Abstract
Objective
To retrospectively analyze the effect of hyperthermic intraperitoneal chemotherapy (HIPEC) on the progression free survival (PFS) of advanced gastric cancer (GC) and colorectal cancer (CRC).
Method
We retrospectively collected all the HIPEC data of GC and CRC in the Chongqing University Cancer Hospital from August 2018 to April 2023. Data were extracted from inpatient records and outpatient examination records. The IBM SPSS statistics 23.0 software was used to analyze the data. We mainly compared the PFS of HIPEC cases with that of non-HIPEC cases (both from our center and from the literature). PFS was analyzed with the Kaplan-Meier method. Log Rank (Mantel Cox), Breslow (Generalized Wilcoxon), and Tarone-Ware were used for univariate analyses.
Result
A total of 342 HIPEC cases were analyzed in this study. Stage IV GC and CRC accounted for 48.5% of the total number of cases. Abdominal pain and distension (47.4%) were the most common side effects from HIPEC. Serious complications were rare (1.8%, including bleeding, perforation, obstruction, and death). The PFS and disease-free survival (DFS) of abdominal malignancy treated with HIPEC were significantly associated with the TNM stage, but not HIPEC numbers nor HIPEC drugs. In stage IV HIPEC cases, adding adjuvant chemotherapy after HIPEC resulted in better PFS. In addition, the association between peritoneal carcinomatosis index (PCI) and PFS of stage IV HIPEC cases was close to significant. Compared with the 33 stage IV (with peritoneal metastases) GC cases without HIPEC in our center from the last 15 years, the PFS of the 56 stage Ⅳ GC cases with HIPEC was not improved significantly (median PFS: 6 ± 2.92 months vs 7 ± 1.63 months for with vs without HIPEC in stage IV GC, respectively; P ≥ 0.05). Compared with the 58 stage IV (with peritoneal metastases) CRC cases without HIPEC in our center from the last 15 years, the PFS of the 86 stage IV CRC cases with HIPEC was not improved significantly either (median PFS: 7 ± 1.68 months vs 7 ± 0.62 months for with vs without HIPEC in stage IV CRC, respectively; P ≥ 0.05). When comparing our HIPEC data with the non-HIPEC data reported by other scholars for the PFS of advanced GC and CRC, the negative results were similar.
Conclusion
The PFS/DFS of HIPEC cases was associated with the TNM stage, but not with the HIPEC numbers or HIPEC drugs. PCI may be related to the PFS of stage IV HIPEC cases. Adding chemotherapy or targeted therapy after HIPEC may improve the PFS of stage IV cases. HIPEC did not significantly improve the PFS of stage IV GC or CRC cases in our center.
Background
Hyperthermic intraperitoneal chemotherapy (HIPEC) is a newly emerging adjuvant therapy for advanced abdominal malignancy. It has unique efficacy in the treatment of peritoneal implantation metastasis and malignant ascites associated with gastric cancer (GC), 1 colorectal cancer (CRC), 2 ovarian cancer, 3 peritoneal pseudomyxoma, 4 and so on. HIPEC refers to the use of perfusion therapy apparatus to inject infusion liquid containing drugs into peritoneal cavity at a precise constant temperature (usually 43°C), and the infusion liquid is eventually drawn out of the body after circulating in the abdominal cavity, thus forming a thermal cycle treatment of drugs in the abdominal cavity.
HIPEC is characterized by precision and standardization, including precise temperature control (the temperature of abdominal heat perfusion is 43 ± 0.1°C), precise flow rate control (perfusion flow rate 300-600 mL/min), precise positioning (the perfusion tube is placed in the abdominal cavity to the subdiaphragm and pelvic floor, so the thermoperfusion fluid fills the entire abdominal cavity), and precise removal (free cancer cells, subclinical lesions, and small cancer nodules can be removed).
The anti-cancer principles of HIPEC maybe include: (1) Thermal effect inhibits angiogenesis in tumor tissues, resulting in tumor degeneration and necrosis. (2). At the cellular level, HIPEC can destroy the stability of tumor cells, activate lysosomes, destroy cytoplasm and nucleus, and induce tumor cell apoptosis. (3). HIPEC denature cancer cell membrane proteins at the molecular level, interfering with DNA, RNA and protein synthesis. (4). Hyperthermia and chemotherapy drugs can play a synergistic anti-tumor effect, which is most obvious at 43°C, and the thermal effect can increase the permeability of chemotherapy drugs. (5). Continuous perfusion cycle can mechanically scour free cancer cells in the abdominal cavity and small metastases in the peritoneum.
