Abstract
Objectives
Stereotactic body radiotherapy (SBRT) is widely used for localized prostate cancer and implementation of MR-guided radiotherapy has the advantage of tighter margins and improved sparing of organs at risk. Here we evaluate outcomes and time required to treat using non-adaptive MR-guided SBRT (MRgSBRT) for localized prostate cancer at our institution.
Methods
From 9/2019 to 11/2021 we conducted a retrospective review of 80 consecutive patients who were treated with MRgSBRT to the prostate. Patients included low (LR) (5%), favorable intermediate (FIR) (40%), unfavorable intermediate (UIR) (49%), and high risk (HR) (6%). Short-term androgen deprivation therapy was used in 32% of patients. Target volumes included prostate gland and proximal seminal vesicles with an isotropic 3 mm margin. Treatment was prescribed to 36.25 Gy in 5 fractions every other day with urethral sparing. Hydrogel spacer was used in 18% of patients. Time on the linac was recorded as beam on time (BOT) plus total treatment time (TTT) including gating. Analyzed outcomes included PSA response and patient reported outcomes scored by the American Urological Association (AUA) questionnaire and toxicity per CTCAE v5. General linear regression model was used to analyze factors affecting PSA and AUA in longitudinal follow up, and chi-square test was used to assess factors affecting toxicity.
Results
Median follow up was 19.3 months (3.8 – 36.6). Median BOT was 4.6 min (2.6 – 7.2) with a median TTT of 11 min (7.6 – 15.8). Pre-treatment vs post-RT median PSA was 6.36 (2.20 – 19.6) vs 0.85 (0.19 – 3.6), respectively (P < 0.001). PSA decrease differed significantly when patients were stratified by risk category, favoring LR/FIR vs UIF/HR group (P = 0.019). Four (5%) patients experienced a biochemical failure (BCF), with a median time to BCF of 20.4 months (7.9 – 34.5). Median biochemical failure free survival (BCFFS) was not reached, with 2-yr and 4-yr BCFFS of 97.1% and 72.1%, respectively. Patients with LR/FIR disease had 100% 2-yr and 4-yr BCFFS, whereas patients with UIF/HR had 95% and 41% 2-yr and 4-yr BCFFS (P = 0.05). Mean pre-treatment AUA was 7.3 (1 - 25) vs 11.3 (1 - 26) at first follow-up; however, AUA normalized to baseline over time. Urethral Dmax ≥35 Gy trended to lower AUA score at all follow-ups (P = 0.07). Forty-one (51%) patients reported grade 1-2 genitourinary toxicities at the 1 month follow up. Grade 3 toxicity (proctitis) was noted in 1 patient. There was no decrease in any grade rectal toxicity with use of hydrogel spacer (3 vs 6, P = 0.2). No grade ≥4 toxicities was observed.
Conclusions
MRgSBRT has the potential for treatment adaptation but this comes at the cost of increased resource utilization. Our experience with non-adaptive MRgSBRT of the prostate highlights its short treatment times as well as efficacy with good PSA control and low toxicity profile.
Keywords
Introduction
Hypofractionated radiation has been widely utilized for patients with localized prostate cancer for many years. The rapid adaptation of this technique has been due to a combination of factors including better exploitation of the hypothesized lower α/β ratio for prostate cancer compared to surrounding normal tissues as well as robust data supporting its efficacy and safety as well as patient convenience.1-4 More recently there was been an shift towards Ultra-hypofractionation (UHF) or Stereotactic Body Radiotherapy (SBRT) approaches fueled by improvements in imaging, motion management and data supporting good tumor control and low toxicity.5,6
Magnetic resonance-guided radiotherapy (MRgRT) offers several advantages including improved soft tissue visualization, real-time tracking, gating and daily treatment adaptation; features which have made this modality be investigated for its use in SBRT for prostate cancer. Early European experiences published data using daily adaptive treatments which were well tolerated but reported treatment times as long as 86 minutes.7,8 The potential benefits from daily treatment adaptation and gating have to be considered against the cost of higher time demand by staff (physician and physicist) and decreased clinical throughput. 9
Our group previously published our early experience using non-adaptive MRgSBRT demonstrating that the regimen was well tolerated and with low toxicity at 1 year. 10 More recently, the randomized MIRAGE trial reported that MRgSBRT reduced the incidence of acute Grade ≥2 gastrointestinal (GI) (0% vs 10.5%) and genitourinary (GU) (24.4% vs 43.4%) toxicity compared to CT-based SBRT. 11 In the current study we report our updated outcomes in terms of PSA control, toxicity and time required to treat using non-adaptive MRgSBRT for localized prostate cancer.
