Abstract
Background
To reveal successes and potential limitations of the lung cancer screening program, we conducted a survey that included both quantitative and open-ended questions to measure patient experiences and satisfaction with screening.
Methods
We report on the five open-ended items related to barriers to returning for screening, experience with other cancer prevention screenings, positive and negative experiences, and suggestions for improving future appointments. The open-ended responses were analyzed using constant comparison method and inductive content analysis.
Results
Respondents (182 patients, 86% response rate for open-ended questions) provided generally positive comments about their lung cancer screening experience. Negative comments were related to desire for more information about results, long wait times for results, and billing issues. Suggestions for improvements included: scheduling on-line appointments and text or email reminders, lower costs, and responding to uncertainty about eligibility criteria.
Conclusion
Findings provide insights about patient experiences and satisfaction with lung cancer screening which is important given low uptake. Ongoing patient-centered feedback may improve the lung cancer screening experience and increase follow-up screening rates.
Introduction
Lung cancer is the leading cause of cancer-related death in the United States, and the five-year survival rate for non-small cell lung cancer is 26% and 7% small cell lung cancer for all SEER stages combined. 1 The high mortality rates and poor survival outcomes of this disease are primarily attributed to most patients diagnosed with late-stage disease when the prospects for cure are limited. 2 Prior research has shown that among patients with lung cancer, those that reported limited finances are associated with an increase in symptom burden, poor quality of life, and are less able to obtain cancer treatment which can cause poor survival outcome.3,4 As such, early detection is critical for improving lung cancer outcomes by diagnosis patients at early stage when local therapy for early-stage disease is associated with substantially improved survival outcomes. Four randomized trials, including the National Lung Screening Trial (NLST) conducted in the United States, have demonstrated that lung cancer screening by low-dose helical computed tomography (LDCT) is efficacious in significantly reducing overall and lung cancer-specific mortality.2,5-7
Following publication of the NLST, guidelines for annual LDCT screening for high-risk individuals in the United States were issued by the National Comprehensive Cancer Network (NCCN), 8 U.S. Preventive Services Task Force (USPSTF) 9 and Centers for Medicare and Medicaid Services (CMS). 10 Despite these guideline recommendations, uptake of lung cancer screening among eligible individuals remains poor at around 6% nationally. 11 Worldwide, less than 5% of individuals who are at a high-risk for lung cancer have undergone screening, which is due to a decrease uptake of lung screening programs. 12 Lung screening implementation is variable in Europe and only 2 countries have implemented lung cancer screening program in high-risk population: Croatia 13 and Poland. 14 The Czech Republic has an ongoing population-based lung cancer screening program and 3 European Union countries have require formal recommendation to embark on lung cancer screening. 13
Current lung cancer screening guidelines do not consider never smokers, despite 10-20% of lung cancer diagnoses occur among this population. A study identified that the current lung screening guidelines misses lung cancer diagnosis in never smokers. 15 The MISCAN lung microsimulation determine that never smokers have a similar relative ratio 15 to 35 compared with ever-smokers. 16 COVID-19 infections can mimic malignant nodules which causes an increased false-positive screening rates and increase lung screening uptake. 17 Although there are no national statistics about adherence to lung cancer screening, published systematic reviews and meta-analyses that analyzed retrospective and prospective cohorts demonstrated that a 55% overall adherence rate across follow-up periods (i.e., 12 to 36 months) 18 and patient adherence increases to 65% when patients were diagnosed with Lung-RADS 3 or 4 compared to Lung-RADS 1 or 2. 19 Prior studies have identified that adherence across each round of screenings declines significantly20,21 which may be attributed to lack of awareness of mortality benefit, receiving a false-positive result, lack of access to screening, and cost.22-30
In 2015, the H. Lee Moffitt Cancer Center & Research Institute (Tampa, FL) launched a formal lung cancer screening program for high-risk individuals who meet NCCN inclusion criteria. 8 To identity potential strengths and limitations of this lung cancer screening, we conducted a survey study to access patient experiences and satisfaction. We present results from the qualitative analyses of the open-ended questions of patient’s responses. These results will inform the development of strategies and interventions to improve patient satisfaction and experience in the lung cancer screening programs as well as help patient adhere to their screening.