Chongqing University Cancer Hospital has been carrying out HIPEC for abdominal malignant tumors for more than 5 years. We want to know the clinical effect of HIPEC for advanced gastric cancer (GC) and colorectal cancer (CRC). So we retrospectively analyze the HIPEC data from our center in recent 5 years (from August 2018 to April 2023). We hope this study may provide reference for clinicians.
Method
The reporting of this retrospective study conforms to the STROBE guidelines. 5 The study was approved by the ethics committee of Chongqing University Cancer Hospital (CZLS2024170-A) and was conducted in accordance with the Declaration of Helsinki. All cases signed informed consent for the HIPEC treatment.
Data Collection
We retrospectively and consecutively collected all the HIPEC data for abdominal malignancy in the Chongqing University Cancer Hospital from August 2018 to April 2023. And the non-HIPEC data are completely random choices from electronic medical record system in our hospital from June 2009 to May 2020 (inclusion criterion is the medical records and follow-up data adequate enough). We have de-identified patient details in this study. Clinical data are extracted from inpatient records and outpatient examination records (the deadline for prognostic data extraction is on August 2023). The literature chosen for PFS comparison in this study are based on some criteria: (1). About PFS of advanced gastric or colorectal cancer, (2). Without HIPEC treatment, (3). With clear and reliable data, (4). From high quality publications.
Therapy
Cytoreductive surgery (CRS, with effort, but may be not strict or standard in this retrospective study) and HIPEC is conducted for all the stage IV (93.4% only associated with abdominal and peritoneal metastases) cases. All the stage IV HIPEC cases in this study have no other treatment prior to CRS and HIPEC. Radical tumor resection and HIPEC is conducted for all the stage II, III cases. Written informed consent of HIPEC from all the cases are obtained for potential therapeutic benefits and side effects. The HIPEC tubes are implanted in abdomen by laparoscopic surgery. The temperature of HIPEC is 43°C, and every HIPEC treatment lasts 60 minutes. The therapeutic fluid is a mixture of normal saline and drugs (refer to the dose of systemic chemotherapy and drug instruction, cisplatin is usually administered with 50-80 mg each time), and the therapeutic fluid capacity is 3000-5000 mL each time. The flow rate is usually 300-600 mL/min. According to “Expert Consensus on Clinical Application of hyperthermic intraperitoneal chemotherapy in China (2019 edition)”, 6 preventive HIPEC is usually 1-2 times; therapeutic HIPEC is usually 1-3 times, but can be increased to 3-5 times.
Staging and PCI
The stage is determined according to the eighth edition of the AJCC/UICC TNM staging system. All the staging II, III cases are pathological stage (surgical specimen). But cancer of stage IV is pathological (for metastatic lesion) or clinical stage (lacking pathological evidence, but imageological examination supporting). The peritoneal carcinomatosis index (PCI) is performed according to “Expert Consensus on Clinical Application of intraperitoneal hyperthermic chemotherapy in China (2019 edition)”. 6 And the PCI is evaluated based on the surgical exploration record and abdominal CT in this study.
Prognosis
Due to the insufficient follow-up time, this study only focuses on the efficacy of HIPEC for DFS (stage II, III) and PFS (stage IV) of GC and CRC. Tumor recurrence (stage II, III) or tumor progression (stage IV) are based on imageological examination (refer to RECIST Standard 1.1), combined with tumor markers and pathology. PFS is defined as the time from start of any treatment to disease progression (recurrence/progression of the abdominal and peritoneal tumors is the most important basis for evaluating patients’ PFS in this retrospective study) or death and DFS is defined as the time from start of any treatment to tumor recurrence, metastasis or death in this study.
Statistic Analysis
We use IBM SPSS statistics 23 to analyze data. We mainly compare the PFS of HIPEC cases with that of non-HIPEC cases (from our center and literature). PFS is analyzed with the Kaplan-Meier method and chi-square test. Log Rank (Mantel Cox), Breslow (Generalized Wilcoxon), Tarone-ware are used for univariate analyse. The PFS is expressed as estimated median ± standard error. If necessary, heterogeneity is analyzed between different clinicopathological feature groups. P < 0.05 is defined as a significant difference.
