Abstract
Objectives
This study aims to describe the clinical outcomes of combination therapy with sarilumab and baricitinib for severe novel Coronavirus-19 (COVID-19) infection in cancer patients. With this study, we aim to evaluate the role of expanded immunotherapy for severely ill patients with COVID-19 respiratory infections with limited options. The secondary objective is to assess the safety of combination therapy with sarilumab and baricitinib for severe COVID-19 infection.
Methods
This was a retrospective cohort study of patients admitted to Moffitt Cancer Center with COVID-19 infection between January 2020 and April 2022. Our research received a waiver to sign consent by the patients according to our institutional IRB because it was free of any risk for the patients and respected the patient’s privacy. Following the Institutional IRB approval and relevant Equator guidelines, we collected information on patients with severe COVID-19 infection and received sarilumab and baricitinib. We evaluated the survival rate and safety of combination therapy. All the patient’s information was de-identified to protect their information according to Health Insurance Portability and Accountability Act (HIPAA).
Results
Four patients were included in the data analysis. Two survived, and two of them died (Table 1). All the patients that survived were previously vaccinated. Among the two patients who died, one was vaccinated, and the other was unvaccinated. All the patients tolerated the combination therapy well, and none of the patients who survived developed secondary infections or COVID-19-associated complications beyond 12 months of discharge.
Conclusion
Our study explores the potential safe combination use of different immune modulators targeting multiple pathways of the inflammatory cascade for severe and refractory COVID-19 respiratory infections in high-risk oncology patients. The small number of patients in our observational study was a limitation. A larger sample of patients will be needed to conclude more precisely the efficacy of the combination therapy of sarilumab and baricitinib for refractory cases of severe COVID-19 respiratory infection. Moreover, exploring other cytokine release signaling pathway targets may be the key to significantly reducing inflammation and further pulmonary fibrosis with chronic unbearable respiratory sequela.
Introduction
On January 30, 2020, an outbreak of a novel Coronavirus-19 (COVID-19) detected in Wuhan, China, was declared a public health emergency of international concern by the World Health Organization (WHO). 1 As of January 21, 2023, over one hundred and three million cases of Coronavirus and over one million and one hundred thousand deaths have been reported in the United States (CDC).
Multiple studies have shown that a massive cytokine storm, more than direct viral damage affecting the lung and other organs, plays a significant role in the pathogenesis and mortality of COVID-19 infection.2,3 Therapeutic options remain under development, and antivirals have only shown reduced hospitalization rates and mortality when given early.4,5 Studies have shown that immunomodulators have a potential treatment role for severely ill patients while targeting specific cytokines.6,7 Immunomodulators are recommended for patients with rapidly deteriorating clinical status while on dexamethasone and remdesivir. Medications in this group include Interleukin-6 (IL-6) receptor antagonists such as tocilizumab or sarilumab, and Janus Kinase (JAK) inhibitors such as baricitinib or tofacitinib. 8
Only a few studies have addressed combination therapy targeting key cytokines to treat severe COVID-19 infection (IL-6) receptor antagonists and (JAK) inhibitors. In these studies, the treatment was successful, and there were no significant adverse events.9-11
Although there are few potentially beneficial outcomes with combination therapy, current guidelines from the National Institutes of Health (NIH) and the Infectious Diseases Society of America (IDSA) do not routinely recommend this practice outside clinical trials. However, because of worse outcomes in the immunosuppressed population than in the general population, clinicians may try to optimize management with limited available options. 12
This study analyzed the outcomes and safety of combination therapy with sarilumab and baricitinib. We hypothesize that the severe inflammatory response against COVID-19 infection will be ameliorated by simultaneously targeting different cytokines involved in the cytokine storm, including IL-6, IL-8, IL-12, and TNF-α among others that play an essential role in tissue injury, multi-organ failure, thrombosis, and ARDS. 13
Materials and Methods
Study Design and Participants
This was a retrospective cohort study of patients admitted to Moffitt Cancer Center with COVID-19 infection between January 2020 and April 2022 conducted following the relevant Equator guidelines. The University of South Florida (USF) Institutional Review Board (IRB# 003954) approved the study with a waiver for informed consent. Our research received a waiver to sign consent by the patients because it was free of any risk for them and respected the patient's privacy.
