Abstract
Cancer treatment optimizations select the most optimum combinations of drugs, sequencing schedules, and appropriate doses that would limit toxicity and yield an improved patient quality of life. However, these optimizations often lack an adequate consideration of cancer’s near-infinite potential for evolutionary adaptation to therapeutic interventions. Adapting cancer therapy based on monitored tumor burden and clonal composition is an intuitively sound approach to the treatment of cancer as an inherently complex and adaptive system. The adaptation would be driven by clinical outcome setpoints embodying the aims to thwart therapeutic resistance and maintain a long-term management of the disease or even a cure. However, given the nonlinear, stochastic dynamics of tumor response to therapeutic interventions, adaptive therapeutic strategies may at least need a one-step-ahead prediction of tumor burden to maintain their control over tumor growth dynamics. The article explores the feasibility of adaptive cancer treatment driven by tumor state feedback assuming cell adaptive fitness to be the underlying source of phenotypic plasticity and pathway entropy as a biomarker of tumor growth trajectory. The exploration is undertaken using deterministic and stochastic models of tumor growth dynamics.
Keywords
Introduction
Despite the expanding space of therapeutic options being available for cancer patients, therapeutic resistance continues to be the most critical challenge in the fight against cancer. The genetic diversity and phenotypic plasticity of cancer cells sustain a near-infinite capacity of cancer for evolutionary adaptation to therapeutic interventions, which ultimately lead to therapeutic resistance. In fact, standard of care (SOC) treatments involving the use of drugs with fixed schedules at maximum tolerated dose (MTD) foster the selection of therapy-resistant populations of cancer cells which maintains a clinically undetectable residual minimal disease (MRD) and would inevitably lead to recurrence and resistance to other lines of therapy. Even when complete response is achieved, the elimination of therapy-sensitive cancer cells promotes the proliferation of therapy-resistant populations due to competitive release. 1 Although cancer treatment at MTD with fixed schedules has been and still is the prevailing therapeutic paradigm, different treatment strategies have been explored, including metronomic therapy, intermittent therapy, model-informed precision dosing, and evolutionary-based treatments. 2 However, the increasing appreciation of the complex adaptive nature of cancer and its evolutionary dynamics is positing the necessity of evolutionary-based treatment strategies, such as adaptive therapy, to address resistance.1,3,4 It is noteworthy to mention that intermittent therapy is presumed to reduce toxicity and exploit evolution to tackle resistance, 5 but so far, the results of clinical testing are not compelling,6-10 which is in clear contrast to the rising clinical success of adaptive therapy.11,12 It has also been suggested that the way evolution is exploited constitutes a point of divergence between traditional intermittent therapy and adaptive therapy. 5 In particular, traditional intermittent therapy starts with a lengthy induction period at MTD which reduces the proportion of therapy-sensitive cancer cells, weakening their collective ability to suppress the proliferation of therapy-resistant cancer cells during the pause period. 5
Inspired by eco-evolutionary principles of competition between species, adaptive therapy (AT) was conceived as a strategy of disease management, 3 where a tumor burden is maintained with a sufficient proportion of therapy-sensitive cancer cells to suppress the proliferation of therapy-resistant cancer cells and prevent their competitive release. 1 A pilot clinical trial (NCT02415621) of adaptive therapy for metastatic castrate-resistant prostate cancer (mCRPC) using abiraterone monotherapy has shown a significant increase in time to progression (TTP) and overall survival (OS) for adaptive therapy compared to SOC treatment.11,12 Other clinical trials of adaptive therapy are currently underway for different cancers, including mCRCP (NCT03511196) and melanoma (NCT03543969). One key ingredient of an effective adaptive therapy is the timely and accurate monitoring of tumor burden. In the case of the mCRPC pilot study (NCT02415621), serum prostate-specific antigen (PSA) was used to estimate the number of cancer cells.11,12 The timely therapeutic action based on the monitored tumor burden is equally critical to an effective adaptation to tumor growth dynamics. Indeed, it was suggested based on computer simulations that progression occurred for most except for 4 trial patients because treatment was not stopped until many months after PSA decline below 50%, which was the treatment protocol’s threshold for stopping the therapy. 12 The third essential component to the success of an adaptive therapeutic strategy is the availability of mathematical/computational models of tumor dynamics. 11 Although such models are rooted in simplifying assumptions about the time-varying nonlinear dynamical behavior of tumor growth, their role is indispensable to the exploration of new adaptive therapeutic strategies, the design of corresponding clinical trials, and the assessment of their results. 4 Furthermore, given the inter-patient tumor heterogeneity,13-21 patient-tailored parametrization of tumor growth models would be needed to achieve personalized optimizations of adaptive therapy. 12