The research route is drawn as follows.
Results
1. Summary of the HIPEC patients
Summary of the HIPEC Cases.

dMMR: mismatch repair deficient.
pMMR: mismatch repair proficient.
Side Effects and Complications of HIPEC

Differentiation poor includes: poorly differentiated carcinoma, adenocarcinoma grade III, sig-ring cell carcinoma, mucinous adenocarcinoma, etc. 2. Association between the DFS/PFS and clinicopathological features of the HIPEC cases
We use Kaplan-Meier method (IBM SPSS statistics 23) to conduct a preliminary analysis of the HIPEC data. The Log Rank (Mantel Cox), Breslow (Generalized Wilcoxon), Tarone-ware are used for univariate survival analyse. The estimated median DFS/PFS of different clinicopathological features of the HIPEC cases are presented in Table 3. We find the HIPEC cases of different TNM stages have significantly different DFS/PFS (DFS is for stage II-III cases, PFS is for stage IV cases. To make results more intuitive, we draw DFS/PFS on the same chart and figure, Figure 1. And the age, tumor differentiation, numbers of HIPEC, PCI also seem to correlate with the DFS/PFS. But due to the proportion differences of stage IV cases in different groups of these factors, the credibility of these correlations is uncertain (For example, 37.8 % of the patients with 1-2 HIPEC treatments are stage IV cancers, while 61.7 % of the patients with 3-4 HIPEC treatments are stage IV cancers). 3. The PFS of stage IV cases with HIPEC Preliminary Analysis of DFS/PFS in Different Clinicopathologic Feature of the HIPEC Cases. Kaplan-Meier survival analysis: Log Rank (Mantel Cox), Breslow(Generalized Wilcoxon), Tarone-ware. The DFS/PFS of GC and CRC in different stages. 2: stage II, 3: stage III, 4: stage IV. Log Rank (Mantel Cox), Breslow (Generalized Wilcoxon) and Tarone-ware, P < 0.05. Due to the small number of stage II GC cases, the survival curve is not drawn.


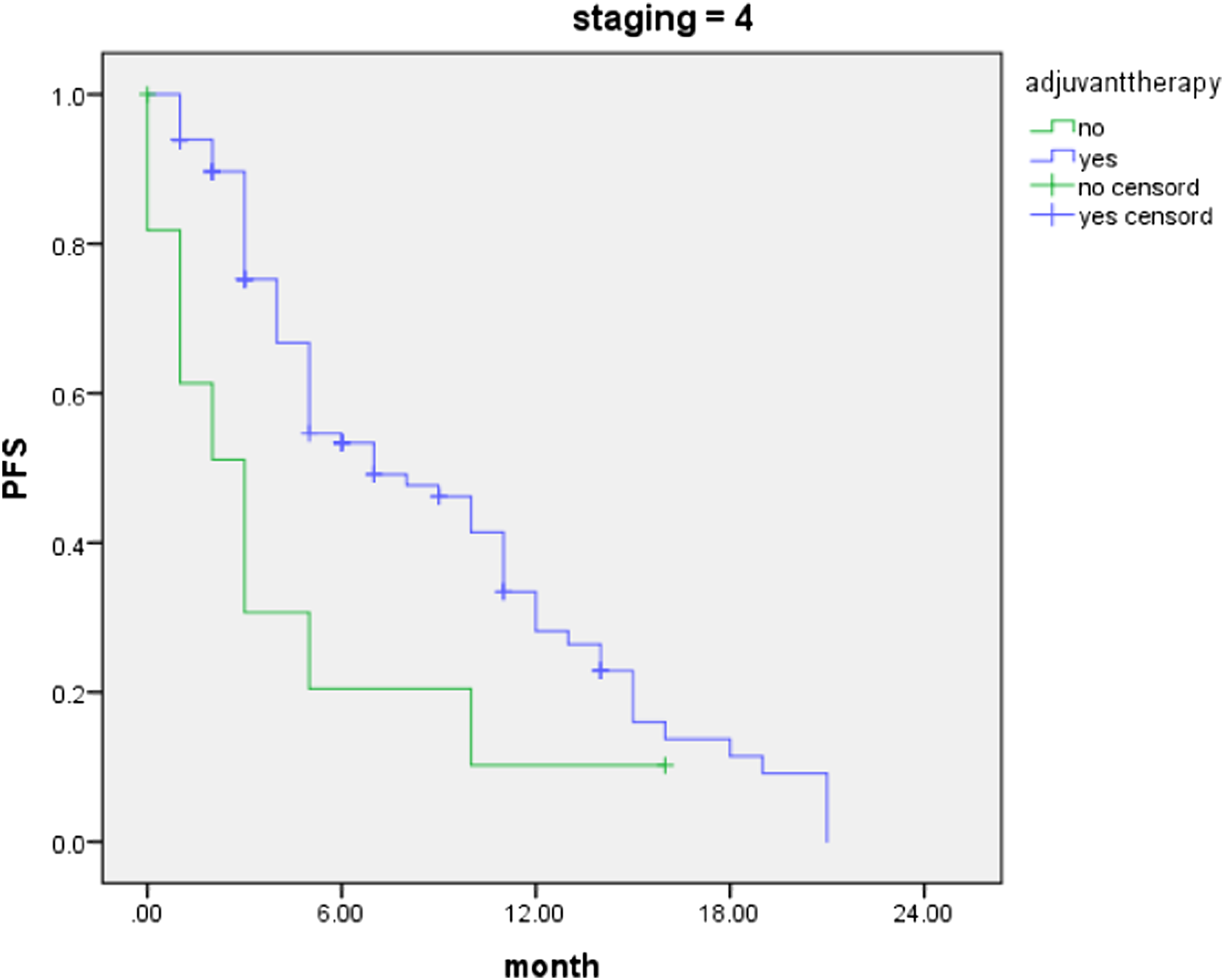
