Abstract
Objective
Advanced gastric cancer (AGC) is a severe malignant tumor, and overexpression of HER2/ERBB2 may play a crucial role in its development. The purpose of this study is to investigate the overexpression of HER2/ERBB2 in gastric cancer through a meta-analysis and examine its relationship with perfusion parameters using dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI) technology.
Methods
We conducted an extensive literature search and collected relevant studies for the meta-analysis. We used a random-effects model and assigned weights using the “inverse variance” method. Additionally, we included 95 AGC patients diagnosed pathologically between April 2018 and October 2021. They all underwent DCE-MRI scans, and the data were subsequently analyzed using the Omni kinetic software. HER2 expression was assessed using immunohistochemistry.
Results
The meta-analysis revealed an overall odds ratio (OR) of .21 for HER2/ERBB2 overexpression in gastric cancer, with a 95% confidence interval of [.14, .30]. DCE-MRI results showed a significant association between high HER2 expression and poor tumor differentiation (
Conclusion
Overexpression of HER2/ERBB2 in gastric cancer is significantly associated with certain radiomic parameters of DCE-MRI, providing a valuable diagnostic tool for clinical practice. Furthermore, the meta-analysis further confirmed the critical role of HER2/ERBB2 in the development of gastric cancer.
Keywords
Introduction
Gastric cancer (GC) ranks as the fifth most prevalent malignant tumor globally, exhibiting the third highest incidence and mortality rate.1,2 Every year, hundreds of thousands of people worldwide succumb to stomach cancer, underscoring the significance of studying its pathogenesis. The subtle onset of early gastric cancer (EGC) often results in patients being in the advanced stage at the time of the initial diagnosis. 3 The development of gastric cancer is influenced by various factors, including genetics, environmental factors, and dietary habits.1,2,4 Currently, the primary treatment modalities for advanced gastric cancer (AGC) include neoadjuvant chemotherapy, immunotherapy, and targeted molecular therapy. Notably, targeted molecular therapy has proven to be an efficacious strategy for extending overall survival.2,5,6 According to the National Comprehensive Cancer Network (NCCN) guidelines for gastric cancer diagnosis and treatment, the utilization of trastuzumab as the sole targeted therapy for gastric cancer is solely determined by the patient’s Her-2 positivity.7-9 It makes accurate evaluation of Her-2 crucial.10-12 The overexpression of HER2/ERBB2 (Human Epidermal growth factor Receptor 2, HER2) is believed to play a pivotal role in the pathogenesis of gastric cancer.13,14 Multiple studies have confirmed that the overexpression of HER2/ERBB2 in certain cancers is associated with the malignancy of tumors, poor prognosis, and resistance to certain treatments.15,16 The HER2/ERBB2 gene encodes the human epidermal growth factor receptor 2, which belongs to the epidermal growth factor receptor (EGFR) family. 17 HER2 plays an important role in cell growth, differentiation, and repair. When HER2 binds with other receptors, it can activate multiple signaling pathways, such as the PI3K/Akt and MAPK pathways, thereby promoting cell proliferation and survival. 18 In many types of cancer, especially breast cancer and gastric cancer, HER2 overexpression is considered a key factor in tumor initiation and progression. HER2-positive tumors usually exhibit higher malignancy and poorer prognosis and may develop resistance to traditional therapies such as chemotherapy.19,20 In gastric cancer, while the specific role of HER2/ERBB2 is still under investigation, increasing evidence suggests its close correlation with the development and progression of gastric cancer. However, current evaluation methods, such as immunohistochemistry (IHC) and fluorescent in situ hybridization (FISH), primarily rely on tissue specimens, which limits their widespread application. 21
In recent years, the Dynamic Contrast-Enhanced Magnetic Resonance Imaging (DCE-MRI) technique has been shown to evaluate the biological characteristics of different tumors accurately.22-24 DCE-MRI enables quantifying contrast agents, such as Gd-DTPA, as they enter the interstitial space. Additionally, DCE-MRI can evaluate target tissues’ vascular density and functional characteristics. By utilizing quantitative analysis software, DCE-MRI can accurately calculate a range of parameters related to pathological tissues, thereby offering supplementary information regarding the characteristics of the lesions. 25 Furthermore, prior studies have demonstrated the effective evaluation of tumor-related protein expression, such as Her-2, in breast cancer and its correlation with treatment response through the use of DCE-MRI.26-28
In summary, this study aims to investigate the overexpression of HER2/ERBB2 in gastric cancer and examine its correlation with perfusion parameters through the utilization of DCE-MRI technology. We posit that employing this combination approach will yield robust scientific evidence for the early diagnosis and treatment of gastric cancer, thereby holding significant implications for clinical practice.
Materials and Methods
Meta-Analysis
Meta-analysis, as a statistical technique, can integrate findings from multiple studies, thereby offering more robust and reliable conclusions.
29
Our meta-analysis adhered to the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA), with the aim of investigating the overexpression status of HER2/ERBB2 in gastric cancer development.
