Abstract
Background
Oral cancer remains 1 of the biggest health care challenges; it has a poor response to treatment, and treatment often results in severe side effects. Nano-targeted drug carrier-assisted drug delivery systems can improve the benefits of targeted drug delivery and treatment efficacy. A systematic review and meta-analysis was conducted to investigate the effect of targeted nano carrier drug delivery systems on the management of oral cancer.
Methods
A comprehensive literature search was performed using PubMed, ScienceDirect, the Cochrane Library, Google Scholar, and Scopus using PRISMA guidelines, to identify relevant in vitro and in vivo (human) studies. Studies evaluating the impact of nanocarrier-based delivery systems on oral cancer cells or human models were selected. Pooled effect sizes were calculated using random-effects models via RevMan 5.4, and heterogeneity among studies was assessed.
Results
After full-text assessment, 15 research articles were included [14 in vitro studies and 1 randomized controlled trial (RCT)]. In the meta-analysis, the pooled data (IC50) for the impact of the nanocarrier delivery system vs control on oral cancer was −7.67 (95% CI: −41.77, 26.43), with a high heterogeneity (
Conclusion
Nanocarrier-based drug delivery has been found to be a superior approach compared to drug delivery in free form, increasing the efficacy and safety of oral cancer treatment.
Keywords
Introduction
Cancer is the leading cause of death and a major health concern worldwide and is the leading cause of death globally. Among head and neck cancers, oral cancer is the most commonly found 1, contributing 40% of all cases.1,2 Meanwhile, increasing trend (2%–3%) was observed annually in oral cancer cases. 3 Oral cancer refers to tumors that occur in various parts of the mouth, including the hard plates, lips, anterior two-thirds of the tongue, buccal mucosa, and other tissues of the oral cavity. 4 In addition, craniofacial tumors are characterized by the presence of many cystic and neoplastic pathoses and are considered unique due to functional impairment and aesthetic defects. 5 As such, the survival rate of oral cancer has plateaued at approximately 50% over the past 30 years, despite great advancements in knowledge and treatment opportunities. 6 Likewise, in a study, 5-year overall survival in oral cancer patients was significantly lower than control (53.7% vs 55.8%). 7 Given that there are no specific symptoms for oral cancer, the majority of patients are diagnosed at an advanced stage, and only one-third of patients are diagnosed at an early stage. 8
The oral cancer therapy for the early stage is usually surgical recession, and the advanced stage can be treated with radiotherapy or chemotherapy combined with surgery. 9 Moreover, utilization of diode laser ablation also has promising functional and aesthetic outcomes. 10 Nevertheless, these modalities do have significant side and antagonistic effects: radiotherapy causes permanent tissue damage to the organs and pathways around the tumor, while chemotherapy leads to vomiting, nausea, hair loss, diarrhea and infections in the patient, causing declining QoL and wellbeing. 11 Another barrier is the development of multidrug resistance. 12 In addition to these problems, the poor solubility of chemotherapeutic agents, low-solubility drugs administered intravenously plug off the blood vessels, a high hydrostatic pressure within the tumor interstitial fluids induces an outward convective interstitial flow, sets up a barrier for the effective transport of drugs, and aggressively drains out the therapeutic agents from the tumor cells. 13 Similarly, acidic microenvironments in tumors can also cause the deterioration of acid-sensitive drugs. 14
Because of the enormous impact of this deadly disease, investigators worldwide are progressively discovering nanotechnology to create advanced drug delivery systems. Which has the potential to revolutionize oral cancer treatment by improving drug targeting, tumbling toxicity, and enhancing therapeutic outcomes. 15 In addition, various advantages of nanocarriers have been investigated compared with conventional delivery systems, such as enhanced and precise biodistribution, prolonged plasma half-life, and targeted delivery of anticancer agents. 16 Nanocarriers expect constant, direct, and controlled drug release to malicious cells as they are programmed to identify malicious cells and provide specific drug release to them without any interference over normal healthy cells. Additionally, many disadvantages have been overcome with the evolution of nano-carrier-based drug delivery systems. 17
