Abstract
Purpose
Splenic metastases (SM) from breast cancer (SMBC) are exceedingly rare. To date, the relevant literature is primarily based on pan-tumour species, with only a few studies exploring SM specifically in relation to breast cancer. As such, the present retrospective study explored the clinicopathological characteristics and prognoses of patients with SMBC at the breast care centre of the authors’ hospital.
Methods
Data from patients newly diagnosed with metastatic breast cancer (MBC) between June 2017 and June 2022 were extracted from medical records at the authors’ hospital. Clinicopathological characteristics and their associations with progression-free survival (PFS [time from diagnosis of initial recurrence and/or metastasis to diagnosis of SM]), first overall survival (1stOS [time from diagnosis of breast cancer to death or last follow-up visit]), and second overall survival (2ndOS [time from diagnosis of SM to death or last follow-up visit]) were analysed in patients with SMBC.
Results
In total, 1009 patients with MBC were identified, of whom 18 (1.7%) had SM. T1 and T2 stages were documented in 15 (83.3%) patients, whereas N2 and N3 were documented in 13 (62.2%). 14 (77.8%) patients were oestrogen receptor and/or progesterone receptor positive. A Ki-67 index ≥ 30% accounted for 72.2% (13/18) of cases, and all patients were histological grade II or III. Liver and/or lung metastases were documented in all 18 (100%) patients. Median PFS was 6.3 months. The median 1stOS and 2ndOS were 41.8 and 10.6 months, respectively. The number of previous treatment lines before diagnosis of SM was a significant adverse prognostic factor for PFS, and disease-free survival was a significant adverse prognostic factor for 1stOS.
Conclusion
SMBC commonly presents with diffuse multiple organ metastases in the terminal stage of malignancy and has a poor prognosis, which may provide deeper insight into SMBC for clinicians.
Plain language summary
Splenic metastasis from malignancies is a rare event, although accumulating evidence supports its occurrence. Nevertheless, few studies have explored splenic metastases (SM) specifically in relation to breast cancer (BC). Moreover, due to the low incidence of SM from breast cancer (SMBC), it is difficult to perform comparative or randomised controlled studies to provide supportive data for SMBC. This retrospective study provides the first comprehensive and detailed elaboration of the clinicopathological characteristics and prognoses of patients with SMBC. Patients with poor biological characteristics were more likely to develop SM. Splenic metastasis indicates a poor prognosis and survival is expected to be < 1 year on patient with BC. These results provide deeper insights into SMBC for clinicians and may facilitate the design and development of individual treatment regimens for those with the disease.
Keywords
Introduction
The spleen is the second largest lymphatic reticuloendothelial organ in the body. 1 Blood-borne metastatic carcinoma of the spleen is defined as carcinoma involving the intraparenchymal splenic tissue and should not be confused with superficial subcapsular foci associated with peritoneal dissemination.2,3 Owing to unique anatomical factors and inhibitory effects of the splenic microenvironment on the growth of metastatic cells, splenic metastasis is a relatively rare occurrence.3-10 As reported in previous studies, the incidence of splenic metastases (SM) of epithelial origin varies widely, based on the nosological profile of each malignant tumour, ranging from 0.2-21%.5,11-13 There are conflicting data in the literature addressing various cancers and identification of the primary tumour. Lam et al 13 reported that the most common primary sites are the lungs, stomach, pancreas, liver, colon, oesophagus, nasopharynx, breast, ovaries, and uterus. Of note, reports addressing the incidence of SM from the same primary carcinoma vary greatly. 7
Breast cancer (BC) has surpassed lung cancer as the most common malignancy diagnosed among women according to Global Cancer Statistics in 2022. 14 BC most frequently metastasizes to the bone, lungs, liver, brain, and lymph nodes, and less frequently to the spleen. Furthermore, SM from BC (SMBC) are generally asymptomatic, making their discovery difficult, although some patients may present with abnormal pain, splenomegaly, anaemia, thrombocytopenia, and bleeding. 11 The incidence of reported cases of SMBC has increased in the past few decades due to advances in diagnostic image resolution, imaging techniques, and long-term patient follow-up. 15 Despite accumulating data describing SM from various solid carcinomas, few studies have focused specifically on SMBC. In the present study, we retrospectively collected data from patients with SMBC treated at our hospital and further investigated the clinicopathological characteristics, growth features, and prognoses of SMBC, aiming to provide deeper insight into SMBC for clinicians to design and develop individual treatment regimens for affected patients.