30
Data was collected from multiple studies, where ‘total’ refers to the total number of events in each study, and ‘event’ represents the number of HER2/ERBB2 overexpression events. We conducted an extensive literature search, including multiple academic databases such as PubMed, Embase, Web of Science, etc., covering the literature from the databases’ inception until 2023. The objective of retrieval strategies is to achieve high sensitivity and comprehensiveness. Following an initial search, we performed a two-stage literature screening process. We excluded irrelevant papers and then conducted a full-text reading of the remaining papers using pre-defined inclusion criteria (Figure 1). From the literature that met the inclusion criteria, we extracted essential information about the studies, encompassing details such as authorship, publication year, country of origin, detection methods, and data related to the expression of HER2/ERBB2 (Table 1). The analysis utilized a random effects model and employed the “inverse variance” approach for weight allocation. The log odds ratio is selected as the effect size because it can continuously measure the effect’s magnitude for proportional data. We used exponentiation to calculate the original Odds Ratios (OR) from the logarithmic ratio. It enables us to interpret OR directly while ensuring that the invalid line (representing no effect) is positioned at 1. PRISMA study flow diagram. The screening involves 2 stages: first, a broad literature search is conducted to identify studies that meet the search criteria. After the initial search, obviously, irrelevant studies are excluded, and the remaining studies are reviewed in full to determine whether they meet the predefined inclusion criteria. Finally, the basic information and data from the studies that meet the criteria are extracted, providing the foundation for further processing in the META analysis. The overall process aims to ensure the accuracy and reliability of the results. Included Studies. Note: IHC: immunohistochemistry.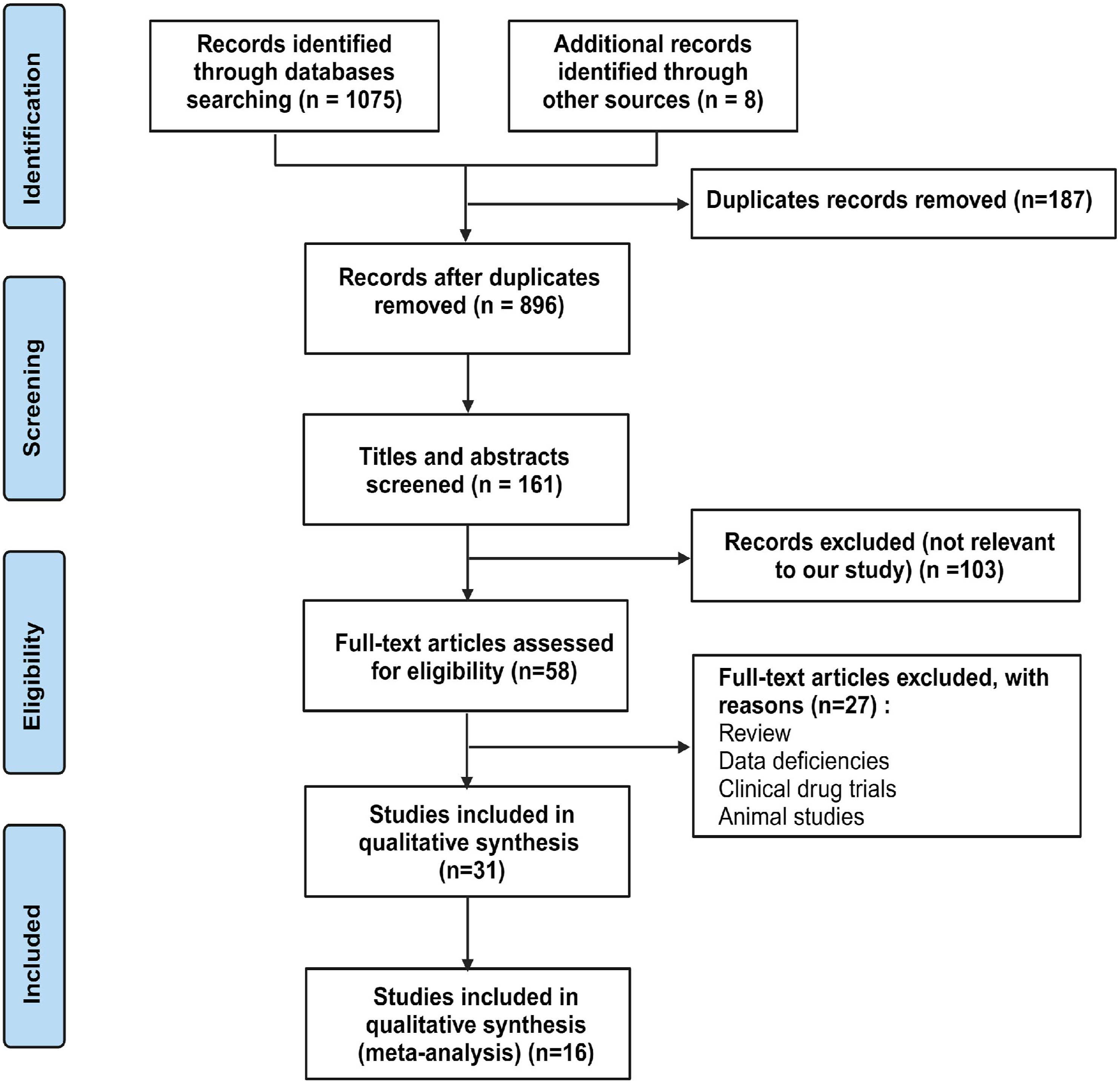

A sensitivity analysis was performed to evaluate the reliability of the findings. This analysis involved excluding low-quality studies and evaluating the impact of any individual study on the results. We also considered potential publication biases, including reporting and language biases, to assess their potential influence on the combined findings. To more accurately assess publication bias, we performed Egger’s and Begg’s tests using the R language. We calculated the pooled effect size and its corresponding 95% confidence intervals (CI) to provide quantitative measurements of the results and estimates of uncertainty. Additionally, we utilized forest plots to represent the pooled results visually and presented comprehensive explanations of the key findings, emphasizing the significance of demonstrating the involvement of HER2/ERBB2 overexpression in the initial stages of gastric cancer. 29
While conducting meta-analysis, we adhered to the relevant ethical and compliance regulations, encompassing research ethics review and data privacy considerations. This study has been registered in PROSPERO (CRD42024593102).
