Abstract
Encapsulated papillary carcinoma (EPC) represents a distinct entity within the spectrum of breast papillary tumors, typically manifesting as a retroareolar mass. This rare subtype can be effectively visualized using ultrasound and magnetic resonance imaging, which reveal characteristic cystic-solid nodules. Histopathologically, EPC is defined by a papillary tumor structure with a well-defined fibrous capsule, devoid of myoepithelial cells both within and around the capsule. Immunohistochemical staining for myoepithelial markers is essential to confirm the absence of these cells, thereby validating the diagnosis of EPC. At the molecular level, EPC exhibits feature similar to estrogen receptor-positive invasive ductal carcinoma (IDC), with a biological behavior that lies between ductal carcinoma in situ (DCIS) and IDC. Generally, EPC has a favorable prognosis, associated with minimal recurrence and metastatic potential. Therapeutic strategies for EPC may parallel those for DCIS, including surgical excision. Adjuvant radiotherapy is recommended following surgery for patients with concurrent DCIS or those who have undergone breast-conserving therapy. In cases with associated IDC, management prioritizes the treatment of the invasive component. High-grade EPC often requires systemic therapies due to its poorer prognosis and increased risk of lymph node involvement.
Keywords
Introduction
Encapsulated Papillary Carcinoma (EPC) of the breast, also known as intracystic papillary carcinoma, is a distinctive and uncommon variant of breast cancer. First described by McKittrick et al, in 1969, EPC was historically considered as a variant form of ductal carcinoma in situ (DCIS), accounting for approximately 0.5% to 2% of all breast cancer diagnoses.1–4 The fifth edition of the World Health Organization (WHO) classification of tumors of the breast recognizes EPC as a distinct subtype within papillary tumors and a specialized form of invasive breast cancer. 5 Histologically, EPC is characterized by a fibrovascular core with papillary structures lined by epithelial cells exhibiting low to intermediate nuclear grades, all encased within a fibrous capsule and lacking myoepithelial cells within the papillae or at the lesion’s periphery. 6 Given its unique histological features and generally favorable prognosis, EPC has garnered significant interest within the oncology community.
Recent investigations suggest that a subset of EPC cases, ranging from 2% to 14%, are of high grade and exhibit a more aggressive biological phenotype.7–9 Advances in molecular biology and clinical research have enhanced our understanding and therapeutic strategies for breast EPC. This review aims to synthesize the current research on EPC of the breast, providing a comprehensive and up-to-date resource for clinical diagnosis and management.
Clinical Presentation
Breast EPC predominantly affects females, with an age of onset ranging from 21 to 99 years.10-13 A study conducted by Grabowski et al, 4 which included 917 EPC patients, revealed a median age of diagnosis of 69.5 years. The prevalence of EPC among male breast cancer patients has been observed to vary between 3.5% and 13.7%,4,12,14-16 a figure that is significantly higher than the incidence of invasive ductal carcinoma (IDC) and DCIS in men.
EPC lesions are typically located in the central portion of the breast, frequently situated beneath the nipple and areola, and are often detected as a palpable mass in closely proximity to the areola.17,18 These lesions can vary significantly in size, with diameters ranging from 0.3 to 15.0 cm.18-20 Approximately one-third of patients present with nipple hemorrhage or discharge,15,18 while axillary lymph node enlargement is an uncommon finding. 19 A minority of patients may not exhibit any discernible clinical symptoms, underscoring the importance of routine screening and diagnostic imaging for early detection. 20
Imaging
Ultrasound and magnetic resonance imaging (MRI) have been shown to be superior to mammography in delineating the papillary architecture of breast lesions.21-24 MRI, in particular, offers significant advantages in visualizing the local infiltration of lesions and detecting any associated secondary lesions. 25
Mammography
In mammography, EPC typically appears as round or irregular masses with high to medium density opacities, often exhibiting smooth or lobulated margins (Figure 1). These characteristics frequently result in EPC being classified as a BI-RADS (Breast Imaging Reporting and Data System) category 3 or 4 lesion.25,26 However, when associated with invasive carcinoma, the margins of EPC may become indistinct,
27
occasionally presenting as irregular, lobulated nodules with spiculated contours and coarse calcifications.
