Abstract
Persistent infection with high-risk human papillomavirus remains the primary factor associated with the progression of cervical squamous intraepithelial lesions and the development of cervical cancer. Nevertheless, a combination of factors, including genetic predisposition, immune response, hormonal influences, and nutritional status, contribute synergistically to the development of cervical cancer. Among the various factors involved in the pathogenesis and therapy of cervical cancer, retinoids have gained considerable attention due to their multifaceted roles in different cellular processes. This review investigates defects within the vitamin A metabolism pathway and their correlation with cervical cancer. Additionally, it integrates epidemiological and experimental findings to discuss the potential utility of retinoid-based therapies, either alone or combined with other therapies, as agents against premalignant lesions and cervical cancer.
Introduction
Infection with high-risk human papillomaviruses (HR-HPVs) is the primary etiological cause of worldwide cervical cancer (CC).1,2 The HR-HPVs 16 and 18 are the most frequent genotypes found in cervical squamous cell carcinomas (CSCCs) and adenocarcinomas, respectively, and HPV16 is the most frequent genotype in cervical lesions (∼50% of CSCCs cases). 3 Epidemiological studies suggest that persistent infection with HR-HPV is a necessary cause for developing, maintaining, and progressing cervical intraepithelial neoplasia (CIN) into CC. 4
The natural history of CC begins with HR-HPV infection at several anatomical sites of the reproductive tract (ie, the exocervical basal stratum and the transformation zone, TZ). 5 It was also proposed that columnar and squamous cervical neoplasms are derived from HR-HPVs infection of a discrete population of cuboidal epithelial cells residing at the interface of the ectocervical or TZ squamous epithelium and the endocervix. 6 Interestingly, most women (∼80%) who had contact with HR-HPVs do not develop CIN. Among those who develop low-grade CINs, only a few percent (∼20%) will progress to pre-invasive lesions, a process that may take decades after a primary infection. 7 These important epidemiological data are also supported in CC mouse models (K14HPV16, K14 × 106/E7, and K14 × 107 mice), in which the sole overexpression of HR-HPV16 oncogenes in the mice's cervical tissue usually does not develop CC. 8 Those pieces of evidence suggest that HR-HPV infection is a necessary cause but insufficient to develop CC. Other factors provide a “second hit” to maintain and progress CIN lesions into CC.9,10 Among the various factors involved in the pathogenesis and therapy of CC, retinoids have gained considerable attention due to their multifaceted roles in different cellular processes. 11
Retinoids, a class of compounds structurally similar to vitamin A, exert their effects by binding to specific nuclear receptors, namely Retinoic acid receptors (RARs) and Retinoid X receptors (RXRs). These receptors are pivotal in regulating gene transcription in various cellular processes such as growth, differentiation, and apoptosis. 12 Due to their regulatory influence on critical cellular pathways, retinoids have emerged as promising candidates for preventing and treating CC.
This review will explore the understanding of retinoids in the context of CC development and therapy. We will discuss their mechanisms of action and therapeutic applications in cervical premalignant lesions and CC.
