Abstract
Background
In the era of immunotherapy, the immune function of patients with cancer has attracted increasingly more attention. The immune scoring system is an important supplement to the classical tumor staging and classification process. The immune system plays a controversial role in the development of cancer. Meanwhile, the prognostic significance of peripheral blood lymphocytes is still controversial. The present study aimed to assess the prognostic significance of peripheral blood lymphocytes in eight types of cancers.
Methods
We performed a retrospective analysis of 32731 patients with cancer hospitalized in Shanxi Cancer Hospital from January 2012 to December 2016. The percentages of CD3+, CD4+, CD8+, CD19+, CD56+, and CD127+ lymphocytes in the peripheral blood of all patients were examined using flow cytometry. The immune cell subsets of patients with cancer were classified into three groups using the K-means clustering method via the R language software. Differences in the overall survival rate were analyzed using the Kaplan–Meier method. The Cox regression model was utilized for univariate and multivariate analysis.
Results
The mean survival time of patients with liver cancer, rectal cancer, gastric cancer, breast cancer, esophageal cancer, colon cancer, ovarian cancer, and lymphoma was 30.25, 21.74, 37.67, 16.28, 21.62, 30.25, 31.43, and 34.27 months, respectively. The survival curves showed that the most prognostically beneficial immune state of the patients was when the expression of the immune cells in the peripheral blood was in equilibrium. Moreover, Cox proportional hazards regression model analysis revealed that the factors affecting the overall survival (OS) of patients with eight different kinds of cancer were not identical. However, CD19+ lymphocytes had the most significant impact on the prognosis of these patients.
Conclusions
Cancer occurrence and development were associated with the density of lymphocyte infiltration. Thus, immune homeostasis could be an effective indicator to evaluate prognosis and judge cancer treatment.
Introduction
Cancer is a chronic disease that endangers human health. On average, approximately six people are diagnosed with malignant tumors every minute in China. 1 A prognostic biomarker is a factor that either positively or negatively influences a patient’s clinical outcome. 1 The immune status of patients with cancer is a significant index for prognosis evaluation and treatment scheme adjustment. The activation of the adaptive immune system may inhibit malignant cells, whereas that of various types of innate immune cells may promote tumor growth. Adaptive immunity is coordinated by antigen-specific T and B lymphocytes, and tumor growth is inhibited by a combination of cytotoxic T lymphocyte (CTL)-mediated direct killing, cytokines, and antibody-mediated tumor cell lysis. 2 Natural killer (NK) cells, essential components of innate immunity, are different from other classical immune cells. They can provide a rapid response without requiring antibodies and major histocompatibility complex (MHC). Foxp3-expressing regulatory T (Treg) cells, which are indispensable for preventing autoimmunity, also suppress effective tumor immunity. Of note, the role of different immune cell types in regulating cancer progression has been increasingly recognized.3-6 Indeed, CD4+ follicular helper T cell infiltration predicted breast cancer survival, and CD8+ cells in malignancies were associated with better survival in small cell lung cancer, 7 endometrial cancer, 8 bile duct cancer, 9 esophageal cancer, 10 and follicular lymphoma. 11 Moreover, NK cells may act as a possible prognostic factor in childhood acute lymphoblastic leukemia. 12 A complex and dynamic interaction between the tumor cells and the immune system has been considered a novel definition of cancer.13,14 In addition, interference in immune cell balance could trigger the recurrence and metastasis of malignant tumors. 15
With the development of modern tumor immunology, it was realized that it is not possible to completely eliminate all tumor cells in the body via the action of drugs alone and that it is important that the body’s own immune cells fight the residual tumor cells. Therefore, immune function monitoring plays an important role in preventing cancer, observing cancer development, and estimating prognosis. The detection method of lymphocyte subsets in the peripheral blood is simple and feasible and can be used as an economical and effective method to monitor the immune status of patients with cancer. For a wider range of cancer types, no systematic and comprehensive analysis of the prognostic value of primary immune cell types has been conducted. The present study aimed to analyze the relationship between the expression of CD3+, CD4+, CD8+, CD19+, CD56+, and CD127+ lymphocytes in peripheral blood and the survival of patients with eight kinds of cancer who were hospitalized in Shanxi Cancer Hospital.