26
Mammographic examination findings: (A). Case of pure EPC, smooth appearance with a clear boundary. (B) Cases of EPC with DCIS: the mass shadow is irregularly shaped with sharp edges and high density. A 5.5 mm diameter nodule with several small calcifications was identified behind and inferior to the mass. (C) Cases of EPC with invasive carcinoma: a nodular shadow with a relatively clear boundary and spiculated shadow can be observed at the local boundary.
Ultrasound Imaging
Ultrasound imaging plays a crucial role in diagnosing of EPC. As a follow-up evaluation for masses identified through mammography, ultrasound can clarify the nature of cystic lesions that contain solid components or have solid areas adjacent to ductal structures. These lesions typically appear as hypoechoic or exhibit mixed echogenicity, with well-defined borders. The cyst walls may present solid projections, and the solid portion may display a slightly irregular contour. Color Doppler flow imaging generally indicates an absence of blood flow signals around the cyst wall, while the solid portion may show linear or sparse punctate blood flow signals28-30 (Figure 2). These sonographic characteristics are instrumental in differentiating EPC from Solid Papillary Carcinoma (SPC) of the breast. However, both mammography and ultrasonography have limitations in sensitivity and specificity when distinguishing papilloma from complex cystic breast lesions, such as abscesses, fat necrosis, and hematomas.
26
A report by Lam WW et al, involving a cohort of 40 patients with 56 papillary lesions indicated that the diagnostic yield for mammography and ultrasound was 37.5% (21/56) and 82.1% (46/56), respectively. The ability to differentiate between malignant and benign papillary lesions was moderate, with mammography and ultrasound demonstrating sensitivities of 69% and 56%, and specificities of 25% and 90%, respectively.
31
Ultrasound examination findings for various types of EPC: A. Pure EPC case: Hypoechoic nodule with regular shape, non-parallel growth, blurred edges, uneven internal echo, enhanced posterior echo, and a clear boundary of the posterior space. Color Doppler flow imaging (CDFI) revealed strip-like blood flow signals. EPC with DCIS: Irregular nodule surrounded by multiple small punctate hyperechoic accumulations, with rich blood flow signals. C. EPC with invasion: Hypoechoic nodule with blurred edges and irregular fissure-like anechoic areas inside. Multiple small strong light spots with enhanced echogenicity were observed behind the nodule.
Magnetic Resonance Imaging
MRI demonstrates EPC as a cystic, encapsulated mass with well-defined solid components. The lesion appears isointense to slightly hyperintense on T1-weighted imaging (T1WI) and exhibits internal hyperintensity on T2-weighted imaging (T2WI). Contrast-enhanced sequences typically reveal ring-like enhancement of the mass, with irregularly enhanced papillary mass shadows evident within most cysts (Figure 3). These distinctive MRI features are crucial for identifying lesions that may be occult on mammography and indistinct on ultrasound imaging.25,31 MRI: (A-C). A simple EPC was identified in the left breast, displaying a hypointense signal on T1WI and a slightly hyperintense signal on T2WI. The lesion exhibited a smooth edge, slight enhancement on the contrast-enhanced scan, and relatively prominent edge enhancement. (D-F). EPC with DCIS presented as an abnormal signal mass shadow in the upper inner quadrant of the right breast, with uneven signal and significant enhancement post-contrast. G-I. EPC with invasion was characterized by an irregular lobulated mass with thick spiculation at the edge and heterogeneous signal intensity. The lesion demonstrated prominent and heterogeneous enhancement in the early phase of dynamic contrast-enhanced MRI.
Furthermore, MRI plays a pivotal role in preoperative lesion localization, evaluating local tumor spread, and identifying concurrent DCIS and invasive lesions.23,24 Tang et al 32 have emphasized that MRI features provide essential supplementary information for risk stratification of breast EPC, with large tumor size, surrounding non-mass enhancement, and axillary lymph node metastasis serving as indicators of higher risk stratification for the tumor.
