Abstract
Breast cancer has the highest incidence among female malignancies, significantly impacting women’s health. Recently, numerous HER2-targeted therapies have achieved excellent clinical outcomes. Currently, anti-HER2 drugs are divided into three main categories: monoclonal antibodies, small-molecule tyrosine kinase inhibitors, and antibody-coupled drugs (ADCs). The main toxic side effects of small molecule TKI-based therapy are diarrhea, hand-foot syndrome, rash, nausea, and vomiting. Diarrhea is a potential predictor of tumor response, affecting up to 95% of cancer patients treated with TKIs. Severe gastrointestinal toxicity can result in the need for dose reductions and treatment interruptions. This not only compromises the efficacy of TKIs but also deteriorates human nutrition and quality of life. The majority of individuals develop diarrhea within 7 days of starting treatment, with approximately 30% developing grade 3 or higher diarrhea within 2-3 days of starting treatment. The severity of diarrhea typically correlates with the dosage of most TKIs. Current prevention and management strategies are primarily empirical, focusing on symptom alleviation rather than addressing the toxicological mechanisms underlying TKI-induced diarrhea. Consequently, anti-diarrheal drugs are often less effective in managing this condition in cancer patients receiving TKIs. Moreover, our understanding of the toxicological mechanisms responsible for such diarrhea remains limited, underscoring the urgent need to identify these mechanisms in order to develop effective anti-diarrheal medications tailored to this specific context. This review aims to elucidate management approaches and mechanisms for diarrhea induced by TKIs during HER2-positive breast cance.
Plain language summary
Breast cancer has the highest incidence among female malignancies, significantly impacting women’s health. In recent years, numerous HER2-targeted therapies have been developed, including lapatinib, neratinib, tucatinib, and pyrotinib. However, second-generation tyrosine kinase inhibitors (TKIs) often cause severe gastrointestinal toxicity, necessitating dose reduction and treatment interruption. This not only compromises the efficacy of TKIs but also deteriorates nutritional status and quality of life. Diarrhea is a potential predictor of tumor response and is the second most common adverse event, affecting up to 95% of cancer patients treated with TKIs. Approximately 30% of these patients experience grade 3 or higher diarrhea within just 2-3 days of initiating TKI treatment. The severity of diarrhea typically correlates with the dosage of most TKIs. Current prevention and management strategies are primarily empirical, focusing on symptom alleviation rather than addressing the toxicological mechanisms underlying TKI-induced diarrhea. Consequently, anti-diarrheal drugs are often less effective in managing this condition in cancer patients receiving TKIs. Moreover, our understanding of the toxicological mechanisms responsible for such diarrhea remains limited, underscoring the urgent need to identify these mechanisms to develop effective antidiarrheal medications tailored for this specific context. This review aims to elucidate management approaches for diarrhea induced by HER2 tyrosine kinase inhibitors during breast cancer treatment.
Introduction
Breast cancer has surpassed lung cancer to become the most prevalent cancer worldwide and holds the highest incidence rate among women. 1 HER2 is used as a therapeutic target and HER2 mutations are present in about 15%–20% of breast cancer patients. 2 HER2 is a tyrosine kinase receptor and abnormal activation of these kinases prevents apoptosis, cell overgrowth, and vascular growth in epithelial cancer. 3 Previously, patients with her2-positive breast cancer had extremely limited treatment options, rapid progression, poor prognosis, and high rates of recurrence and metastasis. 4 Recently, the use of HER2-targeted drug has greatly improved the median progression-free survival of patients, prolonging survival. 5