Materials and Methods
Patient Population
The present investigation was a single-centre, retrospective study. Data from 1009 patients ≥18 years of age, with histologically confirmed metastatic BC (MBC), who attended the National Clinical Research Centre for Cancer/Cancer Hospital & Shenzhen Hospital (Shenzhen, Guangdong, China) between June 2017 and June 2022, were retrospectively collected. All procedures involving human participants were conducted in accordance with the 1964 Declaration of Helsinki and its later amendments, or comparable ethical standards. The protocol for the present study was approved by the institutional review board and ethics committee (approval number: JS2024-21-1) of the National Clinical Research Centre for Cancer/Cancer Hospital & Shenzhen Hospital (Shenzhen, Guangdong, China) on 16 October 2024. All patients provided written informed consent for the use of their anonymized medical information for research purposes. All patients’ personal details were de-identified. All recruited patients fulfilled the following criteria: pathology confirmed of BC; and MBC staged according to the Union for International Cancer Control (UICC, 8th Edition) tumour-node-metastasis (TNM) staging system. The reporting of this study conforms to STROBE guidelines. 16
Follow-Up and Data Collection
All patients with SM underwent telephone and in-person follow-up until 31 August 2023, and death was considered to be a defined event. Metastasis and recurrence were diagnosed based on clinical, imaging, and surgical pathological examinations. Progression-free survival (PFS) was calculated from the date of initial recurrence and/or metastasis to the date of SM diagnosis. The first overall survival (1stOS) was calculated from the date of BC diagnosis to the date of death or last follow-up visit, while the second overall survival (2ndOS) was defined as the time from the date of SM diagnosis to the date of death or last follow-up visit. Clinicopathological data were obtained by reviewing pathology reports, medical records, laboratory examinations, and medical imaging dictations, and primarily included the following: Eastern Cooperative Oncology Group (ECOG) performance status; age; location; T stage; N stage; TNM stage at initial diagnosis; histopathological type; human epidermal growth factor receptor 2 (HER2) status; oestrogen receptor (ER)/progesterone receptor (PR) status; Ki-67 index; histological grade; disease-free survival (DFS); visceral metastases; number of previous treatment lines before diagnosis of SM; number and size of SM; blood routine examination; and D-dimer level during SM.
Statistical Analysis
All data were analysed using SPSS version 26.0 (IBM Corporation, Armonk, NY, USA) and Prism version 5 (GraphPad, Inc., San Diego, CA, USA). Descriptive statistics were used to analyse clinicopathological features. The Kaplan–Meier method was used to estimate PFS, overall survival (OS), and corresponding 95% confidence interval (CI) for patients with SMBC. Cox univariate and multivariate analyses were used to determine the predictive utility of the variables for PFS and OS. Data from patients who did not have any documented events, were lost to follow-up, or died from any cause were censored based on the last date on which the patient was event-free. All reported P-values were two-sided, and differences with P < 0.05 were considered to be statistically significant.
Results
Clinicopathological Characteristics of Patients with SMBC
Clinicopathological Characteristics of 18 Patients With SMBC at Baseline.