Pathologic Findings
Morphology
Microscopically, EPC is delineated from the surrounding breast tissue by a thick fibrous capsule (Figure 4(A)-(C)). The tumors appear as unilocular or multilocular cystic structures with solid areas of variable thickness, predominantly composed of fibrous connective tissue. The fibrovascular papillae within the capsule are lined by a relatively homogenous population of neoplastic epithelial cells (Figure 4(D)). These cells generally exhibit a monomorphic columnar or cuboidal epithelial morphology with nuclei of low to intermediate grade, occasionally forming solid or cribriform arrangements (Figure 4(E)-(F)). A distinctive feature is the absence of a myoepithelial cell layer both within the fibrovascular papillae and at the periphery of the lesion. Furthermore, the tumor cells of EPC may exhibit apocrine differentiation, with over 90% of the cells displaying apocrine secretory characteristics, which are defined by abundant, finely granular, eosinophilic cytoplasm and small round nuclei with prominent nucleoli, thereby classifying this variant as apocrine EPC (Figure 4(G)-(H)).33-35 Microscopic observation of EPC: (A). The tumor is encased within a thick fibrous capsule, distinctly separated from the surrounding tissue. (B) Adjacent to the fibrous capsule is DCIS (indicated by the arrow) (HE, 40×). (C) Invasive carcinoma associated with EPC, located outside the capsule (indicated by the triangle) (HE, 40×). (D) The intracapsular tumor predominantly comprises fibrovascular papillae lined with low-nuclear columnar epithelium (HE, 100×). (E, F) The epithelial cells exhibit cribriform and solid arrangements. (G, H) Apocrine type EPC was characterized by apocrine epithelial cells displaying rich eosinophilic cytoplasm and prominent nuclei (HE, 200×).