Patient Screening is Included in the Process
Dynamic Contrast-Enhanced Magnetic Resonance Imaging (DCE-MRI) is an advanced imaging technique that has been shown to effectively depict the microenvironment and biological characteristics of tumors.22,31 The participants in this study consisted of patients who were diagnosed with AGC from April 2018 to October 2021. The inclusion criteria for this study were as follows: (1) Patients who were confirmed to have gastric cancer through either surgery or gastric endoscopy biopsy; (2) Patients with complete clinical data, which included basic medical history, laboratory test results, and imaging data; (3) Patients who did not receive any anti-tumor treatment, such as chemotherapy or radiotherapy, before the DCE-MRI examination; (4) Patients with no history of other concurrent malignancies; (5) Patients who were 18 years of age or older. The exclusion criteria consist of the following: (1) the presence of contraindications for surgery or gastric endoscopy, such as severe heart, lung, liver, kidney dysfunction, or bleeding tendency; (2) poor quality MRI scan images due to factors like image distortion, blurring caused by patient movement, equipment malfunction, or other reasons, leading to unclear image segmentation; (3) tumors with a maximum diameter smaller than 1 cm, as they may adversely impact the accuracy of DCE-MRI evaluation; (4) patients who were lost to follow-up or non-compliant during the study period. The research process is as follows: AGC patients were retrieved from the hospital’s medical record system during the specified period, and screening was conducted based on the inclusion and exclusion criteria.
Moreover, extensive clinical data recording and DCE-MRI examinations were carried out on eligible patients. Lastly, we conducted post-processing and analysis on the DCE-MRI images and subsequently inputted the gathered data into statistical software for further analysis. Following a thorough screening and evaluation series, 95 eligible patients with AGC were included in this study. The research protocol was reviewed and approved by the Ethics Committee of Shaoxing People’s Hospital (No. 2020-K-Y-147-01).
DCE-MRI Protocol
Before performing surgical treatment or a gastroscopic biopsy, obtaining the patient’s informed consent must be obtained at least 1 week in advance. Moreover, to suppress gastrointestinal motility during the 3.0 T DCE-MRI examination, a muscle injection of 10 mL of Anisodamine should be administered within 20-30 min. During the 5-10 min preceding the examination, it is recommended for the patient to consume 800-1000 milliliters of warm water to have their stomach filled. Patients undergo 30 min of breath-hold training before undergoing an MRI examination.
The MRI examination was conducted using a Verio 3.0 T MR scanner and a 12-channel phased array surface coil manufactured by Siemens in Germany. The subject is supine with the head forwardly tilted and both arms raised for a comprehensive stomach scan. Subsequently, a routine abdominal examination is conducted, followed by DCE-MRI performance using the free-breathing technique. T1-weighted images at multiple reverse angles were acquired using a fast 3D volume-insertion screen gas technique and sequences incorporating fat suppression for transverse section scanning. The scanning parameters were as follows: TR of 3.25 ms, TE of 1.17 ms, reverse angles of 5°, 10°, and 15°, respectively, with a field of view measuring 350 mm × 284 mm, a matrix size of 288 × 164, and a slice thickness of 5 mm. The scanning duration for each reverse angle (5°, 10°, and 15°) is 6.5 s, which adds up to 19.5 s. We employed a gradient echo dynamic contrast-enhanced sequence with the following parameters: a flip angle of 10°, 30 slices, and a temporal resolution of 6.5 s. 34 phases were scanned, and the total imaging time was 227.5 s. One acquisition was performed using the parallel acquisition technique, with a voxel size of 1.7 mm × 1.2 mm × 5.0 mm and the other parameters described above. During multiphase dynamic enhanced scanning, a contrast agent is injected through the elbow vein using a high-pressure injector. The injection is administered at a dose of .1 mmol/kg and a rate of 3.5 mL/s. After completing the injection of the contrast agent, flush with 20 mL of normal saline at the same flow rate.