Materials & Methods
Patient Eligibility
In this retrospective review, patients were part of an institutionally approved IRB (MCC#20383). In this study, 80 patients were consecutively identified from our institutional database who had been treated with MRgSBRT between September 2019 and November 2021. All patients had a biopsy-proven diagnosis of prostate cancer and stratified into low-risk (LR), favorable-intermediate risk (FIR), unfavorable intermediate risk (UIR) and high risk (HR) as per NCCN guidelines. As part of their staging workup, all patients underwent computed tomography (CT) or pelvic magnetic resonance imaging (MRI) and did not have evidence of nodal involvement or distant metastatic disease (based on bone scan or PSMA which were obtained at the discretion of the treating physician). Use of androgen deprivation therapy (ADT) was used at the discretion of the treating physician.
MRgRT planning and treatment
Simulation and treatment planning was performed as previously described by our group. 10 Briefly, patients were instructed to have full bladder and empty rectum before simulation and treatments. Patients then underwent MRI simulation in the 0.35 T MRIdian System (ViewRaw Inc., Mountain View, CA) along with CT for dose calculation, both scans done in the supine position typically with MRI first followed by CT. During MRI simulation a balanced steady-state free progression (TrueFISP) imaging sequence was used to create images weighted by T2/T1 ratio. No additional immobilization devices were utilized during simulation. A representative slice of the prostate was contoured as a tracking structure on a real-time single plane sagittal cine MRI sequence at 4-8 frames per second and a 3 mm gating structure was derived.
The clinical target volume (CTV) consisted of the entire prostate gland and the proximal seminal vesicles with a 3 mm isotropic expansion to planning target volume (PTV). Patients were treated to a total dose of 36.25 Gy in 5 fractions delivered over other day.
Of note, urethral sparing technique was not implemented uniformly until early-2020. In the early experience, the entire prostate was taken to 36.25 Gy. While there is some variation on the technique for urethral sparing based on physician preference, the most commonly employed technique is as described by Bruynzeel et al. 8
Statistical Analysis
Kaplan–Meier survival method was used to estimate BCFFS. All time-to-event analyses were calculated from the date of completion of SBRT. General linear regression model was used for analyzing factors affecting PSA and American Urological Association (AUA) in longitudinal follow up. Chi-square test was used to assess factors associated with toxicity assessed by patient reported AUA questionnaire and physician reported Common Terminology Criteria for Adverse Events (CTCAE) v5.0. Two-sided P < 0.05 was considered to be statistically significant. Data were analyzed using the statistical software SPSS (version 26.0). Additionally, we have followed the relevant EQUATOR guidelines. 12
Results
Patient and Treatment Characteristics
Baseline Patient Characteristics.

Abbreviations: Yrs, years; ADT, androgen-deprivation therapy; Gy, gray; min, minutes.
Outcomes
The median follow-up for the entire cohort was 19 months (4 – 37). All patients were alive at the time of last follow-up. Figure 1A summarizes the PSA trend for entire cohort, where we observed a significant decrease in PSA from pre-treatment (P < 0.001). The median pre-treatment PSA was 6.36 ng/mL (2.20 – 19.64), median PSA at first follow up was 1.73 ng/mL (0.02 – 12.68) and median PSA at seventh follow up was 0.85 ng/mL (0.19 – 3.57). Figure 1B shows PSA trends as stratified by LR/FIR vs UIR/HR and also shows a significant decrease (P = 0.019). PSA trends for A. Entire cohort B. Stratified by risk group. Abbreviations: LR, low risk; FIR, favorable intermediate risk; UIR, unfavorable intermediate risk; HR, high risk.
