Abstract
Purpose
To date, only a few studies have investigated the role of molecular alterations in cancer recurrence. This exploratory study aimed to evaluate the impact of molecular alterations on the time and site of recurrence in patients with stage I–IV CRC and to identify the risk factors predicting recurrence-free survival in colon cancer.
Methods
A total of 270 patients were retrospectively included. We assessed the full RAS status using Sanger and pyrosequencing. MSI status was determined by immunohistochemical analysis. Molecular alterations were correlated with recurrence timing (early or late), recurrence patterns, and recurrence-free survival. Statistical analysis was performed using the Kaplan–Meier method and the log-rank test.
Results
Of the 270 patients, 85 (31%) experienced recurrence, among whom 53% had mutant full RAS status, 48% had KRAS mutations, and 31.4% had KRAS p. G12V mutation subtype. Compared with those with late recurrence, patients with early recurrence were significantly older (P = 0.02) and more likely to have poorly differentiated tumors, a higher rate of positive lymph nodes, KRAS mutations, and especially KRAS p. G12V mutation variant. RAS mutation status, KRAS mutations, and rare mutations are more common in patients with lung cancer recurrence. Multivariate logistic regression analysis revealed that differentiation, perineural invasion, full RAS mutation status, and KRAS codon 13 mutations were independent factors for recurrence-free survival in colon cancer.
Conclusion
In this cohort, the timing and patterns of recurrence appeared to be associated with the patient’s molecular profile. KRAS codon 12 mutations were the worst predictors of recurrence-free survival at all stages in our population.
Introduction
Colon cancer (CC) is one of the most common types of cancer worldwide. It ranks third in terms of incidence, with 1.9 million new cases diagnosed in 2020. 1 In Morocco, CC is a major public health issue given the remarkable and continuous increase in its incidence and mortality rates. According to GLOBOCAN, 4558 new colon cancer cases and approximately 1386 deaths were recorded in 2020. 1 The risk of recurrence raises concerns about postoperative surveillance to improve patient survival through earlier detection of local and metachronous recurrence at a curable stage. In colon cancer, approximately 15%–25% of patients present with synchronous metastasis at the time of diagnosis, and approximately 25%–50% develop metachronous metastasis or distant recurrence.2-5 Previous reports have indicated that 80% of recurrences usually appear in the 3 years following curative surgery, 6 and 95% of them occur in the first 5 years. 7 Early recurrence was defined as recurrence within 2 years of surgery. The most common sites of distant recurrence patterns are the liver (20%-50%), followed by the lungs (5-15%), peritoneum (15%), and bone (3% to 7%).8-11 Local recurrence (LR) occurs in 4%–11.5% of patients. 12 Many studies have demonstrated that recurrence is a major concern in stages I–III CC because it is strongly associated with shorter disease-free survival, and poor prognosis negatively affects patient survival.13,14 Furthermore, in stage IV CC, the majority of patients die from distant recurrence, although advances in targeted therapies. 15
It is well established that a higher risk of recurrence, its typical patterns, and decreased recurrence-free survival (RFS) are profoundly influenced by the clinicopathological features of the tumor, such as primary tumor location, lymph node metastasis, and TNM stage. 16 However, recent studies have demonstrated that the molecular and mutation profiles of tumors are associated with a higher risk of recurrence and may affect the site of relapse and metastatic involvement.4,17,18
