Abstract
Objective
The clinical characteristics and prognosis of primary epithelial-myoepithelial carcinoma of salivary gland (EMC-SG) have not been defined well due to its rarity. The purpose of this study is to assess the proportion of EMC-SG among salivary gland cancers, describe the clinicopathological features and prognosis of this disease, further analyze the factors associated with EMC-SG survival, and establish individual survival-predicting models.
Methods
Data on patients diagnosed with salivary gland malignancy between 2000 and 2020 were collected from the Surveillance, Epidemiology, and End Results database. The Kaplan-Meier method and log-rank test were employed to estimate survival of EMC-SG patients. Univariable and multivariable Cox proportional hazards models were developed to determine the EMC-SG survival-associated factors. Furthermore, EMC-SG nomograms were constructed.
Results
A total of 15 212 patients with salivary gland malignancy were identified. Of these, 310 cases were diagnosed with EMC-SG, representing a prevalence of 2.03% (95%CI 1.82%-2.28%). The overall survival (OS) rates for all 310 EMC-SG patients at 2-year, 5-year, and 10-year were 92.43%, 84.85%, and 73.39%, respectively. Age, primary site, and T stage were independent prognostic factors for OS, while pathological grade and the use of surgery were independent prognostic factors for cancer-specific survival (CSS). The concordance index (C-index) for the OS- and CSS-specific nomograms was 0.72 (95%CI 0.64-0.80) and 0.77 (95%CI 0.67-0.87), respectively. The calibration curve and receiver operating characteristic analysis demonstrated that the predicted values aligned well with the actual observations. Decision curve analysis indicated the superiority of the nomograms over the traditional Tumor Node Metastasis staging system.
Conclusions
This study represents the largest cohort of EMC-SG patients used to investigate the characteristics and prognosis of this disease. EMC-SG patients often have a less aggressive course and favorable prognosis. The established nomograms provide a useful tool for clinicians to predict patient outcomes, and can assist in customizing the counseling approach for this rare disease.
Keywords
Introduction
Epithelial-myoepithelial carcinoma (EMC) is a relatively rare, low-grade malignant tumor characterized by distinctive biphasic histology. 1 While EMC can manifest in various anatomical sites, rare occurrences have been documented in the maxillary sinus, trachea, larynx, hypopharynx, and even the breast.2-6 EMC is most frequently found in the salivary gland, accounting for approximately 1 to 2% of salivary gland malignancies. 7 Previous case reports or series suggested that EMC of salivary gland (EMC-SG) displays less aggressive behavior with a low probability of local recurrence and distant metastases, as well as a favorable survival. 8 For instance, a study in 2015 reported only 4.5% cases with distant metastases, with a 5-year disease-specific survival rate of 91.3%. 9 Since EMC-SG is a rare disease, the understanding of the clinical characteristics and survival of individuals diagnosed with primary EMC-SG is currently limited.
The Surveillance, Epidemiology, and End Results (SEER) is a comprehensive source of population-based information in the United States that collects data on cancer incidence, prevalence, and survival from specific geographic areas representing approximately 28% of the U.S. population.10,11 The comprehensive and valuable information has substantially promoted the analysis of rare cancer. In this study, we aimed to describe the epidemiology, clinicopathological characteristic and prognosis of primary EMC-SG patients using the largest population cohort from SEER database spanning from 2000 to 2020. The purpose of this study included: 1) determining the proportion of EMC-SG in salivary gland cancer; 2) describing the demographic and clinicopathological characteristics of these patients; 3) analyzing the factors associated with survival, further establishing individual survival-predicting model.
