Abstract
Objectives
Brain metastases (BMs) are commonly categorized into cystic and solid. However, the difference in the prognosis of patients with either cystic or solid BMs following radiotherapy remains poorly understood. We used a retrospective design to elucidate the disparities in survival between these two patient groups undergoing radiotherapy and to identify factors influencing the overall survival (OS) of patients with BMs.
Methods
This retrospective study encompasses 212 patients diagnosed with BMs. We meticulously analyzed the clinical characteristics, radiation therapy modalities, and risk factors influencing the OS among these patients, categorized by BMs type, post-brain radiation therapy.
Results
A statistically significant difference in mOS was observed between the two cohorts (Solid vs Cystic: 23.1 vs 14.6 months). Subgroup analysis unveiled distinctions in mOS, particularly in patients with EGFR-mutant lung adenocarcinoma (Solid vs Cystic: 23.1 vs 6.43 months). The volume of BMs and the biological effective dose (BED) emerged as significantly prognostic factors for patients with cystic BMs. For patients with solid BMs, fraction dose, BED, and the number of BMs were identified as independent prognostic factors for survival.
Conclusion
Brain radiotherapy shows superior survival benefits for lung cancer patients with solid BMs compared to those with cystic BMs, particularly in EGFR-mutant lung cancer. In particular, patients receiving BED ≥60 Gy have a more favorable prognosis than those receiving BED <60 Gy, regardless of the type of BM (solid or cystic) in lung cancer.
Introduction
Brain metastases (BMs) are among the most common intracranial tumors in adults and are a significant contributor to mortality in patients with malignant tumors.1,2 Recent advances in medical technology have prolonged the survival of cancer patients and consequently increased the incidence of BMs. A small proportion of patients have BMs at initial diagnosis, and approximately 20-40% of patients develop BMs during the course of their disease. 3 Lung is the most common primary cause of BM, followed by renal cell and breast cancer. 4 Unfortunately, the prognosis for patients with BM is poor, with a median survival of only 1 to 3 months without intervention. 5
Radiographically, BMs can be classified as either cystic or solid. Solid BMs are the predominant pattern and consist primarily of solid tumor cells or tissue. They typically present as solid masses or nodular lesions, occasionally interspersed with small cystic areas. Cystic BMs, on the other hand, manifest as metastatic brain lesions characterized by fluid-filled or cystic structures. These lesions typically result from the spread of cancer cells from the primary tumor to the brain via the bloodstream or lymphatic system, forming cystic sacs or cysts containing fluid or semisolid material. Cystic BMs are recognizable as lesions with cystic or low-density areas on imaging and often show both morphological and clinical differences from solid BMs. Some previous research studies have defined cystic BMs as lesions with a cystic component of more than 50% of the total volume, whereas those with a cystic component of less than 50% are classified as solid BMs.6,7
Treatment options for BMs include surgery, whole-brain radiation therapy (WBRT), WBRT with a simultaneous integrated boost, stereotactic radiosurgery/stereotactic body radiotherapy (SRS/HFRT), chemotherapy, targeted therapy and immunotherapy with immune checkpoint inhibitors (ICIs). These treatments have significantly prolonged the survival of patients with BMs.8,9 Local therapies, particularly radiotherapy (RT), play a pivotal role in the treatment of BMs. For patients with 1-3 BMs, SRS has the benefit of local control, reduced delivery time, and reduced cognitive impairment compared to the WBRT. recent studies agree that WBRT + boost/HFRT and SRS are equivalent in the treatment of single brain metastases.10,11 However, SRS may be with more cerebral distant relapse in comparison with WBRT + boost. 12 Owing to economic reasons or considering poor follow-up compliance in China, WBRT + boost/HFRT treatment still remains possible. Although SRS is recommended by guidelines as a standard radiotherapy method for patients with 1-3 BMs. However, several retrospective clinical studies have reported varying prognosis of radiotherapy for solid and cystic BMs. One study showed that solid BMs in NSCLC had significantly better local control rates than cystic BMs after RT with no difference in mOS. 9 Brigell showed that HFRT results in a higher local control rate compared to WBRT in patients with solid BMs, but the radiotherapy models have a limited effect on the local control rate of cystic BMs. 13 Contrary to the aforementioned studies, Wang reported that there was no difference in prognosis and local control between patients with cystic and solid BMs treated with RT. 6
Currently, reports comparing the prognosis of patients with cystic and solid BMs after radiotherapy are limited and inconsistent. Due to the rarity of cystic BMs, there is still a lack of definitive evidence from large clinical research trials to support the optimal treatment of cystic BMs. Therefore, we aimed to analyze the differences in survival and response to radiotherapy in these two groups of patients and to investigate the factors associated with overall survival in BMs. We have to try to find more appropriate treatments for different types of BMs.
