Abstract
Background
Cancer is the leading cause of death worldwide, and poor adherence to chemotherapy has become a public health issue in developing countries, including Ethiopia.
Objective
To assess chemotherapy adherence and associated factors among patients with cancer in the Amhara Region, Northeast Ethiopia.
Methods
A hospital-based prospective cross-sectional study design was employed, and a total of 460 cancer patients were enrolled using a systematic random sampling method at Felege Hiwot and Dessie comprehensive specialized hospitals from May 15, 2022, to July 15, 2022, in the Amhara region. The data was gathered through a review of medical records and a face-to-face interview and entered into Epi-Data version 4.6 before being exported to SPSS version 26 for analysis. Furthermore, at 95% confident interval (CI), multivariable binary logistic regression was used, and variables with
Result
The overall response rate in this study was 94.1% (433), with 42.3% of patients adhering to chemotherapy. Moreover, having a family history of cancer [AOR = 3.58, 95% CI (2.22, 5.76)], being female [Adjusted Odds ratio (AOR) = 2.17, 95% CI: (1.31, 3.60)], having no history of comorbidity [AOR = 2.74, 95% CI (1.56, 4.81)], having side effects from chemotherapy [AOR = 3.50, 95% CI (1.55, 7.90)], and having social support [AOR = 1.52, 95% CI (1.21, 1.95)] were important predictors of chemotherapy adherence.
Conclusion
and recommendations: Patients' understanding of illness characteristics and treatment should be improved through health education. Involving family members in the treatment plan will also improve adherence to chemotherapy in this context.
Introduction
Cancer is a group of diseases characterized by the uncontrolled proliferation and dispersal of abnormal cells. 1 Cancer is a leading cause of death and a major impediment to increasing life expectancy in every country on the planet. 2
In 2020, an estimated 19.3 million new cancer cases and nearly 10.0 million cancer deaths occurred worldwide. Cancer is an increasing public health burden in every country on the planet, particularly in Africa at large. It is worth noting that the cumulative risk of death from cancer among women in Africa in 2020 is roughly comparable to the risks observed among women in Northern America and Europe. 3 According to hospital records, there are more than 150000 cancer cases in Ethiopia each year, and cancer is responsible for 4% of all deaths. 4 Nevertheless, if the issue is ignored Cancer cases are expected to rise by 65% globally, from 12.7 million in 2008 to 21 million in 2030. 5
WHO adopts the following definitions of adherence from Haynes and Rand: “The degree to which a person’s behavior matches agreed-upon recommendations from a health-care provider about instructions regarding medication intake, medical device use, diet, exercise, lifestyle changes, rest, and attendance at scheduled appointments.”6-8
Although cancer has numerous treatment options, chemotherapy is by far the most widely used. 9 Chemotherapy adherence entails a collaborative effort and silent comprehension between care providers and patients about the level of compliance in day-to-day treatment in such things as dose, frequency, duration of therapy, and cessation. 10
Adherence to cancer treatment is critical for achieving optimal health outcomes such as a cure or an improvement in quality of life. Despite the availability of cancer treatment options, patients are vulnerable to adverse drug reactions for a variety of reasons, including poor drug adherence and clinical conditions. 11 As a result, poor chemotherapy adherence leads to drug resistance, recurrence, and poor quality of life in patients, negatively impacting the nation’s health costs and ultimately resulting in mortality. 12
Chemotherapy adherence is strongly related to patient-related factors, therapy-related factors, condition-related factors, healthcare system factors, and socioeconomic factors.10,13-16
The World Health Assembly’s sixty-sixth session approved the Global Action Plan for the Prevention and Control of Noncommunicable Diseases (NCDs), including Cancer, 2013-2020. 17 Furthermore, Ethiopia has had the National Cancer Control Plan strategy in place since 2015 to promote cancer prevention and early detection, as well as improve diagnosis and treatment, including palliative care. 18 Based on this, cancer treatment centers have been initiated in more than 10 public and teaching hospitals in Ethiopia. 18 The Amhara region has Six treatment centers, particularly Dessie and Felegehiwot comprehensive specialized Hospitals with large catchments and clients were involved in this study.