Methods and Materials
Data Source and Study Population
This retrospective population-based study utilized data extracted from the SEER database. The database (Incidence - SEER Research Data, 17 registries, Nov 2022 Sub, 2000-2020) was searched. To identify all cancer patients with salivary gland cancer in SEER database ranging from 2000 to 2020, the selection criterion for primary site corresponding to salivary glands was set using the International Classification of Diseases for Oncology, Third Edition (ICD-O-3)/World Health Organization 2008 classification. Further, patients with EMC-SG were selected based on the ICD-O-3 histology codes specific to epithelial-myoepithelial carcinoma (ICD-O-3: 8562/3). In addition, inclusion criteria were restricted to individuals presenting with salivary gland malignancy as their only primary malignancy, alongside documented information pertaining to survival time and vital status. The SEER database is publicly available and contains de-identified data, which exempts this study from institutional review board approval. This study complies with the RECORD guidelines. 12
Data Collection
Demographic and clinicopathological characteristics of each patient were extracted including age at diagnosis, gender, race/ethnicity, tumor stage at diagnosis, tumor grade, tumor staging, treatment options, survival time and survival status. Tumor staging was categorized according to the American Joint Committee on Cancer-Tumor, Node, Metastasis (AJCC-TNM) staging system 7th edition, 13 and SEER historic staging. The study endpoints of this retrospective study were overall survival (OS), defined as the time from diagnosis to death from any cause, as well as cancer-specific survival (CSS), defined as the time from diagnosis to death specifically from EMC-SG. The above-mentioned variables were obtained using the SEER*Stat 8.4.3 software.
Statistical Analysis
Descriptive statistics were used to summarize the baseline characteristics of the study population. The Kaplan-Meier method was employed to estimate survival of EMC-SG patients, and differences in survival of subgroup stratified by different variable were assessed using the log-rank test. Univariable and multivariable Cox proportional hazards models were developed to determine the OS-/CSS-associated factors, further Hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated. The nomograms were constructed to predict individual survival probabilities at 2, 5, and 10 years, based on the significant risk-/survival-associated factors from multivariable Cox hazards model. The performance of the nomogram was evaluated in terms of calibration (agreement between predicted and observed outcomes) and discrimination (ability to differentiate between patient outcomes) using the Harrel’s concordance index (C-index) as well as receiver operating characteristic (ROC) curve. Decision curve analysis (DCA) was carried to evaluate the clinical utility of the nomogram model by calculating the net benefit across various threshold probabilities, further ascertaining whether the implementation of the nomogram in clinical practice would yield greater benefits than harms in the context of decision-making. All statistical tests were two-sided, and a P-value of less than .05 was considered statistically significant. Analyses were performed using R software (https://www.r-project.org). C-indices, ROC curves and calibration plots were used to evaluate and validate the utility of the predictive models.
Results
Prevalence of EMC-SG in all Salivary Gland Cancer
A total of 15 212 patients with salivary gland malignancy were identified in SEER database between 2000 and 2020. Of these patients, 310 cases were diagnosed with primary EMC-SG, representing a prevalence of 2.03% (95%CI 1.82%-2.28%).
Demographic and Clinicopathological Characteristics
Demographic and Clinicopathological Characteristics of 310 EMC-SG Patients.

NOS: not otherwise specified. Other in primary site included sublingual gland (N = 1) and overlapping lesion of major salivary glands (N = 2). EMC-SG: epithelial-myoepithelial carcinoma of salivary gland; AJCC TNM: American joint committee on cancer-tumor, node, metastasis.
Survival Analysis
The Overall Survival Rates for all 310 EMC-SG Patients at 2-Year, 5-Year and 10-Year.

OS: overall survival; EMC-SG: epithelial-myoepithelial carcinoma of salivary gland; TNM: tumor, node, metastasis; NOS: not otherwise specified.
Univariate and Multivariate Cox Regression Analysis for Overall Survival Among 310 EMC-SG Patients.

EMC-SG: epithelial-myoepithelial carcinoma of salivary gland.
Univariate and Multivariate Cox Regression Analysis for CSS Among 310 EMC-SG Patients.

CSS: cancer-specific survival; EMC-SG: epithelial-myoepithelial carcinoma of salivary gland.