In colon cancer, common oncogenetic mutations are found in the mitogen-activated kinase (MAPK) pathway genes, including KRAS (40%-45%), BRAF (10%-15%), and the NRAS (2%-6%), and in the phosphatidylinositol 3-kinase (PI3K) pathway member, PIK3CA (20%)19-21 (p. 3). In the field of treatment, patients with metastatic colon cancer harboring KRAS and NRAS mutations do not respond to anti-epidermal growth factor receptor (EGFR) monoclonal antibodies (cetuximab and panitumumab). 22 The positive relationship between full Ras mutation status and poor overall survival is well established in the literature. 19 Furthermore, recent studies have demonstrated that mutated KRAS is associated with worse recurrence-free survival.15,23,24 It has also been reported that KRAS mutations influence not only the time of recurrence but also their patterns. According to Tie et al. KRAS mutation status was associated with increased lung recurrence in stage II–III colorectal cancer (CRC). 23 Yaeger et al. reported that lung, bone, and brain metastases occur significantly more often in patients with KRAS mutations after hepactectomy. 17 In addition, BRAF mutations have been reported to be associated with peritoneal spread, which may explain the poor prognosis of tumors with a BRAF-mutated status. 17
To date, few studies have thoroughly investigated the relationship between RAS mutations, RFS, recurrence timing, and recurrence patterns in colon cancer. All reports have focused on stage II–III CRC with KRAS exon 2 mutations 23 or on patients with liver metastasis (LM). 25 Furthermore, the effect of NRAS mutations on RFS and recurrence patterns remains unknown, and no study has evaluated the association between KRAS and NRAS mutation subtypes (exon/codon) and patient recurrence, which is now mandatory, especially since it has been shown that mutational subtypes of each gene are associated with poor prognosis factors and shorter overall survival.19,26 Therefore, in the present study, we investigated for the first time the impact of molecular alterations on the time and site of recurrence in patients with stage I–IV CRC. We identified the risk factors for recurrence-free survival.
Materials and Methods
Sample Size Calculation
To calculate the number of cases required for our study, we used a formula that has been widely used in cross-sectional studies: - N = sample size - For a 95% confidence level, Z-score = 1.96 - P = the estimated proportion of the population with the characteristic (.50) Margin of error = .05
At risk α = 5%, the N value should be equal to at least 225 patients (N = 225).
Study Design
This study enrolled patients with I–IV CC treated at the HASSAN II University Hospital Center of Fez between 2009 and 2021. A consecutive series of 270 cases was identified. Clinicopathological data, treatment protocols, and follow-up (site and time of the first recurrence) data were collected from the pathology database at the laboratory of anatomical pathology of the hospital. Follow-up was performed at 3-month intervals for the first 2 years and at 6-month intervals thereafter.
Inclusion and Exclusion Criteria
The 270 patients were included according to the following criteria: (a) patients with invasive colon carcinoma (tumor stage T1 and above) and (b) patients who underwent surgical resection at the Department of Surgery of the University Hospital HASSAN II of Fez. The exclusion criteria were as follows: (a) patients with rectal adenocarcinoma, (b) patients aged <18 years, (c) patients with unknown tumor status or tumor site, (d) surgery-related mortality, (e) death within 90 days of surgery, and (f) patients with incomplete follow-up records.
Histopathological Study
All tumor samples were subjected to routine pathological and histopathological examinations at the Department of Anatomical Pathology. All specimens were obtained from primary sites after first-line therapy. Tumor tissues were then fixed in formalin (10%) and embedded in paraffin. Hematoxylin and eosin-stained slides of formalin-fixed paraffin-embedded (FFPE) tumors were reviewed by pathologists to identify the histological characteristics of the tumor (histological type, histological differentiation, perineural invasion, and vascular invasion) and evaluate the percentage of tumor cells in the tumoral zone. All cases were staged according to the tumor, node, and metastasis (pTNM) classification system (AJCC 8th edition).
Molecular Analysis
All biomarkers were assessed using FFPE samples.
KRAS, NRAS, and BRAF Mutation Profiling
DNA Extraction
Formalin-fixed paraffin-embedded tumors were macrodissected from sections containing greater than 30% neoplastic cells. Genomic DNA was extracted using the QIAamp DNA FFPE Tissue Kit (Invitrogen) according to the manufacturer’s instructions and quantified by Qubit analysis.
