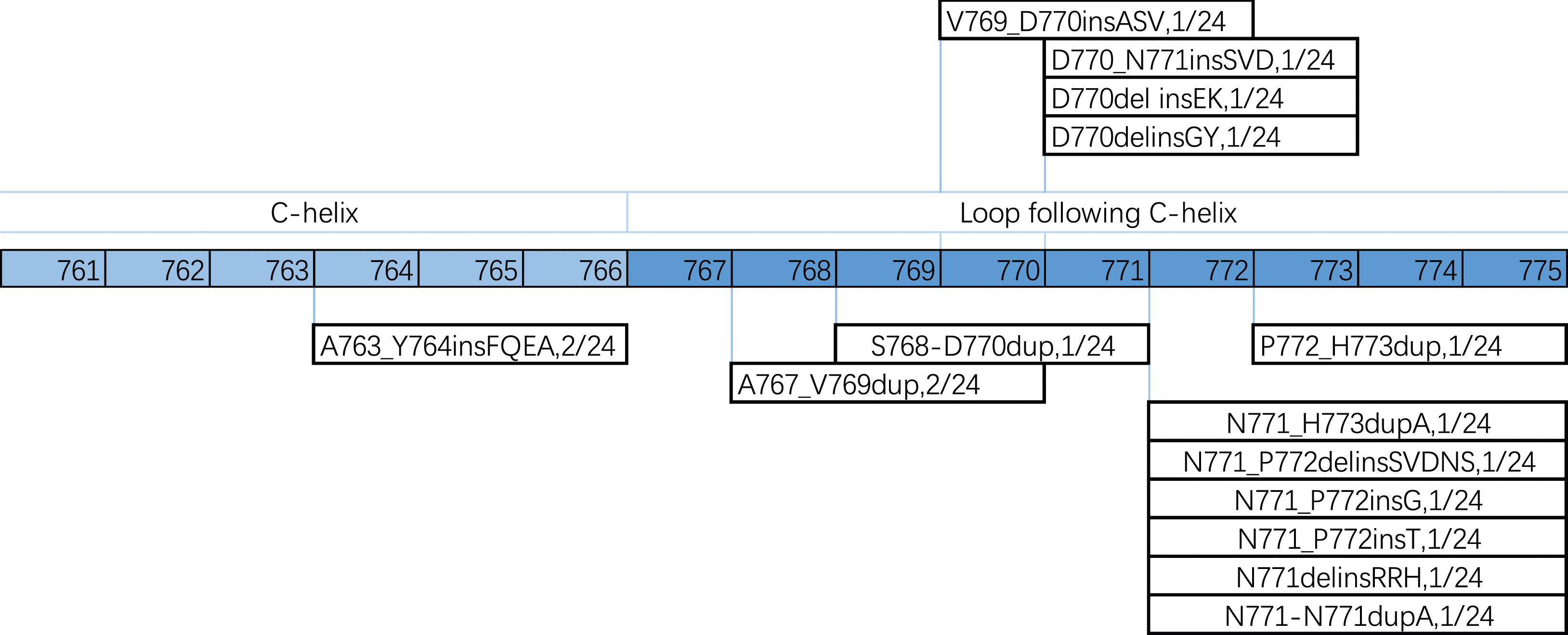
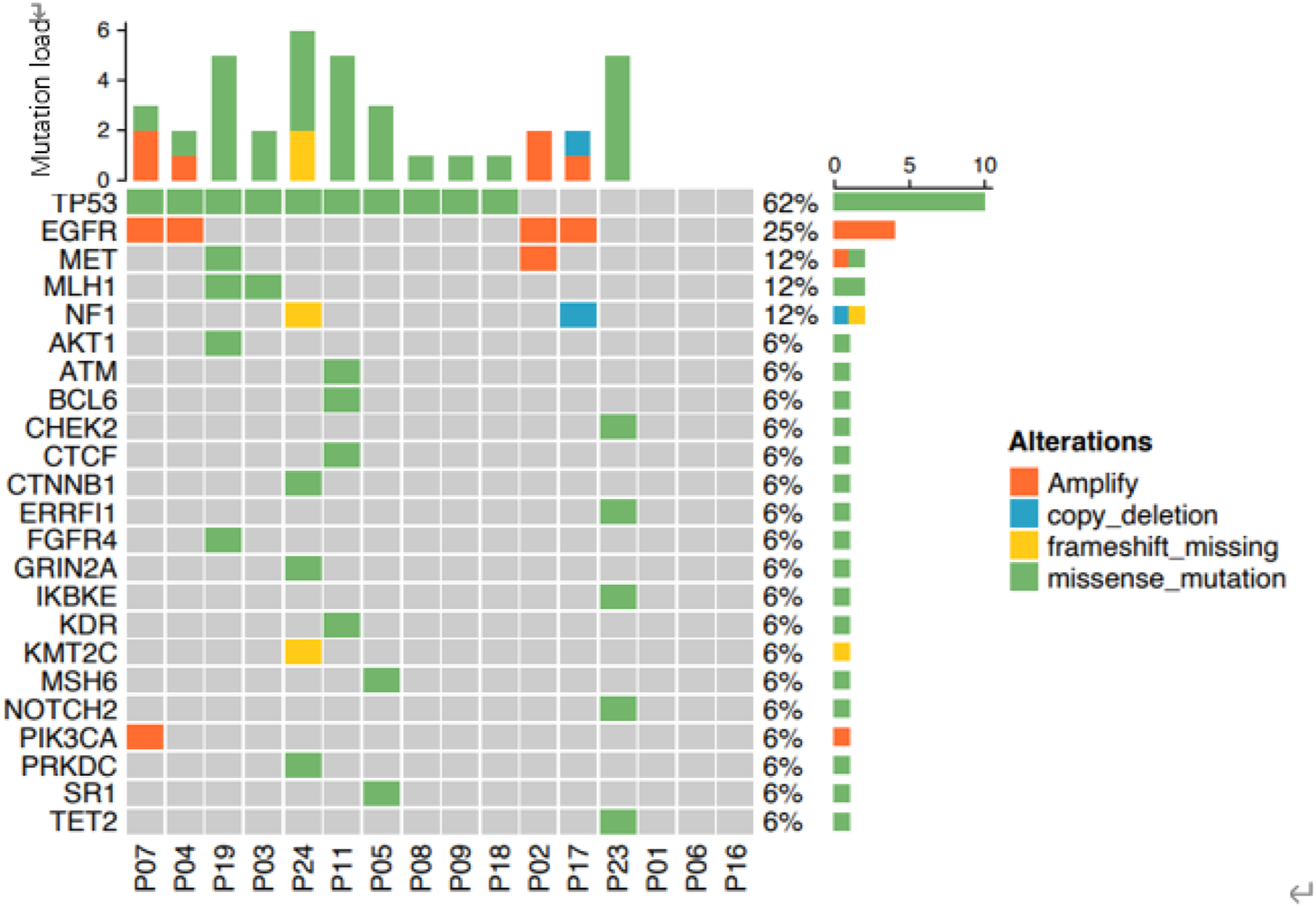
Eight EGFR ex20ins mutations were detected by PCR but the specific subtype was not determined. The remaining 16 patients were examined by NGS. Notably, A763_Y764insFQEA and A767_V769dup were the most common mutation subtypes (12.50% for both), while the other subtypes were reported in 6.25% of the patients, respectively (Figure 1). Further analyses revealed that D770 and N771 were the most frequent locus subtypes and that the majority (15/16, 93.8%) reported insertion mutations at the loop located at the posterior end of the C-helix (Figure 2). TP53 mutation was the common concurrent mutation accompanying EGFR ex20ins (62.50%), followed by EGFR amplification (25.00%), MET mutation/amplification (12.50%), MLH1 mutation (12.50%), and NF1 frameshift deletion/copy deletion (12.50%) (Figure 3). EGFR ex20ins mutation subtypes. Spectrum of EGFR ex20ins mutations in lung adenocarcinoma. Co-existing genetic alterations with EGFR ex20ins mutations in lung adenocarcinoma.