Infusion Parameter Collection
The Omni Kinetics software provides a powerful tool for in-depth radiomics parameter analysis. 32 Utilizing this software can achieve more precise analysis and interpretation of DCE-MRI images, thereby further elucidating the relationship between HER2 and radiomics parameters. We conducted a three-dimensional non-rigid registration preprocessing on the original DICOM images to rectify the displacement of regions of interest (ROIs) caused by involuntary movements, such as the patient’s breathing during DCE. Process patient images using GE Healthcare’s Omni Dynamics software. The motion-corrected dynamic contrast-enhanced sequence images and the images of dynamic contrast-enhanced sequence scans at multiple flip angles (5, 10, 15 degrees) should be imported into the software. Select the central blood vessels of the abdominal aorta to obtain the arterial input function (AIF) curve. The Tolt pattern is utilized to automatically generate voxel perfusion maps through post-processing software, which facilitates the analysis of contrast-enhanced dynamics. Two experienced radiologists participated in this study. The researchers chose the slices that showed the most noticeable tumor enhancement for the manual outlining of the region of interest (ROI). To ensure reliable data measurement and prevent complications such as bleeding, necrosis, and cystic changes, surrounding fat and normal gastric wall tissues are minimized. Special attention is given to reducing the inclusion of these tissues when delineating the lesion. Subsequently, the researchers integrated all the identified regions of interest to form a three-dimensional volume of interest. They performed an analysis to obtain perfusion parameter maps for Ktrans, Kep, and Ve. 33 Each imaging parameter should be calculated, and the extracted radiological features of each patient should be divided into 6 groups: first-order, histogram, Haralick, GLCM, RLM, and morphological indices. Quantitative parameters in all mutations are typically equal and have a minimum value of zero. Therefore, they are excluded from statistical analysis. The final measurement value is determined by calculating the average of 3 calculations.
Pathological Assessment
Immunohistochemistry (IHC) is a commonly used molecular biology technique that provides us with visual information regarding tumor marker expression.34,35 Surgical or endoscopic biopsy tissues are first fixed, sampled, dehydrated, embedded, and stained with hematoxylin and eosin (H&E). Representative wax blocks are then chosen for microscopic examination. Following this, the sample will be cut into 2 μm sections and subsequently undergo IHC, with 3,3′-diaminobenzidine (Sigma-Aldrich, D8001) used as the chromogen. Afterward, 2 expert pathologists will independently analyze and interpret the staining results of all sections. According to the standards set by the American Society of Pathology, 36 the expression of Her-2 can be categorized into 4 groups: (1) when the staining on the tumor cell membrane is less than 10%, it is designated as Her-2 (−); (2) when the staining on the tumor cell membrane is 10% or more, with weak and intermittent staining intensity, it is designated as Her-2 (+); (3) when the staining on the tumor cell membrane is 10% or more, with moderate and continuous staining intensity, it is designated as Her-2 (++); (4) when the staining on the tumor cell membrane is 10% or more, constantly covering the entire membrane, it is designated as Her-2 (++). If the expression of Her-2 is (++), FISH testing is conducted to determine the existence of gene amplification. 36 In this study, the expression of Her-2 was categorized as negative (−), positive (+), and strongly positive (++). The Her-2 negative group included cases classified as (−), (+), and (++), while the Her-2 positive group included cases with amplification (+++) and (++). 37
Statistical Analysis
First, all data is preprocessed before input into SPSS 25.0 and R software for further analysis and the generation of graphics. The normality of the continuous variable is initially evaluated using the Kolmogorov-Smirnov test. To analyze non-normally distributed data, we employed the non-parametric Mann-Whitney U test to compare quantitative parameters and histogram and texture features between 2 groups based on Her-2 status. After preliminary screening, we selected the features that demonstrated significant differences. We then employed lasso regression to further reduce dimensionality, aiming to address multicollinearity issues and ultimately identify the most influential variables. Receiver operating characteristic (ROC) curves were generated to evaluate the diagnostic performance of the selected features in distinguishing between Her-2 positive and negative cases. The corresponding Area under the curve (AUC) was calculated to assess the model’s accuracy. We calculated the pooled effect size (Odds Ratios, OR) and its 95% confidence interval during the meta-analysis. We also assessed heterogeneity using the I2 statistic. If the value of I2 exceeds 50%, we classify it as indicative of substantial heterogeneity and employ a random-effects model for meta-analysis. We have implemented corrections to ensure data accuracy, particularly in cases where the number of events exceeds the total count. Subsequently, we calculated the Z-value to evaluate the significance of the summary effect. The Z value is calculated by dividing the effect size of the merger by its standard error, and it is used for calculating the two-tailed
Results
Meta-Analysis Results
In our meta-analysis, we constructed a funnel plot to evaluate possible publication bias and explored the correlation between the effect size of studies and their precision (Figure 2A). While most research has primarily centered on the overall effect size of meta-analyses, a few studies stand out for their deviation from the central line in the low precision region. This observation suggests the potential existence of small study effects or publication bias. However, the majority of studies demonstrate effect sizes that are in line with the overall effect size. To more accurately assess publication bias, we performed Egger’s and Begg’s tests using the R language. The Summary of the effect size estimation and publication bias assessment of HER2/ERBB2 overexpression in gastric cancer using a random-effects model.
