Abstract
Background
To better understand the clinicopathological features and prognostic profiles of squamous cell carcinoma (SCC) of the breast.
Methods
Information on breast cancer was obtained from the Surveillance, Epidemiology, and End Results database (2004–2016). Comparative analyses were carried out to investigate the heterogeneity in the clinicopathological characteristics and survival outcomes between SCC and invasive ductal carcinoma (IDC), while propensity score matching was conducted to analyze the variations among baseline characteristics. Prognostic factors for SCC of the breast were successively identified using Cox regression analysis.
Results
A total of 382 SCC patients and 561477 IDC patients were identified in this study. Comparatively, the SCC cohort exhibited a higher proportion of male individuals, poor differentiation, an advanced TNM stage, an increasing percentage of triple-negative (TN) subtype, an increasing rate of organ involvement, and less access to therapeutics. The aggressive profile was consistent in the TN subgroup, with a significantly higher proportion in SCC than in IDC (25.7% vs 6.8%). Prognosis of SCC was profoundly poorer than that of IDC (mOS, 78.6 months and 121.6 months,
Conclusions
This study systematically analyzed the heterogeneous characteristics of SCC of the breast in comparison with IDC. Squamous cell breast cancer presented with increasing aggressive behavior and inferior prognosis. Prospective studies should focus on this subgroup and introduce individualized therapeutic protocols in clinical practice.
Keywords
Introduction
Squamous cell carcinoma (SCC) of the breast is a special histologic breast cancer, and its clinicopathological characteristics and survival outcomes are not well understood. Pure SCC of the breast, which accounts for more than 90% of malignant cells originating from squamous metaplasia, accounts for approximately 0.1% of all invasive breast cancer. 1 Due to its very low prevalence, a poor understanding exists regarding its clinical presentation, in association with the absence of less standard treatment of SCC of the breast.
Previous studies have assessed the clinical presentations of SCC of the breast.2-7 However, the majority of them focused on an insufficient sample size with a limited follow-up or discussed this subgroup of patients with the absence of comparators to fully understand the SCC-specific characteristics. The clinical features and prognostic profiles of SCC of the breast remain controversial, and given the undetermined consensus on this special subgroup, increasing evidence deserves thorough analysis to obtain a better understanding of the clinical outcomes in the SCC population. Herein, we conducted this study on a large-scale population, retrieved from the Surveillance, Epidemiology, and End Results (SEER) database, to comprehensively discuss the clinicopathological characteristics and prognosis of SCC of the breast, with the aim of curating an informative reference for practitioners in prospective practice.
Materials and Methods
Population
The information of patients diagnosed with breast cancer ranging from January 01, 2004 to December 31, 2016 was obtained from the SEER database (November 2018 submission), and individuals with the histologic type of invasive SCC were identified for the following analysis. Patients were excluded from the study in case of missing histologic confirmation.
Clinicopathological characteristics were extracted for the following data: age at diagnosis, sex, race, tumor grade, laterality, tumor size, node involvement, distant metastasis, subtype, estrogen receptor (ER) status, progesterone receptor (PgR) status, human epidermal growth factor receptor 2 (HER2) status, bone and visceral metastases, surgical intervention, radiation treatment, and chemotherapeutic delivery. This study was performed according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 8
Outcomes
Squamous cell breast cancer was considered a breast malignancy with the histologic type of SCC (ICD-O-3 8070-8078), while invasive ductal breast cancer was regarded as an invasive breast neoplasm with ductal carcinoma (ICD-O-3 8500). Overall survival (OS) was defined as the interval between the initial diagnosis of breast cancer and death due to any reason or the last follow-up. Breast cancer–specific survival (BCSS) refers to the period from the diagnosis of primary breast cancer to death caused by cancer progression. According to the SEER terminology, visceral metastases consist of lung, liver, and brain involvement. The American Joint Committee on Cancer 7th edition guidelines were adopted to define the TNM stage of breast cancer.