Patient Outcomes
Treatment Outcomes of Patients Receiving First-line Chemotherapy-based Combination or Targeted Therapies
Overall, 7 cases showed PR and 14 cases showed SD, with an ORR and DCR of 29.17% and 87.5%, respectively. No significant differences in PFS and OS were found between the first-line targeted therapy group [PFS: 257 days, 95% confidence interval (CI): 116-397 days; OS: not reached] and chemotherapy-based combination therapy group (PFS: 182 days, 95% CI: 156-207 days; OS: 998 days, 95% CI: 674-1321) (Figure 4). Efficacy of chemotherapy-based combination therapy or targeted therapy for NSCLC with EGFR ex20ins. (A) PFS1 of NSCLC patients with EGFR ex20ins at our center. (B) The OS of all the NSCLC patients with EGFR ex20ins. (C) PFS1 comparison between the first-line targeted therapy and chemotherapy-based combination therapy. (D) OS comparison between the first-line targeted therapy and chemotherapy-based combination therapy.
In the targeted therapy group, the 3 cases were treated with furmonertinib, in which 2 showed PR and one exhibited SD, reporting an ORR of 66.67%, DCR of 100%, and median PFS of 215 days. In the chemotherapy-based combination group, one patient, initially presenting PD-L1 negativity, was further treated with a combined regimen of nedaplatin, pemetrexed, and toripalimab, which led to a state of SD, with PFS1 and OS durations of 334 days and 776 days, respectively.
Treatment outcomes of patients receiving second-line treatments
Sixteen patients who showed disease progression received the second-line therapy, which comprised either a third-generation targeted therapy or chemotherapy-based regimens. Among the cases treated with the second-line treatment, 1 reported PR, 12 SD, and 3 PD, exhibiting an ORR of 8.3%, DCR of 81.3, and median PFS2 of 191 days (Figure 5). PD-L1 was highly expressed in 2 patients who received the second-line chemotherapy combined with immunotherapy, in which 1 patient treated with Impower150 mode (pemetrexed + platinum + bevacizumab + atezolizumab) showed a PFS2 and OS of 652 days and 986 days, respectively, and the other treated with the pemetrexed + carboplatin + sintilimab + anlotinib regimen showed a PFS2 and OS of 101 days and 390 days, respectively. PFS2 of patients treated with second-line treatments.
EGFR ex20ins variants
The most common subtype of EGFR ex20ins, A763_ Y764insFQEA subtype, was reported in 2 cases. Of them, one patient who was treated with first-line chemotherapy showed PD, with a PFS1 of 35 days and OS of 635 days; the other patient treated with a third-generation TKI as the first-line regimen, showed a PFS1 of over 258 days.
Treatment Outcomes of Patients Based on Brain Metastasis
For patients without brain metastasis (18 cases), the median PFS1 was 182 days (95% CI:123, 240), For those with brain metastasis (6 cases), the median PFS1 was 191 days (95% CI: 123, 432). No significant difference in median PFS1 and median OS was found between the 2 groups (Figure 6). The details of the patients with brain metastasis are summarized in Table 2. Median PFS1 and median OS of EGFR ex20ins patients with brain metastasis. Clinical Characteristics and Prognosis of EGFR ex20ins Patients With Brain Metastasis. Abbreviations: F, female; M, male; P, Pemetrexed; C, cisplatin; B, bevacizumab; R, radiotherapy.