Methods
Lung Cancer Screening Program
The NCCN guidelines (v2.2020) establishes that patients are at high-risk if they are a) 55-74 years old with a ≥ 30 pack-year history of smoking and smoking cessation <15 years b) age ≥ 50 years and ≥20 pack-year history of smoking with additional risk factors. 8 Every patient is screened for eligibility prior to scheduling. There are instances that some patients will be schedule who are not eligible due to scheduling services overriding eligibility. This occurs in less than 5 cases per year. Patients who are required to have a shared decision making (SDM) visit are identified upon requesting a lung screening. A thoracic advanced practice provider (APP) or pulmonologist will conduct the SDM visit. In-person or zoom options are available depending on patient preference. Patients can self-refer but a patient may provide their Primary Care Physician information so the lung screening results can be sent. The Lung Screening Committee is responsible for overseeing that the strategy, quality, and program development are racially and ethnically diverse. The imaging technicians, schedulers and providers are also racially and ethnical diverse. The Lung Screening Coordinator within the Department of Thoracic Oncology conducts outreach at patient, provider, and community levels. Community health workers within the Office of Community, Outreach, and Engagement also conduct outreach at the community level.
Study Population
The study was IRB approved (Advarra IRB, Columbia, MD) and deemed exempt (#Pro0042395). As of January 2020, 590 patients had completed at least 1 lung cancer screening who met the NCCN inclusion criteria (v2.2020 and earlier).
8
Supplemental Table 1 provides the demographics information for the 590 screening participants compared to the 212 survey respondents. Among these 590 patients, 576 (97.6%) were alive and 14 (2.4%) were deceased. Starting July 2020, an initial English-language survey and cover letter was mailed to the 576 patients, along with a pre-paid self-addressed return envelope. Following the Dillman method,
31
a follow-up cover letter and the survey were mailed at 3 and 6 months after the initial mailing to prompt non-responders to complete the survey. Upon return, the survey was scanned into a study database using the Participant Research, Interventions, & Measurement (PRISM) core. Completed surveys were received between August 2020 to February 2021. Figure 1 is the CONSORT flowchart for our study. CONSORT flowchart of survey study participants.
Survey
A survey was developed to collect information from study participants regarding their experience and satisfaction concerning lung cancer screening at Moffitt Cancer Center. The cross-sectional survey was modeled according to the standardized questionnaire developed by the Medical Group Management Association in 2009. 32 The questions were adapted to focus on specific domains of the lung cancer screening program and open-ended questions were added to identify barriers and facilitators as a qualitative initiative. Qualitative and behavioral researchers reviewed and modified each question prior to administration to the patients. No incentive was provided for survey completion. The survey collected demographics and asked the following open-ended questions: 1) Please provide details of your barrier(s); 2) What would make it more likely that you return for subsequent lung screenings?; 3) Please tell us about any positive experiences you would like to share about your lung cancer screening visit; 4) Please tell us about any negative experiences you would like to share about your lung cancer screening visit; 5) What could we have done differently that would make you more likely to recommend lung cancer screening services at Moffitt to friends and family? The first question related to provide details of barriers is a follow-up, open-ended item for those who indicated in a quantitative item that they experience a barrier to lung cancer screening.
Data Analysis
Descriptive statistics (counts and percentages) were used to quantify the demographics and clinical variables survey response using Stata v17 (College Station, TX). The open-ended responses were analyzed using the constant comparison method and inductive content analysis. The reporting of this study conforms to COREQ guidelines.33-35 COREQ checklist is presented in Supplementary Table 2. Open coding was used to identify inductively themes within each open-ended question and emergent themes were considered if they represented a meaningful pattern in the data. An Excel file was created with all the responses, which was the divided by the questions to which it corresponds. Two coders reviewed all the open-ended responses to generate a list of codes, noting strength of the pattern in the data (i.e majority, few). Each code was refined via comparison and discussion by the 2 coders and discrepancies was resolved through discussion. The inter-rater reliability was calculated between the 2 coders by using the list of response for each question and identifying the number of times each coder labeled the comment as belonging to a finalized code. Level of agreement between the 2 coders was .88 (kappa coefficient). Representative quotes are included with each theme.
Results
Patient demographics and health status in the lung cancer screening satisfaction survey.