TKIs, which include small molecule inhibitors, offer several advantages such as reduced cardiotoxicity, oral administration, and better tolerability than cytotoxic drugs. 6 However, oral targeted therapies can independently cause a range of gastrointestinal adverse reactions. Although these toxicities, especially diarrhea, are usually well managed clinically, they can still affect patients’ quality of life. 7 Diarrhea typically occurs early in treatment and if severe diarrhea is not treated promptly and effectively, it can cause serious complications, resulting in reduction and interruption of targeted drug therapy doses. 8 Clinicians may overlook the dangers of mild diarrhea, thinking it is tolerable. However, when treatment continues for months or even years, mild toxicity can cause mental and physical pain. 9
Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0

Furthermore, the potential mechanisms underlying molecularly targeted drug-associated diarrhea remain under investigation and likely multifactorial in nature. Initially, some scholars posited that diarrhea induced by molecular targeted drugs shared a similar pathogenesis to that of chemotherapy. 11 However, subsequent studies revealed distinct treatment-specific mechanisms between diarrhea induced by HER2 TKIs and chemotherapy to induce diarrhea. Diarrhea related to molecular-targeted drugs typically arises in the small intestine, while there is no significant change in the colorectal, while chemotherapeutic drugs act in the colorectum, which indicates that the incidence sites of the two are completely different. 12 Although both have the process of direct mucosal damage, intestinal flora disorder and increase of pathogenic bacteria, chloride ion secretion change and increase of pro-inflammatory factors. 13 The incidence of diarrhea varies widely among individuals, and it is uncertain whether it is intestinal cells or enzymes in the gastrointestinal system are HER2 inhibitor-sensitive. Bowen suggested that EGFR-TKIs might induce diarrhea by preventing intestinal epithelial cells from transducing signals, which would result in intestinal mucosa atrophy. 14 In addition, Loriot showed that increased chloride secretion and disruption of barrier function in EGFR-TKIs can lead to diarrhea. EGFR-TKIs may lead to mucosal injury, inflammation, and diarrheal adverse effects by disrupting the epithelial ion transport or barrier. As a result, it is imperative to focus on the pathological mechanisms and management of diarrhea associated with oral TKIs. However, combining targeted medications with traditional chemotherapy medications can make the side effects brought on by the latter worse. 15 Hyperproliferative cells make the gastrointestinal tract vulnerable to acute toxicity from chemotherapy. 16
However, currently, its treatment management follows that of chemotherapy-induced diarrhea. 17 The NCCN in the United States have established guidelines for chemotherapy-induced diarrhea, and current treatment for diarrhea focuses more on symptom severity management than on addressing the pathophysiological mechanisms causing dysfunction. 18 Therefore, gaining a deeper insight into the mechanisms behind diarrhea caused by HER2 TKIs is crucial. This will help in developing robust strategies for the evaluation, prevention, and management of gastrointestinal toxicity. Understanding these mechanisms can lead to more effective approaches to mitigate these adverse effects. 19 The aim of this review is to outline its pathogenesis and therapeutic management.
Potential Mechanisms of Diarrhea
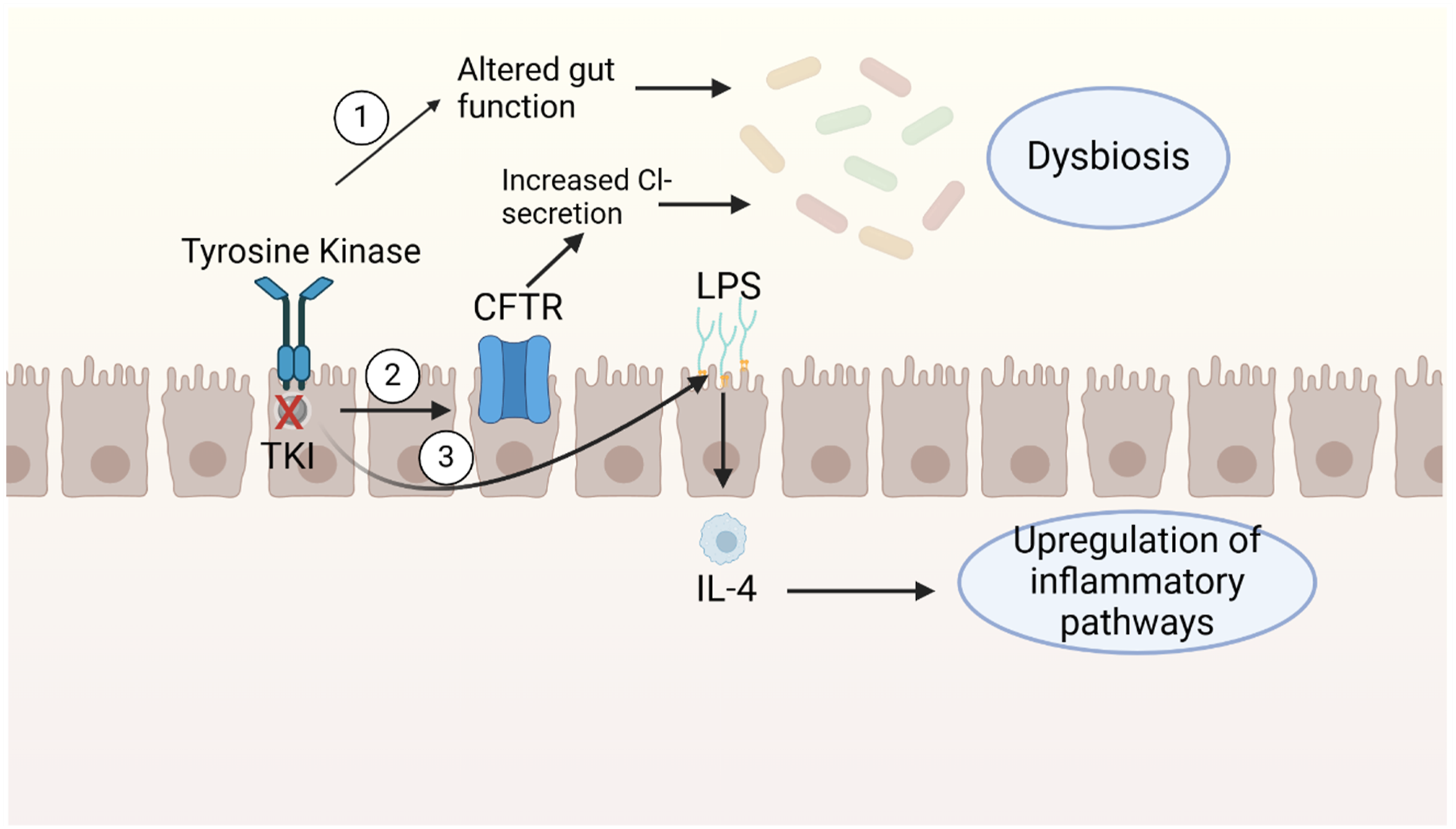