OS- and CSS- Predicting Nomogram Model
All independent prognostic factors for OS and CSS were further incorporated into the establishment of survival-predicting nomogram models. As showed in Figure 1, the primary site contributed to the most to OS prediction, followed by T stage and age. In the CSS-predicting model, pathological grade contributed into the most to CSS prediction, followed by the use of surgery. The C-index of OS-/CSS-nomogram model was 0.72 (95%CI 0.64-0.80), 0.77 (95%CI 0.67-0.87), respectively. The calibration plots show satisfactory performance of the prognostic nomogram’s predicted 2-, 5-, 10- year OS/CSS and actual survival (Supplemental Figure 1). Establishment of prognostic nomograms for OS (A) and CSS (B) among EMC-SG patients. OS: overall survival; CSS: cancer-specific survival; EMC-SG: epithelial-myoepithelial carcinoma of salivary gland.
Comparison of the Nomogram Model and AJCC TNM Staging
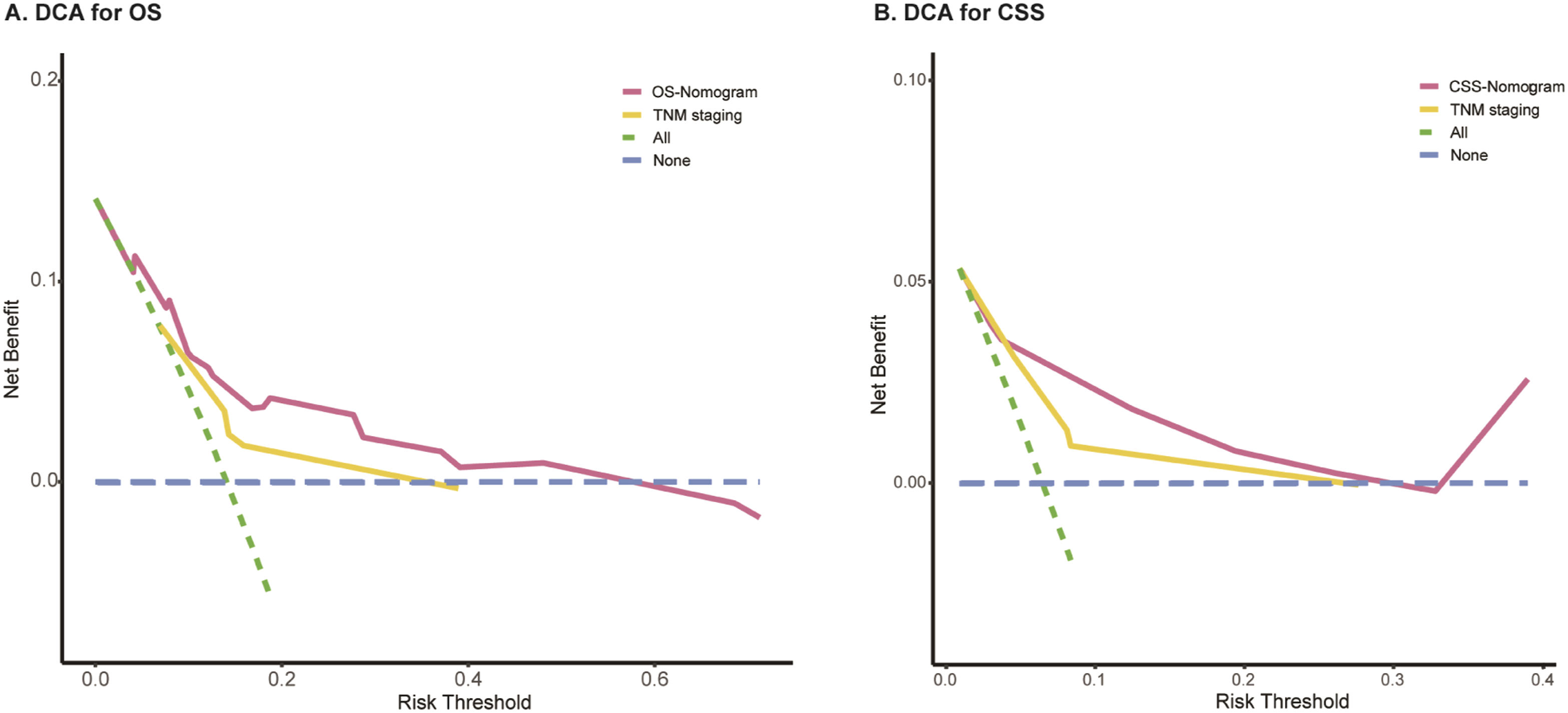
ROCs and DCAs were plotted to compare the prognostic ability of established prognostic nomogram models and AJCC TNM staging. As shown in Figure 2, when using the established nomogram model, the AUC (Area Under the Curve) of the ROC at the 2-, 5- and 10- year OS were 0.70, 0.75, 0.78, and the AUC of the ROC at 2-, 5- and 10- year CSS were 0.82, 0.80, 0.72, respectively. For TNM staging, the AUC of the ROC at the 2-, 5- and 10- year OS were 0.73, 0.67, 0.66, and the AUC of the ROC at 2-, 5- and 10- year CSS were 0.72, 0.77, 0.70, respectively. Additionally, DCAs indicated both novel models have the considerable net benefits and showed greater practicability in predicting OS/CSS when compared with the AJCC staging system (Figure 3). ROC analysis of the established nomograms (A, B) and TNM staging (C, D) for 2-, 5-, and 10- year OS and CSS among EMC-SG patients. ROC: receiver operating characteristic; TNM: tumor node metastasis; OS: overall survival; CSS: cancer-specific survival; EMC-SG: epithelial-myoepithelial carcinoma of salivary gland. DCA of the nomograms predicting OS (A) and CSS (B) compared with TNM staging. DCA: decision curve analysis; OS: overall survival; CSS: cancer-specific survival; TNM: tumor node metastasis.
Discussion
EMC-SG is a rare entity and makes up approximately 2% of salivary gland malignancies. Consistent with previous reports,8,9 the updated finding derived from the SEER database showed that EMC constitutes about 2.03% of all salivary gland malignancies. Due to its rarity, the clinicopathological features and prognosis of the disease remain unclear. This highlights the necessity of conducting population-based studies for this rare disease. The SEER database is highlighted in gathering a large cohort to offer more reliable data and insights than single-institution studies could offer. Thus, the present study is the largest cohort of primary EMC-SG patients to date, investigating the characteristics and prognosis of this disease.