Most models of tumor growth that have been used to explore and support adaptive therapy are deterministic in nature and are based on the dynamics of competition between 2 and 3 cancer cell phenotypes. 22 As such, they abstract away the stochasticity of genetic and epigenetic alterations and tumor phenotypic plasticity, which are drivers of therapeutic resistance.23-25 However, irrespective of the depth and breadth built-in these models to cover all known salient drivers of cancer, the time-varying, stochastic, nonlinear, and patient-specific tumor growth dynamics may only be approximated with a limited accuracy that may be inadequate to yield effective therapeutic strategies. One potential approach to address this challenge is to develop adaptive therapeutic strategies that are driven by tumor state feedback, where the tumor state is estimated based on the continuous monitoring of tumor growth dynamics throughout the span of patient care. Advances in liquid biopsy combined with next generation sequencing (NGS) and imaging are expected to enable the longitudinal monitoring of tumor burden and clonal composition.26-30 The article explores an adaptive tumor control approach that assumes phenotypic plasticity to be rooted in cell adaptive fitness and pathway entropy as a marker of tumor growth trajectory.31,32 The exploration is undertaken using a theoretical lens aided with mathematical modeling as the most appropriate mean to study the wide spectrum of possible treatment strategies and to seek insights about adaptive cancer control as a plausible approach to turn cancer into a clinically manageable chronic disease.
Adaptive Treatment of Cancer
Adaptive cancer therapy3,11 is a control strategy aligned with the methods of adaptive feedback control theory.33-35 Tumors are the dynamical systems under adaptive control, where the parameters of the corresponding controller are adjusted in response to the changes in the state of the system under control, that is, the tumor. The general structure of an adaptive control system
35
may be reframed for the case of adaptive cancer therapy as shown in Figure 1. Adaptive cancer treatment. The treatment controller is based on an implicit or explicit model of the tumor as a nonlinear, time-varying dynamical system under control. The therapeutic actions of the controller are determined by the tumor state feedback and the controller parameters, which are continuously tuned based on treatment outcomes.
Therapeutic actions consist of 1 or more drugs administered in accordance with specific dosing regimens, that is, frequency and dose. An example of adaptive therapeutic strategy could be to modulate the dosing regimen for multiple drugs administered either concurrently or sequentially based on feedback signals such as tumor burden and toxicity. The adaptive control strategy would consist of the regular tuning of the 2 control parameters, namely, (1) the timing of drug dose modulation and (2) the dose for each drug being administered. The tuning may be performed at regular periods of time such as daily or weekly depending on the drugs being used, the treatment monitoring protocol, and the specific omics signature of the patient’s cancer. This simple parameterization of the control problem leads to the following tumor control law:
At least 3 classes of adaptive control approaches for dynamical systems may be readily used as potential adaptive cancer control strategies, namely, gain scheduling, self-tuning control, and model-reference adaptive control.34,35 Gain scheduling consists in choosing the appropriate values of control parameters from a lookup table based on the operating conditions of the system under control. This is akin to the adaptive therapy used in the castrate-resistant metastatic prostate cancer.
11
In this case, a dosing regimen of 1000 mg daily of abiraterone and 10 mg daily of prednisone was used until the PSA is reduced by 50% from the pre-abiraterone patient’s baseline, at which point the abiraterone treatment is stopped. Abiraterone plus prednisone treatment is resumed when the PSA recovers to or exceeds the pre-abiraterone baseline. Referring to the control law
Real-world implementation of adaptive controllers presents several challenges related to the convergence and stability of control algorithms.