Interestingly, drug delivery systems are being used in many ways to transplant therapeutic agents at targeted sites using nanocarriers. Major nano-carriers include drug delivery systems like liposomes, carbon nano-tubes, polymeric, polymeric micelles, solid lipids, quantum dots and magnetic nanoparticles. 13 Both natural and synthetic polymers have been used for the preparation of nanocarriers. In this technique, active therapeutic agents are trapped with polymers by covalent bonds or materially embedded in a polymeric matrix. Polymeric nano-agents can be used for early diagnosis of oral cancer.18,19 Polymer-based drug delivery systems consist of either amphiphilic or branched capsules. Moreover, lipid-based nanocarriers were the earliest carriers to be used in nano-medicine and are considered good delivery systems as they can easily degrade inside the body and their unique property to capture the lipophilic agent outside the lipid layer and hydrophilic agents inside the aqueous environment. 20 Lipid-based drug delivery systems have been approved for the treatment of Acquired Immunodeficiency Syndrome (AIDS)-linked cancers and mammary gland tumors. 21 Similarly, magnetic particles are nanostructures with sizes of 1-100 nm and are divided into a magnetic core and a coating on the surface, that is, the coating of the functional layer. Metal Nanoparticles (MNPs) are composed of pure metals (cobalt, nickel, and iron) and oxide particles based on iron oxide, which are predominantly used in biomedical studies. 13 This formulation of IGF1-IONPDOX (a magnetic nanocarrier) was able to penetrate the barrier erected by the tumor’s stromal membrane, which inhibits the dispersion of the drug in the tumor; therefore, it not only reduced the tumor mass, but also slowed down its proliferation in pancreatic tumors in humans. 22 Similarly, the drug delivery efficiency of the carbon nanotube-based drug delivery system reached 80% in breast cancer cells. 23
Although these approaches have several advantages, some nanomaterials used in nanocarrier drug delivery systems might have some toxic issues, and their clearance from the body might be a challenge. Similarly, oral cancer treatment is a positive approach, and treatment with nanocarriers effectively reduces cancer tumor/cells. However, their clinical benefits in the treatment of oral cancer are still limited, and only a few studies have evaluated the role of nano-carrier-based drug delivery systems in the treatment of oral cancer in humans. The main objective of this cumulative analysis was to gauge the efficacy and impact of a nano-carrier-based drug delivery system vs a conventional drug delivery system and to provide rationale and a compilation of all evidence and complication rates with nanocarrier drug delivery systems, which could aid professionals and investigators as guidance expertise to update the development of novel, safe, and effective nano drug delivery systems or treatment modalities to restore patient outcomes.
Materials and Methods
The protocol for the current systematic review and meta-analysis was adopted according to the Preferred Reporting for Systematic Reviews and Meta-analysis (PRISMA) guidelines for the purposes of reproduction and transparency of the studies. A predefined search process was followed from the beginning to the end of the selection of relevant literature. 24 The PRISMA statement is very useful for transparent and thorough reporting in the literature. 25 The protocol for this systematic review was registered on the International Platform of Registered Systematic Review and Meta-Analysis Protocols (INPLASY) (registration number: (2024100110).
Literature Search
The studies were identified from a systematic analysis of the listed online databases, including PubMed, ScienceDirect, the Cochrane Library, Google Scholar, and Scopus, during the search without any date restrictions. Different keywords such as “nano-particles” OR “nano-carriers” OR “liposomes” OR carbon nano-tubes” “polymeric nano-particles” OR “quantum dots” OR “magnetic nano-particles” OR “dendrimer” AND “drug delivery” OR “drug carrier” AND “oral cancer” OR “oral squamous carcinoma” OR “mouth neoplasm” OR “oral health” were used (see Supplementary Table 1).
Inclusion Criteria
Studies were included on the basis of PICO guidelines: P = Participants/Patients with any type of oral cancer, I = Intervention based on nanoparticle delivery system, C = Control/comparator based on alternative drug delivery system or placebo, O = Outcomes; any outcome regarding survival after treatment. Only randomized controlled trials (RCTs), case-control studies, in vitro studies, and cohort studies published in English were included, without any data restrictions.