Of the 80 patients in the cohort, only 4 (5%) experienced a biochemical failure (BCF). Median biochemical failure free survival (BCFFS) for the entire cohort was not reached, BCFFS at 2-year was 97.1% and 72.1% at 4-year (Figure 2A). We then stratified BCFFS by into low-risk patients including LR/FIR and high-risk patients with UIR/HR. For LR/FIR group, the median BCFFS was not reached and was 100% at both 2 and 4-year (Figure 2B). Whereas for UIR/HR patients, median BCFFS was 35 months (95% CI 25.1 – 44.9), with 2-yr and 4-yr BCFFS of 94.7% and 41.4%, respectively (Figure 2B). There was a significant difference in terms of BCFFS between LR/FIR and UIR/HR (P = 0.05) (Figure 2B). Biochemical failure free survival (BCFFS). (A) Kaplan Meier graph representing BCFFs for the entire cohort in terms of median, 2-year and 4-year control. (B) Kaplan Meier graph representing BCFFS stratified by risk category. Abbreviations: LR, low risk; FIR, favorable intermediate risk; UIR, unfavorable intermediate risk; HR, high risk.
Toxicity
Toxicity of treatment was assessed by patient reported AUA questionnaire and physician scored as per CTCAE v5.0. Mean pre-treatment AUA was 7.3 (1 – 25) and we see a sharp increase at the 3 month follow up, with a median AUA of 11.3 (1 – 26) however in subsequent follow ups the AUA score gradually decreases to 7.8 (1 – 14) by the 12 month follow up, this trend is depicted in Figure 3A. We observed that increase in AUA scores were noted at all follow-up intervals with Dmax ≥35 Gy to the urethra, which was trending towards significance (P = 0.07) (Figure 3B). AUA Scores depicted as estimated marginal means (EMMs) A. AUA trend across the entire cohort over 12 months of follow up. (B) AUA trend according to maximum dose received by urethra and stratified into low dose (<35 Gy) or high dose (≥35 Gy) across 12 months of follow up. Abbreviations: AUA, American Urologic Association; RT, radiation, FU, follow up; Gy, gray.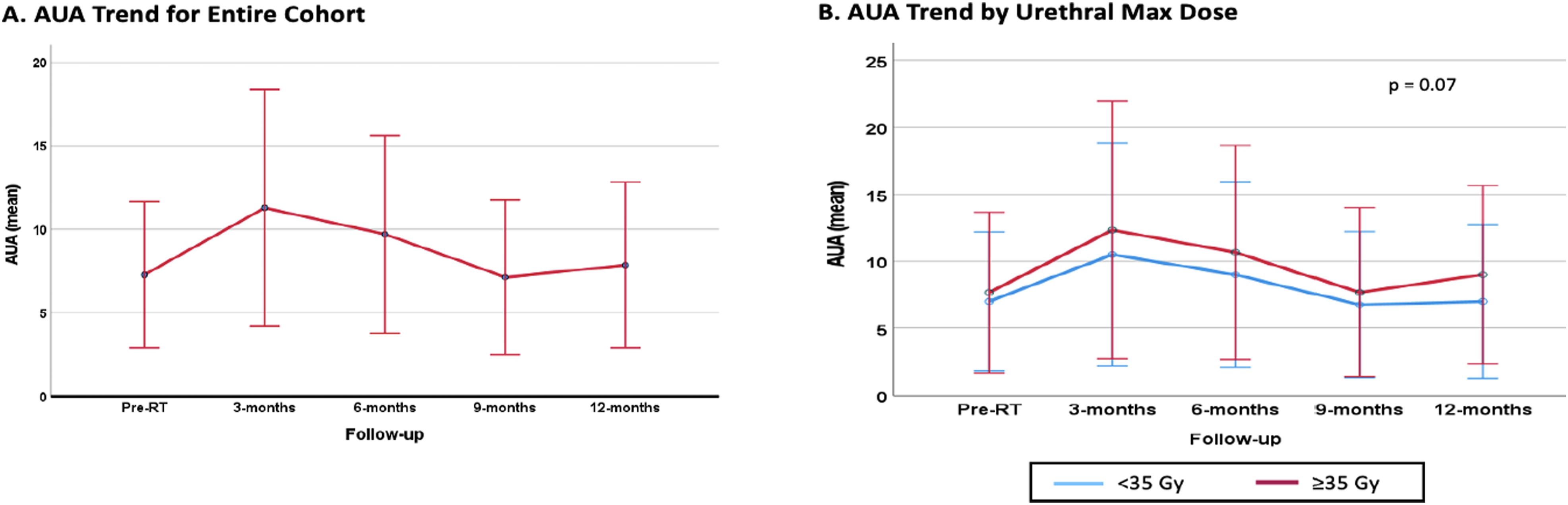
Acute Toxicities Graded as per CTCAE v5.0. Median Time to First Follow up was 1.3 months and Median Time to Second Follow up was 4.5 months From Completion of Treatment.