38
These challenges would apply to cancer control where an accurate model of the nonlinear, time-varying tumor dynamics is lacking. Furthermore, it may not be clinically feasible to modulate drug doses with a sufficient frequency to achieve the “persistent excitation” of the tumor that would be needed for the implicit or explicit estimations of tumor growth dynamics that drive the self-tuning of controller parameters. However, the clinical context of cancer control may still be fertile to the use of “managed” adaptive controllers. For instance, the Lotka–Volterra (LV)-based model of tumor growth parameterized by Gatenby and colleagues
12
may be used to develop a class of explicit adaptive controllers where monitored tumor burden can serve as the feedback signal to control tumor growth. In particular, the LV model may be extended with an additional term representing therapeutic actions as follows: Adaptive PID control of tumor growth. The adaptation of the controller parameters consists in resting the PID gains to zero to stop treatment when the PSA falls below 50% of its pre-treatment level.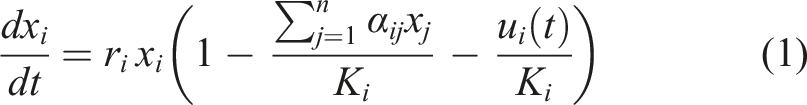

The design of adaptive cancer therapy can, as succinctly reviewed above, benefit from the rich field of control theory and its applications for time-varying nonlinear systems such as manufacturing processes, robots, planes, and rockets. However, unlike these human-engineered physical systems for which application-plausible mathematical models may be derived from first principles, tumor growth dynamics are emergent from spatial and temporal nonlinear interactions of living cells subject to stochastic evolutionary pressures and embody a biological complexity whose control may require mathematical modeling combined with observation-driven heuristics. Therein lies the potential use of data-driven artificial intelligence to narrow the effective scope of adaptive cancer therapy and provide support for longitudinal tuning of treatments based on learning the specific features and patterns of the patient’s disease.
Adaptive Combination Therapy
Cancer cells may be categorized into 3 classes: Entropy-guided Adaptive Combination Therapy. Tumor entropy 
In order to explore its feasibility, the proposed approach to adaptive combination therapy is applied to an extension of the tumor evolution model in Bozic et al.
51
The extension accounts for a clone-dependent selective advantage based on a putative cell adaptive fitness.
31
Tumor growth is modeled using a Galton–Watson branching process
52
starting with a single founder cell, having a single driver mutation, that either divides or “stagnates” (ie, differentiate, senesce, or dies).
51
Cells harboring k driver mutations (k-cells) and making up the k-clone acquire an additional mutation with probability 
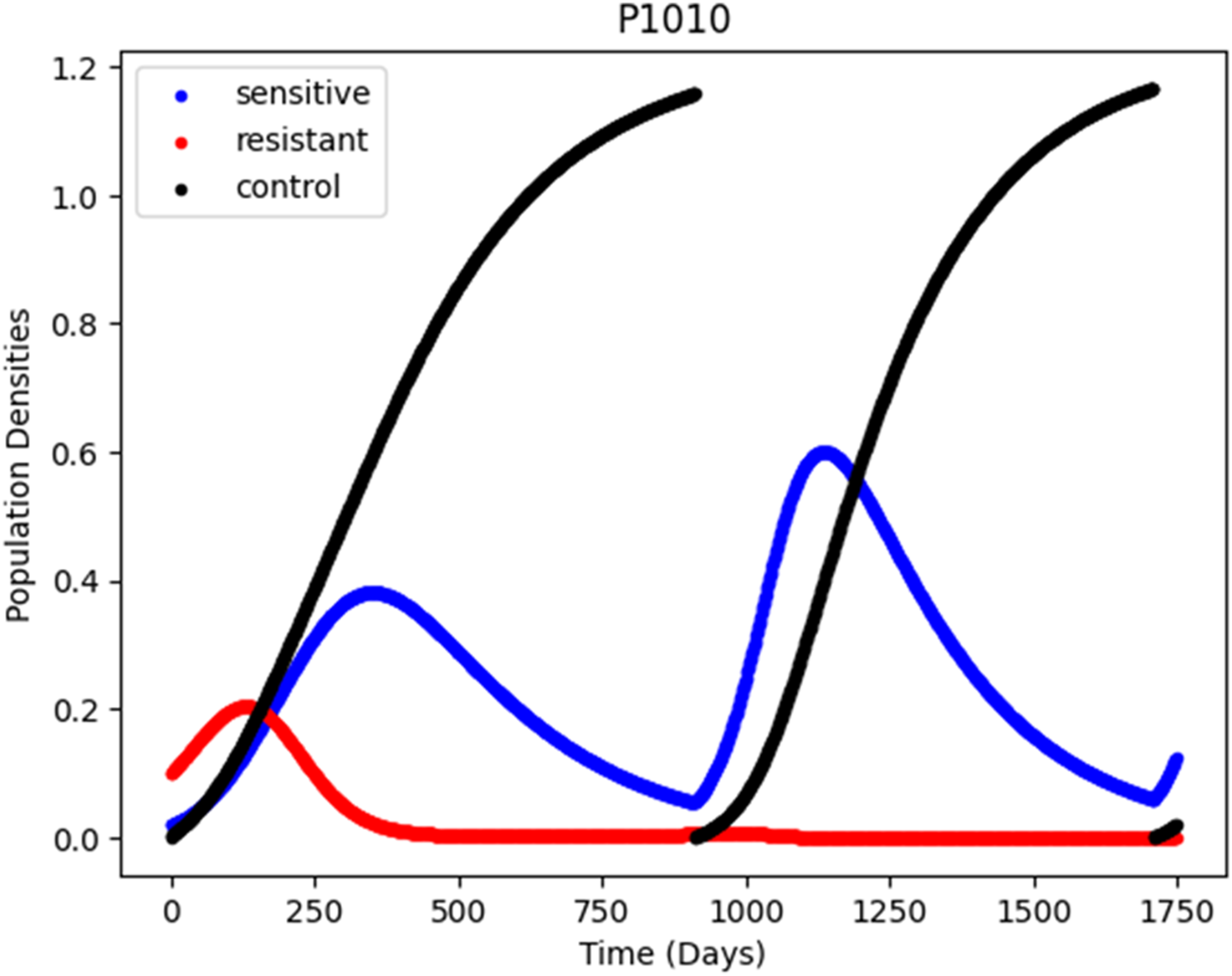