Retinoid Biology and Biosynthetic Pathway
Retinoids comprise a group of natural or synthetic lipophilic compounds structurally related to vitamin A. In nature, retinoids consist of retinol, retinal, and retinoic acid (RA). Examples of RA include all-trans RA (also known as tretinoin or ATRA), 9-cis RA (alitretinoin), and 13-cis RA (isotretinoin). These compounds cannot be produced endogenously, so they are synthesized through plant-sourced carotenoids (pro-vitamin A) or acquired directly by taking retinoids from animal sources (ie, retinol and retinyl esters). Retinyl esters are the storage forms of vitamin A in the liver, whereas retinol is the primary precursor of active retinoids in the bloodstream. 13
Due to its lipophilic nature, most of the blood-circulated retinol binds to Retinol-binding proteins and Transthyretin, whereas free retinol flows through the target tissues’ cell membrane. The Retinol-binding protein 4 (RBP4) is the protein that delivers most of the retinol to target cells via Receptor for retinol uptake STRA6. 14 Once entering the target cells, retinol is rapidly converted to ATRA (the acidic and irreversible form of retinol that isomerizes spontaneously by nonenzymatic processes), to 13-cis RA, or 9-cis RA); ATRA, as well as 9-cis RA isomer, are the most transcriptionally active forms of retinoids. 15
Inside the target cells, transcriptionally active retinoids bind intracellular proteins such as Cellular retinoic acid-binding protein 2 (CRABP2), which delivers active vitamin A to RARs and RXRs, or bind Fatty acid-binding protein 5 (FABP5) (Figure 1). RARs and RXRs are two distinct nuclear receptor families for active vitamin A, which mediates the so-called “canonical pathway”. Each of the two canonical receptors comprises three paralogs (alpha, beta, and gamma) with different ligand-binding affinities (eg, ATRA and its two isomers are bound by all RARs, whereas RXRs only bind 9-cis RA).16,17 Vitamin A metabolism