Abstract
Background
Immune therapy, especially involving PD-1/PD-L1 inhibitors, has shown promise as a therapeutic option for cholangiocarcinoma. However, limited studies have evaluated survival outcomes in cholangiocarcinoma patients treated with immune therapy. This study aims to develop a predictive model to evaluate the survival benefits of immune therapy in patients with cholangiocarcinoma.
Methods
This retrospective analysis included 120 cholangiocarcinoma patients from Shulan (Hangzhou) Hospital. Univariate and multivariate Cox regression analyses were conducted to identify factors associated with survival following immune therapy. A predictive model was constructed and validated using calibration curves (CC), decision curve analysis (DCA), concordance index (C-index), and receiver operating characteristic (ROC) curves.
Results
Cox regression analysis identified several factors as potential predictors of survival post-immune therapy in cholangiocarcinoma: treatment cycle (<6 vs ≥ 6 months, 95% CI: 0.119-0.586, P = 0.001), neutrophil-to-lymphocyte ratio (NLR <3.08 vs ≥ 3.08, 95% CI: 1.864-9.624, P = 0.001), carcinoembryonic antigen (CEA <4.13 vs ≥ 4.13, 95% CI: 1.175-5.321, P = 0.017), and presence of bone metastasis (95% CI: 1.306-6.848, P = 0.010). The nomogram model achieved good predictive accuracy with a C-index of 0.811. CC indicated strong concordance between the predicted and observed outcomes. Multi-timepoint ROC curves at 1, 2, and 3 years validated the model’s performance (1-year AUC: 0.906, 2-year AUC: 0.832, 3-year AUC: 0.822). The multi-timepoint DCA curves also demonstrated a higher net benefit compared to extreme curves.
Conclusion
The nomogram model, incorporating key risk factors for cholangiocarcinoma patients post-immune therapy, demonstrates robust predictive accuracy for survival outcomes, offering the potential for improved clinical decision-making.
Introduction
Cholangiocarcinoma, a malignant tumor group in the hepatobiliary system, is characterized by poor prognosis, and its incidence is increasing globally. Cholangiocarcinoma accounts for approximately 15% of all primary liver cancers and 3% of gastrointestinal cancers. 1 In a study of 2234 patients across 11 countries, 49.6% had unresectable disease at diagnosis. 2 Furthermore, traditional systemic chemotherapy has shown inconsistent efficacy in cholangiocarcinoma treatment. 3 In recent years, immune checkpoint inhibitors (ICIs), particularly PD-1/PD-L1 inhibitors, have shown promise in cancers like gastric cancer and melanoma, gaining interest in cholangiocarcinoma treatment.4,5 In 2022, the US FDA approved PD-L1 inhibitors for advanced cholangiocarcinoma treatment. A randomized controlled trial (RCT) involving 1069 patients found a significant survival benefit with combination chemotherapy and immunotherapy compared to chemotherapy alone. 6 Despite multiple studies highlighting the positive impact of immunotherapy on the prognosis of cholangiocarcinoma patients,7,8 adverse reactions such as hyperprogression and other negative effects have also been reported.9-11 Therefore, developing reliable biomarkers is essential for identifying cholangiocarcinoma patients most likely to benefit from immunotherapy.
Currently, PD-L1, mismatch repair (MMR), and tumor mutation burden (TMB) are the most commonly reported immunotherapy biomarkers. 12 Patients with high PD-L1 expression, MMR deficiency, or high TMB are generally more likely to respond favorably to immunotherapy.12-14 However, sequencing studies indicate that dMMR/MSI-H phenotypes and high TMB are relatively rare in cholangiocarcinoma patients.15,16 Furthermore, the predictive value of PD-L1 for immunotherapy efficacy in cholangiocarcinoma remains debatable. Two large, randomized, double-blind, placebo-controlled phase III trials found no significant correlation between PD-L1 status and immunotherapy efficacy.6,17 This discrepancy may result from factors like differences in detection reagents, variable cut-off values, individual PD-L1 expression levels, and heterogeneous PD-L1 distribution within tumor.18-20 Researchers have used machine learning (ML) models incorporating genetic profiling and age-related genes to classify cholangiocarcinoma patients, with survival models showing promising predictive performance.21,22 However, the clinical application of these models is constrained by the limited availability of genetic sequencing, specialized equipment, and specific reagents in many clinical settings.