Abbreviations:
PFS and OS of Patients with SMBC
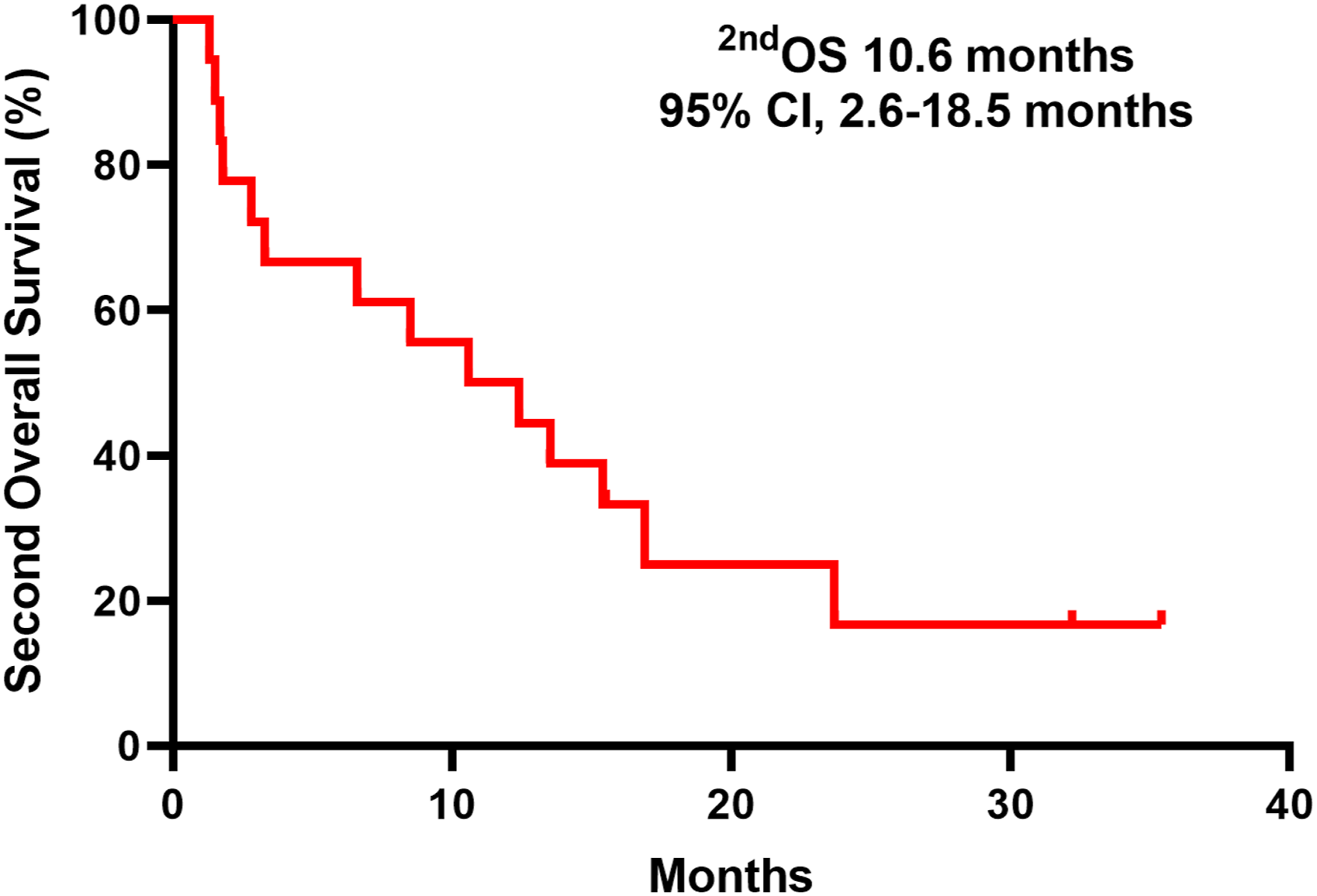
Kaplan–Meier survival analysis was performed to better understand the prognosis of patients with SMBC. By the end of the follow-up period, 15 patients died. The median interval between diagnoses of BC and SM was 25.4 months (range, 0.0-275.0 months). The median PFS was 6.3 months (95% CI, 0.0-13.4 months). The median 1stOS was 41.8 months (95% CI, 2.6-18.5 months) (Figure 1) and the median 2ndOS was 10.6 months (95% CI, 20.0-63.6 months) (Figure 2). Kaplan–Meier estimate of first overall survival in patients with splenic metastases from breast cancer. 1stOS: first overall survival (time from diagnosis of breast cancer to death or last follow-up visit), CI: confidence interval. Kaplan–Meier estimate of second overall survival in patients with splenic metastases from breast cancer. 2ndOS: second overall survival (time from diagnosis of splenic metastases to death or last follow-up visit), CI: confidence interval.