Statistical Analysis
Comparative analyses of demographics and clinicopathological features between invasive ductal carcinoma (IDC) and squamous cell breast cancer were performed using Pearson’s chi-squared and Fisher’s exact probability tests for qualitative data and the t-test or Wilcoxon rank test for quantitative data with and without normal distribution, respectively. Propensity score matching (PSM) was carried out to analyze objective distinctions between baseline characteristics of the two subgroups of breast cancer, including age at diagnosis, sex, race, tumor grade, laterality, TNM stage, molecular subtype, ER status, PR status, HER2 expression, and treatment options. The discrepancy in survival outcomes was explored using the Kaplan–Meier method with log-rank tests, in which the prognostic factors of squamous cell breast cancer were identified using univariate Cox regression analyses. All statistical analyses were two-sided, with a P value less than .05, considered as statistically significant, and performed using IBM SPSS Statistics (version 26.0, Armonk, NY, IBM Corp) and R software (version 3.6.4).
Results
Clinicopathological Characteristics
In this study, 382 patients with SCC of the breast were eligible for inclusion. The differences in the baseline characteristics between the two cohort groups are presented in Table A1. The median age of patients with IDC and SCC was 60.88 and 66.51 years, respectively. In comparisons with IDC, patients with squamous cell breast cancer exhibited a higher proportion of male individual (2.6% vs .8%,
Regarding the patients with the TN subtype, it was denoted that a consistent pattern could be detected in % difference between the two cohorts, including an older age at diagnosis (67.38 years vs 58.63 years,
Prognosis
Substantial differences were detected in the prognosis profiles of patients in the two subgroups. The overall prognosis of the SCC cohort was significantly lower than that of IDC patients, with the median OS (mOS) of 78.6 months and 121.6 months (
To eliminate the uneven distributions in baseline characteristics, the PSM analysis in a 1:5 (SCC/IDC) was performed to investigate the heterogeneity in survival outcomes between the two cohorts, of which the results signified that the inferior tendency of both OS and BCSS remained stable in breast cancer patients with SCC (mOS, 78.9 months vs 97.1 months,
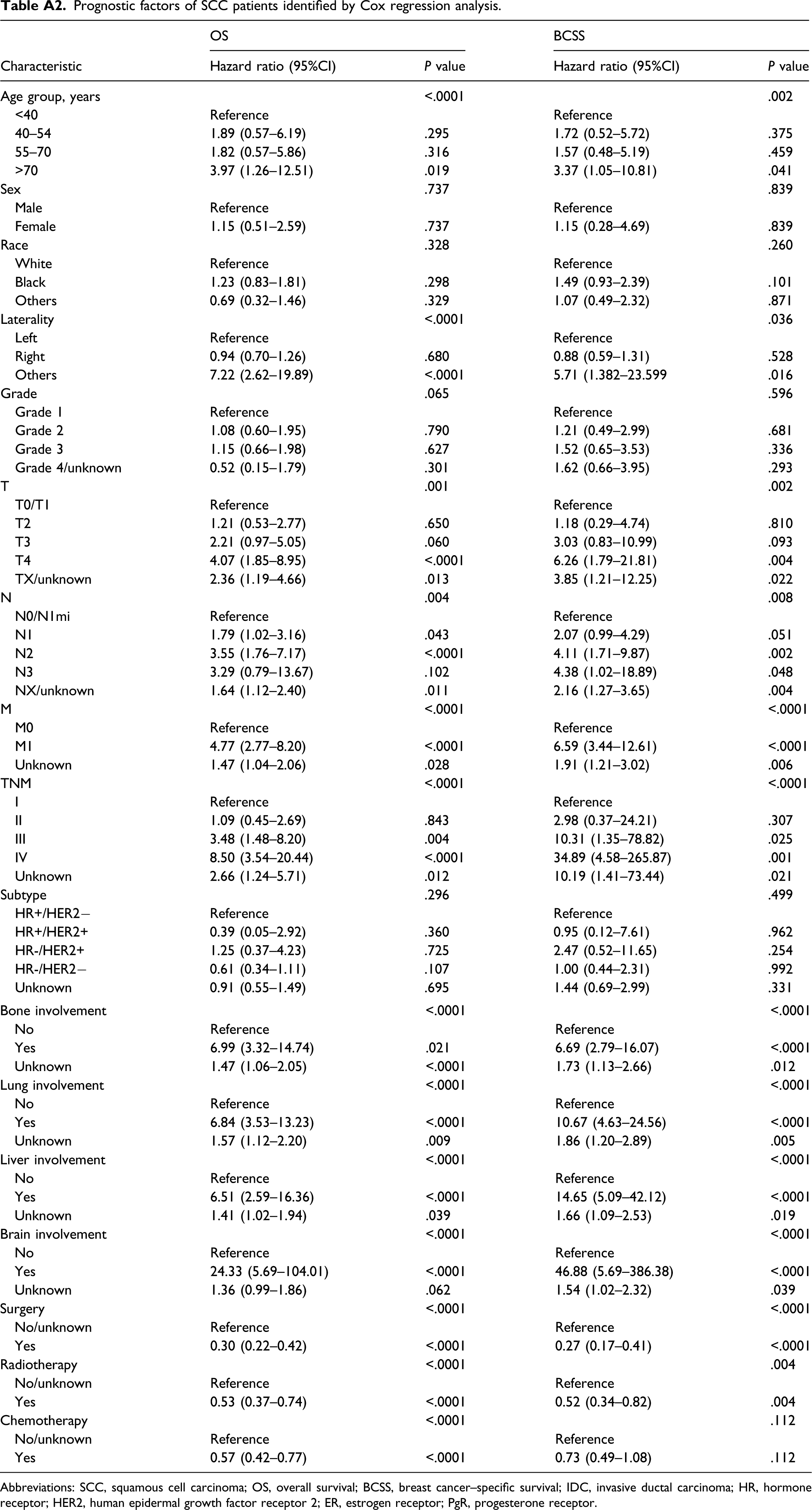
In the entire SCC cohort, the 2-year survival rate was 66.9%, and the 5-year survival rate was 51.4%. Regarding the TNM stage, there were 9.7% (37/382) of stage I, 20.4% (78/382) of stage II, 11.8% (45/382) of stage III, and 6.0% (23/382) of stage IV at initial diagnosis, of which the mOS were 62.8 months, 65.8 months, 37.3 months, and 17.9 months, and the mBCSS were 73.1 months, 70.5 months, 43.9 months, and 16.1 months, respectively (Supplementary Figure S7A and 7B). Comparative analysis of survival outcomes revealed that there was no statistical significance between the OS of stage I and stage II, which was greatly improved compared to that of disease at the advanced stage, including stage III and stage IV (Supplementary Table S2). A consistent tendency was detected in the BCSS of SCC of patients with breast cancer (Supplementary Table S3). The survival outcomes of patients with heterogeneous characteristics are presented in Supplementary Tables S4 and S5. The prognostic factors for OS were successively identified using Cox regression analysis, which included age (