The tumor microenvironment, composed of inflammatory cells, lymphocytes, and signaling molecules, plays a critical role in cancer development at various stages. 23 Immunotherapy, which activates immune mechanisms like effector T cells to elicit antitumor responses, is significantly influenced by the tumor microenvironment. In contrast, tumor progression also affects immunotherapy efficacy by altering the tumor microenvironment. 24 Recent studies have shown that inflammatory indicators, including lymphocyte-to-monocyte ratio (LMR), neutrophil-to-lymphocyte ratio (NLR), and Prognostic Nutritional Index (PNI), correlate with survival outcomes in cholangiocarcinoma patients.25-28 Liver function, another important factor in cholangiocarcinoma progression, is significantly affected by immunotherapy. Patients receiving immunotherapy face a 45% increased risk of elevated alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels. 29 Furthermore, low albumin levels negatively correlate with survival and disease progression in patients receiving immunotherapy. 30 Assessing inflammatory and liver function biomarkers is more straightforward and cost-effective than genomic sequencing or pathological analysis. Therefore, this study aims to develop a nomogram model incorporating these biomarkers to predict the survival benefits of immunotherapy for cholangiocarcinoma patients. This model could serve as a practical clinical tool for guiding personalized treatment decisions in clinical settings.
Materials and Methods
This study was approved by the Institutional Review Board of Shulan Hospital, Hangzhou, China (Reference Number: KY2024049, Approval Date: June 24, 2024), with the requirement for informed consent waived due to the retrospective nature of the study. Between December 2016 and June 1, 2024, 120 patients who received immunotherapy were included in the study. Inclusion criteria were: (1) histologically confirmed cholangiocarcinoma (stage III or IV) as the primary tumor, (2) received at least two cycles of PD-1/PD-L1 therapy (monotherapy or in combination with chemotherapy), (3) complete clinical treatment and follow-up data, and (4) no acute infections or inflammatory conditions before immunotherapy. The exclusion criteria were: (1) cholangiocarcinoma not being the primary tumor (n = 2), (2) treatment cycles fewer than two (n = 24), (3) incomplete clinical treatment data or follow-up information (n = 45), and (4) receiving non-PD-1/PD-L1 immunotherapy (n = 3). Figure 1 illustrates the patient selection and model construction process. This study adheres to the TRIPOD guidelines.
31
Flow chart illustrating the patient screening process for this study. The chart details the number of patients screened and the reasons for inclusion and exclusion criteria. We provided specific numbers for each selection phase to ensure a clear definition of the final cohort included in the analysis.
Follow-Up Study
Follow-up was conducted primarily through outpatient visits and telephone calls at 1- to 2-month intervals. Each follow-up included routine biochemical blood tests, tumor marker assessments, and imaging studies (CT and/or MRI). The final follow-up was done on June 1, 2024, or at the time of the patient’s death. The primary outcome measures were overall survival (OS), defined as the time from PD-1/PD-L1 treatment initiation to death or final follow-up, and progression-free survival (PFS), defined as the time from treatment initiation to tumor progression or death from any cause.
Progressive disease (PD), stable disease (SD), and partial response (PR) were evaluated per the Response Evaluation Criteria in Solid Tumors (RECIST). PR was defined as a reduction of at least 30% in the total diameter of target lesions from baseline. PD was defined as a minimum 20% increase in the total diameter of target lesions from baseline, with an absolute increase of at least 5 mm or the appearance of one or more new lesions. SD was defined as no significant change in tumor size relative to baseline, meeting neither PR nor PD criteria.
Data Collection
Baseline Clinical Characteristics of Patient.

Abbreviations: PNI = Serum Albumin +5*Number of Peripheral Blood Lymphocytes, PLR Numerical Ratio = Platelet/Lymphocytes, NLR = Neutrophils/Numerical, Lymphocyte/Mononuclear Cells (Lymphocytes Numerical, Systemic Immune Inflammation Index (SII) = Platelet Count × Neutrophil Count/Lymphocyte Count, Platelet/Albumin, PAR = APRI = Aspartate Aminotransferase/Platelet, ALT/AST = Alanine Aminotransferase/Aspartate Aminotransferase, and the Cut-Off Values of PNI, PLR, NLR, LMR, SII, PAR, APRI, ALT/AST and Other Variables Were Obtained by the Time Receiver Operating Curve.
Adverse Reactions

ICI Types and Doses.

Statistical Analysis
Single Factor and Multi-Factors Cox Analysis Results.