The PFS of 166 stage IV (93.4% only associated with abdominal and peritoneal metastases) HIPEC cases in different clinicopathological features are shown in Table 4. Recurrence/progression of the abdominal and peritoneal tumors is the most important basis for evaluating patients’ PFS in this retrospective study. In these stage IV cases, PFS is not significantly associated with tumor site, tumor differentiation, HIPEC numbers, or HIPEC drugs. But adding adjuvant chemotherapy after HIPEC results in better PFS, Figure 2. And the association between PCI and PFS is close to significant (the median PCI for these 166 patients is 10). 4. Analyze the PFS of stage IV GC between with and without HIPEC Statistical Analysis for PFS of Stage Ⅳ HIPEC Cases in Different Clinicopathologic Feature. Kaplan-Meier survival analysis: Log Rank (Mantel Cox), Breslow(Generalized Wilcoxon), Tarone-ware. The PFS of stage IV patients with or without adjuvanttherapy after HIPEC 4: stage IV, yes: with adjuvanttherapy, no: without adjuvanttherapy. Log Rank (Mantel Cox), Breslow (Generalized Wilcoxon) and Tarone-ware, P < 0.05.

First, we compare our findings with those of other studies. When we extract the data for comparative analysis (chi-square test), we don’t find the significant differences in PFS of advanced GC between HIPEC group (our center) and Non-HIPEC groups (Yamada’s
7
or Park’s
8
study), Table 5. Then, we continue to compare the PFS of stage Ⅳ GC between with and without HIPEC in our center. The 33 non-HIPEC cases of stage IV GC in our center (randomly selected from June 2009 to May 2020, but combined with abdominal peritoneal metastasis and with complete follow-up data) receive only chemotherapy, targeted therapy, etc. We find the stage IV GC cases with HIPEC have a median PFS of 6 ± 2.92 months, compared with a median PFS of 7 ± 1.63 months for those without HIPEC, Table 6 (There is no significant heterogeneity in age, sex, and tumor differentiation between the 2 groups). HIPEC do not significantly improve the PFS of stage IV GC cases in our center, Figure 3. 5. Compare the PFS of advanced CRC between with and without HIPEC Compare the PFS of Advanced GC With Other Studies. chi-square test. The PFS of Stage Ⅳ GC Between With and Without HIPEC in Our Center. Kaplan-Meier survival analysis: Log Rank (Mantel Cox), Breslow(Generalized Wilcoxon), Tarone-ware. The PFS of stage IV GC with or without HIPEC in our center 4: stage IV, YES: with HIPEC, NO: without HIPEC. Log Rank (Mantel Cox), Breslow (Generalized Wilcoxon) and Tarone-ware, P ≥ 0.05.