Exclusion Criteria
Studies with insufficient data or those that did not focus on oral cancers, case reports, reviews, editorials, commentaries, or published in languages other than English were excluded.
Study Selection and Assessment
Papers were selected based on a review of studies, wherein two reviewers searched, screened, and initially assessed the titles and abstracts for inclusion in the present study, according to their aims. Then, a full-text assessment of all the papers considered eligible was completed. The reviewers’ views were discussed to arrive at a consensus. In case of a contradiction or ambiguity issue, it was forwarded to the third consensual independent reviewer and resolved.
Data Extraction
Data were extracted from the included studies based on the pre-specified data extraction form in an Excel sheet. Each study was independently reviewed by two reviewers for the following characteristics: author ID, country, study design, sample size, participant characteristics (age, gender, cancer type), intervention and control characteristics (type of nanoparticle, drug used, dose delivered, delivery approach), adverse events, key outcomes, and conclusion.
Quality Assessment
To appraise the methodological quality of in vitro studies, the quality index tool (QUIN) was used with 12 items. Each study was assessed for 12 items and scored in the response as “yes” (allocated 1-2 points), “no” (allocated zero points) or “not applicable”. 26 The second step was to rank each study by using a point response. The studies scored below 50% were considered to have a high risk of bias (RoB), those between 50%–70% were the medium RoB, >70% were the low RoB. 26 For RCTs, we employed the Cochrane collaboration tool to perform quality assessment using the Robvis web-based app. 27 Assessment was carried out in the realms of randomization, deviation from the intended intervention, measurement of outcomes, missing outcomes, and reporting.
Data Analysis
A qualitative synthesis combined the outcomes of studies to show the general, intervention characteristics, and key findings. RevMan 5.4 was used to perform meta-analysis that measured the pooled efficacy of intervention on cytotoxicity and test for heterogeneity.