This study showed that EMC typically presents in older adults, with a median age at diagnosis of 63.5 years, and exhibits a slight female predominance (1.17:1). These demographic findings are consistent with those of prior studies.9,14 Vazquez et al reported a female-to-male ratio of 1.34:1, while other reports ranged from 1.5:1 to 2.0:1.8,11 The Cox regression analysis showed a trend toward worse overall survival among those older adults (aged more than 60 years); however, this was not observed in CSS analysis. Furthermore, EMC originating from the parotid gland was noted in 86.5% of cases, aligning with the prior literature where EMC is predominantly observed in the parotid gland.15,16 Other findings of this study were the low incidence of distant metastases (1.3%) and regional lymph node invasion (5.2%), which suggest EMC-SG patients tend to present with less aggressive behavior, consistent with the existing characterizations of EMC as a less aggressive salivary gland malignancy.9,17 Meanwhile, the predominance of well-differentiated and moderately differentiated tumors in our study highlights the generally favorable pathological features of EMC-SG, which correlates with the more indolent course in these patients.
The treatment paradigm for EMC-SG patients heavily favors surgical intervention, with nearly all patients (96.13%) undergoing surgical resection. This approach is largely due to the localized nature of the disease, which allows for effective surgical margins. However, the role of adjuvant therapy, such as radiation and chemotherapy, remains ambiguous. In the present study, almost half received radiation, but no significant difference in survival was observed between patients with and without radiotherapy. In addition, 4.8% cases underwent chemotherapy. The univariate COX analysis showed that patients receiving chemotherapy has lower survival rates than those not receiving chemotherapy. After adjusting confounding factors, multivariate Cox analysis demonstrated the use of chemotherapy was not an independent prognostic factor. The lower survival of patients receiving chemotherapy may be attributed to their advanced tumor stage. However, due to the lack of the information on disease recurrence, surgical margin status, etc., further studies are required to elucidate the selective application of chemotherapy and radiotherapy to better stratify patients who might benefit from these regimens.