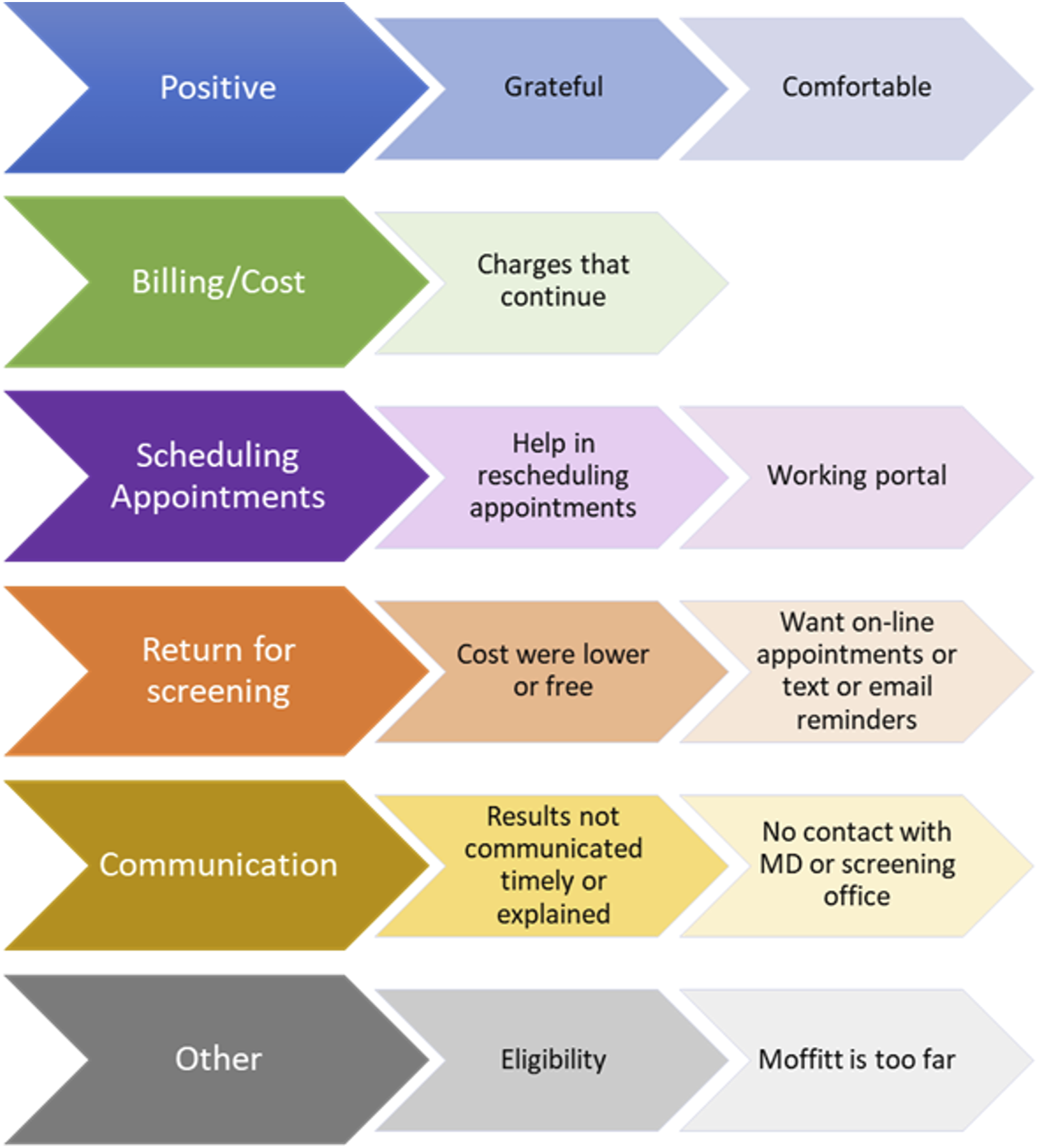
Theme and subthemes of patient’s response to open-ended questions.