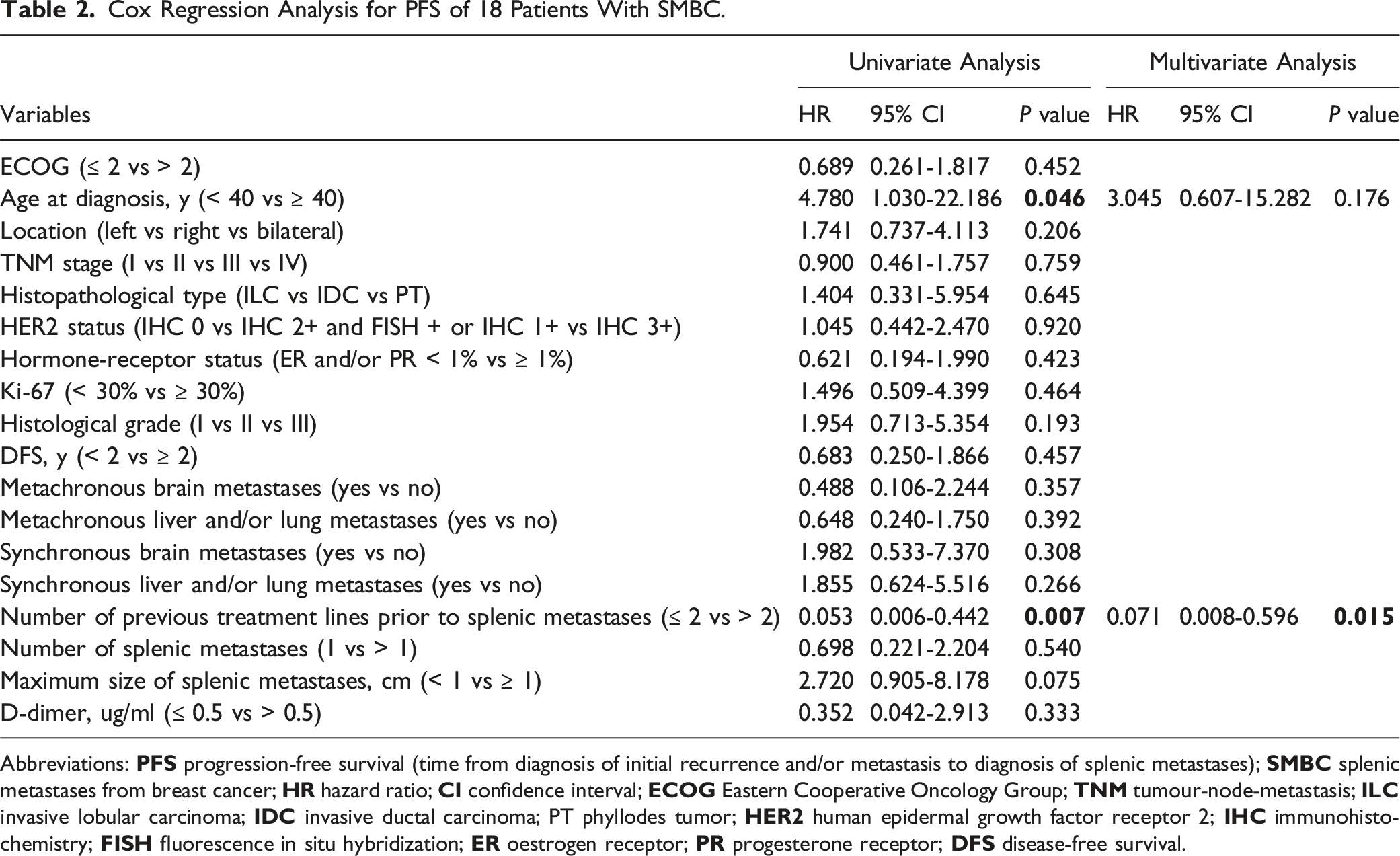
Cox Regression Analysis of PFS and OS in Patients with SMBC
Cox Regression Analysis for PFS of 18 Patients With SMBC.