Material and Methods
Definition and Research Groups of BMs
Cystic BMs were defined as lesions with a low signal center with ring enhancement on contrast-enhanced T1-weighted MRI and high signal intensity on T2-weighted images, with cystic lesion volume exceeding 50% of the total volume. The diagnosis of cystic BMs required contrast-enhanced MRI. Cystic-solid BMs and solid BMs were categorized as solid BMs. In this study, a senior central nervous system radiologist distinguished between cystic lesions and solid lesions.
Patients
This study was approved by the institutional review and ethics committee of Hubei Cancer Hospital (No. LLHBCH2024YN-030). We conducted a retrospective review of all patients with BMs who underwent brain radiotherapy at Hubei Cancer Hospital from February 2018 to August 2019. The study included their basic information, clinical records, treatment regimens, imaging data, and survival outcomes. All patients were evaluated by radiologists, radiation oncologists, and physicists. Inclusion criteria were as follows: (1) a definite histopathological diagnosis of the primary lesion, including: lung cancer, breast cancer, gastrointestinal tumors, and et al; (2) newly diagnosed BMs; (3) patients received brain radiotherapy in our institution; (4) comprehensive clinical and imaging follow-up data available.
Clinical data were collected for all patients, including gender, age, smoking history, performance status (PS) score, presence of brain-related symptoms caused by BMs (seizure symptoms, neurological and psychiatric symptoms, intracranial hypertension symptoms), primary diseases (NSCLC, breast cancer, gastrointestinal tumors, melanoma, etc.), immunohistochemistry and genetic testing status of primary tumors, presence of metastases in other body sites (bone metastases, liver metastases, etc.), locations of intracranial metastases (brainstem, cerebellum, frontal lobe, occipital lobe, temporal lobe, parietal lobe, basal ganglia, thalamus), maximum volume of BMs before treatment (The largest one), extent of peritumoral edema (none, minimal, significant), volume changes of peritumoral edema after brain radiation therapy (no significant change, decrease, increase), radiation therapy modalities and doses, The BED was calculated based on a linear‐quadratic model (BED = nd [1 + d/(α/β)], α/β = 10 Gy), changes in BMs size on follow-up imaging (1 month after radiotherapy, 6 months after radiotherapy, 1 year after radiotherapy), treatments before and after radiotherapy, systemic treatment regimens, use of combination therapy and associated treatment efficacy.
Treatment Regimen and Follow-Up Evaluation
Systemic treatments included chemotherapy (following international standards for regimens and doses), immunotherapy with ICIs before and after radiotherapy, TKI therapy, and anti-angiogenesis therapy (eg,,bevacizumab, endostar). Brain radiotherapy regimens included WBRT (30-40 Gy/10-20F), WBST + BOOST (36.7-60 Gy/10-25F), and HFRT (27-55 Gy/3-6F). Response Evaluation Criteria in Solid Tumors (RECIST v1.1) was used to assess the treatment responses, which were classified as complete response (CR), partial response (PR), stable disease (SD) and progressive disease (PD). The objective response rate (ORR) was the sum of CR and PR, while the disease control rate (DCR) was the sum of objective response rate (CR + PR + SD).
Statistical Analysis
Progression-free survival (PFS) referred to the time from the initiation of intracranial radiotherapy to PD or death. Intracranial progression-free survival (iPFS) indicated the time from the commencement of radiotherapy for BMs to intracranial progression during treatment or death. Overall survival (OS) was calculated from the star of radiotherapy for BMs until death or the last follow-up assessment. The last follow-up date was March 23, 2023.
The reporting of this study conforms to STROBE guidelines. 14 The minimal sample size computed by PASS software was 37 patients with cystic BMs and 150 solid BMs, which indicated that the used sample size in the present study was suitable for preliminary exploration. Statistical parameters were obtained based on previous experimental data.