Compare PFS of Stage IV CRC With Other Studies.

The PFS of Stage Ⅳ CRC Between With and Without HIPEC in Our Center.

Kaplan-Meier survival analysis: Log Rank (Mantel Cox), Breslow(Generalized Wilcoxon), Tarone-ware.

The PFS of stage IV CRC with or without HIPEC in our center 4: stage IV, YES: with HIPEC, NO: without HIPEC. Log Rank (Mantel Cox), Breslow (Generalized Wilcoxon) and Tarone-ware, P ≥ 0.05.
Discussion
In recent 5 years, ChongQing University cancer hospital Gastrointestinal cancer center has carried out a considerable number of HIPEC treatments and accumulated clinical experiences. HIPEC is always the focus and forefront of advanced GC and CRC treatment worldwide.11,12 HIPEC is currently used as a therapy for late-stage GC and CRC, 13 although some trials for mid-stage GC and CRC have been carried out. 14 Due to the insufficient follow-up time, this study only focus on the efficacy of HIPEC for late-stage GC and CRC. Our clinical research experience is not mature, but we carefully analyze the original data in detail, and ensure the authenticity of the results and the objectivity of the analysis. This study is expected to provide a reference for the clinical application of HIPEC for GC and CRC.
In general, our HIPEC cases include more young patients, CRC patients, and late-stage patients. The ratio of pMMR to dMMR is 7:1. Platinum-based drugs are the most common choice for HIPEC in our center. Most patients received palliative chemotherapy, adjuvant chemotherapy, or targeted therapy after HIPEC. HIPEC is a relatively safe technique, and serious complications are not common (1.8%, including blooding, perforation, intestinal obstruction and death). We think some days’ interval (median is 4 days in this study) between operation and HIPEC may be the reason of fewer complication.
In terms of the association between PFS/DFS and clinicopathological features of HIPEC cases, we find there are significant differences in PFS/DFS of HIPEC cases among different stages. This result indicates that the most important factor determining the prognosis of GC and CRC is still the stage (no matter with or without HIPEC), but not other clinicopathologic features (in fact, the association between tumor differentiation and prognosis is also worthy of attention in this study). What’s more, in stage Ⅳ patients treated with HIPEC, the PFS is may associated with adjuvant therapy (chemotherapy-based therapy) after HIPEC and PCI. Adding chemotherapy after HIPEC may significantly improves the PFS. So we don’t think HIPEC may replace the role of chemotherapy. And a higher PCI may means a worse PFS.
In terms of the treatment effect, HIPEC dose not significantly prolong PFS in stage Ⅳ GC patients, no matter compared with the data without HIPEC from other studies or the data without HIPEC from our own. The similar result is found in stage Ⅳ CRC (median PFS is about 7 months). These results indicate HIPEC significantly improving the PFS of advanced GC and CRC is unrealized in our center.
In theory, HIPEC is effective in treating advanced GC and CRC. 15 Unfortunately, we don’t find positive results. There could be several reasons to explain. And we also consider these reasons as limitations of this study. Firstly, in retrospective study, the heterogeneity between groups cannot be ignored. Secondly, the follow-up time of our data is insufficient. Thirdly, there is a lack of clinical experience in the selection of drugs and treatment time for HIPEC. Fourthly, maybe the CRS is not strictly performed for some cases before HIPEC in our centre. Fifthly, the cases included in this study may be insufficient for some special statistics. These factors may negatively affect the efficacy of HIPEC.
We review some literature on HIPEC for GC. For example, 2 separate single-institution phase II single-arm studies evaluating CRS with HIPEC using cisplatin with mitomycin C in 41 GC patients confirmed peritoneal metastasis are analyzed by Green et al. 16 All the 41 patients have synchronous peritoneal metastasis and received systemic chemotherapy as front-line therapy. Green finds the median OS was 24.9 months from diagnosis and 14.4 months from surgery-HIPEC, and the median RFS is 7.4 months. And Green also identifies the number of pathologically positive lymph nodes as an independent predictor of postoperative OS. These results are similar to those of our study, in which stage Ⅳ GC patients treated with HIPEC have the PFS of only about half a year. And the prognosis of these patients is related to pathological features.