Results
Patient Characteristics and Survival Benefits
A total of 120 patients were included in the study cohort. Of them, 31 (25.8%) had died at the last follow-up. The median number of treatment cycles was 6.0, with a median OS of 37.2 months (95% CI: 19.67-37.20) and a median PFS of 9.3 months (95% CI: 8.13-17.63). The OS rates at 1, 2, and 3 years were 73%, 59.6%, and 55.8%, respectively, while PFS rates were 49.8%, 30.5%, and 24.5% at the same intervals. Kaplan-Meier survival curves for OS and PFS are shown in Figures 2 and 3, with immunotherapy response outcomes during the follow-up period in Figure 4. It was observed that most patients had SD at the first review after receiving 2 cycles of immunotherapy. Kaplan-Meier survival curves for overall survival (OS) in patients with cholangiocarcinoma treated with PD-1/PD-L1, with the curves representing the proportion of patients who survived over time. Kaplan-Meier Survival Curves for Progression-free Survival (PFS) in the Study Cohort. The figure shows the percentage of patients who remained progression-free at different time points after treatment. Lane diagram illustrating patient responses to immunotherapy based on follow-up time. Each lane represents an individual patient’s follow-up duration. The black pentagons on the left indicate patients who have died, while the colored shapes represent treatment responses: red squares indicate progressive disease (PD), yellow triangles signify stable disease (SD), and blue circles denote partial response (PR). Responses are evaluated according to the Response Evaluation Criteria in Solid Tumors (RECIST). PR is defined as a decrease of at least 30% in the total diameter of target lesions compared to baseline. PD is defined as an increase of at least 20% in the total diameter of target lesions, with an absolute increase of at least 5 mm or the appearance of one or more new lesions. SD is defined as no significant change in tumor size compared to baseline, meaning it does not meet the criteria for either PR or PD.


Adverse Reactions