Materials and Methods
Patients
From January 2012 to December 2016, patients with eight types of cancer (5019 rectal cancer, 6303 gastric cancer, 6615 breast cancer, 1540 liver cancer, 5832 esophageal cancer, 5019 colon cancer, 1287 ovarian cancer, and 1116 lymphoma) associated with Shanxi Cancer Hospital were searched in the hospital database. All patients had undergone immunologic function testing. The relative counts of peripheral blood lymphocyte subsets were analyzed using flow cytometry. In addition, other clinical data, such as age and sex at diagnosis and the percentage of lymphocytes, were collected from patients’ medical records. The reporting of this study conforms to the AMA Style Guide. 16 All patients signed the informed consent form for the utilization of their clinical data; the personal information of the patients was anonymized and protected.
Follow-Up
Patient follow-up data were obtained by reviewing the hospital records. All the included patients have been followed-up until August 31, 2021. Overall survival (OS) was defined as the interval between the dates of initial hospital admission and either the time of the last follow-up or death. The survival time was measured in months.
Statistical Analysis
The highest survival rate was calculated by dividing the immune cell subsets of patients with cancer into three groups using the K-means clustering method via the R language software. Survival curves were generated using the Kaplan–Meier method. Univariate survival analyses were performed using the log-rank test, and statistically significant variables were entered into Cox proportional hazards regression model analysis, which was used to identify factors associated with OS. A P-value of ≤.05 was considered statistically significant. SPSS software (version 22) for Windows was used for all statistical analyses.
Result
Patient Characteristics
The range of survival time of all patients with cancer is demonstrated in Figure 1. The mean survival time of patients with eight types of cancer was 30.25, 21.74, 37.67, 16.28, 21.62, 30.25, 31.43, and 34.27 months. The mean survival time for patients with eight kinds of cancers.
Liver Cancer
The results indicated that when the expression of CD3+, CD4+, CD8+, CD19+, CD56+, and CD127+ lymphocytes was 60%–70%, 30%–40%, >30%, >10%, 15%–25%, and 5%–7%, respectively, the immune status of the patients with liver cancer was optimal and more prognostically conducive (Figure 2). CD19+ (χ2 = 11.170, P = .004) and CD127+ (χ2 = 1.088, P = .008) lymphocytes were significantly associated with the OS of liver cancer. Multivariate analysis showed that CD19+ (P = .001) and CD127+ (P = .037) lymphocytes were also independently associated with the prognosis of liver cancer (Table 1). Kaplan–Meier curve for OS for CD3+, CD4+, CD8+, CD19+, CD56+, and CD127+ lymphocytes in cancers. *“cum” is the abbreviation of “cumulative.” Cox Proportional Hazards Regression Model Analysis for Overall Survival in Cancers.

Rectal Cancer
The survival curves showed that the immune status of the patients with rectal cancer was optimal and more prognostically beneficial, when the expression levels of CD3+, CD4+, CD8+, CD19+, CD56+, and CD127+ lymphocytes were 65%–75%, 30%–40%, <20%, >10%, 15%–25%, and <4.5%, respectively (Figure 2). The difference in age (χ2 = 7.405, P = .007) and CD3+ (χ2 = 7.773, P = .021), CD8+ (χ2 = 7.714, P = .021), CD19+ (χ2 = 27.033, P = .000), and CD127+ (χ2 = 13.089, P = .001) lymphocytes in patients with rectal cancer was statistically significant. The independent prognostic factors were age (P = .004) and CD19+ (P = .000) lymphocytes (Table 1).