Based on the summary results of the random-effects model, we discovered that the odds ratio (OR) for overexpressing HER2/ERBB2 in the development of gastric cancer was .21, with a 95% confidence interval of [.14, .30]. The confidence interval is above 1, indicating a significant association between overexpression of HER2/ERBB2 and the development of gastric cancer (Figure 2B).
Regarding the heterogeneity among the studies, our findings indicate a high level of heterogeneity between the studies, with an I2 value of 94%. It suggests that the overexpression rate of HER2/ERBB2 may vary across different studies due to specific factors. We conducted a Z-test and found a
Sensitivity Analysis
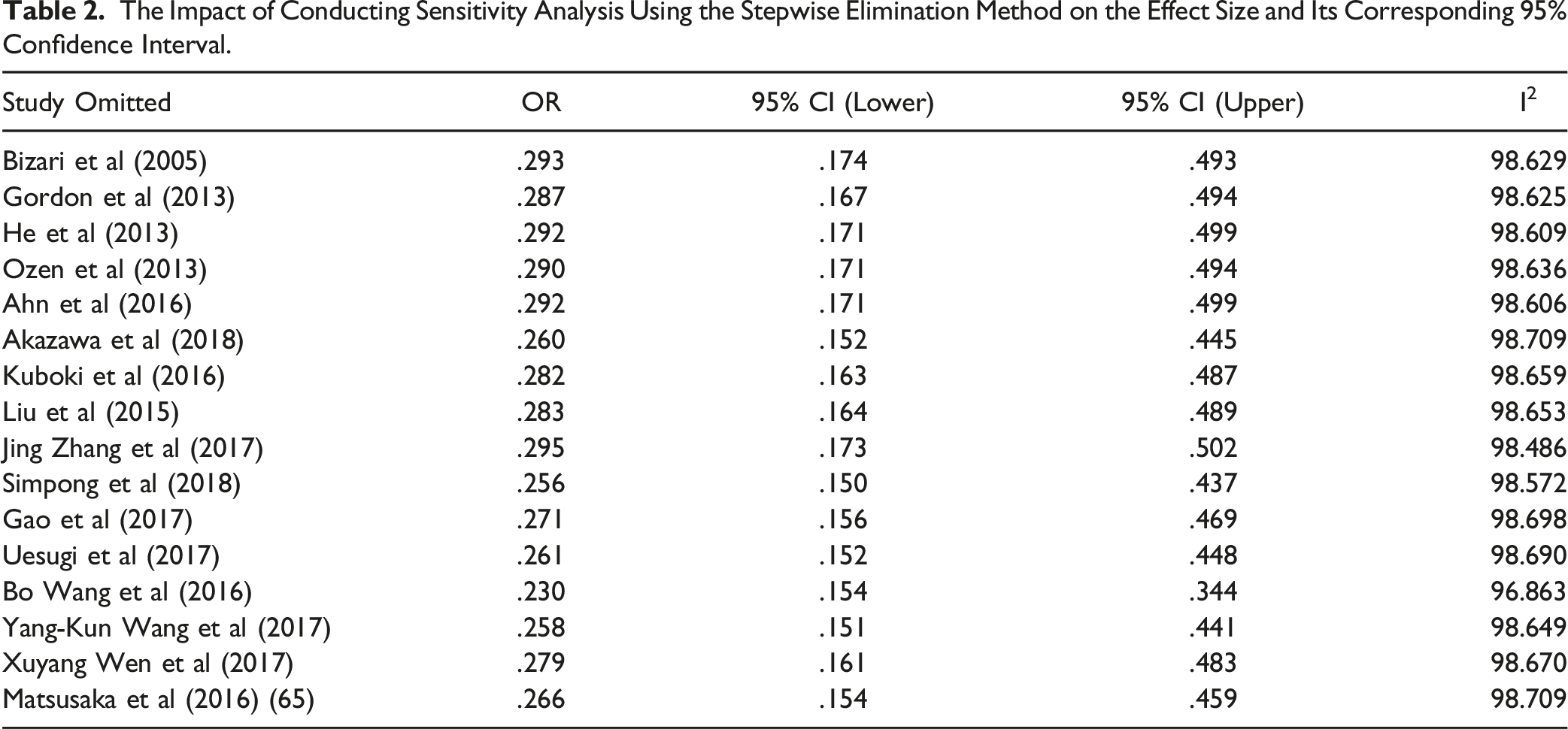
To evaluate the impact and reliability of each study on the combined results, we conducted a sensitivity analysis by applying the one-by-one exclusion method. Including all studies, the overall effect size (OR) is .274 with a 95% confidence interval of [.164, .459], demonstrating strong statistical significance (
The Impact of Conducting Sensitivity Analysis Using the Stepwise Elimination Method on the Effect Size and Its Corresponding 95% Confidence Interval.