Adverse reactions were reported in 115 patients (Table 2), with most classified as grade II. Grade I adverse reactions occurred in 71 cases, with anemia being the most common (42.3%). Grade II adverse reactions occurred in 100 cases, primarily characterized by leukopenia (31%). Grade III adverse reactions were reported in 86 cases, with leukopenia (23.3%), neutropenia (23.3%), and anemia (23.3%) being the most frequent. Grade IV adverse reactions occurred in 29 cases, with neutropenia being the most prevalent (41.4%). No grade V adverse reactions were reported. Immune-related adverse events included three cases of dermatitis (two with sintilimab, one with durvalumab), one case of colitis (sintilimab), and one case of cystitis (pembrolizumab).
Univariate and Multivariate Cox Analyses
Univariate Cox analysis identified treatment cycles, PLR, NLR, SII, CEA, CA199, ECOG performance status, and bone metastasis as potential prognostic factors for cholangiocarcinoma patients receiving immunotherapy. Multivariate Cox analysis identified treatment cycles (<6 vs ≥ 6 cycles, P < 0.01), bone metastasis (P = 0.001), NLR (≥3.08 vs < 3.08, P = 0.002), and CEA (≥4.13 vs < 4.13, P = 0.009) as independent prognostic factors for OS.
Development and Validation of the Nomogram
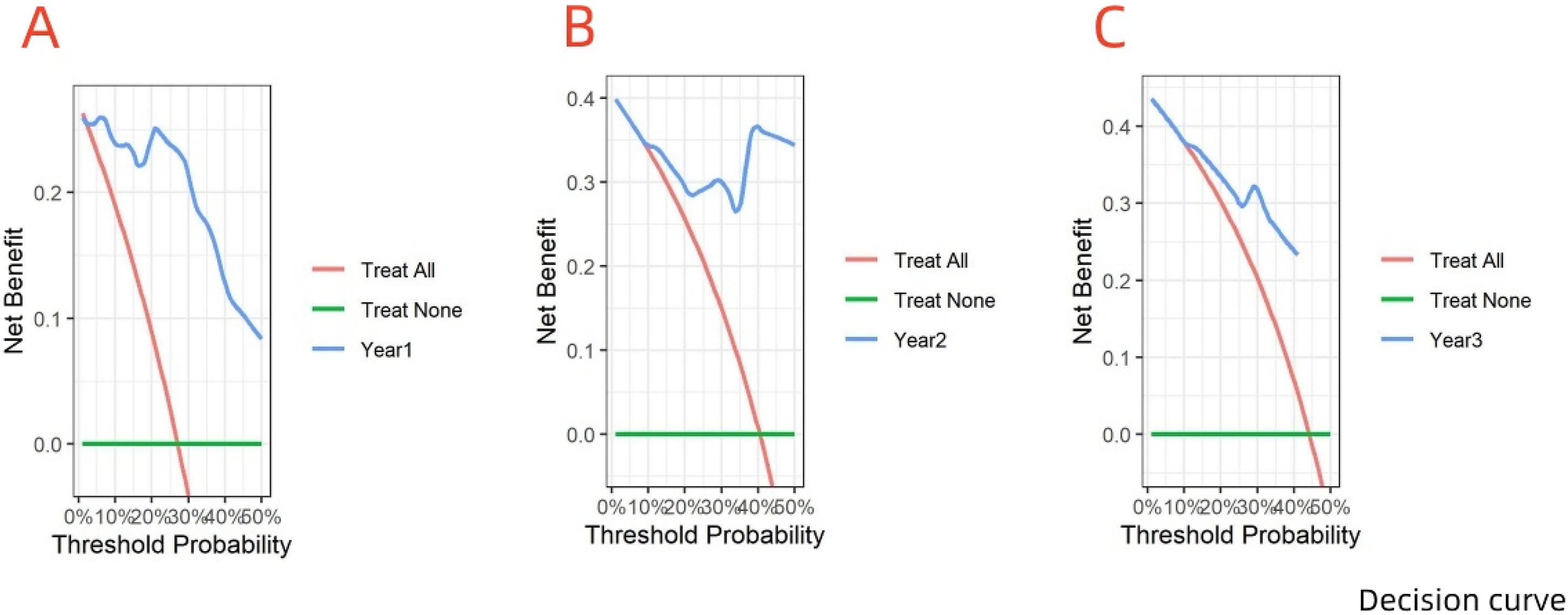
A nomogram model was developed based on the identified independent predictive factors: treatment cycles, bone metastasis, NLR, and CEA (Figure 5). The C-index was 0.811, indicating strong predictive accuracy. The multi-timepoint calibration curves closely aligned with the diagonal, indicating strong agreement between predicted and observed survival. The area under the curve (AUC) values of the multi-timepoint ROC curves exceeded 0.7 across all time points (Figure 5), further confirming the model’s high accuracy. Moreover, DCA demonstrated that the model provided a greater net benefit than the “all” and “none” lines at various threshold probabilities, supporting its significant clinical utility (Figure 6). (A) Nomogram for predicting patient outcomes based on clinical variables. Each variable’s contribution to the risk score is illustrated. (B) Multi-time receiver operating characteristic (ROC) curves for 1-year, 2-year, and 3-year survival predictions, showing sensitivity and specificity, the corresponding AUC values are shown. (C) Calibration curves corresponding to the ROC analysis, assessing the agreement between predicted probabilities and observed outcomes at 1, 2, and 3 years. Decision curve analysis (DCA) for evaluating the clinical utility of the predictive model at different time points: (A) 1 year, (B) 2 years, (C) 3 years. The DCA illustrates the net benefit of using the model compared to the default strategies of treating all patients or none.