Gastric Cancer
The analysis showed that the immune status of the patients with gastric cancer was optimal and more prognostically beneficial when the expression levels of CD3+, CD4+, CD8+, CD19+, CD56+, and CD127+ lymphocytes were 60%–70%, ≥30%, ≤30%, >10%, 15%–25%, and 5%–7%, respectively (Figure 2). The difference in age (χ2 = 52.650, P = .000) and CD3+ (χ2 = 8.909, P = .012), CD4+ (χ2 = 29.137, P = .000), CD8+ (χ2 = 15.108, P = .001), CD19+ (χ2 = 107.492, P = .000), CD56+ (χ2 = 13.777, P = .001), and CD127+ (χ2 = 10.307, P = .006) lymphocytes in patients with gastric cancer was statistically significant. Moreover, CD3+ (P = .001), CD4+ (P = .008), CD8+ (P = .006), CD19+ (P = .000), and CD127+ (P = .004) lymphocytes were the independent prognostic factors of gastric cancer (Table 1).
Breast Cancer
Cox Proportional Hazards Regression Model Analysis for Overall Survival in Cancers.
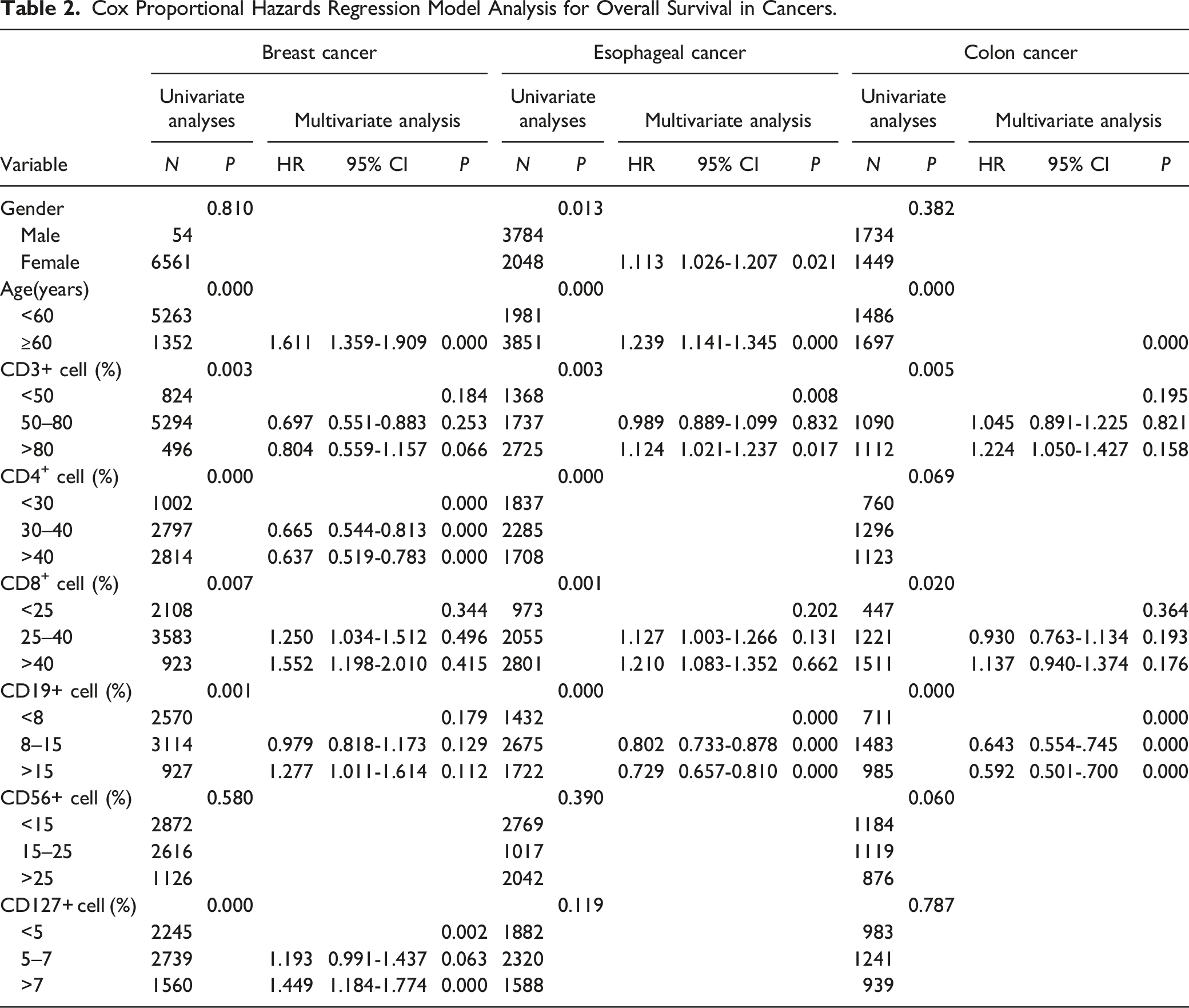
Esophageal Cancer
The survival curve revealed that the esophageal cancer patients’ immune state was the most prognostically beneficial, when the expression of CD3+, CD4+, CD8+, CD19+, CD56+, and CD127+ lymphocytes, respectively, was 60%–70%, 30%–40%, <20%, >10%, 20%–25%, and 5%–7% (Figure 3). In esophageal cancer, the difference in sex (χ2 = 6.156, P = .013); age (χ2 = 18.170, P = .000); and CD3+ (χ2 = 11.417, P = .003), CD4+ (χ2 = 21.212, P = .000), CD8+ (χ2 = 15.113, P = .001), and CD19+ (χ2 = 71.413, P = .000) lymphocytes was statistically significant. The independent prognostic factors of esophageal cancer were sex (P = .021), age (P = .000), and CD3+ (P = .008) and CD19+ (P = .000) lymphocytes (Table 2). Kaplan–Meier curve for OS for CD3+, CD4+, CD8+, CD19+, CD56+, and CD127+ lymphocytes in cancers. *“cum” is the abbreviation of “cumulative.”
Colon Cancer