In the study area, there is a limited document about chemotherapy adherence and associated factors. As a result, the purpose of this study is to assess chemotherapy adherence and associated factors among cancer patients by including some variables (substance use such as cigarette smoking, alcohol consumption, and Khat chewing, as well as the presence of co-morbidities), thereby filling a knowledge gap in the study area. It has the potential to improve patient care and predict patient outcomes. Furthermore, developing and improving chemotherapy adherence protocols, as well as promoting cancer palliative care and research, are critical.
Methods and Materials
Study Area and Period
The research was carried out in the Amhara region from May 15, 2022, to July 15, 2022. The region is divided into 13 administrative zones, with Bahir-Dar serving as the capital city. It is Ethiopia’s second most populous region, with approximately thirty million people, accounting for 27% of the Ethiopian population. There are six comprehensive specialized hospitals in the region, of which two (Felege Hiwot and Dessie Comprehensive Specialized Hospital) provide chemotherapy for cancer patients predominantly in the region. Felege Hiwot and Dessie Comprehensive Specialized Hospital began chemotherapy treatments in 2016 and 2018, respectively; the Dessie Comprehensive Specialized Hospital oncology unit treats approximately 563 patients, while Felege Hiwot Comprehensive Specialized Hospital treats 547 cancer patients. The reporting of this study conforms to STROBE guidelines. 19
Study Design
A hospital-based prospective cross-sectional study design was employed.
Eligibility Criteria
The study included all cancer patients who received at least one cycle of chemotherapy treatment, and all cancer patients who were severely ill and unable to communicate during data collection were excluded.
Sampling Size
A single population proportion formula was used to calculate the sample size. The following assumptions were taken into account: At Tikur Anbessa Specialized Hospital, the proportion of chemotherapy adherence was 77.5%, 20 with a 95% level of confidence and a 4% margin of error. By including a 10% non-response rate, the total sample size increased to 460.
Sampling Procedure
A systematic random sampling method was used to select study participants. First, the total number of cancer patients who visited the oncology unit within two months at Falelege Hiwot Comprehensive Specialized Hospital (567) and Dessie Comprehensive Specialized hospital (371), was obtained, and the total number of patients per two months was calculated. The sample size was allocated proportionally. Based on this, 182 cancer patients on chemotherapy from Dessie Comprehensive Hospital and 278 cancer patients on chemotherapy from Falelege Hiwot Comprehensive Specialized Hospital were included. The first participant was chosen by the lottery method, and the subsequent participants were chosen using systemic random sampling every two intervals.
Data Collection Technique and Instrument
The data was collected through both face-to-face interviews and chart reviews and has four sections. The first section, such as socio-demographic characteristics (age, gender, marital status, educational level, region, place of residence, source of treatment, and employment status), was obtained directly from the patient via face-to-face interviews. Section two deals with clinical variables such as primary cancer diagnosis and type of cancer treatment (i.e., chemotherapy, radiotherapy, surgery, or a combination of these) that were obtained from medical charts at the oncology unit. The data extraction tool was developed based on a routine data registration protocol and the oncology center’s standardized chemotherapy entry and follow-up form. This oncology tool consisted of the weight of patients, diagnosis of disease, co-morbidity, stage of cancer, cycle and type of chemotherapy, appointment date, other medications given for patient problems, and name of physician (supplementary file 1). The medical records for cancer patients were obtained from the oncology ward registration book, and patient charts were obtained from the card room using a patient’s Medical Record Number (MRN). The third section also deals with patient adherence to chemotherapy, and the data was collected through a medication adherence tool. One of the largest and most commonly employed indirect methods of adherence assessment is patient self-report, and of these, the Morisky Medication Adherence Scale (MMAS-8) is an 8-item self-report scale that evaluates medication-taking behavior established from the earlier validated 4-item and augmented with extra questions to more accurately reflect obstacles related to compliance behavior.21,22 As a result, the eight-item (MMAS-8) was used to collect data on chemotherapy in this study. The scale consists of eight questions, the first seven of which have a binary answer (yes or no) coded as 1 or 0, indicating whether the person is adherent or non-adherent in taking their medications. 22 However, one item is different and has a 5-point Likert scale indicating how frequently he or she fails to take his or her medications. 23
The MMAS-8 internal consistency was found to be reliable (r = .88) in the previous study. 24 In this study, Face validity and content validity were assessed by four experts to determine the instrument’s validity. The content validity ratio (CVR) and content validity index (CVI) were calculated and found to be .23 and .84, respectively, indicating that the instrument is valid. And also, the instrument’s reliability was tested using Cronbach’s alpha among the 5% study population, which was .821, indicating that the instrument was reliable. The last section deals with the health-care system, social support, and personal habits like alcohol, chat chewing, and cigarette smoking.