Cancer cell killing is modeled as a reduction of clonal selection as specified by the term Tumor growth under adaptive control. Clone size (number of cells with a given number of driver mutations) vs the age of the tumor. Twelve simulation runs of the stochastic tumor growth, where each run represents a hypothetical patient. The first column shows tumor progression for patients not receiving a treatment. The other columns show tumor progression for patients for whom treatments started at t = 1200 (∼10 years), t = 1800 (∼15 years), and t = 2400 (∼20 years) after the first driver mutation event, respectively. The simulation results are obtained using Tumor total entropy. The decline of tumor entropy for multiple hypothetical patients under adaptive therapy. The leftmost illustration shows tumor entropy for patients not receiving a treatment. The treatments started at t = 1200 (∼10 years), t = 1800 (∼15 years), and t = 2400 (∼20 years) after the first driver mutation event, as indicated in the patients’ identifiers. Adaptive Control of Tumor Growth. Adaptive PID controls (drug dose modulation) and treatment schedules for 9 hypothetical patients. The levels 5, 10, and 15 in the treatment schedule (blue) correspond to type 1 (Chemotherapy), type 2 (Immunotherapy), and type 3 (Targeted Therapy) therapies, respectively. The treatments started at t = 1200 (∼10 years), t = 1800 (∼15 years), and t = 2400 (∼20 years) after the first driver mutation event, as indicated in the illustrations’ titles. Treatments are applied in successive cycles that are 90 days long, with drug holidays of 15 days preceding the switch to a different type of treatment as recommended by the algorithm. The illustrated PID controls are normalized by the carrying capacity.


The proposed control algorithm leads to a control of tumor growth, as measured by tumor entropy, with treatments lasting between 9 and 16 months depending on tumor size at the time of diagnosis (see Figures 4 and 5). The treatments recommended by the proposed algorithm (Figure 6) involve different patterns of combination therapy that may be deemed clinically plausible when compared to current treatment protocols and advances in cancer treatment strategies.53,54 However, these therapy combination patterns need to be continuously monitored and vetted in order to ensure patient safety. This objective may be fulfilled by an artificial intelligence (AI) supervisory control layer guided by the treatment protocols in effect and the continuously monitored patient health state and treatment outcomes. This additional layer of therapy control would add a level of assurance that the recommendations of the adaptive control system are clinically acceptable with respect to toxicity, effectiveness, and efficacy. Reinforcement learning 55 may be one of the candidate AI approaches that could be used to implement an AI-driven supervisory control layer of the proposed model of adaptive cancer therapy.