28
In addition, statistical heterogeneity was assessed using the chi-square test (
Results
Literature Search
Searches of the literature were performed using PubMed, Scopus, ScienceDirect, the Cochrane Library, and Google Scholar, and 613 primary research papers were identified. In the screening phases of PRISMA, namely, identification and title and abstract screening, 54 duplicated research papers were identified and excluded, which were found in the identification phase of PRISMA. Therefore, 559 research papers were considered for the screening of studies to determine which research papers were eligible for inclusion in our study. After screening, 533 studies were deemed unsuitable for this study and were thus excluded. We assessed only 26 research papers for full screening, and we analyzed only 15 qualitative and quantitative research papers because 11 studies were excluded for some reasons, which are presented in Figure 1. PRISMA flow chart and article selection process.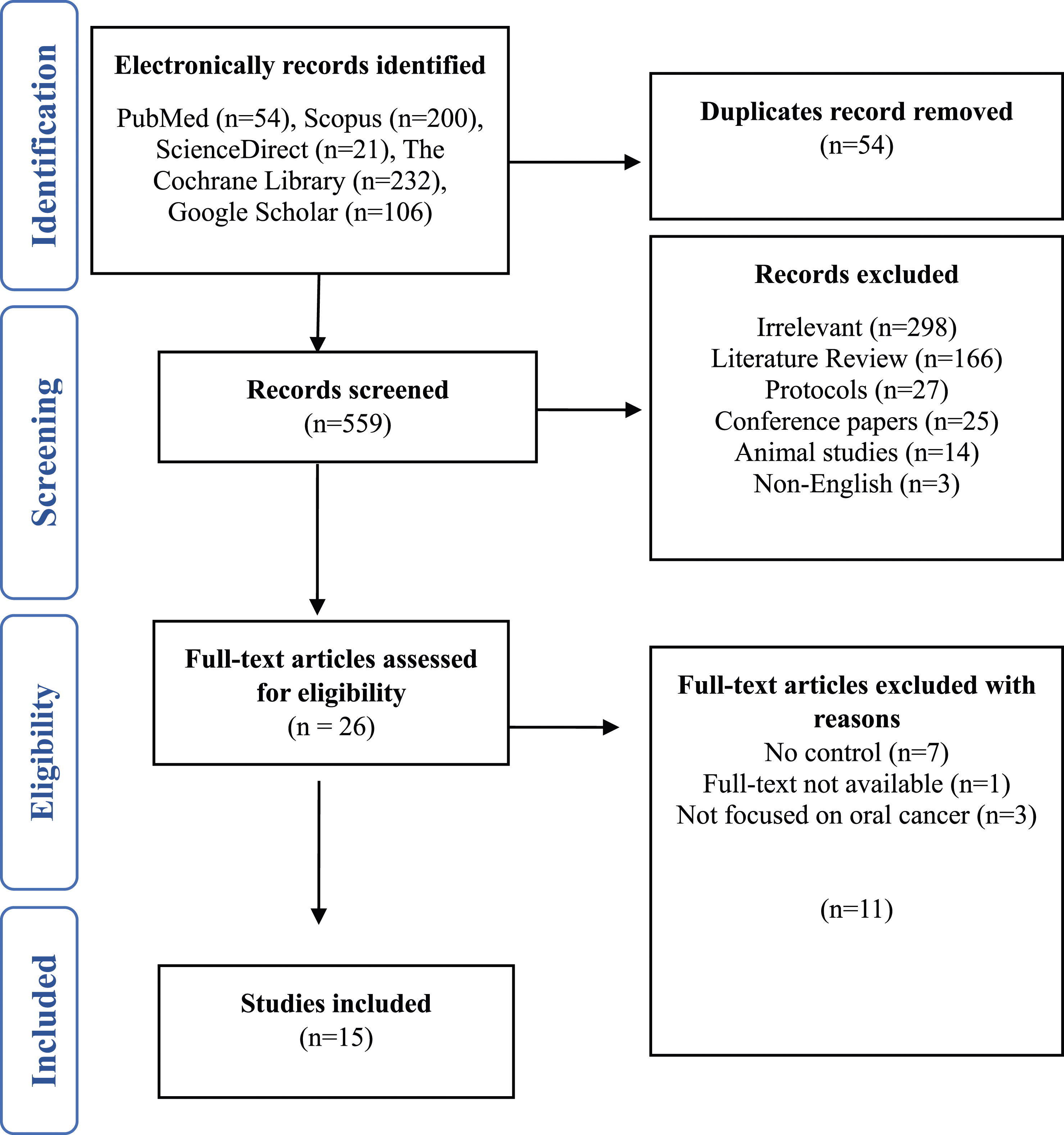
General Characteristics of in Vitro Studies
General characteristics of the included in vitro studies.

Abbreviations: EA = Ellagic Acid, Fu = Fluorouracil, Cu = Curcumin, DOX = Doxorubicin, OCSCC = Oral Cancer Squamous Cell Carcinoma, SCC = Squamous Cell Carcinoma.
General characteristics of RCTs
Summary of the General Characteristics of the Included RCTs.

Abbreviations: RCT = Randomized Controlled Trail, OCSCC = Oral Cancer Squamous Cell Carcinoma, Cu = Curcumin.
Outcomes of in Vitro Studies
Summary of Nanoparticle Characteristics and Key Outcomes of

Abbreviations: EA = Ellagic Acid, IC50 = Inhibitory Concentration, PLGA = Poly (lactic-co-glycolic acid), AuNPs = Gold Nano-particles, Fu = Fluorouracil, Cu = Curcumin, DOX = Doxorubicin, HA = Hyaluronic acid, CS = Chitosancoa, GO = Graphene Oxide, GHHD = DOX@GO-HA-HN-1.
Outcomes of RCTs
Summary of Nano-Particle Characteristics and Key Outcomes of RCTs.

Abbreviations: Cu = Curcumin, OCSCC = Oral Cancer Squamous Cell Carcinoma.