pathway. Carotenoids and retinyl esters are acquired through dietary intake. (1) Digestive enzymatic action can convert retinyl esters into retinol and free fatty acids within the small intestine. After that, the carotenes and retinol form micelles, facilitating their absorption via transporters such as SRB1, CD36, or STRA6. Once inside enterocytes, carotenes undergo cleavage by BCO1, resulting in the formation of retinal, which is subsequently converted into retinol by RDH. Retinol then binds to CRBP2; LRAT esterifies retinol to form retinyl esters. Then, retinyl esters, retinal, and carotenes are transported as chylomicrons to the liver via the lymphatic system.
30
(2) In the liver, REHs convert retinyl esters into retinol, which binds to RBP4. Excess retinyl esters may be stored in hepatic stellate cells. Retinol or retinyl esters bind to RBP4 within a complex with TTR for transport from the liver, forming retinol-RBP4-TTR. (3) This complex enters circulation until reaching target cells, where STRA6 binds to facilitate retinol entry. Retinol binds to CRBP1 within the target cell and is metabolized to retinal by RDH. Subsequently, the retinal is oxidized to ATRA by ALDH. ATRA can bind to CRABP2 or FABP5 and translocate into the nucleus to interact with nuclear receptors, including RAR/RXR and PPAR/RXR.31,32 SRB1: Scavenger receptor class B member 1, CD36: cluster of differentiation 36, STRA6: Receptor for retinol uptake STRA6, BCO1: Beta,beta-carotene 15,15′-dioxygenase, RDH: Retinol dehydrogenase, CRBP2: Cellular retinol-binding protein II, LRAT: Lecithin retinol acyltransferase, REH: Retinyl ester hydrolases, RBP4: Retinol-binding protein 4, TTR: Transthyretin, ATRA: all trans retinoic acid, ALDH: Aldehyde dehydrogenases, CRABP2: Cellular retinoic acid-binding protein 2, FABP5: Fatty acid-binding protein 5,: RAR: Retinoic acid receptor, RXR: Retinoic X receptor, PPAR: Peroxisome proliferator-activated receptor. All genes, protein, and symbols were written according to HUGO Gene Nomenclature Committee (HGNC, https://genenames.org/) and UniProt (https://www.uniprot.org/).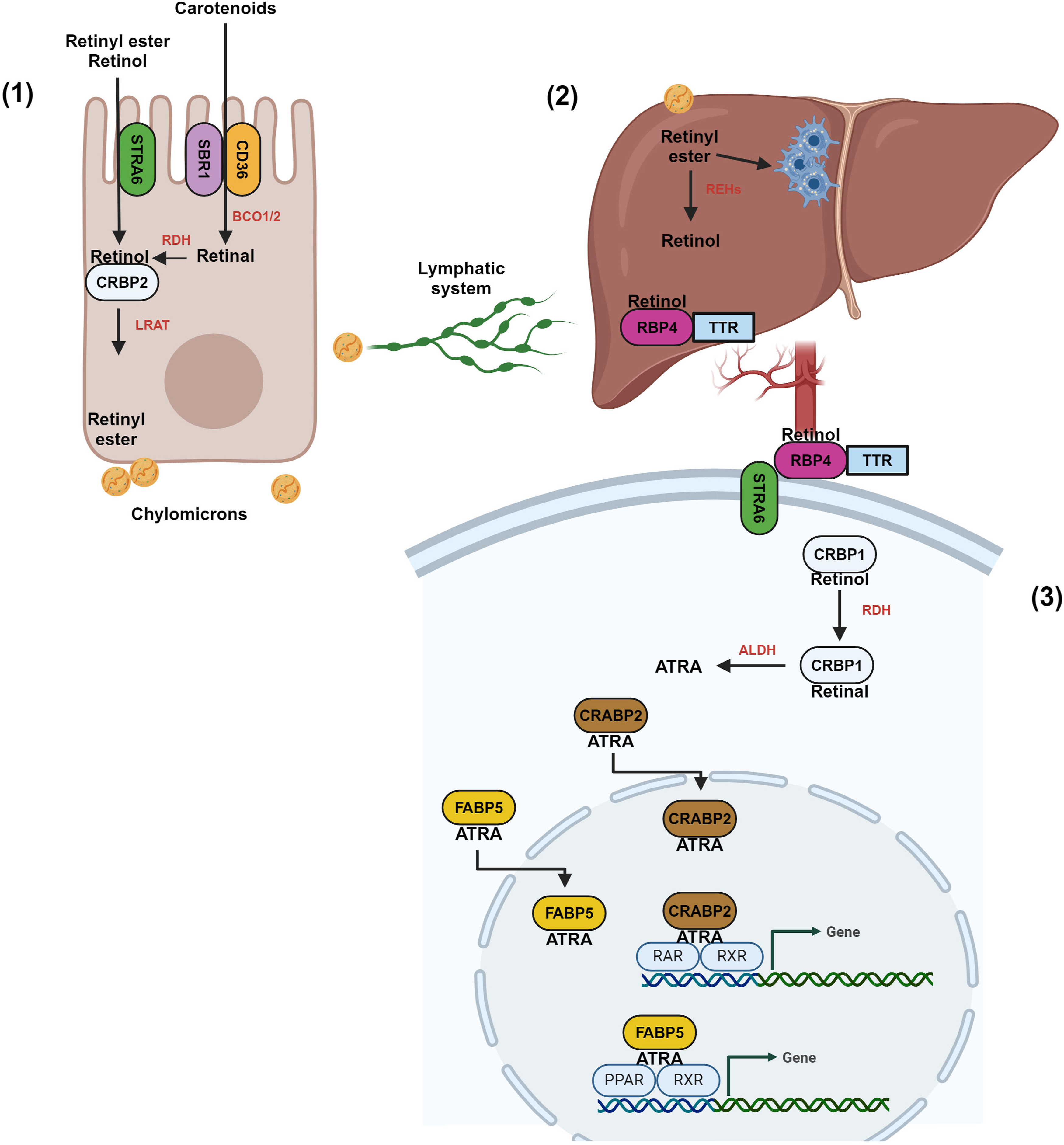