Immunohistochemistry
The histomorphological overlap between EPC and other breast papillary lesions presents a significant diagnostic challenge. Accurate diagnosis is of EPC necessitates the identification of myoepithelial cells and the assessment of their distribution pattern through immunohistochemical staining. This process not only facilitates the differentiation on of benign papillary lesions from malignant papillary carcinomas of the breast but also aids in distinguishing EPC from papillary DCIS.
36
Commonly employed myoepithelial markers in breast pathology include P63, Calponin, CK5/6, CK14, 34βE12, and Smooth Muscle Actin (SMA) (Figure 5(A)-(D)). In routine pathological diagnosis, the use of a combination of multiple markers is often essential to achieve more accurate results.36,37 Immunohistochemical examination: (A-D) respectively revealed that the myoepithelial markers calponin, P63, CK5/6, and SMA were not expressed in the EPC papilla and around the fibrous capsule. In contrast, myoepithelial markers around the DCIS duct outside the fibrous capsule showed positive expression. (E, F). Tumor cells in EPC showed diffuse and strong expression of ER, with some tumor cells weakly to moderately expressing PR (EnVision, 40×).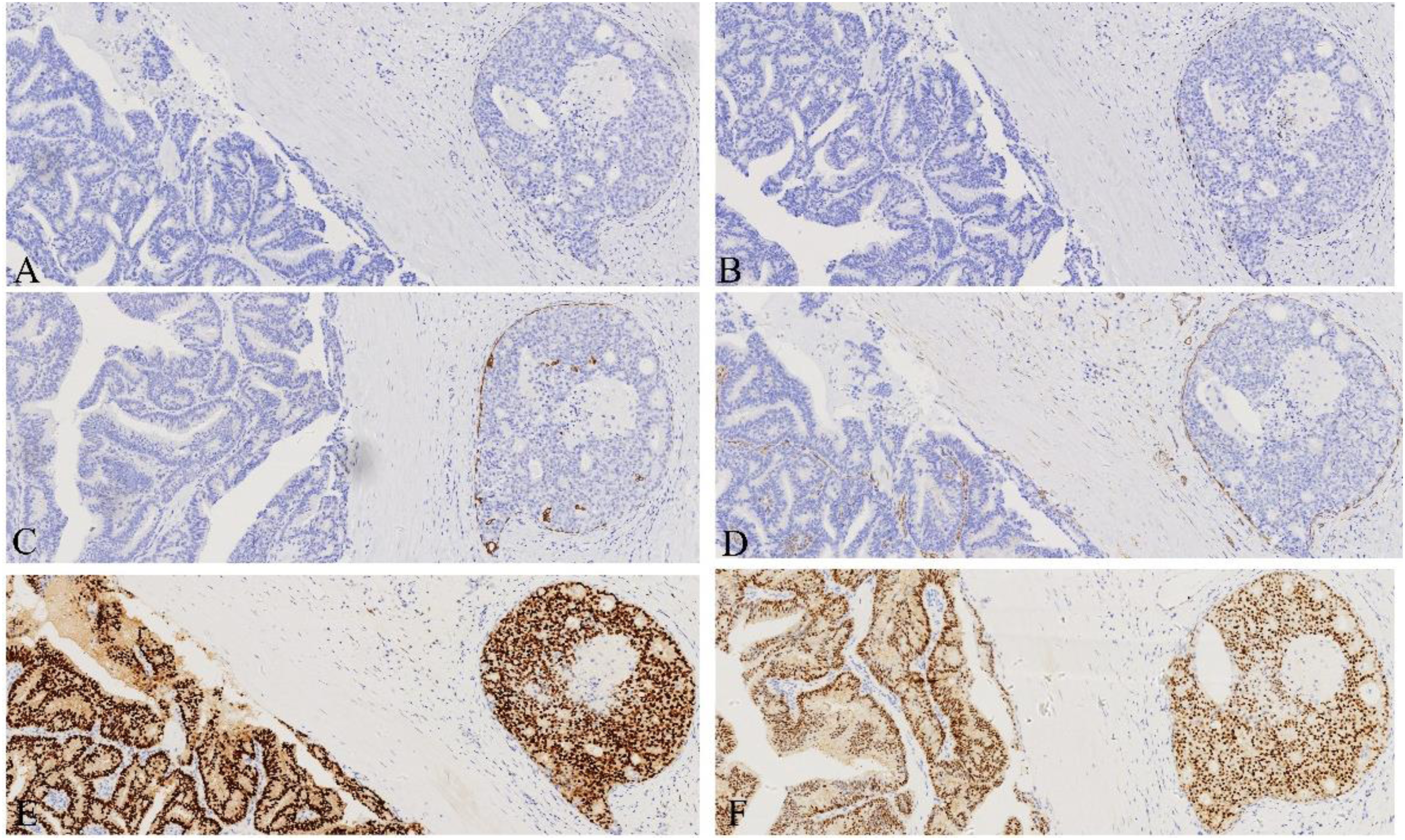
Immunohistochemical Features of EPC.
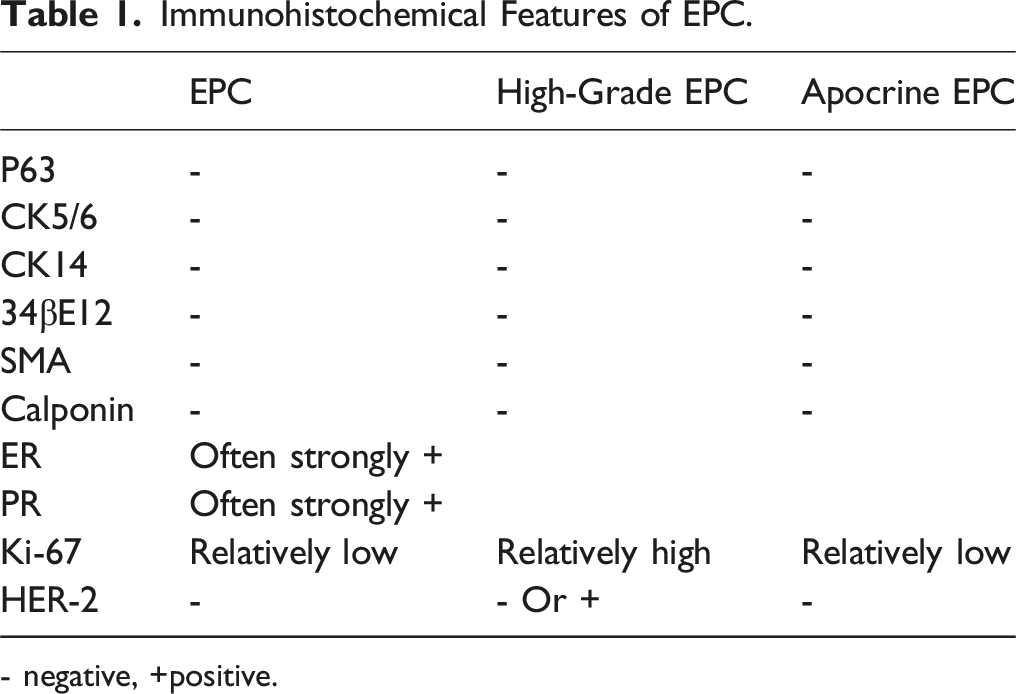
- negative, +positive.