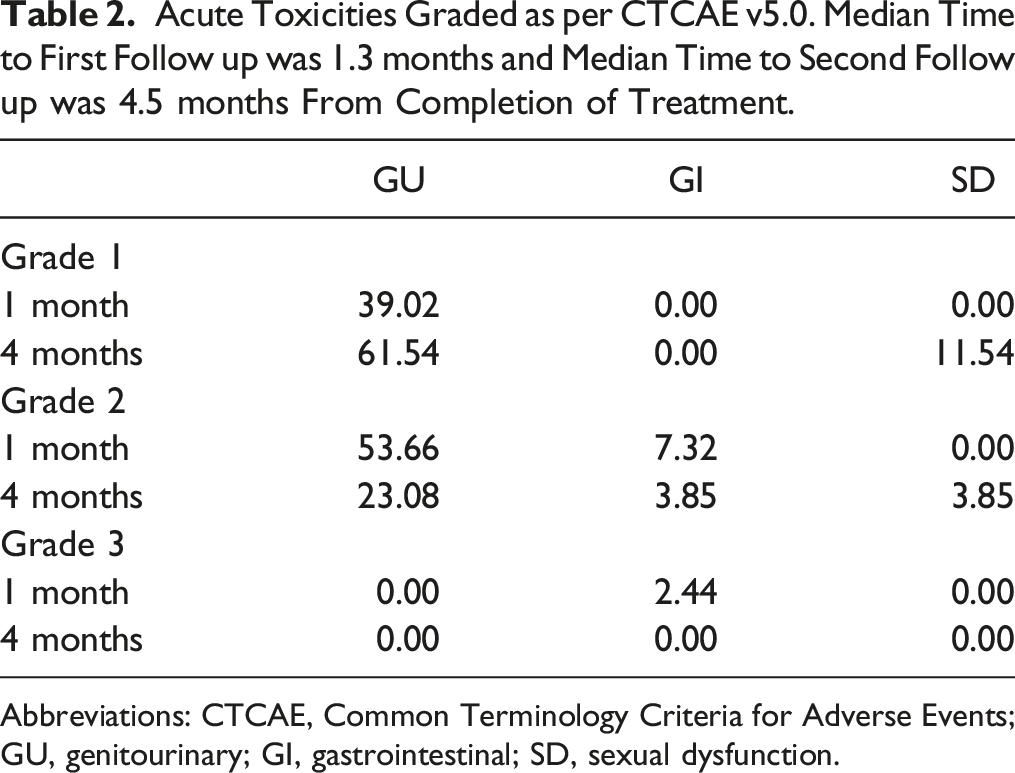
Abbreviations: CTCAE, Common Terminology Criteria for Adverse Events; GU, genitourinary; GI, gastrointestinal; SD, sexual dysfunction.