The survival analysis provided new insights into factors affecting prognosis. Overall, the EMC-SG patients had favorable survival, 18 with more than 78% patients could survive longer than 10 years. Even EMC-SG patients with distant metastases also have a 5-years survival rate of 50%. Age, primary site, T stage, pathological grade, and the use of surgery were identified as independent prognostic factors for OS or CSS. EMC-SG patients mainly occur in older adult, with almost 35.5% patients older than 70 years. Age was identified as an overall survival-associated factor, but not CSS. It remains unclear about the influence of primary site on survival among EMC-SG patients. Although previous reports suggested a more challenging surgical resection of the parotid gland due to its intricate relationship with facial nerve, several studies did not find a significant difference in survival by primary site. 9 Similarly, our study did not observe the significant difference between parotid gland and submandibular. Additionally, surgery and pathological grade were more likely to affecting CSS rather than OS. Because confounding factors influencing CSS are fewer compared with OS, the use of surgery and pathological grade may be more vital for predicting the extent to which this disease could affect the patient’s survival.19,20
Besides, these factors are instrumental in developing the prognostic nomograms, which showed good prediction capability, as evidenced by the C-index values for overall survival and cancer-specific survival (0.718 and 0.773, respectively). The calibration plots validated these models by demonstrating satisfactory agreement between predicted and actual survival. Such tools are beneficial in clinical practice, aiding physicians in personalizing patient discussions and management plans based on individual risk profiles. Comparing the predictive accuracy of the novel nomogram models with the traditional AJCC TNM staging system reveals a significant insight. While the TNM system has been a cornerstone in cancer prognosis, this study’s findings suggest that the established nomograms may provide superior predictive utility for OS and CSS in EMC-SG patients. This could be attributed to the nomograms’ ability to integrate multiple individual prognostic factors, as opposed to the TNM system, which primarily focuses on anatomical disease extent. However, it is crucial to recognize that, despite the promising results, external validation in other patient cohorts is necessary to confirm these findings and to ascertain the universal applicability of the nomograms.
Despite its strengths, several limitations should be mentioned. First, the retrospective nature of the SEER-database-based study may have inherent biases such as missing data and reporting inaccuracies. Additionally, the SEER database does not include all variables of interest that may be relevant to survival, such as performance status, detailed treatment regimens, treatment response, etc. Future research should focus on prospective studies to further validate these results and possibly integrate molecular and genetic data that were not available in this study. Understanding the molecular underpinnings of EMC could lead to targeted therapies, potentially improving outcomes for patients with adverse prognostic factors such as metastatic EMC-SG.
Conclusion
In conclusion, the present study offers a significant contribution to the understanding on the epidemiology, clinical features, and prognosis of EMC of the salivary gland, suggesting its less aggressive course. The development of prognostic nomograms provides useful tools for clinicians to predict patient outcomes, representing a step forward in personalized care for this rare disease. Further research and external validation are warranted to confirm these findings and to continue advancing the understanding of this rare malignancy and refining strategies for its management.
Supplemental Material
Supplemental Material - Clinical Features and Prognosis of Primary Epithelial-Myoepithelial Carcinoma of Salivary Gland: A Surveillance, Epidemiology, and End Results Database-Based Study
Supplemental Material for Clinical Features and Prognosis of Primary Epithelial-Myoepithelial Carcinoma of Salivary Gland: A Surveillance, Epidemiology, and End Results Database-Based Study by Hui Han and Li-Na Pan in Cancer Control
Footnotes
Acknowledgments
The authors would like to thank the SEER database for providing clinical information.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
The raw data downloaded from SEER database. Readers could obtain the data from the corresponding author due to reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