High-Grade EPC
In 2015, Rakha et al delineated a novel subtype of EPC characterized by high-grade nuclei, 8 which is associated with more aggressive biological behavior and a propensity to occur in younger women.9,41,42 This high-grade EPC subtype is often marked by larger tumor volumes 42 and a higher frequency of necrosis compared to its low-grade counterpart. 43 Histologically, high-grade EPC predominantly features solid structures with compact papillary arrangements, sometimes accompanied by micropapillary elements. The tumor cells exhibit pronounced pleomorphism, discohesion, and frequent mitotic figures. Additionally, the stroma may show significant lymphoplasmacytic infiltration.9,43
Immunohistochemically, high-grade EPC demonstrates distinct profiles from low-grade EPC, exhibiting a predominantly hormone receptor-negative phenotype in contrast to the hormone receptor positivity typically observed in low-grade EPC.8,9,42,44 Chinese researchers have reported an increased prevalence of the triple-negative phenotype (negative for ER, PR, and HER-2) in high-grade EPC, with some cases demonstrating a basal-like immunophenotype. Furthermore, the mean Ki-67 proliferation index in these tumors can reach up to 85%, indicating significantly higher proliferative activity. 9 Follow-up data reported by Rakha, EA, et al indicate that approximately 10% (1/10) of patients with high-grade EPC experienced local tumor recurrence and axillary lymph node metastasis. 8
The WHO Working Group recommends that the grading, staging, and clinical management of high-grade EPC, or a triple-negative EPC, or HER-2 positive EPC should be equivalent to that of IDC.5,8,9,44,45 Consequently, precise identification and diagnosis of this EPC subtype are vital for effective clinical management and personalized patient care.
Molecular Mechanisms
The categorization of EPC of the breast as either carcinoma in situ or invasive carcinoma has been a topic of scholarly debate. Initially, Carter et al 46 (1983) characterized EPC as a variant of DCIS due to its lack of interstitial desmoplastic reaction and its characteristic nodular growth pattern. Conversely, Collins et al 6 (2006) argued that EPC should be classified as invasive breast cancer, based on immunohistochemical analyses of myoepithelial markers. They posited that EPC may be analogous to encapsulated papillary thyroid carcinoma, both of which are invasive cancers characterized by well-defined boundaries and capsules. In contrast, Esposito et al (2009) highlighted the presence of an intact basement membrane surrounding the lumen of EPC, as evidenced by a strong positive reaction to type IV collagen. This finding supports the notion that EPC is an in situ ductal lesion. 47 These conflicting findings underscore the necessity for further research and discourse to accurately diagnose and treat EPC in clinical practice.
Duprez et al (2012) employed genomic hybridization comparison and sequencing analysis to reveal that EPC and ER-positive IDC share similar genetic profiles. These commonalities include losses on chromosome 16q, gains on 16p, and 1q, as well as a high mutation rate in the PIK3CA gene. The authors proposed that EPC should be regarded as a subset of the ER-positive breast cancer lineage and emphasized that the favorable prognosis of EPC might be attributed to a lower frequency of tumor gene copy number abnormalities and a higher incidence of PIK3CA mutations. 48
Matrix metalloproteinases (MMPs) represent a family of zinc-dependent proteolytic enzymes primarily responsible for degrading the protein components of the extracellular matrix. 49 In breast cancer, MMPs are upregulated and are intricately associated with tumorigenesis, invasion, and metastasis, thereby categorizing them as invasion-related proteins.50-54 Rakha et al 55 found that the expression profile of invasion-related proteins in EPC lies between DCIS and IDC: MMP-1 and MMP-9 levels in EPC are higher than in DCIS but lower than in IDC, while MMP-7 and MMP-2 levels are comparable to DCIS but lower than IDC. This suggests that the invasive potential of EPC is intermediate between that of DCIS and IDC.
In a study by Salvatore et al, in 2014, integrated genomic and transcriptomic analyses revealed that EPCs exhibit significantly lower expression levels of proliferation-related genes such as CCND2, KIT, and MAP4K4, as well as genes involved in cell adhesion, migration, and motility, compared to IDCs. 56 These findings support the notion that EPCs exhibit a more indolent biological behavior than IDCs, aligning with the observations of Rakha et al.