The survival analysis demonstrated that when the expression of CD3+, CD4+, CD8+, CD19+, CD56+, and CD127+ lymphocytes, respectively, was <65%, 30%–40%, 20%–30%, >10%, >25%, and 5%–7%, the colon cancer patients’ immune state was the most prognostically beneficial (Figure 3). The difference in age (χ2 = 19.137, P = .000) and CD3+ (χ2 = 10.506, P = .005), CD8+ (χ2 = 7.875, P = .020), and CD19+ (χ2 = 47.061, P = .000) lymphocytes in colon cancer was statistically significant. The independent prognostic factors of colon cancer were age (P = .000) and CD19+ (P = .000) lymphocytes (Table 2).
Ovarian Cancer
Cox Proportional Hazards Regression Model Analysis for Overall Survival in Cancers.

Lymphoma
The lymphoma patients’ immune state was the most prognostically beneficial, when the expression of CD3+, CD4+, CD8+, CD19+, CD56+, and CD127+ lymphocytes was 65%–75%, >35%, 20%–30%, >10%, 15%–25%, and >7%, respectively (Figure 3). In lymphoma, there was no significant difference in the tumor infiltration of any lymphocyte (Table 3).
Discussion
Cancer poses a significant threat to public health, and the cancer burden has been rising in China. 17 Gastric cancer, lung cancer, esophageal cancer, liver cancer, and colorectal cancer are the leading malignant tumors in males, whereas breast cancer, cervical cancer, lung cancer, and gastric cancer are the leading cancers in females. 18 According to previous research, patients with malignant tumors usually have immune dysfunction, including the imbalance of immune cells, cytokines, stimulating molecules, and other factors. 19 Multivariate analysis showed that age was an independent prognostic factor in most cancers. So far, approximately 60% of malignant tumors occur in the elderly with the aging of immune dysfunction and accompanied by additional diseases.