Demographic Characteristics of Advanced Gastric Cancer Patients
Clinical Characteristics of Patients With Advanced Gastric Cancers.

Note: *Indicates significance at
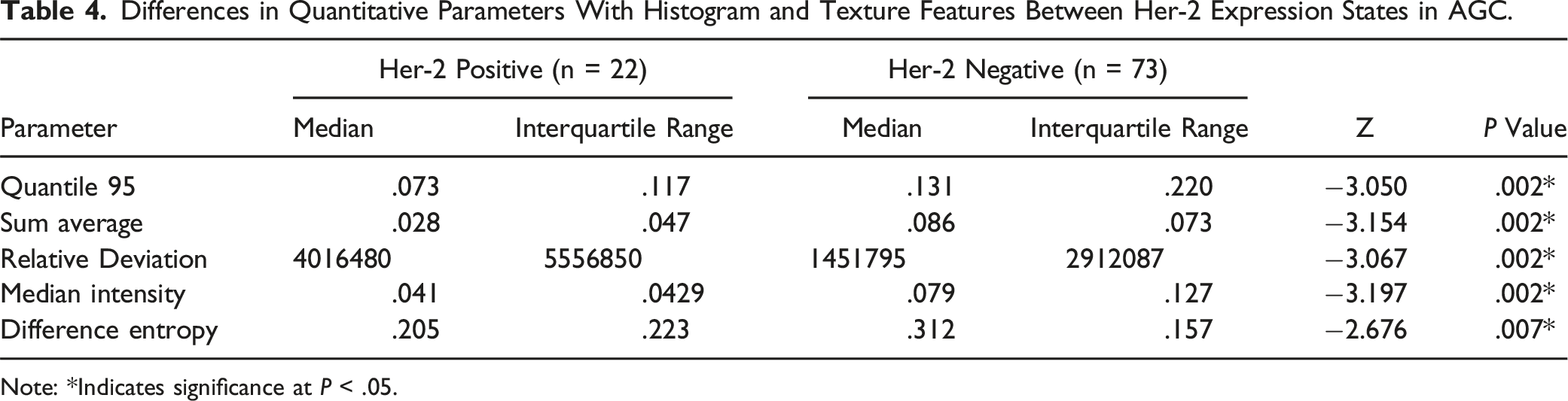
Figure 3 and Table 4 show that out of 228 screened features, only 5 features were retained, namely Quantile 95 of Ve, Sum Average, Relative Deviation, Median Intensity, and Difference Entropy. These parameters were significantly higher in the Her-2 low-expression group than the Her-2 high-expression group. The radiomic parameters of Ktrans and Kep did not show statistically significant differences among the groups. In the radiomic parameters of DCE-MRI analysis in AGC, we conducted ROC curve analysis and calculated the AUC values of significantly different parameters to determine their predictive accuracy. All AUC values were close to .7 (Figure S2 and Table 5). Illustration of the radiomic feature selection process and parameter convergence. Differences in Quantitative Parameters With Histogram and Texture Features Between Her-2 Expression States in AGC. Note: *Indicates significance at ROC Analysis of DCE-MRI Radiomics Features for Differentiating Her-2 Expression in AGC of Poorly Differentiated. Note: AUC: area under curve. *Indicates significance at 
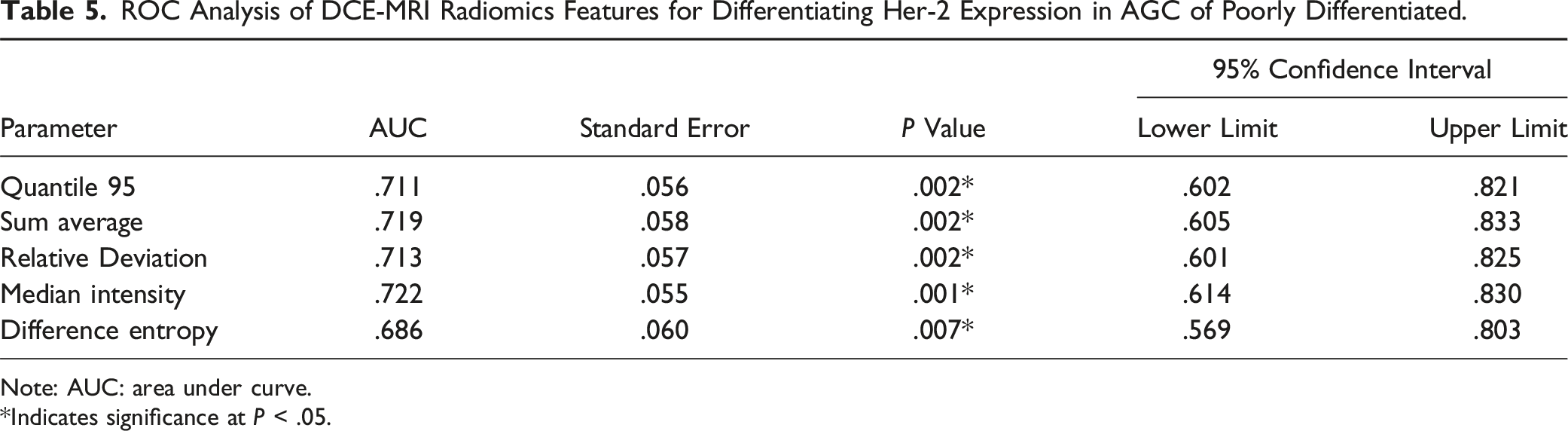
We conducted ROC curve analysis on the radiomics parameters in DCE-MRI for AGC. The purpose was to determine the predictive accuracy of these parameters and calculate the AUC values for all DCE-MRI radiomics parameters with significant differences. These Area Under the Curve (AUC) values are approximately .7 (Table 5).