Discussion
Although various evidence has been proposed from previous studies on the clinical features of SCC of the breast, it was rather insufficient, and this subgroup of breast malignancy deserves to be elaborated in depth. In this study, we systematically discussed the heterogeneity in clinicopathological features and survival outcomes of SCC of breast compared to IDC, taking full consideration of diverse clinical variables and focusing on the TN subtype with an illustration of the specific variability of this subgroup, which could provide a promising reference for the current practice.
First, we investigated the heterogeneous presentations between the SCC and IDC cohorts. It has been suggested that the disease characteristics of SCC tend to be more aggressive than those of IDC with the presentation of an advanced TNM stage and organic metastases, which was consistent with the findings of previous studies.3,9 Comparative analysis of survival profiles demonstrated that the prognosis of squamous cell breast cancer was significantly worse than that of IDC, which was independent of the disease stage and therapeutic applications. This sort of inferior prognosis remained constant after calibrating the uneven distributions among baseline characteristics with the performance of PSM analysis. From this perspective, we provided a landscape of intrinsic heterogeneity in the two subgroups and revealed the poorer survival associated with the aggressive behavior existing in this special histologic type.
Considering the overwhelming proportion of the TN subtype in the SCC cohort (25.7% vs 6.8%), we performed a systematic analysis and investigated the potential features existing in this subgroup. Aggressive disease features of TN subgroup patients were revealed compared to those with IDC, while the patients with SCC of the breast were less accessible to therapeutics, especially for systemic treatment, and the prognosis was significantly worse than that of the IDC cohort. This kind of prognostic discrepancy could be the result of insufficient therapeutic application, which was further confirmed by the undifferentiated survival between the SCC and IDC cohorts after PSM analysis. Although some studies have noted that the positive rate of hormone receptors tended to be lower in SCC, few analyses have been performed based on this subtype. Of note, this study was one of the few studies focused on the TN subgroup and revealed the potential underlying causes of prognostic discrepancy between the two subgroups of patients. Given the fact that there are limited options for TN subtype breast cancer except for systemic therapies, 10 physicians should take full consideration of the SCC of the breast while introducing multi-disciplinary therapeutic protocols with sufficient intensity.