While some potential mechanisms of HER2 TKI-induced diarrhea have been identified, the precise pathogenic mechanism remains unknown. The following figures illustrate some mechanisms (Figures 1–3). EGFR-TKIs can cause diarrhea by affecting the secretion of fluids in the colon. When EGFR receptors are unable to control the secretion of chloride, it can result in the buildup of chloride in the colon, leading to water retention and ultimately causing diarrhea. CFTR, cystic fibrosis transmembrane conductance regulator, EGFR, endothelial growth factor receptor, CACC, calcium-activated chloride channel, EGFR-TKIs, epidermal growth factor receptor-tyrosine kinase inhibitors. Direct damage to normal mucous membranes; Source: Created with Biorender.com. Intestinal dysfunction, chloride channel dysfunction, and inflammation activation result in modifications to the gut microbiota; CFTR, cystic fibrosis transmembrane conductance regulator, IL-4, interleukin-4, LPS, lipopolysaccharide.

Secretory Mechanisms
One proposed theory is that HER2 TKIs associated diarrhea is caused by excessive chlorine secretion and insufficient sodium absorption. 20 While chloride secretion is essential for maintaining moisture in the gastrointestinal tract, excessive secretion can lead to diarrhea. 21 Research indicates that the overall degree of chloride secretion in the intestinal epithelium is influenced by multiple regulatory factors at both intracellular and extracellular levels. 22 In the normal colon, chloride in the lumen promotes the entry of water into the intestinal lumen. Cyclic AMP and intracellular calcium are examples of intracellular messengers that directly stimulate the absorption of sodium and the release of chloride. The primary mechanism behind HER2 TKI-induced diarrhea involves the activation of parietal membrane chloride (Cl -) and basolateral membrane potassium (K+) channels in the intestinal epithelium. 23 EGFR functions as an inhibitory controller of chloride secretion. However, EGFR inhibitors enhance chloride secretion by obstructing this controlling mechanism and stimulating the potassium (K+) channels in the basolateral membrane. This leads to the occurrence of chloride-secretory diarrhea.11,13 EGFR TKI therapy (containing gefitinib, lapatinib and afatinib) directly activated potassium channels and CFTR chlorine channels. 23 Two key pathways control chloride secretion in intestinal epithelial cells: The cAMP-dependent system activates delayed reactions, while the Ca2+-dependent pathway triggers quick and immediate responses. 24 EGF suppresses the activity of Ca2+-dependent chloride secretion by attaching to the EGFR receptor. 22 Based on these studys, EGFR inhibitors interfere with the inhibitory mechanism of EGFR on chloride ion secretion, leading to an accumulation of excessive chloride in the endolumen23,25 (Figure 1). The surplus chloride creates an electrochemical stimulus for the passage of sodium down the paracellular pathway, leading to the buildup of water (secretory diarrhea). 26 There was one animal study that lapatinib-induced diarrhea may be attributed to the loss of chloride ions in the gastrointestinal tract. 27
Direct Damage to Mucous Membranes
Another theory suggests that diarrhea may result from direct damage to normal mucous membranes, including damage to the colonic crypts, which affects water absorption in the colon. This damage can impair the absorption of water, electrolytes, and other substances, including lipids. 