Discussion
In this research, our meta-analysis revealed a significant association between overexpression of HER2/ERBB2 in gastric cancer with the malignancy degree and prognosis of tumors. This finding not only confirms the critical role of HER2/ERBB2 in gastric cancer but also provides new insights and directions for future therapeutic strategies. Additionally, we successfully detected HER2 expression in 95 patients using immunohistochemistry. The results indicated a notable correlation between HER2 overexpression and the malignant degree and prognosis of gastric cancer, further emphasizing its importance in gastric cancer. We observed a significant association between high HER2 expression in gastric cancer patients and specific radiomic parameters, potentially linked to the activation of the HER2/ERBB2 signaling pathway and alterations in the tumor microenvironment. This study marks the first utilization of radiomic analysis based on 3 pharmacokinetic parameter maps obtained from DCE-MRI in AGC patients, investigating its capability to predict Her-2 status in AGC patients. The outcomes suggest that this radiomic approach may differentiate the Her-2 status of AGC patients.
The overexpression of HER2/ERBB2 is closely associated with the malignancy and prognosis of gastric cancer patients. High levels of HER2 expression typically indicate increased tumor aggressiveness and are often correlated with poorer survival rates. 20 It makes HER2 a crucial biomarker for gastric cancer, guiding clinical treatment strategies. The frequency of HER2 overexpression varies significantly across different types of cancer. For example, approximately 20%-30% of breast cancer cases exhibit HER2 overexpression, while in gastric cancer, this proportion is typically around 10%-20%. 38 This difference underscores the varying clinical significance and treatment strategies associated with HER2 in different tumor types. HER2-positive tumors in gastric cancer often display unique histological characteristics, such as higher lymphocyte infiltration and specific molecular genetic features. 39 It suggests that HER2’s role may be influenced by the tumor microenvironment and other genetic backgrounds in different types of cancer.
Pre-processing DCE-MRI perfusion parameters, specifically vascular endothelial (VE) parameters, can be valuable imaging biomarkers in assessing Her-2 expression status for patients with AGC. This study elucidates the significant correlation between HER2/ERBB2 overexpression and the radiomic parameters of DCE-MRI in gastric cancer, specifically focusing on the alterations in perfusion parameters (Figure 4). This association further clarifies the essential involvement of HER2/ERBB2 in the development of gastric cancer. The conclusions drawn from Omni Kinematics software and immunohistochemistry techniques are statistically robust and reliable. This study introduces a novel diagnostic tool for use in clinical practice. It aids in assessing patients’ HER2 status in advance, facilitating the implementation of personalized and effective treatment strategies. This study aims to investigate the overexpression of HER2/ERBB2 in gastric cancer through a meta-analysis, and to explore its relationship with perfusion parameters using DCE-MRI. The goal is to provide a scientific basis for the early diagnosis and treatment of gastric cancer. Schematic representation of the relationship between HER2/ERBB2 expression and DCE-MRI radiomic parameters in gastric cancer. The significant relationship between HER2/ERBB2 and DCE-MRI perfusion parameters in gastric cancer, particularly highlighting VE, serves as an effective imaging biomarker for evaluating HER2 expression status in patients with AGC.
The study emphasizes the critical role of HER2/ERBB2 in gastric cancer, suggesting that future treatment strategies should be more individualized. By integrating radiomic parameters from DCE-MRI, a more comprehensive assessment of tumor characteristics can be made, allowing for the development of personalized treatment plans. The overexpression of HER2 is closely associated with the effectiveness of molecular-targeted therapies. 36 Future studies could consider combining HER2 status with other molecular markers to explore the potential of combination therapies, which may improve treatment outcomes and reduce the risk of resistance.
Multimodal evaluation through non-invasive imaging techniques, combined with traditional immunohistochemistry, will help identify high-risk patients at early stages and provide real-time monitoring of tumor response. This integrated diagnostic tool will advance precision medicine. Based on HER2/ERBB2 status data and radiomic parameters, new predictive models could be constructed to help clinicians predict patient survival and disease progression. These models will provide valuable insights for clinical decision-making, especially in selecting treatment plans. Follow-up using imaging techniques such as DCE-MRI for dynamic monitoring of HER2 expression and related radiomic changes will aid in timely treatment adjustments, ultimately improving the quality of life for patients.