The Kaplan-Meier method was used for survival estimation, and Cox regression analysis was used for univariate and multivariate survival analysis of cystic and solid BMs and prognostic factors. A two-tailed test with
Results
Characteristics of the Patients
Characteristics and Comparison in Patients With cystic or Solid BMs.

Prognosis of Cystic and Solid BMs after Radiotherapy
The median follow-up time was 53.7 months. As shown in Figure 1(A), patients with solid BMs had a superior mOS compared to patients with cystic BMs (23.1 vs 14.6 months, Kaplan–Meier estimates of OS and iPFS in patients A: The OS of solid BMs and cystic BMs in all patients (23.1 vs 14.6 months, 
Prognosis of Cystic and Solid BMs Derived from Lung Cancer after Radiotherapy
Patients’ Characteristics and Comparison Between Cystic and Solid BMs Derived From Lung cancer.

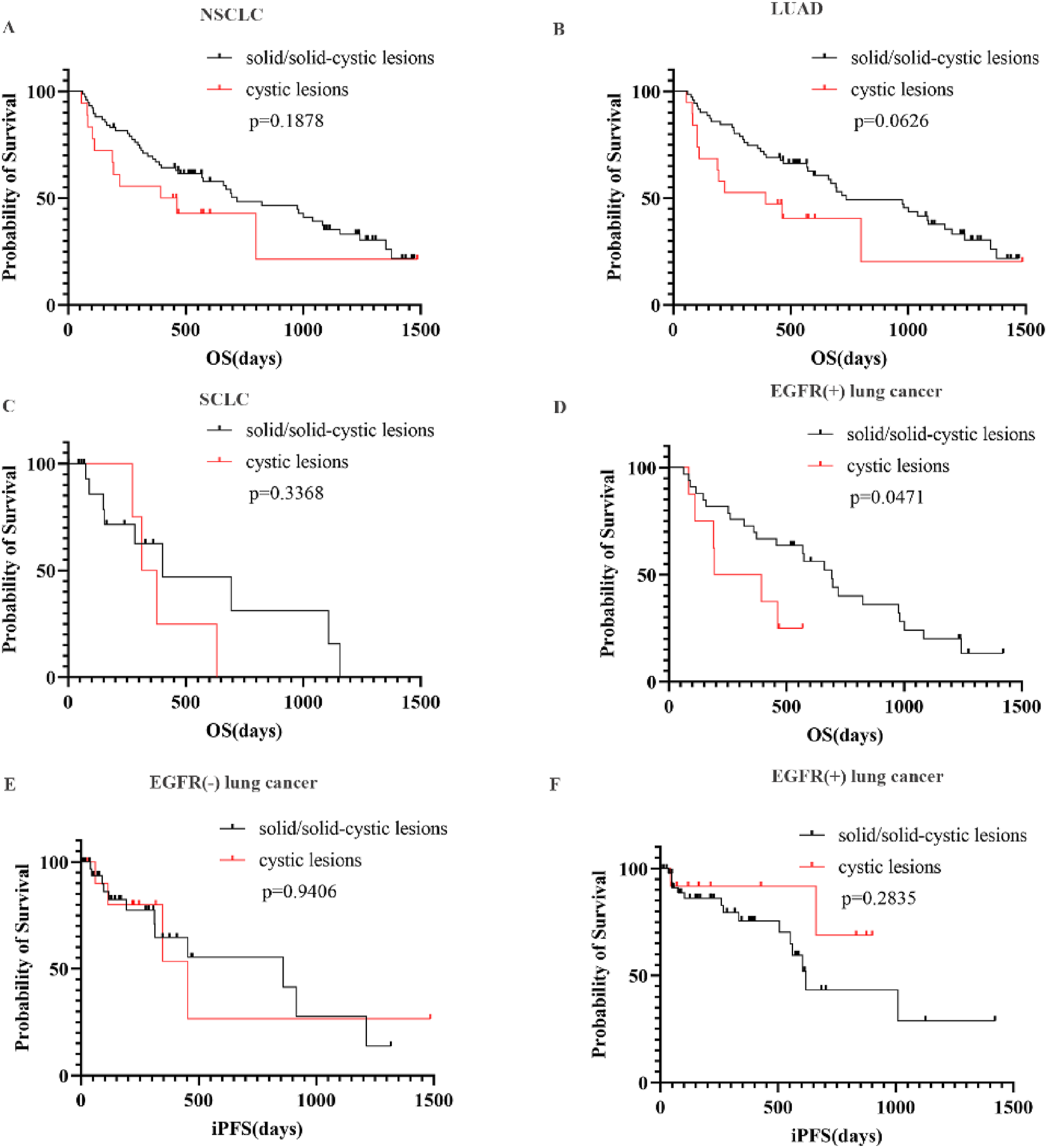
Kaplan–Meier estimates of OS and iPFS in patients of lung cancer A: The OS of solid BMs and cystic BMs in patients with NSCLC (23.9 vs 14.3 months,
Factors Affecting Prognosis in Patients with Lung Cancer BMs
Univariate Analysis of Prognostic Factors of Cystic BMs
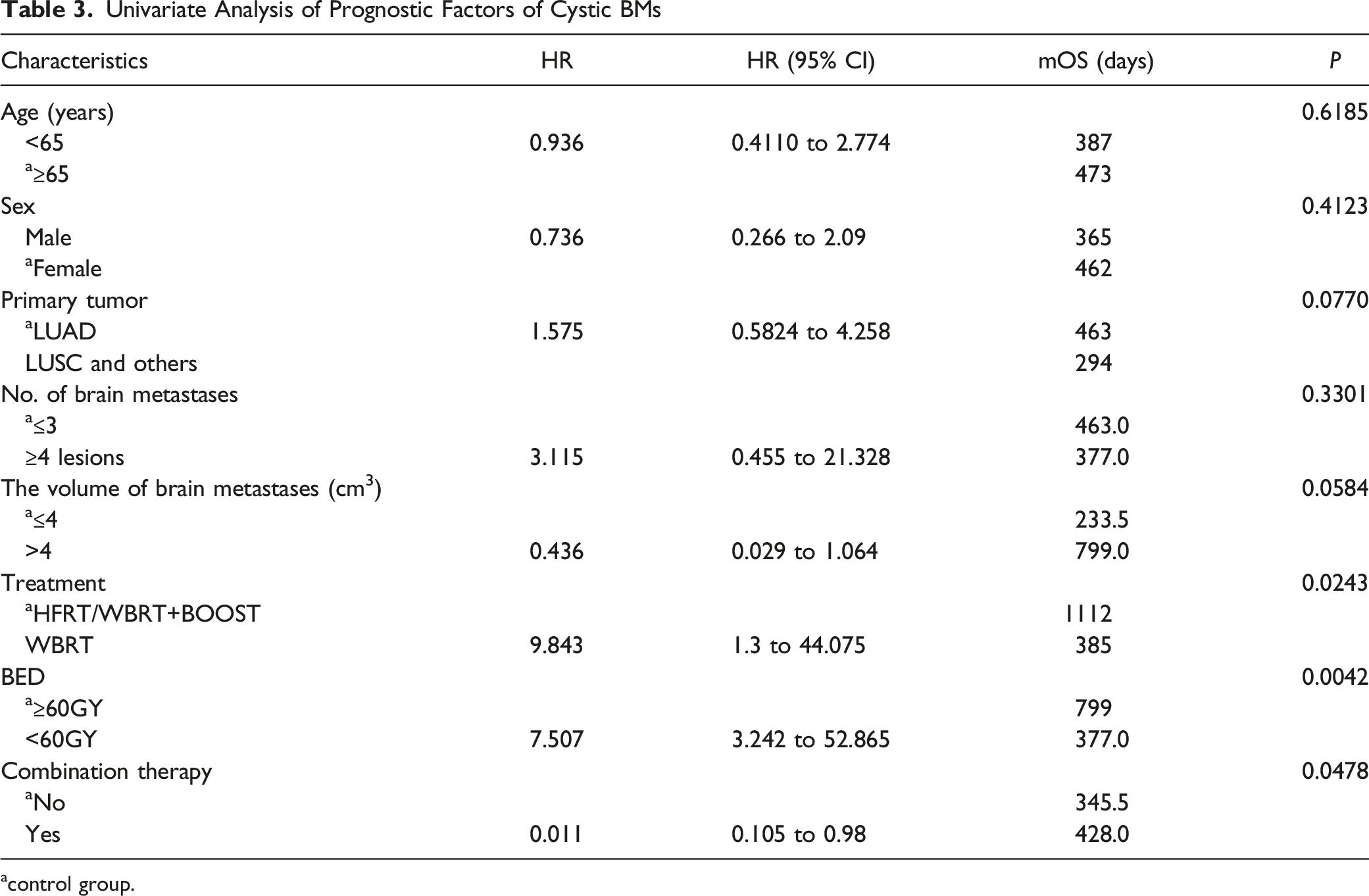
acontrol group.