Abbreviations:
Cox Regression Analysis for 1stOS of 18 Patients With SMBC.

Abbreviations:
Cox Regression Analysis for 2ndOS of 18 Patients With SMBC.

Abbreviations:
Discussion
As previously reported, splenic metastasis is a rare phenomenon. However, based on an autopsy case series and according to existing evidence, MBC involves the spleen in as many as 11-18% of patients. 17 The incidence of SMBC in the present study was much lower than previously reported. Due to the low incidence of SMBC, it is difficult to perform comparative or randomised controlled studies to provide more convincing data regarding the clinicopathological features and prognoses of patients with SMBC. In the present study, patients with low or negative HER2 expression (88.9%), hormone receptor-positive (77.8%), Ki-67 index ≥ 30% (72.2%), grade II or III (100%), and DFS < 2 years (61.1%) accounted for the majority of patients with SMBC. More specifically, in terms of molecular subtypes, patients with low or negative HER2 expression may be more likely to develop SM. Moreover, patients with poor biological characteristics, such as a high Ki-67 index, poor differentiation, and short DFS, may be more likely to develop SM. These findings are, in part, consistent with existing reports. Bartolotti et al 11 observed a trend toward both younger age and a higher number of metastatic sites in patients with SM. Berge et al 18 found that SM were associated with age and tumour biological behaviour in various solid carcinomas; more specifically, the younger the patient and the more aggressive the primary tumour, the more likely SM occurred.
It is widely known that metastases from solid tumours to the spleen most commonly occur as a result of haematogenous dissemination.7,17 Under most circumstances, SMBC develop in the context of late-stage, multivisceral, metastatic disease with either synchronous or metachronous patterns; however, in extremely rare instances, they can also present as a solitary pattern. 3 In the present study, all included cases were accompanied by visceral dissemination to the liver and/or lung(s), which further confirmed that SM are typically indicative of a patient’s cancer being in the terminal stage. It is especially noteworthy that, of the 18 patients, 17 experienced liver metastases and SM simultaneously or metachronously, indicating a common occurrence in this cohort. Harman et al 19 demonstrated that when ≥ 1 organ(s) in the thoracic and abdominal cavities were involved, the rate of SM was 43%. Portal hypertension secondary to diffuse liver dissemination of BC and, both the spleen and liver being part of the retroendothelial system, could partially account for combined metastases to the spleen and liver.1,20 Another noteworthy phenomenon observed in the present study was that nearly three-quarters of the included patients experienced bone metastases before they exhibited SM, which may be partly associated with the high proportion of molecular types with hormone receptor-positive tumours, in which 65-75% could develop bone metastases. Additionally, bone is one of the primary potential sources of further metastatic dissemination of BC and belongs to the reticuloendothelial system, which may be attributed to the development of bone metastases before SM.
In most cases, SM are asymptomatic and present as symptoms secondary to the primary tumour(s), although in rare cases, they can result in splenomegaly, severe pain, haematological disorders such as anaemia or thrombocytopenia, and rupture with massive bleeding.3,11 Among the cases included in the present study, all SM were diagnosed using computed tomography during regular follow-up of patients with no SM-related symptoms. According to previous reports, marked splenomegaly was not common in patients with SM from melanoma, probably because the average size of the splenic nodules was 1.5 cm. 21 Approximately four-fifths of the patients in the present study presented with multiple SM, while the remainder presented with a single lesion. The average size of the SM in the present study was 1.8 cm, ranging from 0.4-8.5 cm in diameter. Hematologically, leukopenia, anaemia, and thrombocytopenia were observed in only a small subset of patients, which may have been secondary to SM, or caused by bone marrow metastasis and chemotherapy. Consequently, the hematologic examination conducted at the time of SM diagnosis was insufficient to accurately reflect true bone marrow function.