Patients were included if they were older than 18, had a solid and hematological malignancy, confirmed severe or critical COVID-19 infection, and received combination therapy with sarilumab and baricitinib at any point in addition to standard of care. 8 A confirmed case was a positive nasopharyngeal swab for COVID-19 by reverse-transcriptase-polymerase chain reaction (RT-PCR) tested by Moffitt Cancer Center clinical microbiology laboratory. Severe infection was defined as SpO2 <94% on room air, a ratio of arterial partial pressure of oxygen to fraction of inspired oxygen (PaO2/FiO2) <300 mmHg, a respiratory rate >30 breaths/min, or lung infiltrates >50%. Patients were considered to have critical illness due to respiratory failure, septic shock, and multiple organ dysfunction. 8
Study Procedures
Hospitalized patients received care per our institutional and national guidelines, including supportive therapy, supplemental oxygen, dexamethasone, remdesivir, sarilumab, or baricitinib if indicated. The institutional guidelines were developed by consensus based on the most current IDSA, NIH COVID-19 Guidelines, and current medical literature. 8
Patients were eligible to receive sarilumab or baricitinib if they had severe COVID-19 respiratory infections with elevated C-reactive protein (CRP) > 7.5 mg/dL and oxygen saturation less than 92% in room air, rapidly progressive respiratory decline, or on high flow oxygen (>30 L/minute). In patients who received sarilumab and had worsening clinical symptoms or were rapidly deteriorating, baricitinib was added. Worsening clinical symptoms were determined collaboratively by the patient’s treating physician in consultation with the Infectious Diseases team. We used sarilumab instead of tocilizumab because of supply disruptions in our institution. The dose of sarilumab was 4 mg IV x1, followed by baricitinib 4 mg daily for up to 14 days by the oral or enteral route.
Data Collection
Patients with diagnosed COVID-19 infection who received sarilumab and baricitinib at Moffitt Cancer Center during the study period were reviewed. Baseline patient characteristics, socio-demographic data, malignancy status, treatments received, concomitant infection status, and clinical outcomes were extracted from the electronic medical record. All the patient’s information was de-identified to protect their information according to Health Insurance Portability and Accountability Act (HIPAA).
Results
Patients Who Received Combination Therapy.

Abbreviations: ANC, absolute neutrophil count; ALC, absolute lymphocyte count; BMI, body mass index; CLL, chronic lymphocytic leukemia; HTN, hypertension; HLD, hyperlipidemia; CRP, C-reactive protein; HFNC, high-flow nasal cannula.
All the patients received remdesivir and dexamethasone in addition to sarilumab and baricitinib. Two patients had received monoclonal antibody therapy for asymptomatic or mildly symptomatic infection before hospital admission. Mortality attributable to COVID occurred in 2 of the 4 patients. The surviving patients were all vaccinated. One patient passed away 26 days post-baricitinib and 36 days after the initial diagnosis with progressive respiratory failure after he pursued comfort care measures. He was found to have Microbacterium aurum and Phenylobacterium hematophilum in 1 of the 2 blood cultures (peripheral sample) 72 hours after the sample. However, this was suspicious for a contaminated blood sample.
The unvaccinated patient died after 11 days of baricitinib therapy and 21 days after the initial diagnosis after electing comfort care. The 2 patients that died were lymphopenic with a lymphocyte count less than 500 cells/μL.