28 Multiple pieces of evidence substantiate this notion. Oral drugs are more likely to cause diarrhea than monoclonal antibodies, indicating a direct effect on lining of digestive system or inhibition of specific tyrosine kinase being targeted. Additionally, The expression of EGFR in the gut is significantly high. Blocking these receptors in the colon results in reduced cell proliferation and the number of blood vessels in the intestinal villi. These structural changes may result in mucosal atrophy and reduced intestinal absorption capacity 23 (Figure 2). Studies have shown that this diarrhea is associated with slower growth and recovery of the intestinal lining, resulting in mucous membrane decreased. This is caused by the suppression of the ErbB pathway, which impacts the growth of intestinal cells, the transportation of nutrients and electrolytes, the production of brush border enzymes, and Regeneration of epithelial tissue. 13 Currently, The relationship between damaged mucosal barriers and intestinal inflammation is uncertain, as it is unknown if the inflammation causes poor barrier function or if poor barrier function contributes to the inflammation.
Changes of Intestinal Microbial Community
Another mechanism associated with the onset of diarrhea is changes in the normal gut microbiome that microbial metabolism affects absorption and other gut functions. 29 During SM-TKI treatment, preliminary evidence suggests alterations in the microbial composition of those who are suffering more severe diarrhea. 30 Microbial communities in nature have a crucial function in assimilating diverse proteins and inhibiting the colonization of harmful bacteria in the gastrointestinal system. Certain substances can cause diarrhea by altering the natural microbiome. It has been observed that when the microbiome is disrupted by antibiotics, both diarrhea and treatment outcomes are affected. 31 Preclinical studies of lapatinib have shown that rats treated with lapatinib exhibit significantly reduced microbial diversity. 32 Additionally, a reduction of beta-proteobacteria in the gut found after lapatinib treatment. Therefore, various factors such as impaired intestinal function, pelvic radiation therapy (RT), gastrointestinal (GI) surgery, dietary choices, antibiotic usage can lead to this outcome.
The gut microbiota are connected to the inflammatory response and the release of chloride ions. 33 However, there is relatively weak evidence that modifications to the normal gut microbiome might result in chloride channel malfunction, despite some research suggesting that pathogenic bacteria or probiotics can affect chloride ion releas. 34 Further evidence that alterations in gut microorganisms impact SM-TKI-induced diarrhea is provided by the possibility that diarrhea may result from elevated chloride production in the intestinal lumen of patients taking the drug. Excessive growth of harmful bacteria and toxin buildup can result in diarrhea, according to studies. 32 An investigation of lapatinib revealed a greatly reduction in microbial diversity in rats receiving lapatinib.34,35 This suggests a close relationship between microbes and inflammatory activation and chloride secretion. The microbiota has been employed as a gauge for SM-TKI treatment response. These data demonstrate the close relationship between gut flora disruption and diarrhea incidence. 36
Inflammation
A preclinical study on neratinib-induced diarrhea revealed that budesonide effectively decreased colon damage and diarrhea while elevating anti-inflammatory interleukin 4 levels, suggesting a potential association with inflammation. Data from Liu and Kurzrock supported these findings, demonstrating a significant alleviation of diarrhea following budesonide administration. 37 Additionally, studies have shown that cells treated with lapatinib express less JAM-A which is widely prevalent in healthy epithelial cells. Lapatinib’s influence on TJP integrity results in increased intestinal permeability. 38 This heightened permeability can lead to lipopolysaccharide (LPS) recruitment, triggering the release of inflammatory cytokines that activate the immune response. 39 Moreover, the imbalance between symbiotic and pathogenic bacteria further contributes to the pro-inflammatory state.