In this study, the survival probability was estimated at a 5-year OS rate of 51.4%. Similarly, based on the SCC population, Yadav et al. reported the 5-year survival rate of SCC of the breast as 52.9%, in which the divergence might be the result of the different period for population enrollment (1998–2013 vs 2004–2016). 3 However, the 5-year survival probability significantly varied among previous studies with a range of 34.5–84.0%,1,4,11-13 which could be attributed to the discrepancies in sample size and study design. Risk factors for SCC were identified, including older age, advanced disease TNM stage, and organ involvement, which were similar to the data reported by prior studies focusing on breast cancer with other histologic types.14,15
Our study highlighted the substantial heterogeneity in the clinical features of breast cancer with squamous differentiation. Currently, clinical trials designed for breast cancer tend to ignore the histological classifications, which probably results in bias regarding clinical significance. Under these circumstances, histological features should be adopted to study protocols for unbiased design and promising survival benefits. In addition, given the profound heterogeneity between the two subgroups, whether therapeutic patterns were supposed to be introduced following the histologic type remained undetermined. This study focused on the differences between SCC and IDC, which was the most frequent subtype in breast cancer, and illuminated that treatment options should be applied in accordance with individualized factors. However, this study has several limitations. First, there is a proportion of missing data among the clinical variables, which might result in misestimation and potentially weaken the power of our findings. Second, some information regarding disease characteristics and therapeutics are not recorded in the SEER database, such as the KI-67 index and lymphovascular invasion, in addition to the implementation of endocrine and targeted therapies; therefore, we cannot elucidate the associations between these factors and clinical presentations. Last, given the fact that the specific coding based on ICD-O-3 are not given in the SEER database, thus the “pure” SCC of which the proportion of malignant cells differentiated as squamous type is usually more than 90%, 16 could not be fully identified in this study.
Conclusions
In conclusion, this study revealed profound heterogeneity in the clinicopathological characteristics and prognosis of SCC of the breast in comparison with IDC. Squamous cell breast cancer presented with increasing aggressive behavior and inferior prognosis as well as a notable proportion of the TN subtype. Prospective studies should focus on this subgroup and introduce individualized therapeutic protocols in clinical practice.
Supplemental Material
sj-pdf-1-ccx-10.1177_10732748211044355 – Supplemental Material for Clinicopathological Characteristics and Prognosis of Squamous Cell Carcinoma of the Breast: A Population-Based Analysis
Supplemental Material, sj-pdf-1-ccx-10.1177_10732748211044355 for Clinicopathological Characteristics and Prognosis of Squamous Cell Carcinoma of the Breast: A Population-Based Analysis by Yiqun Han, Jiayu Wang, Zijing Wang and Binghe Xu in Cancer Control
Footnotes
Author’s Note
The present study was performed in compliance with the Declaration of Helsinki. Permission was obtained to access the SEER database.