All the patients dramatically reduced their CRP after baricitinib addition, and all the patients tolerated the combination therapy well. None of the patients who survived developed secondary infections after 12 months of treatment, and their respiratory status and pulmonary images revealed dramatic resolution of symptoms (Figures 1-4). CT of patient 1 while having acute COVID-19 pneumonia. CT of patient 1 almost 3 months after COVID-19 pneumonia. Chest X-ray of patient 4 while having COVID-19 pneumonia. Chest X-ray of the patient 4 37 days after combination therapy.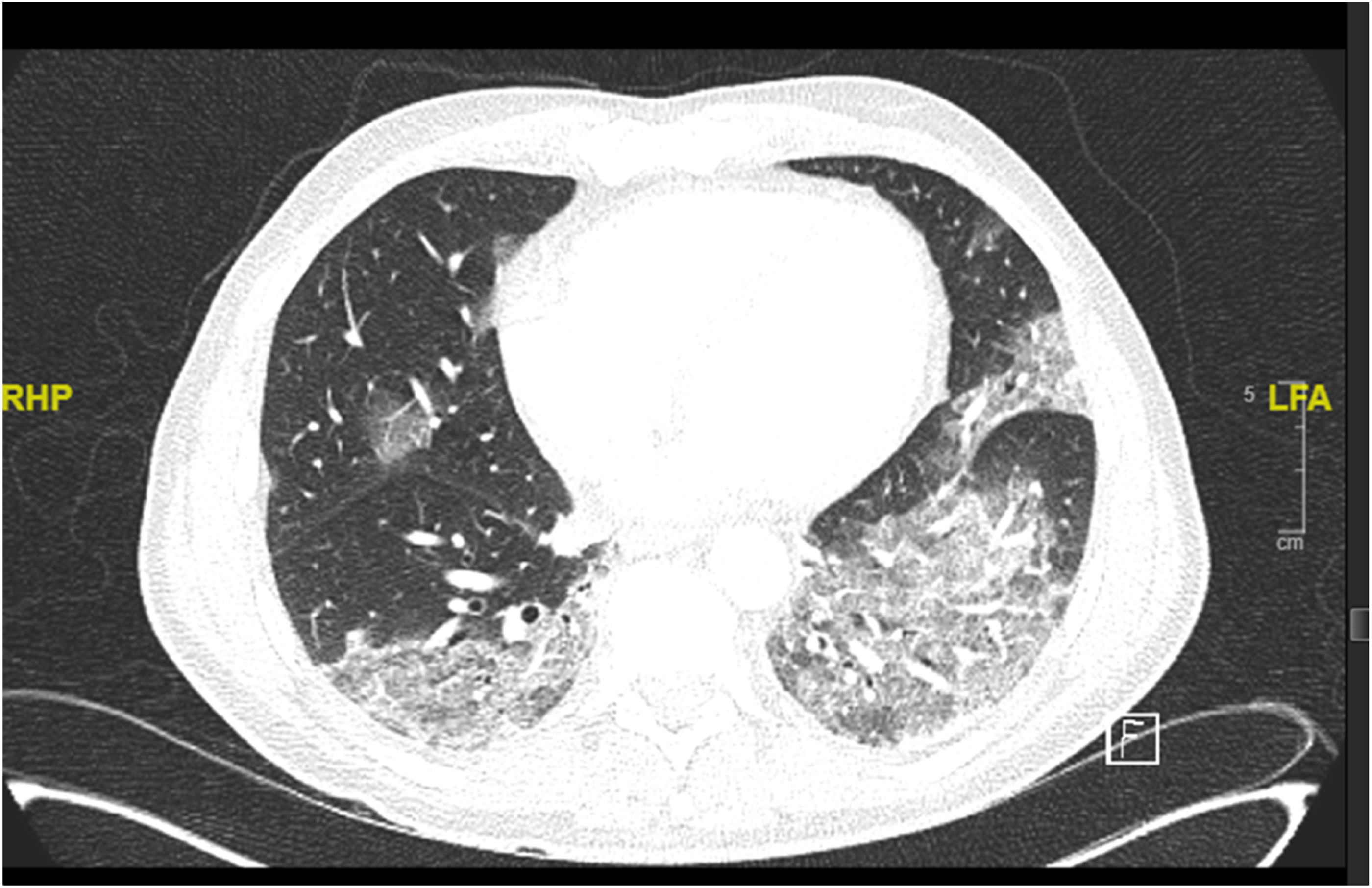



Discussion
Management of COVID has evolved significantly since the start of the pandemic. The development of vaccines may be attributed to the most significant mortality reduction in high-risk patients. 14 However, managing immunosuppressed patients remains challenging, particularly in patients not responding to remdesivir plus dexamethasone. A recent open-label multicenter randomized controlled trial including hospitalized patients with severe comorbidities showed no significant reduction in the risk of all-cause mortality 28 days after using nirmatrelvir/ritonavir. 15
Few case reports demonstrate complete resolution of COVID-19 symptoms after combination therapy with remdesivir and nirmatrelvir/ritonavir.16,17
NIH COVID-19 Treatment guideline suggests using either IL-6 inhibitors such as tocilizumab or baricitinib in hospitalized patients receiving dexamethasone who have rapidly increased oxygen needs and systemic inflammation. 8
Reducing the cytokine release syndrome by the host immune response is critical in managing patients with hypoxic respiratory failure from severe COVID-19. In fatal COVID-19 infection, the pro-inflammatory cytokine IL-6 and the Janus Kinases Signal Transducers and Activators of Transcription (JAK-STAT) pathway are elevated. The JAK-STAT is the mediator of several interferons and cytokines, including IL-6.18-20 In the baricitinib RECOVERY group analysis, 23% of patients received tocilizumab or sarilumab in addition to baricitinib. The trial reported that the benefit of baricitinib was consistent regardless of the use of the Il-6 receptor blocker. Such combination therapy was also not associated with significant infection differences. 21
Another study published by A. Cingolani described a patient who received sarilumab and later received baricitinib as rescue therapy, given the lack of response and improvement in the inflammatory markers. The patient tolerated the medication well and was discharged home.
In our study, two patients died and two survived with dramatic clinical improvement without evidence of secondary infections, including fungal pneumonia. One of the patients who died had advanced stage IV lung cancer with widespread metastatic disease and was not vaccinated. The other patient who died was severely immunosuppressed from CLL and current therapy with ibrutinib. Based on this fact, concluding that COVID was the sole cause of death in these patients would be challenging.
Conclusion
Our study explores the potential safe combination use of different immune modulators targeting multiple pathways of the inflammatory cascade for severe and refractory COVID-19 respiratory infections in high-risk oncology patients. The small number of patients in our retrospective observational study was a limitation. A larger sample of patients will be needed to conclude more precisely the efficacy of the combination therapy of sarilumab and baricitinib for refractory cases of severe COVID-19 respiratory infection. Moreover, exploring other cytokine release signaling pathway targets may be the key to significantly reducing inflammation and further pulmonary fibrosis with chronic unbearable respiratory sequela.
Footnotes
Acknowledgments
The authors thank the Information Technology Pharmacy and the Department of Microbiology.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