Prognostic Analysis
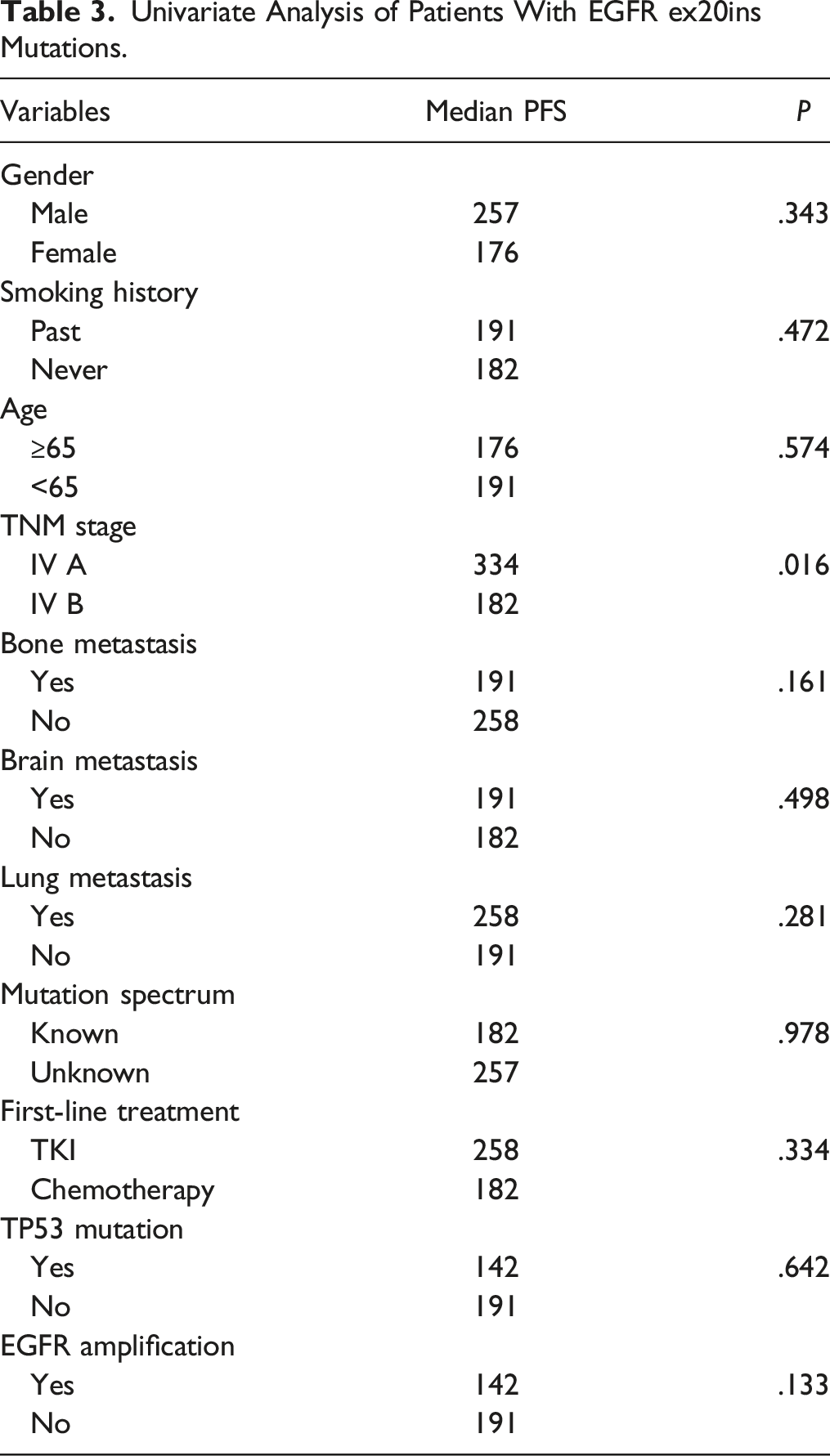
Univariate Analysis of Patients With EGFR ex20ins Mutations.