The vitamin A canonical pathway comprises RARs homodimerization or heterodimerization with RXRs after ligand binding. This ternary complex interacts with retinoic acid response elements (RAREs) or with retinoid X response elements (RXREs) at the promoter regions of the target genes.17,18 As early as 1 h of incubation with ATRA, more than 3000 genes are differentially regulated on cultured keratinocytes directly or indirectly19,20; these genes are involved in cell cycle inhibition [eg, by up-regulating cyclin-dependent kinase inhibitor 1B (p27), 1C (p57), 2B (p15); growth arrest specific 1, 6, 7 (GAS1, GAS6 and GAS7)],21,22 promote differentiation [eg, by up-regulating transglutaminase 1 (TGM1), keratin 1, 3, 6B, 9, 10 16, 18, and 23 (KRT1, KRT3, KRT6B, KRT9, KRT10, KRT16, KRT18 and KRT23)],22,23 cell adhesion induction [(eg, by up-regulating claudin 4 (CLND4)], 24 transcription factors [eg, by up-regulating retinoic acid receptor beta (RARB), cellular retinoic acid binding protein 2 (CRABP2), and down-regulating retinoic acid receptor gamma (RARG), and retinoic X receptor gamma (RXRG)],21,22 cell migration inhibition [eg, by down-regulating plakophilin 1, 3 (PKP1, PKP3); desmoglein 1 (DSG1), metalloproteinase 2, 9, and 14 (MMP2, MMP9 and MMP14)], 22 UVB-induced apoptosis [eg, by up-regulating tumor protein p53 (TP53) and caspase 3, 6, 7, and 9 (CASP3, CASP6, CASP7, and CASP9)], 25 wound healing promotion (eg, by up-regulating of fibroblast growth factor 7 (FGF7), heparin binding EGF like growth factor (HBEGF)],26,27 among others. 22
Non-canonical pathways also involve binding RXRs with other nuclear receptors such as thyroid hormone receptor, the estrogen receptor, and vitamin D3 receptor,28,29 this vastly increases the cell’s vitamin A-mediated transcriptional landmark.