Efflux Transporter Dysfunction
The majority of EGFR-TKIs have strong affinity for ATP-binding cassette (ABC) efflux transporters, particularly those belonging to subfamily B (ABCB), which, when dysregulated, can lead to altered cellular availability of these drugs. 26 Both first and second-generation inhibitors have been shown to interact with ATP-binding cassette (ABC) transporters. 40 When drug transporters become saturated and EGFR inhibitors share efflux transporters or enzymes with other therapeutic agents, drug accumulation in the intestine can occur, resulting in gastrointestinal toxicity. Lapatinib and neratinib are known to exert inhibitory effects on ABC transporters, with Lapatinib functions on these transporters as both a substrate and an inhibitor. In cancer cells, lapatinib has been shown to reverse ABCB1 and ABCG2-driven drug resistance. 41 It has also been discovered that neratinib reverses ABCB1-mediated drug resistance by preventing the ATPase activity of ABCB1. 42 Additionally, with the exception of erlotinib, diarrhea incidence has been observed to increase with escalating doses of oral kinase inhibitors. 43
Changing Transport in the Colon
Loperamide has been observed to reduce the number of bowel movements, suggesting that dyskinesia may be the cause of induced diarrhea. Different substances may target the Cajal cells, which control colonic motor activity. The intestinal epithelium’s basolateral (K+) and chloride (Cl-) channels are activated, which is the main mechanism by which HER2 TKI-induced diarrhea occurs. 23 Through membrane hyperpolarization, enhanced K+ conductance stimulates Cl-channels. 44
TKI Associated with Diarrhea
Lapatinib
HER2-Targeted TKI-Induced Diarrhea in Key Clinical Trials in Breast Cancer.

“NA” means unknown; “AI” means Aromatase Inhibitors.
Pyrotinib
Pyrotinib is a second-generation, well-absorbed, irreversible tyrosine kinase inhibitor that targets EGFR, HER-2, and HER-4. 85 The Chinese National Drug Administration has approved it for use alongside capecitabine in patients who have previously undergone treatment with anthracyclines or taxanes. By covalently engaging ATP binding sites, it inhibits self-phosphorylation, hence disrupting the activation of G1 cycle of tumor cells and limiting tumor development. 86 In a Phase I study, the dose-limiting toxicity (DLT) was grade 3 diarrhea. CBR was 61.1% and ORR was 50.0%. PFS was 35.4 weeks on average. The ORRs of individuals who had not treat with trastuzumab and those who had received it were 33.3% and 83.3%. 28 Furthermore, PHEEBEE, which included 267 patients who had previously received trastuzumab, revealed that patients treated with pyrotinib plus capecitabine have a greatly mPFS than those received lapatinib plus capecitabine (12.5 months vs 6.8 months) and individuals received pyrotinib plus capecitabine had an ORR that was greater (67.2% vs 51.5%) than those received lapatinib plus capecitabine or any combination of two. 55 Compared to lapatinib, pyrotinib increased PFS despite showing higher toxicity. Diarrhea accounted for 96.9% of common AEs, followed by palmar and plantar swelling, vomiting, nausea, anorexia, oral mucositis. 86 As shown in Table 2, according to a Phase II trial, individuals receiving pyrotinib alone experienced an incidence of diarrhea of 44.7%, whereas those receiving capecitabine experienced a higher rate of up to 95%. 87 Up to 95% of patients receiving capecitabine experienced diarrhea and hand-foot syndrome as their most frequent side effects; 31% of individuals experienced grade 3 adverse events in PHOBE study. 55 The incidence of all grades increased with the administration of neratinib and pyrotinib (83% and 97%, respectively), probably as a result of effects on EGFR. 88 In PANDORA trials, the incidence of ≥3 diarrhea is 38.2%. 63 Patients receiving pirotinib, the median time to start diarrhea is 8 days, whereas in the group receiving a placebo, it was 71 days (23.0-178.0). Both groups experienced grade 3 diarrhea for 1.0 (1.0-1.0) days on average, and each group experienced 3.0 (2.0-6.0) and 1.0 (1.0-2.0) days on average cumulatively. 64
Neratinib