Operational Definition
Good adherence: Patients were considered to have good adherence if their MMAS-8 sum score reached 8. 24
Poor adherence: Patients were considered to have poor adherence if they responded with a sum score of 1-7 on the MMAS-8. 24
Adverse effect: if the patient reported at least one of the following chemotherapy-related adverse effects: nausea, vomiting, fatigue, loss of appetite, taste changes, hair loss, mouth dryness, loss of weight, sleep disorder, hot flashes, and constipation.25,26
Social support: is the perceived and received support of a patient’s social network, such as familial relations, friends, neighbors, colleagues, fellow patients, or even pen pals and internet relationships, to reduce the emotional strain response, alleviate mental strain, and enhance social adaptability. 27
Data Quality Management and Analysis
The tool has been translated into the local Amharic language. Data collectors and supervisors were trained, and a pre-test of 5% of the sample size was conducted. The data was cleaned, encoded, and entered into Epi-Data software version 4.7 before being exported to SPSS version 26 of the statistical package for analysis. The level of chemotherapy adherence was calculated and assessed using descriptive statistics. A bivariable logistic regression analysis was performed to select variables for multivariable analysis. A variable with a
Result
This study included 433 cancer patients from a total of 460 study participants, with a response rate of 94.1%.
Socio-Demographic Characteristics of the Respondents
Socio-demographic characteristics of cancer patients on chemotherapy in the Amhara region, north-east Ethiopia, 2022 (n = 433).

Keynote: Others
Disease-Related Characteristics
Disease-related characteristics of cancer patients on chemotherapy in the Amhara region, north-east Ethiopia, 2022 (n = 433).

NHL stands for (Non-Hodgkin’s Lymphoma) and HL (Hodgkin’s Lymphoma).
Medication-Related Characteristics
Medication-related characteristics of cancer patients on chemotherapy in the Amhara region, north-east Ethiopia 2022 (n = 433).

Note: Polypharmacy refers to using five or more medications at the same time.
Healthcare System and Personal-Related Characteristics
Health-care system and personal-related characteristics of cancer patients on chemotherapy in the Amhara region, north-east Ethiopia, from May 2022 to July 2022 (n = 433).

Proportion of Chemotherapy Adherence
Of the total participants, 42.3% (95% CI, 37.6-46.7) of cancer patients adhered well to their chemotherapy regimens. Among participants with good chemotherapy adherence, 135 (73.8%) were female (See Figure 1). Proportion of chemotherapy adherence among cancer patients on chemotherapy in the Amhara region, North-East Ethiopia 2022.
Factors Associated With Chemotherapy Adherence
Factors associated with chemotherapy adherence among cancer patients Amhara region, North-East Ethiopia, 2022 (n = 433).