T cells play a controversial role in the tumor microenvironment. CD3 is expressed on all human T cells, which are divided into Th1/helper T helper cells (CD4+ lymphocytes) and Tc/cytotoxic T cells (CD8+ lymphocytes). In general, during tumor development, T cells, which are activated by dendritic cells, gather near the tumor and enhance the performance and function of other tumor-infiltrating lymphocytes to provide immune surveillance. 20 It was reported that immune cells, particularly the CD8+ lymphocytes, have a more potent positive effect on the prognosis of patients. 21 Tumors with tumor-infiltrating lymphocytes are a strong marker of prognosis in ovarian and colorectal cancers. 22 A detailed immune map of CRCs at diagnosis revealed that a large area of tumor lesions infiltrated by memory T cells with a Th1 orientation (expressing IFN-γ) along with cytotoxic CD8+ T cells (expressing granzymes and granulysin) indicated the most favorable prognosis. 23 In the current survival curves analyses, the group of patients with <25% CD8+ lymphocytes had a significantly better OS than those with 25%–40% and >40% CD8+ lymphocytes in breast cancer. The group of patients with lymphoma with 20%–30% CD8+ lymphocytes had a significantly better OS than those with <20% and >30% CD8+ lymphocytes. Nevertheless, multivariate analysis showed that CD8+ lymphocyte level was an independent prognostic factor in gastric cancer (P = .006). Although T cells can have antitumor effects, tumor cells can still escape clearance by directly or indirectly inhibiting the aggregation of T cells; escaping immune cell surveillance; or immune tolerance. Cancer cells evade T cell-mediated destruction through different mechanisms. The close relationship between CD8+ lymphocytes or CD3+ lymphocytes and a more prolonged disease-free survival or OS has been verified in both the primary and metastatic settings of most squamous-cell carcinomas and large cell lung cancers. 24 The present study results regarding survival curves demonstrated that the group of patients with 60%–85% CD3+ lymphocytes had a significantly better OS than those with <60% and >85% CD3+ lymphocytes in breast cancer. The group of patients with cervical cancer with 60%–85% CD3+ lymphocytes had a significantly better OS than those with <60% and >85% CD3+ lymphocytes. However, multivariate analysis showed that CD3+ lymphocyte level was an independent prognostic factor in gastric cancer (P = .001) and esophageal cancer (P = .008). This may be because when a malignant tumor appears in the body, the CD3+ lymphocyte level is affected by certain tumor-secreted factors, leading to immune response disorder.
A broad range of helper T (CD4+) cells, including the different subsets of cells of helper T1 (Th1) cells, T helper 2 (Th2) cells, T helper 17 cells (Th17), Treg cells, and T follicle helper cells, have a wide range of functions and highly variable prognostic values. 25 Because of their different spatial distributions, varied prognostic values among the specific subsets of immune cells were observed. Recent research has shown that the high frequency of Th1 cells was associated with reduced 2-year survival after surgery in non-small-cell lung cancer (NSCLC). 26 The existence of TH2 cells is closely related to invasive tumors. For example, TH2 lymphocytes were associated with shorter survival in patients with pancreatic cancer. 27 Th17 cells have been shown to be associated with poor prognosis in colorectal cancer, 28 NSCLC, 29 and hepatocellular carcinoma 30 and may improve survival in gastric 31 and cervical cancers. 32 The current study revealed that the group of patients with <30% and 30%–40% CD4+ lymphocytes had a significantly better OS than those with >40% CD4+ lymphocytes in gastric cancer. However, multivariate analysis showed that CD4+ lymphocyte level was an independent prognostic factor in gastric cancer (P = .008), breast cancer (P = .000), and ovarian cancer (P = .000). CD4+ cells can inhibit tumor growth by directly lysing or recruiting other cells. CD4+ lymphocytes in the peripheral blood of patients with cancer are reduced, suggesting that their immune system is extremely weakened at this stage. However, these cells are a mixture of CD4+/CD25+Foxp3 + Treg cells and CD4+/Foxp3− cells. The first subtype is known to inhibit tumor immunity and promote cancer growth and metastasis,33,34 which has been verified in animal studies.35,36 Without the help of CD4+ cells, the role of specific CD8+ lymphocytes would not be prominent. In fact, CD4+ lymphocytes are critical for CD8+ lymphocytes to be transformed into specific effector cells. 37 A reduced CD4+/CD8 + ratio indicates that the immune function of patients with cancer is immunosuppressed. This means that the ability of the body to identify and kill cancer cells decreases, leading to immune state imbalance in the body. The growth rate of tumor cells is high, thereby worsening the disease and causing metastasis.