RAR-RAR, or RAR-RXR heterodimers, operate as “transcriptional switches”; in the absence of ligands, both dimeric combinations bind to corepressors such as the negative coregulator or the silencing mediator for retinoid and thyroid hormone receptor which recruits histone deacetylases, or DNA methyltransferases to repress the transcription of vitamin A-target genes.33,34 However, the presence of active vitamin A provokes a conformational change in the RARs’ helix 12 receptors, causing the release of corepressors and the binding of coactivators, some of them, with histone acetyltransferase activity (ie, p300-PCAF, DRIP/TRAP/ARC, or NCOA1-PCIP); that eventually decondensed chromatin, and allows the further exposition of core promoters to activate gene transcription. 35
As mentioned, active vitamin A is bound by CRABP2, which is the most abundant subtype in the skin and mucosa. 36 However, active vitamin A can also be bound by the FABP5 to produce effects opposite to those of the canonical pathway. FABP5 delivers active vitamin A to the “orphan” Peroxisome proliferator-activated receptor (PPAR), a receptor member that also includes PPAR-apha and PPAR-gamma. 37 The selectivity of active vitamin A for PPAR-beta/delta has a Kd of ∼15 nM, which corresponds to an order of magnitude than those for PPAR-alpha and PPAR-gamma; indeed, reporter gene assays propose that 9-cis-RA regulates transcription via PPAR-beta/delta-RXR heterodimerization. 38
The Association Between Retinoids, Cervical Intraepithelial Neoplasia, and CC
Epidemiological Evidence
Epidemiological studies have examined the connection between vitamin A, HPV infection, premalignant lesions, and CC. 39 A significant inverse relationship between serum ATRA levels and different grades of cervical lesions has been observed. 40 A case-control study revealed that women with serum retinol levels in the lowest quartile were more likely to develop CIN1 than those with sufficient levels (>318 μg/l; OR = 2.3, 95% CI = 1.3-4.1, P = 0.002). 41
In the context of CC, women with lower levels of serum retinol are 4.5 times more likely to progress to carcinoma in situ or CC compared to women with higher levels of serum retinol. 42 A meta-analysis indicated that total vitamin A intake and blood vitamin A levels were inversely associated with the risk of CC (OR = 0.59, 95% CI = 0.49-0.72 and 0.60, 95% CI = 0.41-0.89, respectively) 43 and a recent meta-analysis also reported an inverse association with high vitamin A intake or high blood vitamin A levels and the CC risk; however, there was no statistical significance (OR = 0.77, 95% CI 0.59-1.02 and OR = 0.93, 95% IC = 0.71-1.22). 44 Additionally, a cross-sectional study using the NHANES population-based database reported that women with CC potentially had a lower intake of vitamin A [362 μg (interquartile range:198.00, 612.25] in comparison with women without CC [435 μg (interquartile range: 240.00, 717.00) (P = 0.018)]. However, after adjusting for potential confounders, the intake of vitamin A (OR = 0.999, 95% CI = 0.998-1.000) showed no association with CC. 45
Concerning HPV infection, findings from a comprehensive analysis of the NHANES database indicated that an appropriate amount (95% CI: 0.9-1.0, <10.5 of log2 transformer, ie, 1448.155 μg) of dietary vitamin A may be beneficial to prevent HPV infection. In comparison, an excessive intake (95% CI: 1.1-2.8, ≧10.5 of log2 transformer, ie, 1448.155 μg) could potentially elevate the risk of infection. 46
Molecular Evidence
Tumor development and progression are closely related to vitamin A signaling pathway disruptions. 47 Molecular study of this pathway have also revealed that vitamin A deficiency is associated with an increased risk of cervical carcinogenesis. For example, in a preclinical model that expresses the E7HPV16 oncoprotein, it was reported that chronic dietary vitamin A deficiency produces severe cervical dysplasia, cancer in situ, and CC. 48
Extensive molecular research has highlighted the role of RARs in the initiation and progression of CC. Specifically, hypermethylation of the RAR-beta promoter has been documented in cervical cancer cells, leading to transcriptional repression and loss of tumor suppressor function.49–51 Furthermore, it has also been reported that the decrease in RAR-beta expression results in significant inflammation and development of cervical precancerous lesions. 52 Moreover, ectopic RAR-beta expression has shown a substantial ability to inhibit cervical cancer cell growth by downregulating HPV18 E6/E7 oncogenes and inducing cell cycle inhibitory proteins.53,54 Regarding RXR-alpha, the work by Ocadiz-Delgado et al 55 has effectively demonstrated that the absence of RXR-alpha, in conjunction with HPV16 E6/E7 expression, is sufficient to initiate the development of cervical malignant lesions in a mouse model. Additionally, Shi et al 56 showed that blocking the long noncoding RNA LINC00511 expression suppresses Phospholipase D1 transcription via RXR-alpha, leading to apoptosis and autophagy in cervical cancer cells.
It has also been investigated that the hemimethylation of the CRABP1 and CRABP2 promoters and elevated expression of the RA-metabolizing enzyme CYP26A1 could be potentially associated with CC.57,58 Recently, it has been established that the upregulation of CRABP2 by HPV16 E6/E7 and overexpression of FABP5 contribute to enhanced cell proliferation, migration, and invasion while suppressing cell apoptosis in CC. 59 The malignancy exhibited by CRABP2 depends on the aberrant activation of the oncogenic β1/FAK/ERK integrin signaling pathway. 60
Retinoids and Their Prophylactic and Therapeutic Role in Cervical Intraepithelial Neoplasia and CC
Use of Retinoids as Therapy: in Vitro Studies and Preclinical Models
In Vitro and Preclinical Therapeutics Approaches Using Retinoids Against CC.

ATRA: all-trans retinoic acid; 9cAR: 9-cis-retinoic acid, IFN: Interferon; C33 CM: NR, non-response; IP, increase in proliferation; VPA: valproic acid; anti-PD-L1 Ab: monoclonal antibody directed against programmed cell death-1 ligand 1; GI: growth inhibition; TV: tumor volume; RA: retinoic acid.