Meta-Analysis
In vitro studies
A total of four studies were utilized in the meta-analysis, and the pooled data (IC50) for the impact of nano-particle delivery system vs control on oral cancer was −7.67 (95% CI: −41.77, 26.43), as indicated in Figure 2. High heterogeneity ( Forest plot of IC50 values of experimental and control groups against different cells of OCSCC.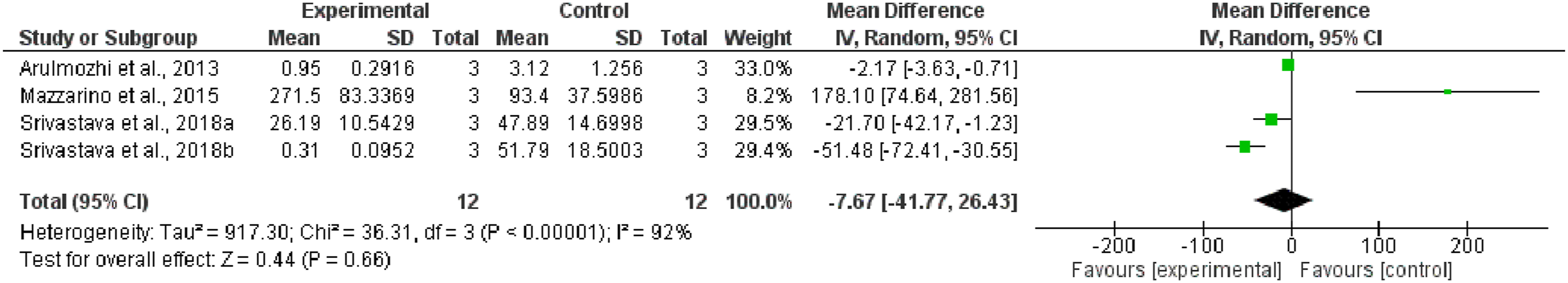
Quality assessment
Quality Assessment of Included in vitro Studies.
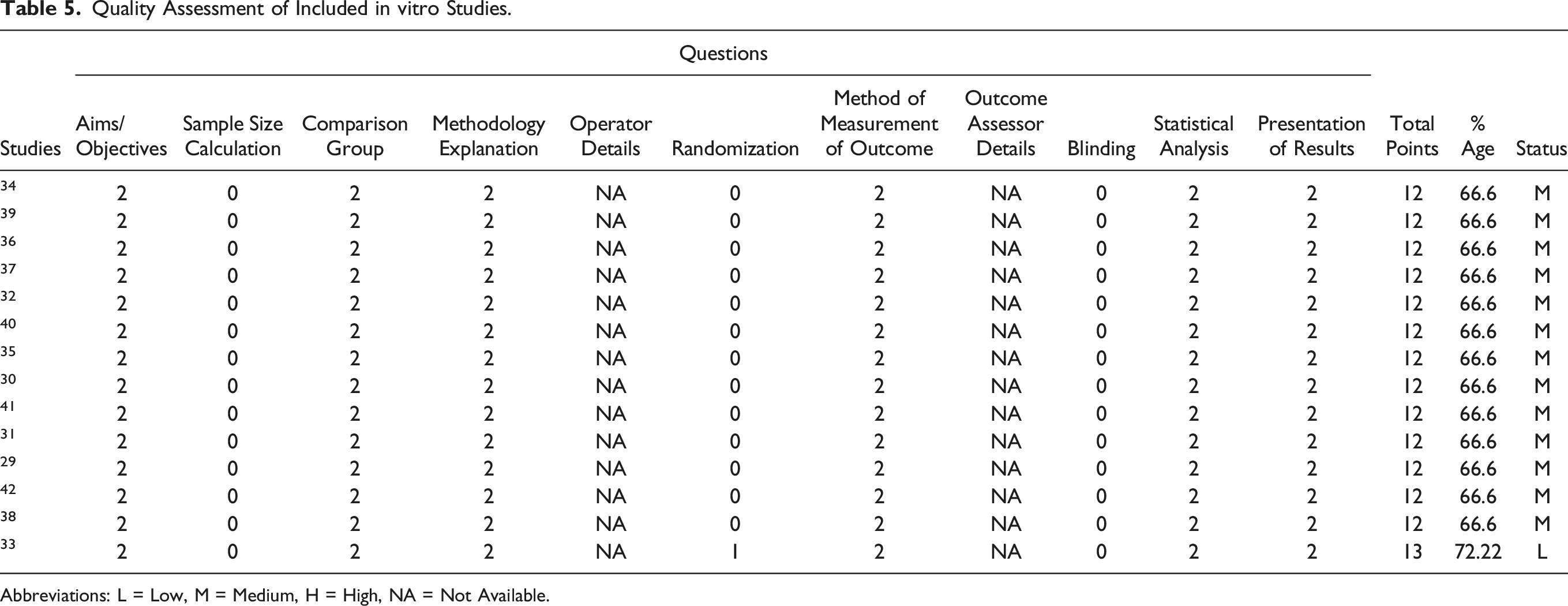
Abbreviations: L = Low, M = Medium, H = High, NA = Not Available.