Discussion
The current study represents an expansion on our original cohort demonstrating the feasibility and safety of non-adaptive MRgSBRT for localized prostate cancer. In our prior experience, we reported our findings in a population of 35 patients where we noticed a significant decrease in PSA as early as 1-month follow up (P < 0.005). 10 In the current study we evaluated 80 patients with a median follow up of 19.3 months (3.80 – 36.57). In our non-adaptive workflow, median BOT was 4.60 minutes (2.59 – 7.20) and median TTT 10.99 minutes (7.57 – 15.77) (Table 1), a time that is comparable to standard CT-based SBRT treatment times and significant shorter than reported adaptive workflows with median fraction time of 56 minutes (34 – 86). 7 Despite, the shorter treatment time in this non-adaptive workflow, we continued to observe a significant decrease in PSA from pre-treatment (P < 0.001) that was maintained until last follow up (Figure 1A).
Of the 80 patients, only 4 (5%) experienced BCF. For entire cohort, BCFFS was 97% at 2-year and 72.1% at 4-year (Figure 2A). For patients with LR/FIR the median time to BCFFS was not reached, 2-year and 4-year BCFFS were 100% (Figure 2B), suggesting that this modality provides durable control for patients with low and intermediate risk disease and is consistent with other series reporting 5-year biochemical control for low and intermediate risk patients ranging between 97 – 91%. 13
SBRT has been traditionally used in patients with low and intermediate risk prostate cancer, given the potential for smaller volumes and omission of elective nodal irradiation (ENI). However, as recently as 2020, SBRT has been added to NCCN guidelines for use in high-risk prostate cancer. 14 Several studies have reported retrospective and prospective efficacy of SBRT in these high-risk patients with 5-year biochemical recurrence free survival of 81%.15,16 For patients with UIR/HR, median BCFFS was 35 months and the 2-year and 4-year BCFFS was 94.7% and 41.4%, respectively (Figure 1B). The 4-year BCFFS in our cohort is lower than reported in the literature. While only a minority of our patients had HR (6%, n = 5), two of the 5 developed BCF, both of whom received 4-month of ADT. The other two patients with BCF had UIR and did not receive ADT due to medical comorbidities and/or patient preference. The most common site of failure was pelvic lymph nodes (75%) followed by bone (50%) and finally prostate (25%).
The low BCFFS seen in patients with UIR and HR suggests that additional considerations should be made regarding management of these patients. The use of SBRT in men with HR prostate cancer has seen a significant increase, largely driven by utilization in men not receiving ADT (0.7% in 2004 to 8.3% in 2016, P < 0.001) and in those whose only risk factor for HR disease is PSA >20 ng/mL (1% in 2004 vs 4.3% in 2016, P < 0.001). 16 Additionally, other factors associated with increased use of SBRT include clinical stage T1 – 2, Gleason 6-7, medical comorbidities and long travel distance.15,16 In our cohort, all the HR patients had PSA <20 ng/mL and only 1 patient had GGG 5. All were offered short-term ADT (4 – 6 months) and only 1 patient refused. In this early report at almost 4 years, there was good PSA control however long term follow up is essential to further elucidate durability of control.
In our SBRT approach for localized prostate cancer, ENI is not offered. Pelvic nodal recurrences were the most common site of failure in 3 of the 4 patients. While the role of ENI in HR prostate cancer patients remains controversial, there are several studies looking at the role of hypofractionated 17 and UHF 18 approaches to ENI. Early results from a prospective study including high, very high risk and node positive patients treated with SBRT to 35 – 37.5 Gy to the prostate and 25 Gy to elective nodal regions showed a 3-year BCFFS of 77% 19 in combination with long-term ADT. Taken together, these results highlight the importance of patient selection and suggests that for these higher risk patients long-term ADT and/or ENI should be considered.
The use of SBRT for treatment of localized prostate cancer has increased over threefold in the US from 2004 to 2016. 16 While SBRT has been primarily delivered using CT-based approaches, the advent of MRgRT in 2014 provided certain advantages that made it an attractive modality for use in prostate cancer. MR-guidance has the advantage of improved soft tissue visualization and real-time tracking which can lead to a reduction in treatment margins and potential decrease in toxicity for patients. 20 To assess patient-reported toxicity we used AUA questionnaires where we observed a similar trend as reported in our initial experience, where there was an immediate increase in AUA from pre-treatment (mean 7.3) to 1 month after treatment (mean 11) and a subsequent decline out to 12 months from treatment (mean 7.8) (Figure 3A). With the widespread use of high-dose per fraction approaches, the importance of limiting dose to normal organs becomes paramount.