Neratinib is an oral, irreversible tyrosine kinase inhibitor of HER1, HER2, and HER4. Kinase activity is inhibited when neratinib covalently binds to cysteine residue. Inhibition of phosphorylation of ErbB family and ERK and Akt.89,90 It shares structural similarities with the strong EGFR inhibitor EKB-569, however it has a stronger inhibitory effect on EGFR and HER2. When neratinib and target tyrosine kinase are combined, the result is permanent inhibition that lasts long after the medication leaves the cellular environment. 91 During the G1-S phase transition, neratinib’s control over downstream signal transduction causes standstill, ultimately leading to downregulation of cell proliferation. 89 According to the neratinib study, 28% of individuals with CTCAE-grade 3/4 diarrhea and 85% of those with metastatic receiving neratinib experienced full CTCAE grade diarrhea. 92 Neratinib considerably increased DFS in the Phase III ExteNET study as compared to placebo. 66 The side effects of neratinib monotherapy in a randomized Phase II trial were diarrhea, nausea, vomiting, decreased appetite. Grade III–IV diarrhea was experienced by 28% of individuals receiving neratinib. 60 The neratinib plus capecitabine group was linked to a higher frequency of diarrhea compared to the lapatinib plus capecitabine group: full grade diarrhea was 83% vs 66% for lapatinib plus capecitabine; Level III-IV was 24% vs 13%. When it came to toxicity, 96% of individuals receiving neratinib in the Phase III ExteNET study developed diarrhea. Of these, 55% and 41%, respectively, experienced grade 1-2 and 3-4 diarrhea, which is believed to be secretory diarrhea brought on by EGFR suppression. 66 Typically, adverse effects are more common during the first cycle and become less frequent after, especially with the addition of loperamide treatment, which greatly reduces the incidence of diarrhea. For most patients, diarrhea resolves after about 2 weeks. 93 Up to 30%-53% of patients in the original Neratinib trial experienced stage G3 diarrhea in the absence of preventative treatment, and 10% of those receiving Lapatinib with Capecitabine developed grade III–IV diarrhea.60,66 In the MBC monotherapy and combination therapy studies, the incidence of diarrhea in neratinib appeared to be high (83% vs 93%), suggesting that diarrhea is a targeted effect of neratinib.59,94 This negative outcome could be explained by EGFR’s role in calcium-dependent chloride ion transport, and the hypothesized mechanism of neratinib action includes EGFR inhibition that may lead to secretory diarrhea, with EGFR antagonism considered to be the most likely mechanism.95-97 It is currently advised to treat neratinib-associated diarrhea during the first one to two cycles of treatment because Phase II control studies have demonstrated that these cycles minimize the frequency, intensity, and length of diarrheal incidents when compared to those seen in ExteNET. 98 A higher incidence of all grades of diarrhea (83% and 97%, respectively) was observed with the use of neratinib and pyrotinib in comparison to other TKIs. This led to more frequent and severe diarrhea, especially when adjuvant therapy was used, which may have an impact on treatment adherence. The impact on EGFR could be the cause of this. Budesonide or coletepol added to loperamide prophylaxis may lessen the likelihood that neratinib may need to be stopped owing to diarrhea. 9
Afatinib
Afatinib (formerly BIBW 2992) is a novel oral bioavailable irreversible TKI that binds to the entire HER protein family, including HER-1, HER-2, and HER-4. 99 The Phase I trial demonstrated that afatinib at a daily dose of >50 mg increased drug-associated AEs, including diarrhea. In patients who had previously received gefitinib or erlotinib, the trial compared afatinib with a placebo. The findings indicated that rash, diarrhea, stomatitis, and nail disease are prevalent grade 3 adverse events. 100 In LUX-Lung 2, individuals receiving 50 mg/day of afatinib, the frequent AES were diarrhea and rash, both percentages decreased by around 7% when the dose was lowered to 40 mg/day. About 95% and 14.4% of cases of G1-2 and G3-4 diarrhea, respectively, are reported (Table 2). 101 There are reports of around 95% and 14.4% of instances with G1-2 and G3-4 diarrhea, respectively. 70 In one study, 77% of individuals, diarrhea is the most prevalent adverse event connected to medication. 102 Despite the encouraging results of Phase I and Phase II studies, the PFS for the afatinib and trastuzumab groups in LUX-Breast1 study was 5 months and 6 months, respectively. The IDMC conducted an adverse risk-benefit analysis, which led to the recruitment of candidates starting in August 2010 and ending in April 2013. The recommended course of treatment for patients with metastatic breast cancer that is HER2-positive is still trastuzumab-based therapy. 71 LUX-Breast 3 evaluate the treatment of afatinib as a monotherapy or in combination with vinorelbine vs individuals with progressive brain metastases after failure of investigator-selected trastuzumab or lapatinib. The results showed no difference in benefit between afatinib-treated individuals and those given the investigator-selected treatment. However, adverse events occur frequently and treatment with afatinib appears to be less well tolerated. 73 There are currently no plans to further develop afatinib for breast cancer.