Declaration of Competing Interests
The authors declared no potential conflicts of interests with respect to the research, authorship, and /or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Appendices
Baseline characteristics of IDC and SCC breast cancer. Abbreviations: SCC, squamous cell carcinoma; IDC, invasive ductal carcinoma; HR, hormone receptor; HER2, human epidermal growth factor receptor 2; ER, estrogen receptor; PgR, progesterone receptor. OS (A) and BCSS (B) of IDC and SCC cohorts. Abbreviations: SCC, squamous cell carcinoma; IDC, invasive ductal carcinoma; OS, overall survival; BCSS, breast cancer–specific survival. OS (A) and BCSS (B) of IDC and SCC cohorts after a 1:5 PSM analysis. Abbreviations: SCC, squamous cell carcinoma; IDC, invasive ductal carcinoma; OS, overall survival; BCSS, breast cancer–specific survival. Prognostic factors of SCC patients identified by Cox regression analysis. Abbreviations: SCC, squamous cell carcinoma; OS, overall survival; BCSS, breast cancer–specific survival; IDC, invasive ductal carcinoma; HR, hormone receptor; HER2, human epidermal growth factor receptor 2; ER, estrogen receptor; PgR, progesterone receptor.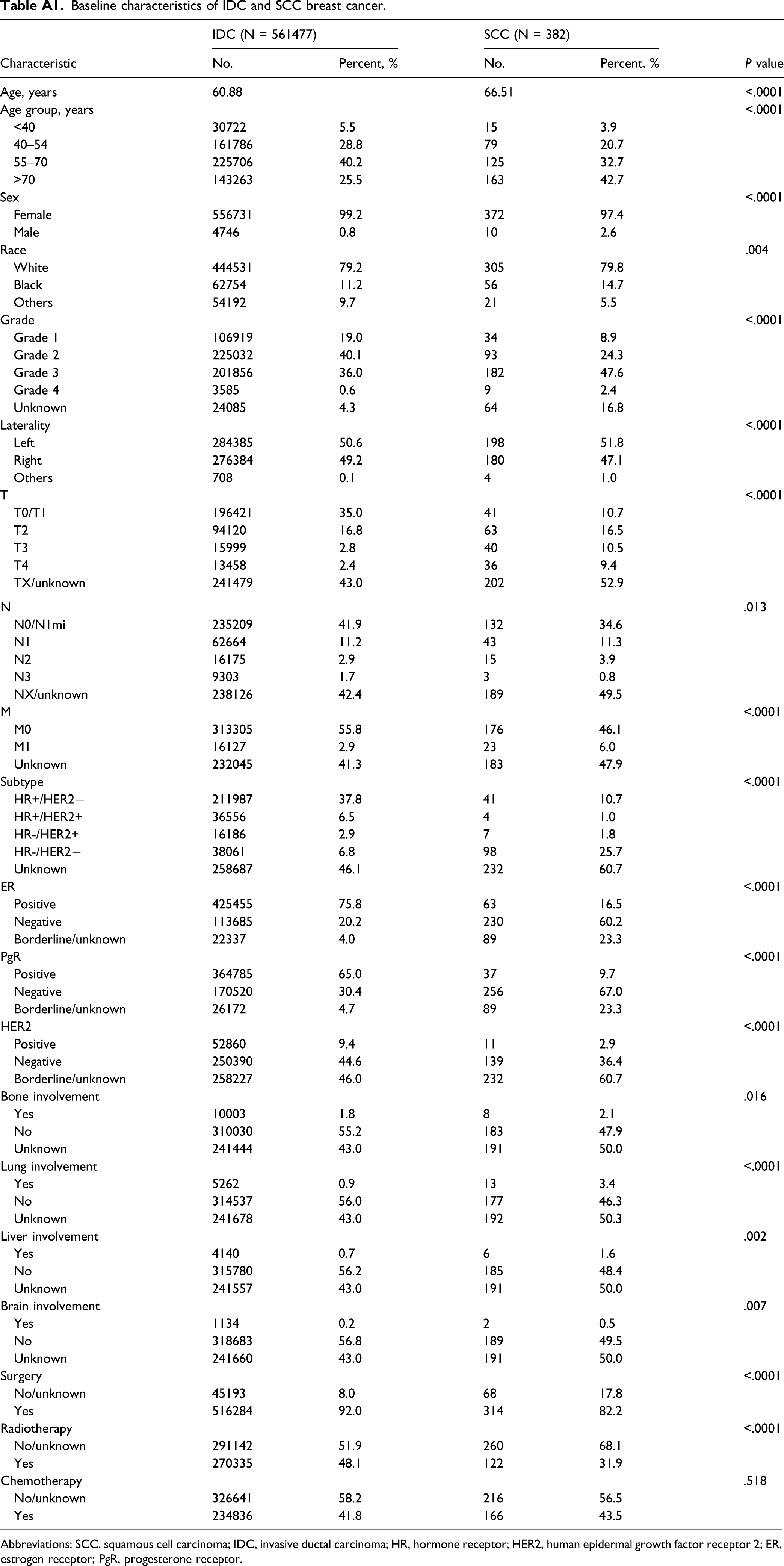
Characteristic
IDC (N = 561477)
SCC (N = 382)
No.