The hazard ratio plot of factors associated with OS A: multivariate Cox regression hazard ratio for OS in patients with cystic BMs with lung cancer. B: multivariate Cox regression hazard ratio for OS in patients with solid BMs with lung cancer.
Multivariate Analysis of Prognostic Factors of Solid BMs.
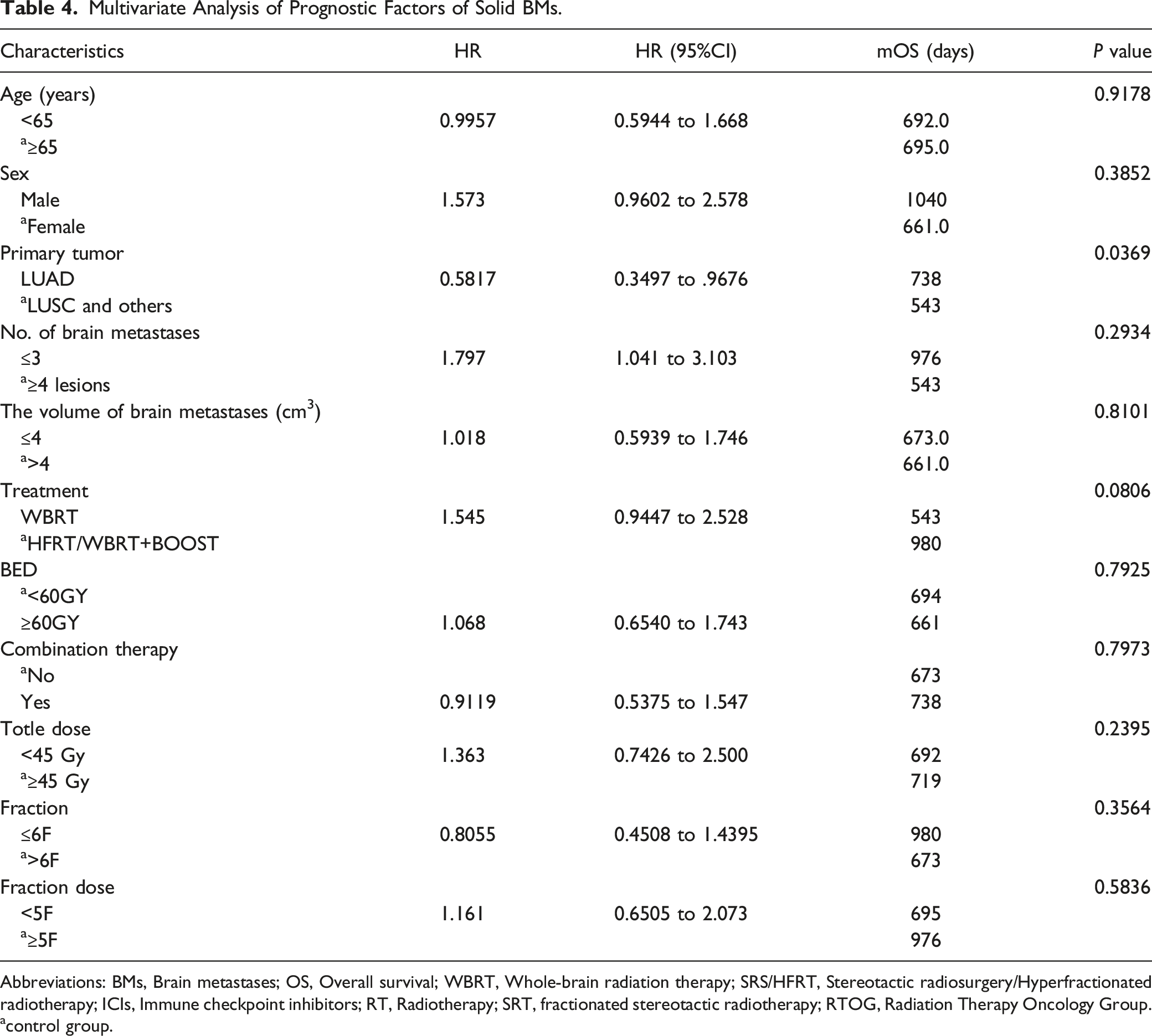
Abbreviations: BMs, Brain metastases; OS, Overall survival; WBRT, Whole-brain radiation therapy; SRS/HFRT, Stereotactic radiosurgery/Hyperfractionated radiotherapy; ICIs, Immune checkpoint inhibitors; RT, Radiotherapy; SRT, fractionated stereotactic radiotherapy; RTOG, Radiation Therapy Oncology Group.
acontrol group.