ATRA also exerts an antineoplastic influence by inducing insulin-like growth factor-binding protein 5 (IGFBP-5) overproduction in the HPV-negative C33 cell line. 67 Furthermore, ATRA and RAR-selective ligands lead to reduced Epidermal growth factor (EGF) levels 68 and decreasing HPV18/16 E6/E7 expression69–71 in the ECE16-1, CaSki, and HeLa cell lines.56,57 Recently, ATRA was also shown to inhibit the proliferation and invasiveness of cervical cancer cell lines (HT-3 and Caski) through pFADK1. Additionally, in this study, the administration of ATRA in a mouse model of cervical cancer was associated with reduced tumor growth. 72
The effectiveness of retinoids in combined therapeutic approaches has also been investigated in CC cell lines. A significant decrease in cell growth is observed when ATRA treatment is combined with Interferon alpha-1/13 (IFN-alpha) or Interferon gamma (IFN-gamma).73–75 This phenomenon is elucidated by the induction of p21, which depends on IRF-1 and p53 in the HeLa cell line. 76 Advancements in the treatment of CC have also extended to the exploration of retinoids as radiosensitizing agents. A recent study by Fan et al 77 evaluated the combination of cisplatin and ATRA, revealing a considerable reduction in cell proliferation and migration of the HeLa stem-like population through inhibiting Wnt signaling. Moreover, 9-cis-RA has been evaluated, uncovering its ability to enhance radiosensitivity in SiHa and CC-1 cell lines. 78
Another strategy to enhance the antitumoral efficacy of retinoids involves expressing tumor suppressor genes (TSGs) in CC. For instance, the ectopic overexpression of Fra-1 in HeLa and CaSki cell lines has been demonstrated to augment the effectiveness of ATRA in inhibiting the metabolism and growth of these cervical cancer cells. 79 Furthermore, the utilization of histone deacetylase inhibitors (HDACi) has gained widespread acceptance as a strategy for modifying gene expression by inducing histone hyperacetylation and reinstating the expression of silenced TSGs. In a study conducted by Feng et al, 80 TSGs such as RAR-beta-2, E-cadherin, p21, and p53 were reactivated by combining HDACi (valproic acid, VPA) with ATRA. Moreover, significant growth inhibition of cervical tumor xenografts was observed due to VPA and ATRA combined treatment. In a previous study, Feng et al 81 demonstrated that combining VPA and ATRA reinstates RAR-beta-2 expression and enhances growth inhibition in HeLa and SiHa cell lines through the PI3K-alpha/PKB pathway.
In recent years, combination therapies involving immune checkpoint inhibitors have been promising across diverse tumor types. 82 Some studies have emphasized the potential of ATRA in targeting myeloid-derived suppressor cells (MDSCs) to enhance the effectiveness of anti-PD1, anti-PD-L1, or anti-CTLA-4 therapies.83–85 Notably, ATRA has been reported to impair the function of murine MDSCs and enhance the efficacy of anti-PD-L1 cancer immunotherapy in CC. 86
Use of Retinoids in Clinical Trials
In patients, retinoids have shown efficacy in treating specific types of cancer, such as acute promyelocytic leukemia in adults87,88 and neuroblastoma in children.89–91 However, their potential as systemic treatments for most solid tumors remains partially untapped. 88 In the context of CIN, five studies explored the use of retinoids in randomized controlled trials, employing four different retinoids administered through oral route (fenretinid 92 ; aliretinoin 93 ; and isotretinoin 94 ) and vaginal route (ATRA95,96). Most of the studies agree that retinoids did not significantly affect the progress of CIN to higher grades or the regression of CIN3. However, within the CIN2 subgroup, partial or complete regression rates were observed when alitretinoin was administered, though this outcome was not statistically significant. 97 Moreover, worse outcomes were also reported in women who received fenretinide. 92
Retinoids have also been used in clinical trials for CC, particularly in combination therapy approaches. For instance, a phase II trial using 13-cis-RA with IFN-alpha-2a showed a 50% response rate, mainly in cases of locally advanced CC.98,99 However, this treatment was not effective for patients previously treated with radiotherapy and/or platinum-based therapy for CC. 100 Additionally, the combination of radiotherapy, chemotherapy, 13-cis-RA, and IFN-alpha-2a or IFN-alpha-2b for CC has been studied in other clinical trials, but the response has not been sufficient to justify large-scale phase III trials.101–104 Moreover, vitamin A has been evaluated in combination with neoadjuvant chemotherapy (NAC) in clinical trials for advanced stages of cervical cancer. Sanusi et al reported significantly reducing CC volume by adding vitamin A to NAC. 105
Discussion
This review evaluated evidence from studies on molecular defects in vitamin A signaling and CC, epidemiological studies regarding the association between vitamin A and the risk of CIN and CC, and its potential therapeutic use.