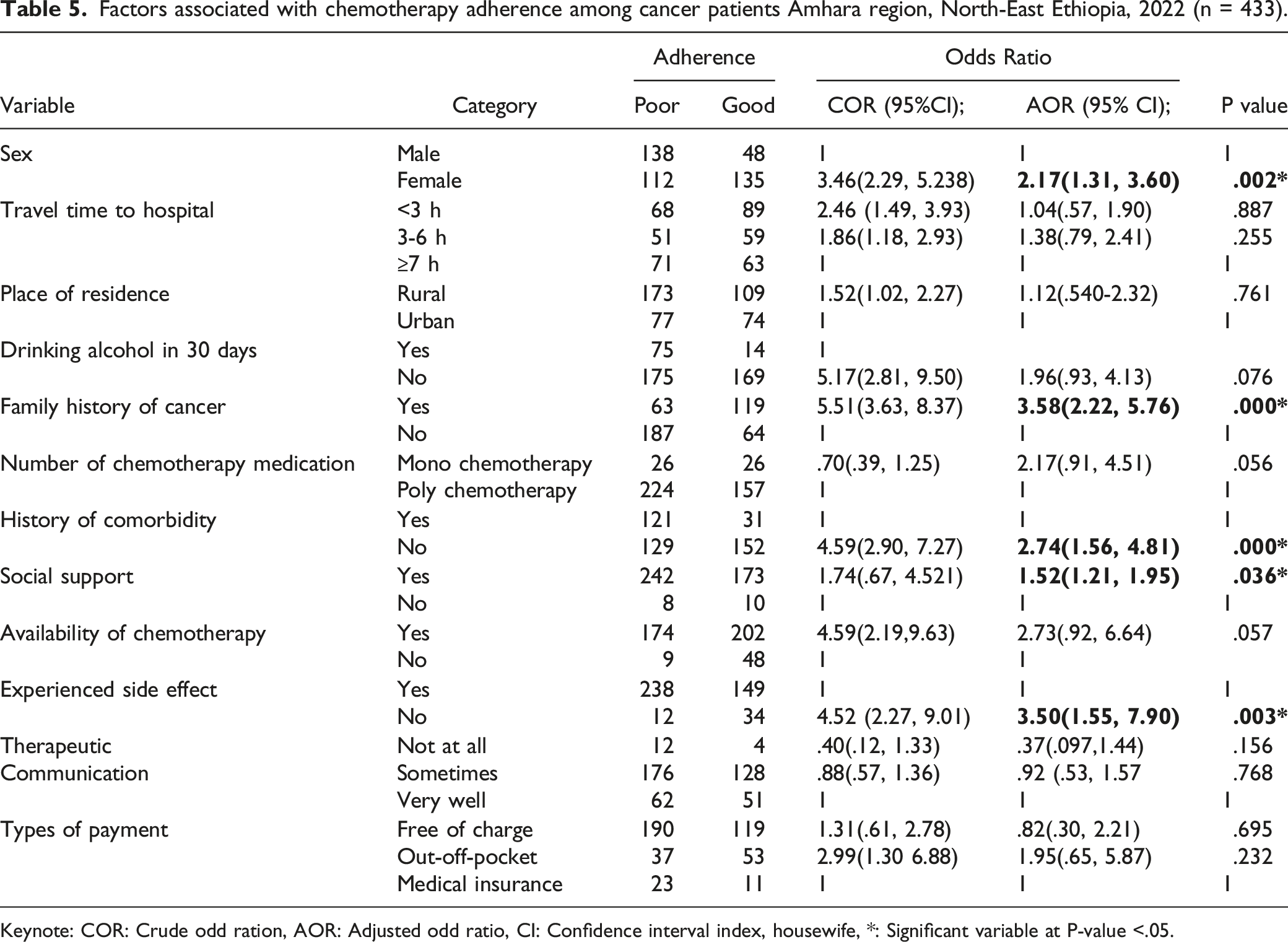
Keynote: COR: Crude odd ration, AOR: Adjusted odd ratio, CI: Confidence interval index, housewife, *: Significant variable at P-value <.05.
Discussion
The magnitude of cancer patients who adhered to chemotherapy was relatively low in this study. Sex, cancer family history, co-morbidity history, side effects experienced, and social support were positive predictors of chemotherapy adherence. Accordingly, the overall proportion of chemotherapy adherence among cancer patients in the Amhara region of north-east Ethiopia was [42.3% (95% CI, 37.6-46.7)] in this study. Chemotherapy adherence was higher among cancer patients in the Amhara region of North-East Ethiopia compared to studies conducted in Nigeria (19.1%) and India (21.1%).28,29 One possible explanation is that the majority of participants in this study were female, and female cancer patients were more adherent than males, so the proportion may be higher.
However, the results were lower when compared to studies conducted in the United States (85.57%), Japan (56.4%), and Brazil (85.2%).29-31 This difference could be explained by a change in assessment tools; in this study, the Morisky Medication Adherence Measurement Scale-8 was used to assess medication adherence, whereas in most other studies, the Morisky Medication Adherence Measurement Scale-4 was used to assess medication adherence. However, the results of this study were consistent with the results of a study conducted in Brazil (46.43%). 32
In this study, a significant relationship between sex and adherence level was discovered. As a result, men were found to be less adherent than women. This finding is consistent with a study conducted in India 33 in which men had a nearly threefold increase in the risk of non-adherence when compared to women. This is due to the fact that men are burdened by outdoor activities that keep them busy and cause them to forget their medications. Alcohol consumption, which is common among men, could also be a barrier to treatment adherence.