Additionally, we attempted to elaborate on the developmental course and prognoses of patients diagnosed with SMBC included in the present study. The median interval between initial recurrence and/or metastasis and detection of SM was 6.3 months. This should serve as a reminder to clinicians to be vigilant for the development of SM within the first year after the first recurrence and/or metastasis and closer follow-up for SM may be necessary in such patients. The median interval between the initial BC diagnosis and SM detection was 25.4 months. Agha-Mohammadi et al 22 found that the relapse interval of solitary SM among various solid tumours was inconsistent, ranging from the time of initial diagnosis to 144 months. Furthermore, we found that the number of previous treatment lines before the detection of SM was an independent factor of PFS, suggesting that patients with MBC who underwent ≤ 2 lines of treatment may be more likely to develop SM.
Relevant literature addressing the survival of patients with SMBC is scarce. In the present analysis, the 1stOS for patients with SMBC was 41.8 months, irrespective of molecular type. Notably, DFS emerged as an independent adverse factor for 1stOS, suggesting that patients with longer DFS may experience a shorter 1stOS. The finding indicated that the 2ndOS for patients with SMBC was merely 10.6 months despite receiving anti-tumour therapy, and no independent predictive factors were identified. Numerous animal studies and several observational studies have reported that the spleen can prevent tumour progression and lead to a better prognosis, acting as protection for itself. 7 Therefore, as mentioned previously, clinical SM are characterised by diffuse dissemination, insensitivity to chemotherapy and radiotherapy, and poor prognosis.
While SM is not a new emergency phenomenon, it remains rare. Although an increasing number of studies have reported on SM, few have specifically focused on SM arising from a single primary cancer type, particularly BC. The present study focused on patients with SMBC and attempted to elucidate their clinicopathological features and prognoses in order to enrich the available data on SMBC while sharing our clinical experiences in managing these cases.
The present study had several limitations. A definitive diagnosis of SM is based on the pathological examination of specimens obtained via fine-needle aspiration, biopsy, or splenectomy. However, in the present study, histological confirmation was not provided, and the diagnosis of SM was based on clinical and radiographic data. All included patients had multiple SM in the context of multiple organ metastases and terminal-stage disease, and more than one-half presented with poor ECOG performance status. Due to the long-term physical and mental discomfort caused by the disease itself and the risk for bleeding associated with invasive procedures on an organ rich in blood, it is difficult for these patients to undergo biopsy or splenectomy; as such, there is a lack of pathological confirmation. As previously mentioned, this was a single-centre retrospective study with a small sample size; as such, future trials aiming to validate these results are warranted.
Conclusion
Splenic metastasis from BC is rare, and few studies have focused specifically on it. This study is the first to focus on SMBC. The results suggest that SMBC are frequently present in the setting of diffuse multiple organ metastases, especially liver metastases, in the terminal stage of malignancy, indicating a poor prognosis. Although the present study was conducted retrospectively at a single institution, it may provide valuable insights into progress made in patients with SMBC and assist oncologists better in addressing future clinical challenges within this unique patient population.
Footnotes
Acknowledgments
The authors of this paper are grateful to the patients and all the researchers, including the physicians, pathologists, and technicians, who participated in this study.
Authors Contributors
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funds from National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital & Shenzhen Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Shenzhen (No. E010322029), Shenzhen Science and Technology Program (2018, JCYJ20180306171227129), Shenzhen Key Medical Discipline Construction Fund (No. SZXK013) and Shenzhen High-level Hospital Construction Fund.