Polymerase Chain Reaction and Direct Sequencing
DNA amplification of KRAS (exons 2, 3, and 4), NRAS (exons 2, 3, and 4), and BRAF (codon 15) was assessed by PCR using primers on either side of the regions of interest. The PCR products were then purified and sequenced using Sanger sequencing technology with a BigDye Terminator V3.1 Cycle Sequencing Kit (ABI Prism). The sequenced reaction products were subsequently purified using a BigDye® Xterminator™ KIT and analyzed on an Applied Biosystems 3500Dx Genetic Analyzer (Applied Biosystems).
Pyrosequencing
Pyrosequencing technology is the second method used in our laboratory to assess the RAS status of patients with colon cancer. The regions of interest (codons 12, 13, 59, 61, 117, and 146) were amplified by PCR using specific primers. After immobilizing the PCR products on the workstation, single-stranded DNA was prepared, and the sequencing primers were annealed according to the manufacturer’s instructions. The samples were analyzed using Pyromark Q24 software in AQ analysis mode to determine the mutation status of each tumor.
Analysis of the Microsatellite Status
The microsatellite instability (MSI) status was determined by immunohistochemistry as described previously. 27
Recurrences
Follow-up data, including patient outcomes and recurrence information, were collected for each patient. Tumor recurrence was studied according to the following parameters: cancer recurrence site (local, liver, lung, periteon, and other sites) and timing. The patients were divided into two periods of recurrence: early recurrence (<2 years) and late recurrence (≥2 years). RFS was defined as the time interval from initial treatment until the first locoregional and/or distant recurrence. Censoring was performed when the patient died or survived without recurrence at the last follow-up. Recurrence was confirmed by histological examination or imaging.
Statistical Analysis
Statistical analysis was performed using SPSS version 21(SPSS Inc., Chicago, IL, USA). The chi-square test or Fisher’s exact test was used to define the links between each mutation and recurrence pattern, as appropriate. An independent t-test was used to identify the potential association between mutation status and time of recurrence.
RFS was estimated using the Kaplan–Meier method, and differences in RFS were assessed using the log-rank test. The Cox proportional hazard regression model was used to evaluate the association between the relevant molecular and clinicopathological variables and RFS. All P-values were two-sided, and P < .05.
Results
Patients and Recurrence Characteristics
Patients and Recurrence Features.


Recurrence patterns in our cohort.
As shown in Table 1, compared with patients without cancer recurrence, those with cancer recurrence were more likely to have mucinous and poorly differentiated tumors (P = 0,05, and P = .03, respectively), less than 12 total lymph nodes (P = .05), presence of positive lymph nodes (P = .01), and perineural invasion (P = .005). Additionally, patients with cancer recurrence were more likely to be diagnosed at an advanced TNM stage (P = .04) and have undergone adjuvant chemotherapy (P < .001). Moreover, in our population, the development of cancer recurrence tended to be associated with the left tumor site (P = .08), with a difference that was close to significant.
Frequencies of Molecular Alterations and Recurrence
Molecular Alterations and Recurrence.