Discussion
We found in this study that compared with solid BMs, cystic BMs are relatively rare in patients with solid cancers. Cystic BMs tend to have a worse prognosis and are often more malignant than solid BMs.
Traditionally, surgery has been accepted as a reliable treatment for large, single BMs. 15 However, in recent years, stereotactic radiosurgery (SRS) and fractionated stereotactic radiotherapy (SRT) have emerged as safe and effective treatments for BMs.16,17 Hongwei Wang and his team found that the mOS for patients with cystic BMs treated with stereotactic cyst aspiration combined with SRS was 19.5 months. 18 However, most reports on the treatment of cystic BMs have been individual case reports or retrospective reviews with small samples. There are not many clinical cohort studies directly comparing the prognosis of solid and cystic BMs after brain radiotherapy.
Previous reports have shown that cystic brain metastases predominantly originate from the lung and breast cancer.
9
This was confirmed in our sample. 85% of cystic brain metastases in our study originated from lung or breast cancer. However, compared with solid BMs, there were some differences within subgroups. The proportion of breast cancers is higher in cystic BMs than in solid BMs (28% vs 13%), while it is relatively lower in lung cancers (57% vs 82%) (
These findings provide some insight into the therapeutic options. Previous data suggest that the OS of cystic BMs is relatively better than that of solid BMs after systematic treatment, especially in patients undergoing
Cystic BMs is frequently detected in patients with
These preliminary findings prompted us to further investigate the role of radiotherapy and provide a basis for the choice of RT model in different types of lung BMs. Multivariate analysis showed that BED was an independent prognostic factor for both cystic and solid BMs. In addition, after adjustment for confounders, BED, total dose and fraction were independent risk factors for OS of solid BMs. The Radiation Therapy Oncology Group (RTOG) recommends the standard practice of using SRS (stereotactic radiosurgery, 18-24 Gy) for solitary BMs, typically delivering a high dose in a single session.
26
For multiple BMs, the standard approach is whole brain radiotherapy (WBRT) combined with or without SRS. WBRT is typically delivered in fractionated treatments, with daily doses of 1.8-2.0 Gy and a total dose typically in the range of 30-40 Gy.
27
The results of this study suggest that a biologically effective dose (BED) of ≥60 Gy provides a survival benefit for patients with solid BMs after brain radiotherapy. Thus, HFRT and WBRT plus stereotactic boost may also improve survival in patients with BMs due to high BED. We recommend concurrent
In addition, in the cystic BMs group, smaller BM size (
Due to its retrospective nature, our study inevitably has the following limitations. Firstly, the number of patients is limited, especially for diseases other than lung cancer. Secondly, some patients did not have sufficient follow-up imaging data, which prevented evaluation of the local control rate in this study and may have affected the results of iPFS.
Conclusion
The prognosis of brain radiotherapy for patients with
Footnotes
Author Contributions
Conceptualization, Zilong Yuan, Desheng Hu and Guang Han; Data curation, Lu Sun and Shouyu Lin; Funding acquisition, Guang Han; Investigation, Wei; Methodology, Yi Peng; Software, Lu Sun, Jianping Bi, Zilong Yuan, Ying Li and Yi Peng; Supervision, Desheng Hu and Guang Han; Validation, Wei; Writing – original draft, Lu Sun and Shouyu Lin; Writing – review & editing, Jianping Bi and Ying Li.
Declaration of Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Chinese Society of Clinical Oncology (CSCO) Cancer Research Fund Project (Y-Young2022-0325), PARP Inhibitor Oncology Research Funding of China Anti-Cancer Association (CACA) (CETSDHRCORP252-4-004) and Cancer Precision Radiotherapy Spark Program of China International Medical Foundation (No. 2019-N-11-02).