The relationship between vitamin A and CC risk is controversial with epidemiological evidence suggesting significant and nonsignificant associations. A meta-analysis by Zhang et al 43 reported that higher total vitamin A intake and blood vitamin A levels were associated with a reduced risk of CC. Conversely, a meta-analysis by Han et al 44 found a nonsignificant marginal inverse association between high vitamin A intake and blood vitamin A levels with CC risk. These discrepancies might be attributed to the different number of cases and controls included in each analysis. For instance, Zhang et al included 3108 cases and 5574 controls, while Han et al involved 2682 cases and 5305 controls. The differences observed could also be due to variations in population characteristics and the methods used to assess vitamin A levels. The Han et al study also analyzed vitamin A intake in breast cancer (OR = 0.84, 95% CI = 0.76-0.93) and ovarian cancer (OR = 0.81, 95% CI = 0.72-0.92), finding a significant inverse association with these cancers. However, blood vitamin A was not significantly associated with the risk of these types of cancer, suggesting that the source of vitamin A measurement (dietary intake vs serum levels) might influence the observed associations.
Different methods for assessing vitamin A status, such as dietary intake and serum levels, 106 provide distinct insights. Dietary intake reflects recent consumption and adherence to dietary recommendations, 107 while serum levels indicate the body's current vitamin A reserves and metabolic status. The variations in these measurement approaches can lead to inconsistency across studies. 108 For instance, reporting biases may affect dietary recall methods, while physiological factors like inflammation can influence serum levels. 109 Differences in population demographics, including age, socioeconomic status, and health behaviors, could also contribute to the observed discrepancies. 110 Additionally, confounding factors such as other micronutrient deficiencies, lifestyle factors (eg, smoking, alcohol consumption), and genetic predispositions111,112 might affect the relationship between vitamin A and CC risk.
Although the studies mentioned previously report the therapeutic potential of retinoids in CC, it is interesting to note that there are differences in their antineoplastic potential in different CC cell lines. For example, a poor therapeutic response has been observed in the HeLa cell line when 1 μM ATRA is used for 3-5 days.62,76,77,81 Moreover, the response in CaSki is heterogeneous; some studies report a considerable inhibition of growth after 5 days of treatment,62,69 while others even report an increase in proliferation.73,76 In the case of the SiHa cell line, the therapeutic effect has been more homogeneous when doses starting at 10 μM ATRA are used63,75,81; however, few works used this cell line. On the other hand, when ATRA is administered in a combination therapy scheme, the therapeutic potential increases substantially.73,75,81,86 These differences may be explained by variations in treatment strategies, including the cell type utilized, dosage administered, treatment duration, and the schedule of combined treatments (Table 1). Furthermore, it is essential to recognize that ATRA exhibits inadequate solubility, considerable toxicity, decreased affinity, and adverse effects. In contrast, synthetic retinoids demonstrate excellent stability, exceptional selectivity, efficacy, and reduced toxicity and side effects.13,113 Therefore, conducting more research on the effectiveness of the newest synthetic retinoids in CC is crucial.
In random clinical trials (RCTs), retinoids have been used for preventing the progression of CIN. Based on the available evidence, RCTs generally indicate that retinoids do not significantly impact the regression of CIN3 or the progression of any CIN but may have some effect on CIN2 regression. In a previous discussion, Helm et al highlighted that the outcomes of retinoid treatment were influenced by factors that made it difficult to determine its benefits reliably. For example, in two studies, a higher proportion of participants with CIN3 were included in the treatment groups.92,93 In another study, there was a higher proportion of smokers in the treatment group. 92 Additionally, four studies reported high spontaneous regression rates of CIN2 or CIN3 in the placebo groups, undermining the studies’ power.92,93,95,96 Possible suboptimal dosing and short treatment duration masked the potential treatment effects of retinoids. 114
Retinoids have demonstrated potential in treating cancer through various RCTs, mainly when used in combination therapies. 11 In CC, Lippman et al, reported a rate of 50% in cases of locally advanced bulky disease when 13cRA and IFN-alpha-2a. Further studies have explored the combination of radiotherapy, chemotherapy, 13-cis-RA, and IFN-alpha-2a or IFN-alpha-2b. While these multi-modal approaches have shown some promise in enhancing treatment responses, the results were not compelling enough to warrant progression to large-scale phase III RTCs. The limited efficacy in these studies could be attributed to several factors, including the specific sequence and timing of therapy administration, the inherent biological differences in tumor response to multi-agent treatments, and the potential for increased toxicity.