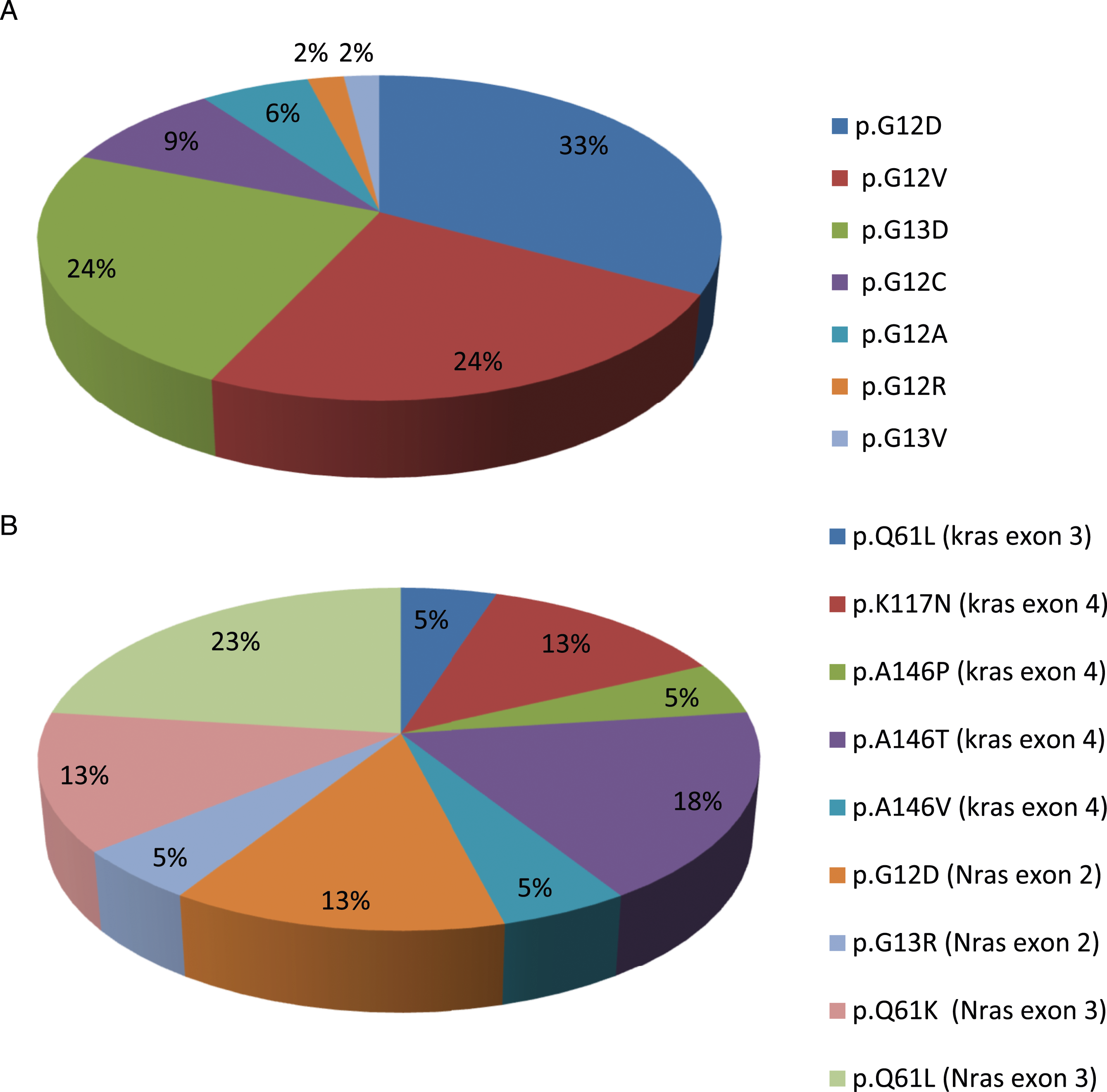
Frequencies of RAS mutation subtypes in our population. (A) Mutation subtypes in KRAS exon2. (B) Mutation subtypes outside KRAS exon 2 (Rare mutations).
Regarding the correlation between cancer recurrence and molecular alterations, patients with cancer recurrence were more likely to have RAS mutation status (n = 45; 53%; P = .002) and to carry KRAS mutations (n = 41; 48%; P = .002). Interestingly, KRAS p. G12V mutation was significantly more common in patients with cancer recurrence than in those with other KRAS exon 2 amino acid transitions (n = 11; 31%; P = .04). In contrast, there were no significant differences in the distribution of NRAS mutation status, KRAS codon 12 vs KRAS codon 13 mutations, rare mutation subtypes, and MSI status between patients with and without cancer recurrence.
Correlation between molecular alterations and clinical pathological features
Association Between KRAS and NRAS Mutation Subtypes and Clinicopathological Features.

Association of Rare RAS Mutations With Clinicopathological Features.