Discussion
This study demonstrated that patients with EGFR ex20ins responded poorly to EGFR-TKIs when compared with those with classical mutations like EGFR 19 del or EGFR L858R mutations. The findings suggest that chemotherapy combined with immunotherapy may be an effective treatment for patients with EGFR ex20ins who report a high PD-L1 expression. To our knowledge, this study, the first time, demonstrates that patients with EGFR ex20ins may benefit from chemotherapy combined with immunotherapy but not EGFR-TKIs.
EGFR ex20ins mutations have been reported in 4%-12% of EGFR-mutated NSCLC patients and approximately 2% of all NSCLC patients. 10 The incidence of EGFR ex20ins mutation was significantly lower in our cohort when compared with findings in western countries (3.49% vs 13.90%, P < .001). 24 In this study, EGFR ex20ins mutations were more common in women and non-smokers, which similar to the classical EGFR mutation in NSCLC. 8 The median age of enrolled patients at diagnosis was 62 (51-71) years. According to a previous study, 25 each subtype may be detected at varying ages. For example, V769_ D770insASV mutation is more common in patients aged over 65 years, while A763_ Y764insFQEA is more common in young people (≤65 years).
EGFR ex20ins mutations can be divided into 2 subgroups according to the amino acid insertion position: Ala767 to Cys775 affecting the C-helix tyrosine kinase domain of EGFR; and Glu762 to Met766 associated with insertion of amino acids in the C-helix of EGFR. In the present study, 15 cases (93.8%) developed mutations in the ring at the back end of the C-spiral, which is consistent with findings from a previous study. 13 In this study, we found that the TP53 mutation was the most common concomitant mutation (62%) associated with EGFR ex20ins. According to previous studies, 10 the most common co-occurring genomic alterations were TP53 (65%),10,26 cyclin-dependent kinase inhibitors 2A and 2B (CDKN2A and CDKN2B) (22% and 16%, respectively), NK2 homeobox 1 (NKX2-1) (14%), RB transcription corepressor 1 (RB1) (11%), 26 and PIK3CA (5%).25,27
A previous study suggests that NSCLC patients with classical EGFR mutations who reported a co-mutation in TP53 may respond more poorly to therapies when compared with those with sole EGFR mutations. 28 In this study, TP53 mutations did not significantly impact prognosis (P = .642). The inconsistency may be attributed to the small sample size recruited. As established, the tumor suppressor gene TP53 is the most frequently mutated gene in human tumors.29-31 Studies have demonstrated that TP53 mutations-induced dysfunctions are strongly correlated with the process of tumor development.32,33 Given that TP53 is an important factor in determining TKI sensitivity, in vitro studies have showed that wild-type p53 is an important factor for gefitinib-induced apoptosis in NSCLC cell lines, whereas p53 mutant reduces gefitinib-induced apoptosis. 34 The presence of p53-responsive elements in FAS promoter region 35 and the evidence that gefitinib treatment induces an up-regulation of FAS on lung cancer cells 36 further suggest a link between TKI response and TP53 status. Future studies are awaited to validate our finding by enrolling a large sample size to elucidate the impact of EGFR ex20ins combined with TP53 mutation on the efficacy of therapies.
EGFR amplification has been documented as an unfavorable predictor for EGFR ex20ins-positive NSCLC patients receiving EGFR-TKI treatment. 37 The study also revealed that EGFR-associated gene amplification accounted for 25% of cases, consistent with earlier research.26,27,37 Among the 24 patients treated with EGFR-TKI, the non-EGFR amplification group showed a longer median PFS when compared with the EGFR amplification group (110 vs 31 days, P = .030). A previous study reveals an association between EGFR amplification and therapeutic response to EGFR TKIs and anti-EGFR monoclonal antibodies. 38 EGFR-TKIs exert a therapeutic effect by inhibiting the EGFR signal transduction pathway. Therefore, EGFR amplification results in reduced therapeutic response to drugs that block the EGFR signaling pathway. Anti-EGFR monoclonal antibodies inhibit the EGFR signal transduction pathway. A combination of an anti-EGFR monoclonal antibody with cetuximab has been shown to produce a favorable therapeutic effect in patients with EGFR ex20ins mutation and EGFR amplification.