Percent, %
No.
Percent, %
Age, years
60.88
66.51
<.0001
Age group, years
<.0001
<40
30722
5.5
15
3.9
40–54
161786
28.8
79
20.7
55–70
225706
40.2
125
32.7
>70
143263
25.5
163
42.7
Sex
<.0001
Female
556731
99.2
372
97.4
Male
4746
0.8
10
2.6
Race
.004
White
444531
79.2
305
79.8
Black
62754
11.2
56
14.7
Others
54192
9.7
21
5.5
Grade
<.0001
Grade 1
106919
19.0
34
8.9
Grade 2
225032
40.1
93
24.3
Grade 3
201856
36.0
182
47.6
Grade 4
3585
0.6
9
2.4
Unknown
24085
4.3
64
16.8
Laterality
<.0001
Left
284385
50.6
198
51.8
Right
276384
49.2
180
47.1
Others
708
0.1
4
1.0
T
<.0001
T0/T1
196421
35.0
41
10.7
T2
94120
16.8
63
16.5
T3
15999
2.8
40
10.5
T4
13458
2.4
36
9.4
TX/unknown
241479
43.0
202
52.9
N
.013
N0/N1mi
235209
41.9
132
34.6
N1
62664
11.2
43
11.3
N2
16175
2.9
15
3.9
N3
9303
1.7
3
0.8
NX/unknown
238126
42.4
189
49.5
M
<.0001
M0
313305
55.8
176
46.1
M1
16127
2.9
23
6.0
Unknown
232045
41.3
183
47.9
Subtype
<.0001
HR+/HER2−
211987
37.8
41
10.7
HR+/HER2+
36556
6.5
4
1.0
HR-/HER2+
16186
2.9
7
1.8
HR-/HER2−
38061
6.8
98
25.7
Unknown
258687
46.1
232
60.7
ER
<.0001
Positive
425455
75.8
63
16.5
Negative
113685
20.2
230
60.2
Borderline/unknown
22337
4.0
89
23.3
PgR
<.0001
Positive
364785
65.0
37
9.7
Negative
170520
30.4
256
67.0
Borderline/unknown
26172
4.7
89
23.3
HER2
<.0001
Positive
52860
9.4
11
2.9
Negative
250390
44.6
139
36.4
Borderline/unknown
258227
46.0
232
60.7
Bone involvement
.016
Yes
10003
1.8
8
2.1
No
310030
55.2
183
47.9
Unknown
241444
43.0
191
50.0
Lung involvement
<.0001
Yes
5262
0.9
13
3.4
No
314537
56.0
177
46.3
Unknown
241678
43.0
192
50.3
Liver involvement
.002
Yes
4140
0.7
6
1.6
No
315780
56.2
185
48.4
Unknown
241557
43.0
191
50.0
Brain involvement
.007
Yes
1134
0.2
2
0.5
No
318683
56.8
189
49.5
Unknown
241660
43.0
191
50.0
Surgery
<.0001
No/unknown
45193
8.0
68
17.8
Yes
516284
92.0
314
82.2
Radiotherapy
<.0001
No/unknown
291142
51.9
260
68.1
Yes
270335
48.1
122
31.9
Chemotherapy
.518
No/unknown
326641
58.2
216
56.5
Yes
234836
41.8
166
43.5


Characteristic
OS
BCSS
Hazard ratio (95%CI)
Hazard ratio (95%CI)
Age group, years
<.0001
.002
<40
Reference
Reference
40–54
1.89 (0.57–6.19)
.295
1.72 (0.52–5.72)
.375
55–70
1.82 (0.57–5.86)
.316
1.57 (0.48–5.19)
.459
>70
3.97 (1.26–12.51)
.019
3.37 (1.05–10.81)
.041
Sex
.737
.839
Male
Reference
Reference
Female
1.15 (0.51–2.59)
.737
1.15 (0.28–4.69)
.839
Race
.328
.260
White
Reference
Reference
Black
1.23 (0.83–1.81)
.298
1.49 (0.93–2.39)
.101
Others
0.69 (0.32–1.46)
.329
1.07 (0.49–2.32)
.871
Laterality