KRAS-mutated tumors were significantly correlated with female gender and the presence of positive lymph nodes. In total, 37% of these tumors had fewer than 12 lymph nodes removed, compared with 24% in RAS wild-type tumors. NRAS mutations were correlated with the presence of vascular invasion and <12 of removed lymph nodes.
As shown in Table 4, rare RAS-mutated tumors compared with WT tumors were more frequently poorly differentiated (82% vs 53%) and were associated with the presence of vascular invasion and a lower rate of removed lymph nodes (<12 resected lymph nodes).
Clinicopathological Features and Molecular Alterations Between Early and Late Recurrence of Colon Adenocarcinoma
Clinicopathological and Molecular Differences Between Early and Late Recurrence.

Regarding molecular alterations, full RAS status and KRAS gene mutations were more common in patients with late recurrence than in those with early recurrence (P = .03; P = .05, respectively). As a point of interest, the KRAS p. G12V mutation subtype was more likely to be found in the late recurrence group than in the early recurrence group (P = .03). There were no differences in the distribution of MSI status and NRAS mutations between the two groups.
Effect of KRAS and NRAS Mutation Status on the Recurrence Pattern
Recurrence Pattern According to Molecular Alterations.

Recurrence-Free Survival According to Clinicopathological Features and Molecular Biomarkers
Recurrence-Free Survival Analysis.


Kaplan–Meier recurrence-free survival curves according to molecular alterations. (A) Full RAS status, (B) KRAS gene status, (C) rare mutation status, and (D) KRAS exon 2 mutant subtypes.
Multivariate logistic regression analysis revealed that differentiation, perineural invasion, full RAS mutation status, and KRAS codon 13 mutations were independent factors for RFS. Table 7 presents the results.
Discussion
The majority of studies investigating the relationship between recurrence and patient molecular profiles have been conducted in local colon cancer patients and have focused only on KRAS gene status and liver recurrence. To the best of our knowledge, the present study is the first to report the impact of a patient’s molecular profile on cancer recurrence, time of recurrence, recurrence patterns, and recurrence-free survival in the largest cohort in Morocco and the Middle East and North Africa region (MENA region).
First, our results showed that full RAS status and the presence of KRAS mutations were significantly associated with cancer recurrence at all stages. These findings support the results of a previous study demonstrating that RAS mutations, especially KRAS mutations, correlate with poor prognostic factors and have worse oncologic outcomes with a higher risk of recurrence in both locally and advanced colon cancer stages.4,15,19 The strength of this study is that the KRAS G12V mutation subtype was significantly more frequent in patients developing recurrence after first-line therapy . According to the KRAS amino acid substitution, different signaling pathways are activated, which leads to distinct tumor progression and invasion.
Many studies have explored novel strategies that could predict early recurrence by integrating clinicopathological features and molecular alterations, which could be useful for the surveillance of colon cancer patients.28,29 In the present study, RAS and KRAS mutations were more common in patients with early recurrence than in those with late recurrence. Lan et al. reported that KRAS was among the top mutated genes in patients with both early and late recurrence. 30 Another study showed that KRAS mutations were detected in the cell-free DNA of CRC patients at all stages. Based on these results, the KRAS status may serve as a tool for the early detection of tumor recurrence. 31 The main strength of this study is that our results demonstrated that patients with KRAS G12V amino acid substitution were more likely to develop early recurrence than those with other KRAS exon 2 mutant subtypes, which has not yet been reported. These results will be helpful in providing personalized follow-up strategies for KRAS exon 2 mutations based on amino acid substitutions.