Tucatinib
Tucatinib is a oral HER-2-TKI that is more than 1000 times more potent against HER-2 than EGFR, contains little inhibition of EGFR and is very specific to the kinase domain of HER2, thus showing reduced EGFR-related toxicity. 103 Tucatinib, when combined with trastuzumab and capecitabine, can lower the risk of intracranial progression or death by two-thirds, as well as the risk of death by almost half in patients with HER-2 positive breast cancer and BM. 104 Tucatinib approved to treat patients with advanced HER2 unresectable or MBC. 105 More than half of individuals receiving tucatinib monotherapy developed diarrhea, while 81% of patients treated with tucatinib in combination with trastuzumab and capecitabine developed diarrhea, indicating that both Tucatinib and capecitabine caused diarrhea. Tucatinib combined with trastuzumab and capecitabine has shown good anti-tumor effects.74,105 According to studies, tucatinib increases the risk of diarrhea and raises transaminase levels. It may be that Tucatinib is highly selective to the HER2 kinase domain while little inhibiting EGFR could change the toxicity profile. 106 When compared to medications that simultaneously inhibit EGFR, tucatinib offers the possibility of dual HER2 inhibition with possibly less harm. 66 When compared to a placebo combination, the tucatinib combination significantly decreased the risk of disease progression and death in patients with brain metastases from metastatic breast cancer. 105 The diarrhea in the Phase III NALA trial was more severe than previously noted (24% of grade 3 diarrhea, Table 2). 74
Treatment and Management
HER2-TKIs induced diarrhea is a significant yet often overlooked side effect requiring effective management. Currently, the management of EGFR-TKIs associated diarrhea typically involves fluid rehydration and the use of anti-diarrheal agents like loperamide. In extreme circumstances, changing the dosage or temporarily stopping TKIs may be required. The 2019 Chinese Expert Consensus on EGFR-TKIs ADR Management suggests loperamide, montmorillonite, and probiotics as preferred treatments for TKIs-associated diarrhea, with codeine (30 mg BID) being an option for severe cases. 10 Additionally, in 2021, the CSCO Breast Cancer Expert Committee issued an expert consensus on adverse event management in HER2 family TKIs-treated breast cancer patients. 107 Moreover, the Breast Cancer Treatment Management chapter of the CSCO Guidelines, updated in 2024 with safety data from the PANDORA study, provides insights into the primary prevention of pyrotinib-induced diarrhea.
Non-Drug Management Strategies
EGFR-TKI-induced diarrhea is frequently treated with dietary modifications and a reduction in the targeted drug’s dosage, and does not result in treatment interruption in most patients. 108 For instance, in patients with recurrent NSCLC, the study of erlotinib showed that diarrhea resulted in a 5% dose reduction and a 6% cessation of treatment. 109 Similar results were shown in pooled analyses of diarrhea in patients receiving lapatinib monotherapy; only 2% of reported episodes of diarrhea resulted in treatment stoppage, and the majority of cases were of low CTCAE grade. Such diarrhea is generally controllable with dietary modifications and the use of antidiarrheal medications. 84 However, early symptom management and adequate fluid intake are crucial to prevent symptom escalation and avoid the need for EGFR TKIs dose reduction or discontinuation.110,111 Recommendations for dietary adjustments to manage ErbB TKIs-induced diarrhea include adopting a whole grain diet, incorporating items like bananas, rice, applesauce, and toast, increasing water intake, and beverages containing caffeine or lactose. Also, patients should avoid foods like fatty, spicy, and fried foods that aggravate their symptoms. To avoid dehydration, it is advised to up fluid consumption to two liters per day. To reduce the risk of hyponatremia and hypokalemia from electrolyte loss via diarrhea, these fluids should contain sugar or salt. 112 After symptoms improve, dietary adjustments can be gradually reverted to a normal regimen. 113
Furthermore, close monitoring of patients during the initial weeks of treatment is crucial for diarrhea management, as diarrhea typically manifests within the first week of TKI therapy. Early symptom control and adequate fluid intake are imperative to forestall symptom exacerbation and avoid the need for EGFR TKIs dose reduction or discontinuation.