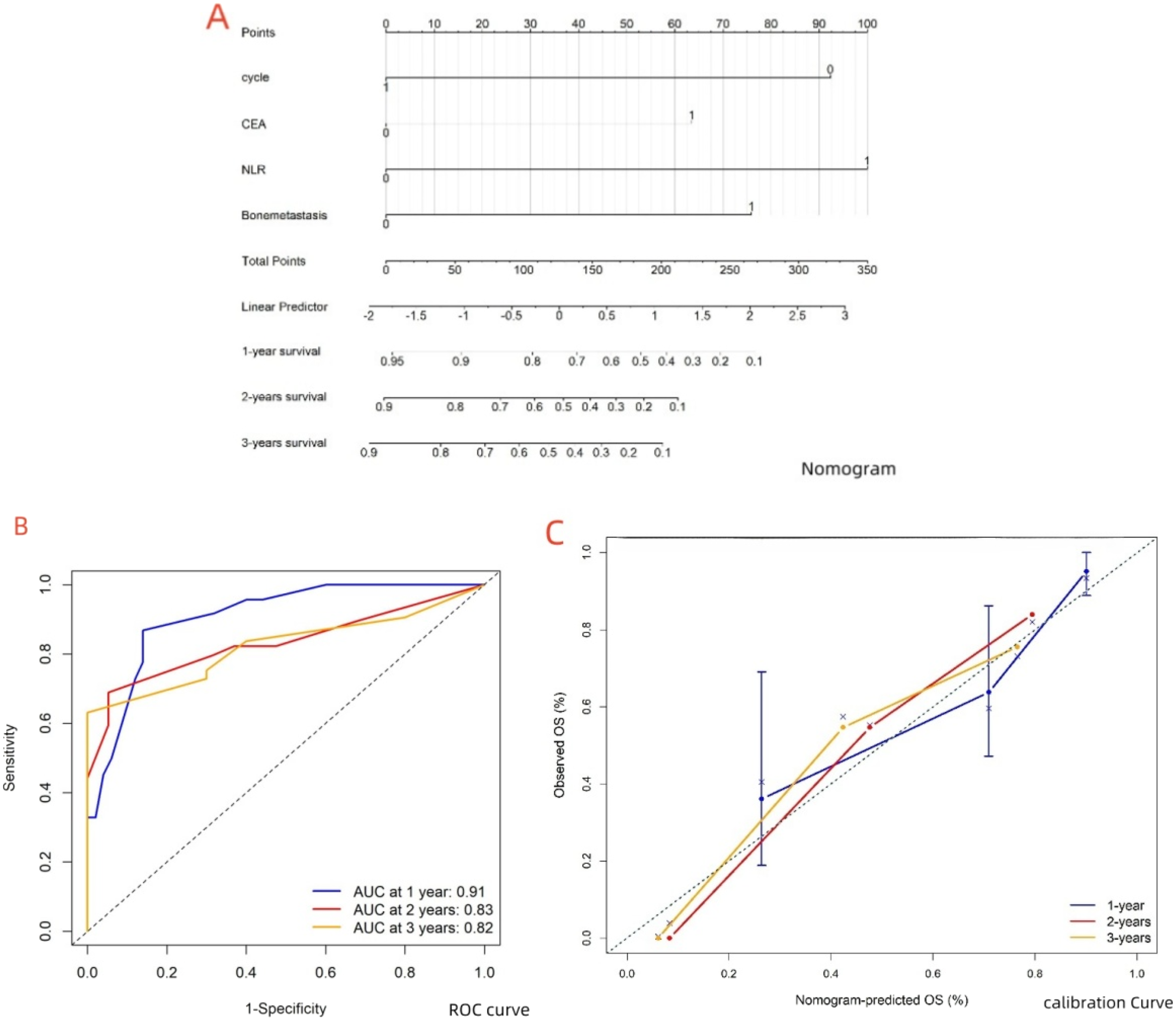
Discussion
The incidence and mortality of cholangiocarcinoma are increasing worldwide, with a 5-year survival rate below 20%. 32 CEA, CA19-9 levels, and inflammatory markers have been identified as potential prognostic factors for survival in cholangiocarcinoma patients.33,34 Studies have also highlighted the predictive role of inflammatory markers, CEA, and CA19-9 in lung cancer patients receiving immunotherapy.35,36 Localized inflammation has been shown in animal models to hinder PD-1 treatment efficacy and promote tumor progression. 37 Bone metastasis and impaired liver function also contribute to reduced efficacy of immunosuppressive treatments.38-40 Typically, two treatment cycles are considered standard for assessing the efficacy of immunotherapy, though delayed responses in some patients introduce uncertainty about the optimal duration of PD-1/PD-L1 therapy. Notably, no studies have yet discussed these influencing factors in the context of cholangiocarcinoma patients receiving immunotherapy. Meanwhile, nomogram models, which facilitate multivariate quantitative analysis, have demonstrated strong performance in predicting survival rates for patients with other cancer types undergoing immunotherapy.41-43 This study incorporated these factors into the analysis. Univariate and multivariate Cox regression analyses identified treatment cycles (<6 vs ≥ 6 cycles), presence of bone metastasis, neutrophil-to-lymphocyte ratio (NLR ≥3.08 vs < 3.08), and CEA levels (≥4.13 vs < 4.13) as significant factors associated with OS in cholangiocarcinoma patients receiving PD-1/PD-L1 inhibitors. By visualizing these factors through the construction of a nomogram, the model’s performance was evaluated using the C-index, CC, and DCA. The findings suggest that the developed nomogram performs well, potentially providing a valuable reference for predicting survival in cholangiocarcinoma patients undergoing immunotherapy.