However, our study demonstrated no difference in the distribution of NRAS mutations between patients with early and late recurrence. Lan et al. reported the same result regardless of tumor location; however, they showed that among left-sided colon cancer patients, NRAS mutations occurred more frequently in patients with early recurrence. 30 In the present study, MSI status was not correlated with the timing of recurrence, which may be related to the good prognostic value of MSI tumors reported in previous studies.27,32
Regarding clinicopathological features, we reported that patients with early recurrence were significantly older than those with late recurrence. However, Lan et al. did not find any association between tumor grade and recurrence. 30 In addition, patients with poorly differentiated tumors were more likely to develop early recurrence, which is similar to the findings of Luo et al. 13
Many studies have demonstrated that RAS mutations, especially KRAS mutations, not only affect the time of recurrence but also their patterns. Vauthey et al. reported that RAS mutation status is associated with an increased incidence of lung recurrence. 33 This result is supported by Kemeny et al. 34 Tie et al. showed that KRAS mutations in stages II and III CRC were more prevalent in lung recurrences but less prevalent in liver recurrences. 23 We also investigated the association of KRAS and NRAS mutation subtypes and MSI status with recurrence patterns in our cohort. We found that patients with full RAS mutation status and KRAS mutations were more likely to develop lung recurrence. Our study confirmed that the RAS mutation status was predictive of the involvement of this site. A novel finding of our study is that rare RAS mutations are significantly more common in patients with lung recurrence. On the basis of these results, it appears that rare RAS mutations could be used as predictive factors for lung recurrence at all stages. Further investigations using a large sample of patients with rare RAS mutations are required to validate these results. However, we did not find any association between the NRAS gene status and recurrence patterns. Our findings are supported by those reported by Tie et al. 23
This study is the first to report RFS according to full RAS status, KRAS mutation subtypes (codon/exon), and rare mutations. The majority of studies have evaluated the prognostic impact of only KRAS gene status and have been conducted in patients with II–III CC.15,35 Our cohort analysis revealed that RAS mutation status was a predictive factor for worse RFS. Similar results were obtained in a large cohort of 2720 patients by Sinicrope et al. 36 In addition, KRAS mutant status was associated with a shorter RFS (P < .001) in our population. In line with our study, Vauthey et al. found that patients with KRAS mutant CRC experienced worse RFS (P = .005). 33 This result has been confirmed by many studies.20,37,38 Furthermore, it has been suggested that KRAS mutations reflect a more invasive tumor process that tends to recur early and after metastatic resection, which explains their impact on RFS. 39
We found that rare mutations appeared to have worse RFS compared with the wild-type status. However, this finding has not yet been reported in the literature. In this study, we also documented that KRAS codon 12 mutant status was associated with the worst RFS compared with the KRAS codon 13 mutant and wild-type statuses. Based on our analysis, it appears that the prognostic value of KRAS codon 12 mutations is related to the association between KRAS G12 V substitutions and early recurrence. Consequently, patients with this mutation require specific surveillance modalities. Our findings are consistent with those of previous reports. 40 Yoon et al. evaluated a cohort of 2478 patients receiving adjuvant FOLFOX alone or in combination with cetuximab and reported that KRAS mutations in codons 12 or 13 were associated with shorter DFS. 41 The mutational status of KRAS codons 12 and 13 appears to be a predictive biomarker for the benefit of adjuvant chemotherapy.
This study has both strengths and limitations. First, this was a single-institution study, which may limit the generalizability of our results to the entire population of our country. Second, detailed data on chemotherapy were not available in this study. Patients who received oral chemotherapy, intravenous chemotherapy, or incomplete chemotherapy were classified as having received adjuvant chemotherapy. Third, other molecular biomarkers strongly involved in CC carcinogenesis (PI3K and P53…), were not tested in the present study; hence, we did not analyze the effect of these variables on the prognosis of our patients. Above all, our study is the first and largest in Morocco and the MENA region.
Conclusion
Our study demonstrated that patients with early recurrence had specific clinicopathological features and harbored more KRAS G12 V substitutions. Patients with KRAS mutations are more likely to develop lung cancer recurrence. Cox regression analysis demonstrated that KRAS codon 12 mutations were the worst predictors of RFS at all stages in our cohort. In contrast, there was no correlation between MSI status and recurrence risk in cases of colon cancer.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