Drug Management Strategies
The conventional first-line treatment for diarrhea caused by oral EGFR-TKIs drugs is the opioid loperamide, is the most effective treatment for cure diarrhea. 114 A synthetic oral opioid Loperamide acts via several different mechanisms, including decreased fluid secretion and peristalsis, which lengthens the time that food travels through the gastrointestinal tract and increases the absorption of fluids and electrolytes. 115 The recommended standard dose of loperamide is initially 4 mg, followed by 2 mg per diarrhea episode and a maximum dose of 16 mg per day, which depends on the degree of diarrhea experienced by the patient. 116 Patients with grade 1-2 diarrhea should begin loperamide medication and remain on EGFR-TKIs at the same dosage. In the event that grade 2 diarrhea lasts over 2 days, it is recommended to interrupt EGFR-TKIs or reduce the dose despite antidiarrheal therapy. If grade 3 or 4 diarrhea occurs, the individuals should begin aggressive intravenous rehydration. Patients should continue to be treated with loperamide, but if a patient develops neutropenia, prophylactic antibiotics may be considered and EGFR-TKIs therapy must be discontinued. 117 For individuals with grade 3 or greater diarrhea leading to the suspension of pyrotinib, prophylactic therapy with loperamide is recommended for 21 days when treatment is resumed again. The CONTROL study suggested that patients receiving neratinib adjuvant therapy should start loperamide at the time of the first dose, continue for 2 courses, adjust the frequency of medication to control the daily defecation to 1-2 times, and prophylactic use of loperamide for 56 days will not significantly increase the incidence of intestinal obstruction. 107 In the recently published PANDORA Phase II study, implementation of loperamide as a primary prophylaxis measure to control underlying diarrhea significantly reduced the incidence of ≥ grade 3 diarrhea in patients receiving prophylaxis (8.9%; 4/45) compared to those who did not (38.2%; 13/34). In addition, treatment interruption rates (6.7%, 3/45) and dose reduction rates (17.8%, 8/45) were lower in patients receiving loperamide prophylaxis than in patients not receiving prophylaxis, 14.7% (5/34) and 35.3% (12/34), respectively. 63
Additionally advised as a therapeutic or prophylactic approach for diarrhea brought on by cancer treatment are probiotics. Probiotics have varied degrees of success in lowering diarrhea during chemotherapy.118,119 Examples include Lactobacillus acidophilus and Bifidobacterium, which competitively prevent pathogens from invading the intestinal mucosa and activate immunity by stimulating cytokine release. 120 However, until now, there has been no substantial evidence that utilizing probiotics during HER2 TKIs treatment leads to a definite improvement in diarrhea.121,122
Conclusion
Diarrhea induced by HER2 TKIs treatment is garnering increasing attention due to its impact on patients. The frequency, severity, and underlying causes of diarrhea symptoms are closely linked to the specific drugs and dosages administered. Even mild cases of diarrhea can persist over a prolonged period. However, the precise mechanism by which EGFR-TKIs trigger diarrhea remains uncertain, necessitating further research to elucidate the pathophysiological mechanisms underlying and explore the potential need for specific pharmacological interventions. It has been suggested that diarrhea occurring in patients treated with the EGFR inhibitor lapatinib may correlate positively with clinical benefits. However, additional investigations are warranted to explore potential associations between diarrhea and clinical efficacy in patients received other HER2-targeted TKIs. Therefore, the identification and management of adverse reactions associated with HER2 TKI drugs are crucial for improving clinical outcomes, enhancing patient adherence to treatment regimens, and ultimately enhancing quality of life.
Footnotes
Acknowledgments
I wish to thank Dr Wang for helping review and edit the article and another three partners-- MSc Zhang, MSc Lin and Dr Jiang for preparing figures and tables. They all helped a lot.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The study was supported by China Anti Cancer Association Her2 Target China Research Fund Project (No.CETSDSSCORP239-11).
Ethical Statement
Data Availability Statement
Date sharing not applicable to this article as no datasets were generated or analysed during the current study.