Billing/Cost
Most of the respondents mentioned billing and cost as barriers and negative experiences. The first theme centered on insurance billing issues such as being billed beyond what the respondent originally paid or was told the cost would be. 1. “Lung scan cost is supposed to be $150 to patient. No other costs are supposed to happen. Almost every year for past 5 years I receive additional bills [in addition to $150 bill for the lung scan]. My insurance does not cover any of this [additional cost]. This lung screening program was promoted as $150 complete to patient” 2. “Charges [additional cost in addition to $150] that continue after I pay my $150 each year” 3. “Communicate better at billing, what potentially I may have to pay [for the lung scan]”
Scheduling Appointments
The second theme centered on frustrations with appointments cancelled by the center, the need for assistance in rescheduling appointments and difficulties with making an appointment on the patient portal. 1. “Appointments kept getting rescheduled and with no explanations given” 2. “Making appointments as too many steps to get a verified appointment date” 3. “Make appointment online live and confirm like I do for (redacted) lab”
Return for Screening
Two concepts arose from the responses of patients regarding what would increase their likelihood of returning for screening. These were lower costs and the ability to make on-line appointments and receive text or email reminders. 1. “With no insurance it would be helpful [to received] help with the cost” 2. “Call me for another screening [appointment] when due” 3. “Appointment reminder 30 days ahead [of scheduled lung scan appointment]” 4. “…Designate single point of contact for [lung scan] appointments and busy communication with screening doctor”
Communication
Respondents noted communication as a barrier to screening reporting issues related to understanding and receiving results in a timely manner and establishing a relationship with a physician. 1. “If the [lung scan] results were explained to me and follow up screenings were recommended. I kind of felt like I was left hanging [after receiving the results]” 2. “After my scan, [I was] informed via phone I probably had stage 1 lung cancer …after my appointment… [I was] informed….[that they] could not operate on me. Why [did it] take an hour to tell me-plus say [that] I probably had stage 1. [I] Had surgery…[and it turned out that I had] no cancer-non cancer nodule” 3. “I would like to receive my lung screening (results) the day I have it completed. I have had to change doctors [in thoracic oncology], a few times unforeseen circumstances were cause but feel I don’t really have a Dr./Patient relationship with anyone”
General Comments
Several responses focus on eligibility and distance to the screening center. Some patients would like to be informed if they still fit the criteria for screening and how often it is recommended. 1. “Not sure if [I] still qualify [for lung scan screening] due to age or current/past medical conditions” 2. “My only concern will be scheduling another [lung scan] test now that I will be living in (different country). I hope flexibility and priority will be given to out of state and country patients…” 3. “difficulty of long drive [to Moffitt]” 4. “Open a satellite office [for lung scans]…”
Positive Comments
Respondents provided generally positive comments about their lung cancer screening experience and at Moffitt Cancer Center.
1. “The Doctors and Staff at Moffitt saved my life; thus, [scheduling] yearly screenings. I am forever grateful.”
2. “The medical staff and administrative staff consistently deliver the highest level of care and attention to myself and other family members at Moffitt’s facilities.”
3. “This year was my 8th year at Moffitt. There is nothing you need to do to improve your great services. The research department is excellent. Thanks for what you do!”
4. “All of my experiences have been very positive. Your staff is always courteous, and professional/They display genuine care for the client. I appreciate all of the caring, professional staff at Moffitt, from the bathroom cleaners to the specialist doctors, all work to ensure the health of their patients.”
5. “Easy directions, valet parking, pleasant employees, comfortable testing, clean facility, last screening during COVID, felt extremely safe”
Discussion
In this analysis of patient satisfaction and experiences in a lung cancer screening program, we found that patients provided generally positive comments about their lung cancer screening experience. However, in questions related to barriers, several complaints were determined such as billing confusion, lack of information about meaning of results, problems with scheduling appointments, and overall communication. Patients also gave recommendations on ways to improve the lung cancer screening program. They mentioned having appointment reminders a day ahead of the appointment which would help them adhere more to screening.
Other studies have reported nonadherence rates in lung cancer screening ranging from 15% to 77.6%.20,36-39 A recent study identified factors influencing nonadherence to lung cancer screening and found that distance, cost, and communication with physicians for follow-up orders were the primary causes. 36 This is in line with our results identifying that communication issues and cost concerns were the most frequently cited barriers. Studies have reported that inability to obtain a physician follow-up order is a barrier for patients to adhere to screening recommendations. 40 Three studies reported that nonadherence significantly decreases with implementation of appointment reminders from a coordinator.38,40-42
Several studies have explored individual attitudes and experiences in lung cancer screening programs in community settings. In Australia, the Cancer Care Coordination Questionnaire for Patients, adapted to lung cancer patients, reported that patient experience was positive overall, but there were areas of improvement related to administrative coordination, communication, as well as information provided to patients. 43 Delmerico et al. developed a survey to address patients’ willingness and barriers to receiving a LDCT for lung cancer and found that insurance coverage was a burden and a main barrier for returning for a screening. 44 Studies have identified that patients who have insurance coverage are more likely to follow-up on annual lung cancer screening.45,46 In our study, respondents focus on barriers related to billing issues and high costs with comments ranging from “help with cost” and “no insurance”. The majority of our respondents had Medicare coverage (62.1%) compared to commercial (24.7%) and self-pay (12.1%).