Merboth 17 investigates the complications associated with CRS + HIPEC and OS, DFS in advanced GC (15 patients). Twenty-five percentage of these patients suffer from serious postoperative complications. And surgical and medical complications, rate of reoperation, and mortality does not differ from the control group. Also, the recurrence pattern, median survival, and 1- and 2-year survival rates show no differences between HIPEC group and control group. HIPEC dose not improve the prognosis of advanced GC, which is consistent with the results of our study. But there are not so many serious complications of HIPEC in our study. The reason may be HIPEC is not performed immediately after surgery, but performed several days after surgery in our study (with a median of 4 days).
We also review some literature on HIPEC for CRC. Arjona-Sánchez 18 assess the efficacy and safety of intraoperative HIPEC in patients with locally advanced colon cancer. Enrolled patients are locally advanced primary colon cancer diagnosed preoperatively (cT4N02M0). Patients are randomly assigned 1:1 to receive cytoreduction plus HIPEC with mitomycin C (investigational group) or cytoreduction alone (comparator group), both followed by systemic adjuvant chemotherapy. A total of 184 patients are recruited and randomized (investigational group, n = 89; comparator group, n = 95). The 3-year locoregional control rate was higher in the investigational group (97.6%) than in the comparator group (87.6%). But no differences were observed in DFS (investigational, 81.2%; comparator, 78.0%; log-rank P = 0.22) or overall survival (investigational, 91.7%; comparator, 92.9%; log-rank P = 0.68). The definitive subgroup with pT4 disease showed a pronounced benefit in 3-year locoregional control rate after investigational treatment (investigational: 98.3%; comparator: 82.1%; log-rank P = 0.003). No differences in morbidity or toxic effects between groups are observed. This result also confirms that HIPEC has a limited effect on the prognosis of advanced colorectal cancer, although HIPEC is a relatively safe treatment.
And Ba 19 investigates the clinical efficacy and safety of different timings of CRS with HIPEC for malignant ascites caused by peritoneal carcinomatosis from CRC. The patients are randomized to CRS + HIPEC group (n = 14), and HIPEC + delayed CRS group (n = 14). Malignant ascites in all patients show complete remission; the total effective rate is 100%. Overall survival is 14.5 (95%CI: 7-19 months) and 14.3 months (95%CI: 4-21 months) for 2 groups, respectively. The timing of CRS and HIPEC does not prolong the survival of patients with peritoneal carcinomatosis from CRC. These results indicate HIPEC is effective in controlling malignant ascites caused by CRC in short term, but has limited effect on the prognosis of advanced CRC. This finding has something in common with our results.
And Quénet et al 20 research the CRS plus HIPEC vs CRS alone for colorectal peritoneal metastases. They find the absence of an OS benefit after adding HIPEC to CRS and more frequent postoperative late complications with the combination. Their data suggest that CRS alone should be the cornerstone of therapeutic strategies with curative intent for colorectal peritoneal metastases. This result is also negative and disappointed.
So we think our findings may be objective and unsurprising.
Conclusion
In conclusion, the prognosis of abdominal malignancy treated with HIPEC is closely related to the TNM stages, but neither the numbers of HIPEC nor drugs of HIPEC in our center. Adding chemotherapy or targeted therapy after HIPEC may improve PFS of stage Ⅳ cancers. HIPEC therapy may be not a substitute for intravenous chemotherapy. The association between PCI and PFS of late stage HIPEC cases deserves attention. HIPEC does not significantly improve the PFS of advanced GC and CRC in this retrospective study. Future studies of HIPEC should focus on the control of local symptoms and local lesions. Clinical studies of drug selection for HIPEC have to go on.
Footnotes
Acknowledgments
Authors agree on the order of attribution. FAN Jing and SUN Hao are the co-corresponding authors of this article. We thank LI Xiao-dong for providing detailed data records. We thank Professor WANG Zi-wei for his guidance on the analysis of this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is Supported by Scientific and Technological Research Program of Chongqing Municipal Education Commission (Grant No. KJQN202300121), Chongqing Natural Science Foundation General Project (cstc2021jcyj-msxmX0102) and Postgraduate education teaching reform research project of Chongqing University (cquyjg240306).
Ethical Statement
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the author’s email