As a receptor of IL-7, CD127 plays an essential role in the development of T lymphocytes along with the maintenance of homeostasis in T cells and differentiation of memory T cells. 38 As previously reported, CD127, which acts as an excellent marker of Treg cells, is expressed at low levels on most of Treg cells. 39 Some of the strategies that cancer uses to evade the immune system are tolerance to peripheral T cells, including Treg and T cell anergy; fatigue; and aging, which impair T cell-mediated immunity. 40 Treg is a subclass of helper T cells whose most important function is to inhibit autoimmune cell-mediated pathological immune response as well as to establish and maintain immune homeostasis in tissues. In the current study, the group of patients with <5% CD127+ lymphocytes had a significantly better OS than those with 5%–7% and >7% CD127+ lymphocytes in breast cancer. The group of patients with cervical cancer with <5% and 5%–7% CD127+ lymphocytes had a significantly better OS than those with >7% CD127+ lymphocytes. Nevertheless, multivariate analysis showed that CD127+ lymphocyte level was an independent prognostic factor in gastric cancer (P = .004), breast cancer (P = .002), and liver cancer (P = .037). Studies have shown that there are a large number of Treg cells in tumor tissues, which promotes the occurrence and development of tumors by inhibiting the antitumor immune response. Moreover, the number of Treg cells is closely related to the prognosis of patients. According to reports, the increase in the number of Treg cells in tumors may be due to tumor antigen stimulation and the resultant clonal expansion. Moreover, there is a certain specificity. The targeted removal of tumor-specific Treg cells or inhibition of their function can effectively stimulate the antitumor reaction in vivo, produce a strong antitumor response, effectively prolong the survival time of patients with cancer, and improve their prognosis.
NK cells are essential in innate and adaptive immune response and tumor immune surveillance, and they recognize and kill tumor cells. CD56, an extracellular marker, is mainly expressed on human NK cells and a few MHC-mediated T lymphocytes. NK cells expressing CD56 are a critical component of the natural immune system of humans. They are capable of controlling tumor growth and the initial dissemination.41-43 Although infrequently found in immune tumor infiltrates, tumor-associated CD56+ lymphocytes have been associated with increased survival rates in patients with HCC115, 44 CRC,45-47 ccRCC, 48 and prostate cancer. 49 In the present study, the group of patients with 15%–25% CD56+ lymphocytes had a significantly better OS than those with <15% and >25% CD56+ lymphocytes in cervical cancer. On the one hand, CD56+ NK cells can restrict the growth and spread of cancer cells. On the other hand, under the action of inflammatory chemokines, circulating CD56+ NK cells can be recruited to the tumor site. CD56+ NK cells exhibit antitumor cytotoxicity without presensitization and production of cytokines and chemokines that regulate various immune responses.