<.0001
.036
Left
Reference
Reference
Right
0.94 (0.70–1.26)
.680
0.88 (0.59–1.31)
.528
Others
7.22 (2.62–19.89)
<.0001
5.71 (1.382–23.599
.016
Grade
.065
.596
Grade 1
Reference
Reference
Grade 2
1.08 (0.60–1.95)
.790
1.21 (0.49–2.99)
.681
Grade 3
1.15 (0.66–1.98)
.627
1.52 (0.65–3.53)
.336
Grade 4/unknown
0.52 (0.15–1.79)
.301
1.62 (0.66–3.95)
.293
T
.001
.002
T0/T1
Reference
Reference
T2
1.21 (0.53–2.77)
.650
1.18 (0.29–4.74)
.810
T3
2.21 (0.97–5.05)
.060
3.03 (0.83–10.99)
.093
T4
4.07 (1.85–8.95)
<.0001
6.26 (1.79–21.81)
.004
TX/unknown
2.36 (1.19–4.66)
.013
3.85 (1.21–12.25)
.022
N
.004
.008
N0/N1mi
Reference
Reference
N1
1.79 (1.02–3.16)
.043
2.07 (0.99–4.29)
.051
N2
3.55 (1.76–7.17)
<.0001
4.11 (1.71–9.87)
.002
N3
3.29 (0.79–13.67)
.102
4.38 (1.02–18.89)
.048
NX/unknown
1.64 (1.12–2.40)
.011
2.16 (1.27–3.65)
.004
M
<.0001
<.0001
M0
Reference
Reference
M1
4.77 (2.77–8.20)
<.0001
6.59 (3.44–12.61)
<.0001
Unknown
1.47 (1.04–2.06)
.028
1.91 (1.21–3.02)
.006
TNM
<.0001
<.0001
I
Reference
Reference
II
1.09 (0.45–2.69)
.843
2.98 (0.37–24.21)
.307
III
3.48 (1.48–8.20)
.004
10.31 (1.35–78.82)
.025
IV
8.50 (3.54–20.44)
<.0001
34.89 (4.58–265.87)
.001
Unknown
2.66 (1.24–5.71)
.012
10.19 (1.41–73.44)
.021
Subtype
.296
.499
HR+/HER2−
Reference
Reference
HR+/HER2+
0.39 (0.05–2.92)
.360
0.95 (0.12–7.61)
.962
HR-/HER2+
1.25 (0.37–4.23)
.725
2.47 (0.52–11.65)
.254
HR-/HER2−
0.61 (0.34–1.11)
.107
1.00 (0.44–2.31)
.992
Unknown
0.91 (0.55–1.49)
.695
1.44 (0.69–2.99)
.331
Bone involvement
<.0001
<.0001
No
Reference
Reference
Yes
6.99 (3.32–14.74)
.021
6.69 (2.79–16.07)
<.0001
Unknown
1.47 (1.06–2.05)
<.0001
1.73 (1.13–2.66)
.012
Lung involvement
<.0001
<.0001
No
Reference
Reference
Yes
6.84 (3.53–13.23)
<.0001
10.67 (4.63–24.56)
<.0001
Unknown
1.57 (1.12–2.20)
.009
1.86 (1.20–2.89)
.005
Liver involvement
<.0001
<.0001
No
Reference
Reference
Yes
6.51 (2.59–16.36)
<.0001
14.65 (5.09–42.12)
<.0001
Unknown
1.41 (1.02–1.94)
.039
1.66 (1.09–2.53)
.019
Brain involvement
<.0001
<.0001
No
Reference
Reference
Yes
24.33 (5.69–104.01)
<.0001
46.88 (5.69–386.38)
<.0001
Unknown
1.36 (0.99–1.86)
.062
1.54 (1.02–2.32)
.039
Surgery
<.0001
<.0001
No/unknown
Reference
Reference
Yes
0.30 (0.22–0.42)
<.0001
0.27 (0.17–0.41)
<.0001
Radiotherapy
<.0001
.004
No/unknown
Reference
Reference
Yes
0.53 (0.37–0.74)
<.0001
0.52 (0.34–0.82)
.004
Chemotherapy
<.0001
.112
No/unknown
Reference
Reference
Yes
0.57 (0.42–0.77)
<.0001
0.73 (0.49–1.08)
.112
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