Zinc Finger Protein Multiplexer 1 (ZFPM1) is a gene that encodes a protein featuring a Zinc Finger Domain and belongs to the PRDM (Positive Regulatory Domain with Zinc Finger) gene family. It has been implicated in the development of human cancers by influencing epigenetic modifications and gene reprogramming. 57 In a study by Liu et al. in 2021, somatic mutations in ZFPM1 were identified as a significant feature of EPC and were found to co-occur with mutations in the PI3K-AKT-mTOR pathway, thereby providing genetic evidence for the unique histological structure and biological behavior of EPC.
Furthermore, Schwartz et al 58 conducted an analysis of the genomic characteristics of pure EPC and EPC associated with IDC, revealing similarities in morphology, gene phenotype, and transcriptomics. However, EPC with associated IDC displayed specific mutations in PIK3CA and an enrichment of matrix-related genes, which are closely associated with the progression to IDC.
Recently, Ghannam et al 59 have further explored the question of whether EPC possesses a native basement membrane. By examining the distribution, density, and types of collagen fibers in the capsule wall of EPC, they have demonstrated that the EPC capsule results from a reactive process, rather than representing a thickened native basement membrane of associated with normal and in situ lesions. This research further supports the notion that EPC is an indolent invasive carcinoma characterized by its distinctive capsular features.
These collective findings suggest that EPC constitutes a distinct subtype of IDC with unique biological characteristics that may partially explain the clinical indolence observed in EPC.
Differential Diagnosis
Main Differentiation Points of Breast Neoplasms With Papillary Features.

EPC encapsulated papillary carcinoma, IP intraductal papilloma, DCIS ductal carcinoma in situ, IPC invasive papillary carcinoma, SPC solid papillary carcinoma, MEC myoepithelial cells.
Intraductal papilloma (IP) of the breast typically presents with bloody or serous nipple discharge. Histologically, IP is characterized by densely branched papillary structures with varying degrees of ductal epithelial cell proliferation, but without atypia and rare mitotic figures. Immunohistochemical staining demonstrates the presence of myoepithelial cells in the papilla and surrounding duct, which aids in differentiating from EPC.
Papillary DCIS of the breast, which is more prevalent in elderly women, commonly manifests as a multifocal tumor located within the duct or terminal duct. Microscopically, papillary hyperplasia of neoplastic cells with atypia within dilated ducts is observed. Unlike EPC, myoepithelial cells are present around the affected lumen in intraductal papillary carcinoma. Immunohistochemical markers such as P63, SMA, Calponin, and CK5/6 can facilitate the diagnosis.
Invasive papillary carcinoma (IPC) of the breast may present a diagnostic challenge, as the fibrous capsule of EPC can contain buried tumor epithelium, which may be mistaken for invasive carcinoma. However, true invasive carcinoma of EPC is distinguished by the presence of cancer cell foci outside the fibrous capsule, forming irregular epithelial nests with clear boundaries that invade the outer stroma, whereas myoepithelial immunohistochemical markers are negative. 60 True IPC is a rare occurrence and typically presents as invasive carcinoma with a simple papillary growth pattern. Many cases of IPC reported in the literature are actually misdiagnosed instances of EPC, SPC, or metastatic papillary carcinomas from other sites.61-63
SPC of the breast shares morphological, clinical, and prognostic similarities with EPC. SPC exhibits expansive nodular growth along terminal ductal lobular units with well-defined boundaries. Tumor cells demonstrate solid growth around fibrovessels, with a uniform oval-shaped or spindle-shaped morphology, sometimes polygonal, plasmacytoid, or signet-ring. Abundant granular cytoplasm and intracellular or extracellular mucus are common features. The majority of SPCs exhibit neuroendocrine differentiation, with positive neuroendocrine immunolabeling.
The accurate distinction between these lesions is contingent upon the integration of histological characteristics and immunohistochemical markers.