Unfortunately, only 1 RCT compared nanoparticle delivery systems with a control and found some concerns in the randomization domain. 43
Discussion
Oral cancer remains a major global health concern, as it presents with high incidence and mortality rates, especially in the developing world. 44 Mainstream treatment modalities, such as surgery, chemotherapy, and radiotherapy, are often hindered by systemic toxicity, drug resistance, and poor targeting of tumor cells. 45 One of the most promising approaches to improve the efficacy and specificity of oral cancer therapy is the use of drug delivery systems based on nanocarriers. 45 These delivery systems include nanoparticles, liposomes, and micelles, which can provide advantages, such as improved bioavailability, targeted drug release, and reduced toxicity. This systematic review and meta-analysis aimed to evaluate the efficacy of nano-carrier-based drug delivery systems for the treatment of oral cancer with respect to therapeutic outcomes.
In the meta-analysis of the in vitro studies of the present study, a significant difference (
Nanocarrier-based drug delivery systems offer important clinical advantages in the treatment of oral cancer. Because it can increase the efficiency of treatment and reduce toxicity to the system. These advanced systems enable targeted delivery of anticancer agents. Adding a higher concentration of drug to the tumor site ensures that it can be reduced, and it can also improve the absorption and controlled release of drugs, which can lead to a prolonged treatment effect. In addition, it provides a better quality of life for patients, making them a transformative innovation in oral cancer management.
This study has several strengths and weaknesses. This review summarizes data from different studies to provide solid evidence that nanoparticle systems are more effective, targeted, and safer for drug delivery than conventional systems in humans. The aggregation of data from the meta-analysis via pooled data allows the quantification of therapeutic benefits, which contributes to the contemporary evidence base for the use of these advanced delivery systems. Some limitations include variability of the studies in terms of study design, nanoparticle formulations, and outcome measures, which could affect the generalizability of the results across studies. Furthermore, the observed statistical heterogeneity in
Conclusion
Collectively, the results of this study revealed that these new delivery systems significantly improved therapeutic outcomes over free drug delivery, despite variability across individual studies and statistically insignificant differences in humans on a study-by-study basis, as demonstrated in the human-focused meta-analysis. Overall, nanoparticle systems systematically improve drug efficacy and decrease toxicity, and the reduction in cancer lesions and enhancement of apoptosis with nano-particle systems points to their potential as an adjunct to conventional treatment regimens. However, challenges like, fabrication complexity and outlining potential solution should also be considered. Further validation of these benefits by conducting such studies with larger sample sizes and better-designed methodologies is needed to maximize the clinical usefulness of nanoparticle drug delivery systems in the management of oral cancer.
Supplemental Material
Supplemental Material - Cytotoxicity of Nanocarrier-Based Drug Delivery in Oral Cancer Therapy: A Systematic Review and Meta-Analysis
Supplemental Material for Cytotoxicity of Nanocarrier-Based Drug Delivery in Oral Cancer Therapy: A Systematic Review and Meta-Analysis in China by Mohammad A. Saghiri, Ravinder S. Saini, and Artak Heboyan in Cancer Control.
Footnotes
Acknowledgments
All the authors are thankful to King Khalid University, Saudi Arabia, for the financial Support.
Author Contributions
Conceptualization and Methodology: M.S. Data Curation and Formal Analysis: R.S. Investigation and Resources: R.S. Validation and Visualization: A.H. Original draft preparation: R.S. Writing, Reviewing and Editing: M.S., A.H. Supervision and Project Administration: A.H. Funding Acquisition: R.S.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by The authors extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for funding this work through the Review Article Project under grant number RA.KKU/7/45.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