History Co-morbidities can worsen a patient’s condition and make it difficult for them to take their chemotherapy medications. According to this study, the number of co-morbidities among cancer patients had a significant impact on adherence behavior. Patients who had no or one co-morbidity were more likely to stick to their treatment than those who had two or more co-morbidities. This finding is consistent with the findings of a Brazilian study. 32 This association may be due to the fact that patients with a greater number of co-morbidities are more likely to experience serious complications and complex treatment regimens, which are favorable conditions for patients not to adhere to their medications.
This study also discovered that social support is linked to medication non-adherence. Cancer patients who were satisfied with their family and friends' support were 1.5 times more likely to adhere to their medication than those who were not. This study’s findings are consistent with previous research conducted in southern Brazil and other parts of the world.34,35 This could be because those who received motivation and encouragement were psychologically well and were able to strictly adhere to prescribed medication.
According to this study, having a history of side effects from chemotherapy drugs was associated with non-adherence to medication. According to this study, cancer patients who do not experience side effects from chemotherapy are at least 3.5 times more likely to have good adherence than those who do. Previous studies on cancer patients in Japan, Taiwan, and India found similar results.29,36,37 A possible explanation for this association could be that participants had a history of side effects that caused treatment interruptions due to concerns about worsening symptoms, which increased the burden of taking prescribed medications and their side effects. As a coping mechanism for the diminished symptoms combined with worsening side effects, some cancer patients decide to discontinue treatment; as a result, some of the patients in the medication non-adherence group may have decided to stop taking their medications to cope with side effects, as in the past.
This study also found a link between a family history of cancer and adherence to chemotherapy. Cancer patients with a cancer family history were about 3.5 times more likely to have good adherence than those without a cancer family history. This finding is consistent with a study conducted in Brazil. 31 One possible explanation is that patients with a cancer family history have more information about chemotherapy medication and the importance of good adherence, which increases adherence among cancer patients with a family history.
Conclusion
In this study, the proportion of cancer patients who adhered to chemotherapy was relatively low. Sex, family history of cancer, history of co-morbidity, experienced side effects, and social support were the main factors associated with the development of chemotherapy adherence. Health education about the disease and its treatment is important for improving patient adherence, and involving family members in the treatment plan and improving patients' understanding of the illness are critical in achieving high adherence to medication in this context.
Strengths and limitations
The Morisky Medication Adherence Scale-8 (MMAS-8) was used in this study as a standardized, expert-reviewed, and pre-tested data collection tool with high reliability and validity. There were some additional variables (alcohol consumption, cigarette smoking, and khat chewing) to assess associated factors for chemotherapy adherence. However, this study may have methodological flaws such as face-to-face interviews, which may lead to social desirability bias by overestimation or underestimation of the result, and recall bias may be exposed to obtain appropriate information from each participant, and cause and effect relationships for the factors dealt with in the study were not possible to establish because it is difficult to know which occurred first, the exposure or outcome variable.
Footnotes
Appendix
Acknowledgments
The author would like to thank Dessie and Felegehiwot Comprehensive Specialized Hospital staff, study participants, and the Merton office for their kind cooperation. We are also thankful to data collectors.
Author Contribution
The authors made substantial contributions to the conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the only institutional support from the hospitals and Alkan health science college.
Ethical Approval
The review committee of the College of Medicine and Health Science, Wollo University, Dessie, Ethiopia, vetted and approved the study procedures (WU/CMHS/140/2022) on April 20, 2022, and ethical approval was obtained. Dessie and Felegehiwot hospitals in the Amhara region also provided explicit consent letters. Having fully understood the purpose of the study, each respondent provided written consent prior to the interview. In addition, the study was carried out in accordance with the Declaration of Helsinki. Recalcitrant study participants were excluded from the survey.
Informed Consent
Anonymized data was collected, and participant information was kept confidential.