Consensus on dosimetric parameters for the urethra vary across institutions, while some guidelines recommend a Dmax to urethra between 38 – 42 Gy for a 5-fraction regimen. 21 As previously described in the methods section, urethral sparing approach was not uniformly implemented initially, therefore the early cohort of patients were not limited to Dmax of 35 Gy. With our current urethral sparing approach, the Dmax is limited to 35 Gy. While this threshold is lower than some guidelines, we noted that patients with Dmax>35 Gy had persistently elevated AUA scores at all time points however it was not statistically significant (P = 0.07, Figure 3B). More recently, given better imaging capabilities and better visualization of the urethra, a further reduction to Dmax <32 Gy has been implemented in a subset of patients. Our group is planning to report the dosimetric comparisons in a separate paper.
We also evaluated toxicity as scored by CTCAE v5.0, at a median time of 1.3 months from completion of treatment (follow up 1) approximately half (n = 41) reported some degree of toxicity. The most common adverse events recorded at this point are listed in Table 2, and are primarily Grade 2 GU (cystitis) in 54% of patients and Grade 1 GU (proctitis and tenesmus). In our initial publication, we did not report any Grade 3 events at a median follow up of 11 months. 10 Here, we report a single event of Grade 3 GI toxicity that occurred at first follow up. This was a case of proctitis that occurred in patient with history of internal hemorrhoids that required hospital admission for rectal steroids and made a full recovery after discharge. As highlighted in Table 2, at the second follow up, median of 4.5 months the incidence of Grade 2 GU events decreased to 23%. Our results, are comparable to those in the recently published MIRAGE trial, where they reported Grade ≥2 GU toxicity of 24% using MRgSBRT within 90 days of treatment completion. 11 No Grade 4 or 5 toxicities were recorded. Finally, there was low rate of sexual dysfunction, 12% Grade 1 and 4% Grade 2 (Table 2) both seen at the second follow up (median time from SBRT 4.5 months), this low toxicity could be attributed to that only a minority of patients (30%) received ADT however longer term follow up will be needed to assess for late toxicity.
Daily treatment adaptation and tracking are promising features of MRgSBRT that can allow for better motion management and sparing of organs at risk, this in turn can obviate the need for invasive procedures including placement of fiducial markers and/or SpaceOAR. 22 In our study, we did not see a significant decrease in toxicity with placement of SpaceOAR (P = 0.2), however only 15 patients had this placed. While there is data showing that placement of SpaceOAR has dosimetric advantages in terms of reducing rectal and bladder doses, patient-reported questionnaires showed no difference. 23 While these results are encouraging, some limitations of our study must be considered including limited number of patients, single institution data, ADT use as a confounding factor, variations in urethral constraints as well as short follow up time. Nonetheless, our study provides evidence that for patients who are unwilling or unable to undergo invasive procedures, non-adaptive MRgSBRT provides a good alternative with low toxicity profile.
Conclusion
This study adds to the growing body of evidence showing that non-adaptive MRgSBRT for localized prostate cancer is an effective treatment modality with good PSA control and low toxicity burden for patients. By using a non-adaptive workflow, it is possible to give access to patients to a safe and effective treatment modality. Nonetheless, longer term follow will be needed to asses for long term efficacy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Yamoah receives grant funding from NCI and American Cancer Society, consulting fees from Janssen R&D. Dr. Latifi receives consulting fees from ViewRay. Dr. Rosenberg receives grant funding and consulting fees from ViewRay. The remaining authors have no disclosures.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
This is a retrospective review so patients are part of an IRB to collect their information from an institutional database. Therefore, there is no informed consent collected per se for the patients in the study.
IRB Approval
IRB (MCC#20383) was used for institutional database for retrospective collection of patient data.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