Discussion
Adaptive control of tumor growth consists in modulating treatments based on treatment response feedback through repeated monitoring of tumor growth dynamics, to achieve desired patient outcomes. As such, tumor monitoring and the synthesis of therapeutic actions are primary aspects of focus in the clinical translation of adaptive therapy. This would require an initial genomic profiling of the tumor in order to subclassify patients, identify mutations that are clinically actionable or indicative of potential drug resistance, and assess the likelihood of patient response to targeted, cytotoxic, and immunotherapy drugs. 56 Screening for proteomic biomarkers57-59 would also be needed in addition to genomic profiling to inform the optimal selection of patient-tailored baskets of drugs to be combined in accordance with the explored model of adaptive combination therapy. The model has 2 levels of adaptation, namely, (1) the switching between therapy types (ie, immune, targeted, and cytotoxic) and (2) the modulation of drug doses and timing. Both levels of adaptation are based on the repeated estimation of tumor entropy which is presumed to be a proxy biomarker for tumor mutational burden and an indicator of the tumor’s stages of evolutionary progression. 31 Tumor entropy would be estimated, for the oncogenic pathways relevant to the cancer subtype,31,32 based on genetic alterations, which could be tracked using liquid biopsy28,60-63 and radiogenomics29,64 techniques. The non-invasive nature of these techniques being reliant on imaging and the sampling of patient blood, urine, and other body discharges makes the frequent estimation of tumor entropy clinically feasible during cancer treatment. Considering the example of lung cancer, which is the leading cause of cancer deaths and a prime target for combination therapy,65-69 there is a spur of research interests in the continuous monitoring of therapy using liquid biopsy.70-74 For instance, NSCLC tumors are known to harbor various genetic alterations, implicating EGFR, ALK, MET, ERRB2/3, ROS, BRAF, KRAS, RET, and NRTK,75-77 and corresponding signaling pathways, including RTK-RAS/ERK, PI3K-AKT-MTOR, Genome Integrity (P53-DNA repair), and cell cycle.78-80 Initial genomic profiling of NSCLC tumors at the time of diagnosis would identify actionable genetic alterations and the associated oncogenic signaling network. FDA-approved liquid biopsy assays such as FoundationOne® liquid CDx 81 could be used to monitor genetic alterations during treatment. 81 In addition, methods such as liquidCNA, 63 which rely on low-pass whole-genome-sequencing of circulating cell-free DNA (cfDNA), could be used to track tumor subclonal fractions that would be needed to estimate tumor entropy. Tumor monitoring using liquid biopsy would also reveal information about the spatial heterogeneity of the tumor,61,82 which would reflect intra-tumor heterogeneity in the treatment response feedback driving adaptive therapy.
The clinical translation of adaptive cancer therapy is faced with numerous open questions 83 that need further research investigations. Adaptive feedback control of cancer is ultimately and unavoidably necessary given cancer evolutionary dynamics and the consequent nonlinear, time-varying dynamics of tumor response to treatments. The availability of reliable biomarkers of tumor burden and clonal composition that can be continuously monitored with repeated non-invasive sampling is a key ingredient to the clinical success of adaptive therapy. Clinically parameterized, calibrated, and validated mathematical models are also essential to the clinical success of adaptive therapeutic strategies.83,84 On the other hand, leveraging the competition between drug-sensitive and drug-resistant requires maintaining a high tumor burden which would entail risks of manifested disease symptoms and new metastases. 83
While the framing of adaptive therapy in terms of competition between 2 phenotypic classes of cancer cells leads to manageable mathematical models of tumor treatment response, it abstracts away phenotypic plasticity and the role of the tumor microenvironment and the immune system. The notion of cell adaptive fitness 31 provides a theoretical lens through which tumor growth dynamics and response to treatments can be understood in the context of a co-evolving tumor microenvironment. This perspective views the dysregulated coupling between cell signaling and metabolic pathways as the primary driver of cancer cells’ continuous adaptation to their tumor microenvironment, where pathway entropy in concert with metabolic and information processing capacities determines cancer cell phonotypic plasticity. 31 The conjectured relationship between pathway entropy and the capacity of cancer cells to successfully replicate 31 provides a model for a one-step-ahead estimation of tumor growth trajectory through a continuous monitoring of tumor entropy. Although real-time predictions of tumor growth and patient health state are operationally and clinically challenging to achieve, 83 continuing advances in combining imaging with liquid biopsy26-29,62-64 to monitor tumor burden and treatment response are bound to overcome these hurdles. This would address one of the main assumptions of the proposed model of adaptive feedback control of tumor growth and would pave the way for the design of future clinical trials of adaptive combination therapy.
Conclusions
Adaptive therapy has the potential to meet cancer treatment challenges emanating from the time-varying, nonlinear treatment response, including drug-induced resistance and the underlying tumor evolutionary dynamics. However, more theoretical, experimental, and clinical investigations are needed to address the many open questions facing clinical translation of adaptive therapeutic strategies. In this respect, the proposed model of adaptive feedback control of tumor growth constitutes an alternative perspective to adaptive treatments that leverage the competition between drug-resistant and drug-sensitive cancer cells. It avoids the need to maintain a high tumor burden during treatments through the inclusion of a one-step-ahead estimation of phenotypic plasticity and tumor growth dynamics through the continuous monitoring of tumor entropy. Mathematical modeling and simulation are used to illustrate the proposed adaptive feedback control of tumor growth using a stochastic, evolutionary tumor growth model as a first step towards further experimental and clinical investigations to explore its clinical feasibility and effectiveness as a cancer therapeutic strategy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work and its publication were supported by Toronto Metropolitan University.