Detection methods for HER2 are crucial in assessing HER2 status in cancers, particularly breast and gastric cancer. Each detection method has its strengths and limitations. Specifically, IHC is relatively simple, more cost-effective than other methods, and provides clear visualization of protein expression levels, but the results may vary depending on the pathologist’s expertise. 40 FISH offers high specificity for detecting gene amplification and provides precise quantitative data on HER2 gene copy number per cell, but the process is more time-consuming than IHC and requires fresh or properly preserved tissue samples. 41 Silver in situ hybridization (SISH) produces more stable signals, which are easier to observe with standard optical microscopy, but it requires specific skills and experience to interpret the results accurately. 42 Chromogenic in situ hybridization (CISH) uses conventional microscopy for visualization and is easily integrated into routine pathology workflows, but it has lower sensitivity, potentially leading to false-negative results. 43 Dual-color dual-hapten in situ hybridization (DDISH) allows simultaneous assessment of HER2 gene amplification and chromosome 17 polysomy in the same sample, providing comprehensive information. However, it requires higher technical expertise, greater laboratory resources, and advanced equipment for accurate interpretation. 44
There are still some limitations in this study. Despite utilizing an extensive literature search and applying strict selection criteria, it is important to acknowledge that the results of a meta-analysis can still be susceptible to potential biases and the quality of the studies included in the analysis. Furthermore, our study consisted of only 95 patients diagnosed with gastric cancer, representing a relatively limited sample size that could potentially undermine the robustness and generalizability of the statistical findings. In analyzing dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI), radiomic features may present a limitation as they uncover a plethora of quantitative information related to lesions that are imperceptible to the naked eye or not amenable to routine measurements. Through non-invasive quantitative radiomic analysis, early assessment of the HER2 status can be crucial for guiding patient treatment strategies. While the findings of this study are valuable, limitations such as sample size and methodological choices exist. Future endeavors should encompass larger-scale investigations and delve deeper into mechanistic explorations to further elucidate the role of HER2 in gastric cancer, consequently enhancing therapeutic interventions and prognostic outcomes for patients. Despite the utilization of advanced Omni-Kinetics software, a more profound investigation is still warranted to interpret radiomic parameters.
Moreover, the accuracy of immunohistochemical results may be influenced by sampling bias in the submitted specimens. 45 Not all patients are eligible for immunohistochemical examination to assess the HER2 status in routine clinical diagnostics and therapy.46,47 Certain patients face challenges acquiring tumor tissue samples, while others encounter constraints due to limited financial resources.
Despite these limitations, this study offers a fresh perspective on the early diagnosis and treatment of gastric cancer. Particularly, the association of HER2/ERBB2 overexpression with gastric cancer provides clinicians with more precise treatment targets. Integrating radiomic parameters from DCE-MRI enables a more comprehensive evaluation of patients’ conditions and prognoses, facilitating personalized treatment recommendations. Furthermore, immunohistochemical testing for HER2 expression allows physicians to accurately classify the molecular subtypes of gastric cancer, providing robust support for precision medicine. Advancements in technology and further research endeavors are expected to lead to a deeper understanding of the specific mechanisms of action of HER2/ERBB2 in gastric cancer. Enlarging sample size in future studies is expected to enhance the generalizability and accuracy of research findings. Coupling modern molecular biology, genomics, and radiomics holds promise for introducing more innovations and breakthroughs in treating gastric cancer.
Conclusions
This study demonstrates a significant association between the overexpression of HER2/ERBB2 and the degree of tumor differentiation, as well as DCE-MRI perfusion parameters in gastric cancer patients. This finding provides a theoretical basis for further investigating the role and mechanisms of HER2 in gastric cancer development. Future research can delve deeper into the mechanism of HER2 in gastric cancer progression and integrate multimodal imaging technologies to develop more precise diagnostic and treatment strategies.
Supplemental Material
Supplemental Material - HER2 in Gastric Cancer: A Comprehensive Analysis Combining Meta-Analysis and DCE-MRI Radiomics
Supplemental Material for HER2 in Gastric Cancer: A Comprehensive Analysis Combining Meta-Analysis and DCE-MRI Radiomics by Liyijing Shen, Yaoqing Li, Huizhen Huang, Zengxin Lu and Bo Chen in Journal of Cancer Control.
Footnotes
Author Contributions
Liyijing Shen and Bo Chen designed the study and contributed to the methodology. Yaoqing Li and Huizhen Huang conducted the literature search and data collection for the meta-analysis. Zengxin Lu and Liyijing Shen performed the DCE-MRI scans and analyzed the imaging data using Omni kinetic software. Bo Chen supervised the overall research project and provided critical revisions to the manuscript. All authors contributed to the writing and revision of the manuscript and approved the final version for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Key Laboratory of Functional Molecular Imaging of tumour and Interventional Diagnosis and Treatment of Shaoxing City (No. 2020ZDSYSO1, Shaoxing People’s Hospital, Shaoxing, Zhejiang, China); General of Health Science and Technology Plan of Zhejiang Province (2021KY1150, 2023KY1234 and 2023KY1239).
Ethical Statement
Data Availability Statement
The original contributions presented in the study are included in the article/supplementary materials, further inquiries can be directed to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