B cells are a key cell type for adaptive humoral immunity because they produce antibodies. The value of humoral immune response mediated by B cells in cancer prognosis however remains controversial. Research has indicated that B cell infiltration is closely correlated with poor prognoses; patients with epithelial ovarian cancer displaying CD19+ lymphocytes in pleural effusions showed poor prognosis. 50 By contrast, the better clinical outcome of gastric cancer in the tertiary lymphoid structure stage is usually related to the density of infiltrating B cells. 51 Large amounts of B cells were associated with advanced clinical outcomes in CRC, 23 HCC, 52 melanoma, 53 HGSOC,54,55 and NSCLC. 56 Whether B cell infiltration produces good or bad clinical outcomes in breast cancer has not reached a consistent conclusion. In the current study, the group of patients with >10% CD19+ lymphocytes had a significantly better OS than those with <5% and 5%–10% CD19+ lymphocytes in gastric cancer, and the group of patients with esophageal cancer with >10% CD19+ lymphocytes had a significantly better OS than those with <5% and 5%–10% CD19+ lymphocytes. The group of patients with colon cancer with >10% CD19+ lymphocytes had a significantly better OS than those with <5% and 5%–10% CD19+ lymphocytes, whereas the group of patients with rectal cancer with >10% CD19+ lymphocytes had a significantly better OS than those with <7% and 7%–10% CD19+ lymphocytes. However, multivariate analysis showed that CD19+ lymphocyte level was an independent prognostic factor in gastric cancer (P = .000), liver cancer (P = .001), rectal cancer (P = .000), colon cancer (P = .000), and ovarian cancer (P = .000). These results suggest that CD19+ lymphocytes have the most substantial impact on patients’ survival. Studies have shown that the tumor-promoting effect of B cells is generally related to the immune-suppressed B cell subgroup (Breg). Further, locally or systemically produced tumor-specific antibodies may also drive tumorigenic effects. In particular, it has been observed that persistent immune complexes formed by tumor-specific antibodies may be associated with adverse clinical outcomes, possibly caused by the regulation of the activity of myeloid cells harboring Fc receptors. Meanwhile, B cells can resist tumor development under certain conditions, primarily by producing tumor-specific antibodies and presenting tumor antigens.
Because of the intricate characteristics, there is a complex and dynamic interaction between the tumor and the immune system. The combined assessment of immune cells or immune factors on the prognosis of patients with cancer is probably better reflected by their relative contribution at the tumor site. 57 Regarding function, NK cells and CD8+ cytotoxic T cells exhibit similar cell-destroying mechanisms; however, NK cells function without the recognition of a somatically rearranged antigen. 58 Sometimes NK cells need to induce a tumor-specific solid CD8+ lymphocyte response via the perforin-dependent selection of the more immunogenic dendritic cell subpopulation and through the early recruitment of conventional cytotoxic T cells to activate dendritic cells, a mechanism that was identified recently. 59 Immunologic markers comprising CD68hi, CD4hi, or CD8low cells are more predictive of decreased survival in breast cancer than individual cell types. 60 The simultaneous occurrence of intratumoral CD8+ lymphocytes and CD20+ B cells in tumors is related to the prolonged survival of metastatic melanoma, independent of other clinical variables. 61 Therefore, the interaction and mutual restriction among peripheral blood immune cells affect the occurrence and development of diseases. A major result of this study suggested that the immune state in which the peripheral blood immune cells are in equilibrium is the most prognostically beneficial. This finding will help further understand that immune homeostasis could be an effective indicator to evaluate prognosis and judge cancer treatment. Another important result was that the sample size of patients with cancer in this study was large, with several different kinds of cancer. However, an accurate prognostic evaluation should consider immune cells and other factors that regulate their activities, including the surgical approach, postoperative chemoradiotherapy, and other related clinical factors. One limitation of this study was that the aforementioned other related factors were not analyzed. Hence, a comprehensive analysis was not possible. Thus, multicenter studies are needed to identify the influence of peripheral blood immune cells on the prognosis of patients with cancer.
Conclusions
In the course of occurrence and development, cancer is associated with the density of lymphocyte infiltration. The present study results showed that CD19+ lymphocytes had the most significant impact on the prognosis of patients with eight different types of cancer. Moreover, in all eight types, the patient’s prognosis reached an optimal state when the immune cells in the peripheral blood were in an equilibrium. Thus, immune homeostasis could be an effective indicator for evaluating prognosis and judging cancer treatment.
Footnotes
Acknowledgments
The authors would like to thank the members of department of clinical laboratory, Shanxi Province Cancer Hospital.
Author Contributions
Conception or design of the work: XZ.
Experiment and data collection: XZ.
Data analysis and interpretation: XZ.
Drafting the article: XZ.
Critical revision of the article: JJ.
Final approval of the version to be published: JJ.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