Most patients experienced grade I or II adverse events, with bone marrow suppression as the most common manifestation. A meta-analysis of 161 studies identified bone marrow suppression as the most frequently observed adverse reaction following immunotherapy. 44 Bone marrow suppression may result from abnormal cytokine secretion induced by immunosuppressants, reactivation of CD8+ cytotoxic T cells, and the production of specific antibodies. 45 While mild bone marrow suppression minimally affects survival, complications such as infections and bleeding require careful monitoring. The effects of bone marrow suppression may accumulate over prolonged treatment cycles, necessitating long-term follow-up. 46 Several cases of immune-related adverse events (irAEs) were identified in this study, including one cystitis, one colitis, and three cases of dermatitis. Several studies have associated irAEs with prolonged survival following immunotherapy.47-49 Research suggests that irAEs may indicate enhanced inflammatory cytokine-mediated immune regulation. 50 Some evidence also suggests that irAEs could serve as biomarkers for immunotherapy.51,52 Due to the limited sample size, irAEs were not included as factors influencing survival in this analysis. Further research with larger sample sizes is needed to investigate the impact of irAEs on survival outcomes.
Advancements in imaging technology, particularly PET-CT and DCE-MRI, have improved survival predictions after immunotherapy by providing supplementary data for assessing treatment efficacy. Studies indicate that PET-CT parameters reflect the tumor microenvironment and provide insights into immunotherapy efficacy.53,54 A phase II clinical trial investigating the combination of immunotherapy with radiotherapy and chemotherapy demonstrated that high baseline SUVmax and total lesion glycolysis (TLG) on PET-CT were significantly associated with poorer PFS. Moreover, high TLG in the tumor microenvironment correlated with an immunosuppressive landscape. 55 Increased FDG uptake has also been shown to correlate with immune activation in the tumor microenvironment. 56 By dynamically observing the perfusion of contrast agents within tumor tissues, DCE-MRI can also assess the efficacy of immunotherapy. Metrics such as DKI and Ktrans values have been shown to correlate with both PFS and OS in patients undergoing immunotherapy.57,58 Moreover, emerging artificial intelligence techniques, such as ML and deep learning, offer significant advantages over traditional regression analyses. These techniques facilitate multi-dimensional feature extraction and capture both linear and non-linear relationships between variables and outcomes. ML models based on radiomics and radiogenomics have shown potential in identifying tumor immune microenvironment phenotypes and predicting survival and PFS.59-61 However, most current research focuses on lung and breast cancer patients undergoing immunotherapy, with fewer studies involving cholangiocarcinoma patients. Future research should aim to integrate clinical factors and imaging characteristics to develop predictive models tailored specifically for cholangiocarcinoma patients receiving immunotherapy.
This study has several limitations. First, it is a single-center, retrospective study with limited sample size. Larger, multicenter cohort studies are needed to validate the findings and improve the robustness of the nomogram model. Second, the nomogram is based primarily on blood samples collected before immunotherapy. Recent studies suggest a non-linear relationship between changes in NLR during ICI treatment and patient survival, with moderate decreases in NLR associated with the longest survival, whereas significant increases or decreases in NLR are associated with shorter survival outcomes. 62 Sample selection limitations restrict the generalizability of this nomogram. Integrating dynamic blood marker assessments, trend analyses, or ML models may enhance predictive accuracy and improve long-term efficacy evaluations. Third, as durvalumab has recently been approved by the FDA as a first-line treatment option for unresectable cholangiocarcinoma, this retrospective study unavoidably included patients treated with various PD-1/L1 agents. However, studies have discussed the differential efficacy of specific PD-1/L1 agents.63-69 Furthermore, the study cohort included patients who received combination therapies, as well as those treated with PD-1/L1 monotherapy following chemotherapy failure. Future studies should include larger cohorts and conduct stratified analyses to refine the model and improve its applicability across diverse therapeutic regimens.
Conclusion
This study developed and validated a nomogram incorporating predictive factors, including treatment cycles, bone metastasis, NLR, and CEA levels. The nomogram demonstrated strong stability and accuracy, providing a valuable tool for predicting survival in cholangiocarcinoma patients undergoing immunotherapy.
Footnotes
Acknowledgments
We would like to thank Dr Haibo Mou from the Shulan (Hangzhou) Hospital for his guidance in the development of this manuscript.
Author Contributions
Haibo Mou and Jianan Jin were responsible for the idea, design, quality control, review, and editing of the manuscript for this study. Jianan Jin was responsible for the conception, design, quality control, statistical analysis, and manuscript preparation of this study. Yibin Zhou, Shiqi Zhang, and Jianan Jin were responsible for data collection, proofreading, and patient follow-up. All authors approved the final version of the manuscript and all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