Our study has some limitations that are worth noting. First, our findings are limited to participants at Moffitt Cancer Center. Thus, results may not be generalized to other populations and institutions but may provide guidance about lung cancer screening participants. Nevertheless, this study may act as a baseline for ongoing studies about satisfaction of patients in different screening programs. This study was conducted from July 2020 to February 2021, during the COVID-19 pandemic, which may have impacted our response rate. Another potential limitation is recall bias for which participants may not remember previous events or experiences accurately or omit details. 47 This qualitative study focused on a small sample size with no ability to comment on survey non-respondents for the open-ended questions. However, this data may be helpful in developing future strategies for the lung cancer screening program. Our study population did not allow for examination of results by race (96.2% White) or ethnicity (96.7% Non-Hispanic/Latino). Another limitation is that there is a potential self-selection bias whereby fewer current smokers choose to participate in the survey compared to the former smokers that participate at a higher proportion.
Despite these limitations, data collected from the qualitative analysis offers a unique perspective on the satisfaction and experience of lung cancer screening program patients.
This qualitative analysis found that patients who participated in the lung cancer screening program at Moffitt Cancer Center viewed the screening program in a positive manner. These positive comments focus on the highest level of care and attention to patients. However, some barriers arise such as cost, communication, and scheduling. Suggestions for improvement centered on better scheduling systems and appointment reminders as well as the ability to speak to a clinician to better understand results and future eligibility. Another suggestion is to incorporate the survey in the Press Ganey survey to collect annual data to enhance participation and measure ongoing feedback. Ongoing patient-centered feedback may improve the lung cancer screening experience and increase adherence to annual lung screening rates.
Conclusion
This study found that patients reported positives experiences in the lung cancer screening program and suggestions that can serve as a baseline to monitor improvements in the screening program. The ongoing patient-centered feedback such as a scheduling systems and better communication can improve the screening experience and increase adherence and follow-up screening rates.
Supplemental Material
Supplemental Material - Qualitative Findings From a Survey on Patient Experiences and Satisfaction with Lung Cancer Screening
Supplemental Material for Qualitative Findings From a Survey on Patient Experiences and Satisfaction with Lung Cancer Screening by Jaileene Pérez-Morales, Rashmi Pathak, Monica Reyes, Haley Tolbert, Rajwantee Tirbene, Jhanelle E. Gray, Vani N. Simmons, Matthew B. Schabath, Gwendolyn P. Quinn in Cancer Control
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.E.G. reports receiving commercial research grants from AstraZeneca, BI, Bristol-Myers Squibb, Genentech, G1 Therapeutics, Ludwig Institute of Cancer Research, Merck, Novartis, and Pfizer. J.E.G is a consultant/advisory board member for AbbVie, AstraZeneca, Axiom HC strategies, Blueprint Medicines, Celgene Copr, Daiichi Sankyo (DSI), EMD Serono, Genentech, Inivata, Janssen, Jazz Pharmaceuticals, Loxo Oncology, Merck, Novartis, OncoCyte Biotechnology Company, Sanofi Pharmaceuticals, Takeda Phamaceuticals, and Triptych Health Partners. M.B.S. is a consultant for Bristol-Myers Squib. The remaining authors declare no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by a U01-CA200464 (to MBS) and in part by the Participant Research, Interventions, and Measurement Core (PRISM) at Moffitt Cancer Center at the H. Lee Moffitt Cancer Center & Research Institute, a comprehensive cancer center designated by the National Cancer Institute and funded in part by Moffitt’s Cancer Center Support Grant (P30-CA076292).
Ethical Approval
The study was approved by the IRB (Advarra IRB) and it was deemed exempt (#Pro0042395).
Data Availability
MBS had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