In this study, 11 patients received targeted therapy, of which 7 patients were treated with osimertinib in combination with JMT101, 1 with a first-generation EGFR-TKI, and 3 with a third-generation EGFR-TKI. A real-world study of furmetinib for the treatment of EGFR ex20ins showed that the ORR was higher in patients at the 240 mg once-daily dose (42.9%) than in the 80 mg once-daily dose group (25.0%) and the 160 mg once-daily dose group (39.5%). 39 In this study, of the 3 cases treated with furmonertinib, 2 showed PR and one exhibited SD, with an ORR of 66.67%, DCR of 100%, and median PFS1 of 215 days. EGFR ex20ins is a promising therapeutic target that deserves further research and exploration. According to the current research, the third-generation TKI is better than the first-generation TKI and the combination of the third-generation TKI may offer a better efficacy, which is worthy of further exploration.
In this study, we found that a combination of chemotherapy and immunotherapy achieved a better therapeutic response than monotherapy. The Phase III clinical trials of IMpower130 and IMpower150, which recruited patients with EGFR mutations,40,41 reveal that atezolizumab combined with bevacizumab and chemotherapy produces a better prognosis than bevacizumab combined with chemotherapy alone. The elevated PD-L1 expression in NSCLC patients with EGFR ex20ins may be effectively addressed through a combination of chemotherapy and immunotherapy. According to previous studies, the expression of PD-L1 in patients with EGFR ex20ins varies from 37% to 80%,25,42 with a high-expression of ≥50% detected in 10% of the cases (N = 9/88). Furthermore, the expression of PD-L1 may vary depending on the type of EGFR ex20ins variants. In addition, compared with other EGFR mutations, EGFR ex20ins features a lower median tumor mutation (TMB) (3.5 vs 5.9, P < .001) and a lower proportion of PD-L1 expression ≥1% (22 vs 60%, P < .001). 43 Therefore, further studies need to recruit large sample sizes to validate the expression of PD-L1 in patients with EGFR ex20ins and the value of immunotherapy for such patients.
This study has some limitations. Firstly, this study is a retrospective, single-center study involving Chinese people, and the sample size is small, which may limit the generability of our findings to other populations. Secondly, a touch of bias may be present in the treatment plan for the population included in this study and there are individual differences among patients, which may influence the interpretation of the efficacy of the treatment. Finally, this study had a high proportion of patients with missing data, resulting in poor data completeness.
Conclusion
In this real-world analysis, either EGFR-TKIs or chemotherapy-based combination therapy can only confer limited benefits for NSCLC patients with EGFR ex20ins.
Supplemental Material
Supplemental Material - Evaluating the Clinical Characteristics and Prognosis of Advanced Non-Small Cell Lung Cancer with Exon 20 Insertions
Supplemental Material for Evaluating the Clinical Characteristics and Prognosis of Advanced Non-Small Cell Lung Cancer with Exon 20 Insertions by Haibo Wang, Yiquan Xu1, Jinlan Lin, and Yunjian Huang in Cancer Control
Synopsis for Table of Contents
This study retrospectively analyzed the clinical characteristics and prognosis of NSCLC patients with EGFR ex20ins in our center, we found that either EGFR-TKIs or chemotherapy-based combination therapy can only confer limited benefits for NSCLC patients with EGFR ex20ins.
Footnotes
Acknowledgments
The authors thank the patients, their families and the study personnel who participated in this trial.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Startup Fund for scientific research, Fujian Medical University (Grant number:2021QH1147), Fujian Provincial Health Technology Project (Grant number: 2021GGA042), Natural Science Foundation of Fujian Province (Grant number: 2021J05084).
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