Treatment and Prognosis
EPC is regarded as a low-grade infiltrating cancer characterized by relatively indolent biological behavior, and it generally exhibits a favorable prognosis.7,16,18,38,64-67 Although some studies have documented instances of EPC metastasizing to lymph nodes and distant sites in the absence of invasive cancer components,7,14,18,20,67 the overall rates of local recurrence, lymph node metastasis, and distant metastasis remain very low.1,4,10,68,69 The median metastasis rate or recurrence rate is 4.3% (ranging from 0% to12%).4,9,14,18,19,45,55
When EPC is associated with DCIS or EPC with IDC, the risk of local recurrence may be elevated (about 8.8% higher than pure EPC). 46 However, a large retrospective study conducted in California by Grabowski et al, which involved 917 EPC cases, revealed that the 10-year relative cumulative survival rates for the non-invasive EPC group (pure EPC or EPC with DCIS) and the invasive EPC group (EPC with IDC) were comparable (96.8% and 94.4% respectively, with no statistically significant difference). This finding suggests an excellent long-term prognosis for EPC patients. 4 Additionally, Hiang Jin Tan et al 20 reported on 54 cases of EPC, with a median follow-up duration of 58.5 months (ranging from 1 to 241 months), Kaplan-Meier curve analysis demonstrated no statistically significant difference in overall survival rates among the 3 groups: pure EPC, EPC with DCIS, and EPC with IDC, further supporting the notion that the overall survival rate is favorable across all EPC groups.
The consensus of the WHO working group is that the clinical staging and treatment of pure EPC should align with that of DCIS. Surgical options include local excision (breast conserving surgery) and simple mastectomy. 5 Given the risk of lymph node metastasis, some researchers advocate for sentinel lymph node biopsy, while routine axillary lymph node dissection is generally avoided.4,19,20,45,68,70-72 In particular, invasive EPC exhibits aggressive biological characteristics and metastatic potential, making preoperative diagnose is as invasive challenging. 45 However, many researchers argue that compared to IDC, EPC has significantly higher rates of lower staging, lower grading, and lower metastasis rates, as reported in the study by Atif A. Hashmi et al, 38 with the respective ratios being 100%:24%, 94%:54%, 0%:24.8%. The long-term survival rate exceeds 90%.38,68 There is no clear or established evidence supporting the benefit of undergoing SLNB treatment. 68 Therefore, further clarification of management regarding the necessity of SLNB in EPC surgery may be required. Adjuvant radiotherapy is recommended for patients with EPC and DCIS, particularly those undergoing breast-conserving surgery to mitigate the risk of local tumor recurrence.1,5,7,20 A multicenter retrospective study conducted in Turkey evaluated the treatment efficacy and prognosis of 80 patients with EPC. The findings indicated that patients who received radiotherapy had a lower local recurrence rate compared to those who did not, thereby underscoring the significance of postoperative radiotherapy (P = 0.025). 10 Additionally, the study by Mogal et al 73 supports the advantages of adjuvant radiotherapy, revealing that patients who received such treatment had a reduced risk of death (HR 0.5, CI 0.3-0.7; P < 0.001).
When invasive carcinoma is present, the assessment of tumor ER, PR, HER-2 status, Nottingham grading, and tumor staging should focus solely on the invasive component.5,68 In instances where EPC is accompanied by microinvasion within the capsule, this does not impact clinical prognosis and should not be classified as invasive carcinoma. Consequently, clinical treatment should continue to reference DCIS. 60
Conclusion
Breast EPC represents a unique and rare subtype of breast cancer, characterized by a relatively favorable prognosis. The emergence of molecular biology techniques and ongoing clinical research has enhanced our understanding of the molecular and biological characteristics, diagnostic methods, and therapeutic strategies associated with EPC. As our knowledge base continues to grow, future research efforts should be directed toward the development of personalized treatment regimens for breast EPC, with the aim of improving the quality of life and long-term outcomes for patients.
Footnotes
Acknowledgments
We would like to thank He Xiangyun for his assistance in this paper.
Author Contributions
Conceived and designed the study: Wang Changrong, Zuo Zhibo, Guo Jufeng, Yan Hongju, You Feng and Xiang Jingjing. Administrative support: Xiang Jingjing. Collection and assembly of data: Wang Changrong, Zuo Zhibo, Guo Jufeng, Yan Hongju, You Feng. Drafted the manuscript: Wang Changrong, Xiang Jingjing. All authors interpreted the data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Traditional Chinese Medicine and Science and Technology Program Project of Zhejiang Province under Grant Number 2024ZL719.