In recent years, combination therapies involving neoadjuvant chemotherapy (NCT) or immunotherapy have been promising to treat various types of tumors, including CC.115–118 Considering the potential of retinoids in combination with NCT or immunotherapy for CC treatment could be an effective strategy and warrants further investigation through RCTs. RCTs aiming to use vitamin A to prevent the progression of CIN must be meticulously designed to control for confounding factors as spontaneous regression rates of CIN in different populations, HPV vaccination status, and other cofactors (eg, smokers)
This review recognizes certain limitations that distinguish it from a comprehensive systematic review. First, our adherence to the PRISMA guidelines was partial, affecting the methodological rigor of our work. Unlike a systematic review, which exhaustively searches, appraises, and synthesizes all relevant studies on a given topic, our literature review may have missed some lesser-known but potentially relevant studies due to less stringent search and selection criteria. Therefore, while our review provides valuable insights, it should be considered a narrative rather than a comprehensive systematic review.
We must also consider the scarcity of recent references on retinoids and CC. The above may indicate a partial absence of ongoing research but could reflect several factors influencing the publication trends and research focus in this field. Previous retinoid studies have shown mixed results in effectiveness and side effects. Prophylactic options like HPV vaccines, improved screening techniques, 119 effective CIN surgery or novel treatment strategies 120 may decrease emphasis on retinoids. The focus in cancer research might have shifted towards more advanced and targeted therapies, such as immunotherapies 121 and personalized medicine.
Conclusions
Vitamin A deficiency may contribute to the development of CIN and CC. Ensuring adequate levels of vitamin A could help prevent these conditions, particularly among high-risk groups such as women with persistent HPV infection and low serum retinol levels. Defects in the vitamin A signaling pathway also play a crucial role in CC development, highlighting the importance of understanding these mechanisms for clinical prevention and treatment.
Further investigation is warranted for both therapeutic and prophylactic uses of vitamin A. For therapeutic use in CC, the approach should be refining combination therapies, optimizing dosing, and identifying responsive patient subgroups through smaller, targeted trials. For prophylactic use in CIN, conduct well-designed, sufficiently powered RCTs, with attention to confounding factors and optimal treatment.
Abbreviations
All-Trans RA
Cervical cancer
Cervical intraepithelial neoplasia
Cytoplasmic retinol-binding protein 2
Fatty Acid Binding Protein 5
High-risk human papillomaviruses
Retinoid acid
Retinoic acid response elements
Retinoic acid receptors
Retinol-binding proteins
Retinoic X response elements
Retinoid X receptors
Footnotes
Acknowledgments
We thank Elizabeth Álvarez Ríos for her excellent technical support.
Author Contributions
The first author was responsible for the comprehensive drafting and structuring of the manuscript. The first and second authors were dedicated to conducting an literature review and acquiring data. The second author further contributed significantly to refining and revising the manuscript's content. The third author was involved in data collection and realized the ![]() . The last author was responsible for designing and supervising the research. He was actively committed to drafting and revising the manuscript. He also assumed responsibility for the modifications and submission process of the paper.
. The last author was responsible for designing and supervising the research. He was actively committed to drafting and revising the manuscript. He also assumed responsibility for the modifications and submission process of the paper.
Declaration of Conflicting Interests
The authors declared no conflicts of interest regarding this article's research, authorship, and publication.
Funding
The authors received no financial support for the research, authorship, and publication fees.
