Abstract
Background
Mixed neuroendocrine and non-endocrine neoplasms (MiNENs) are challenging to diagnose and manage clinically. The current understanding of MiNENs’ pathobiology, molecular mechanisms, and management is incomplete. Though microsatellite instability (MSI) is known to impact carcinogenesis, reports examining MSI mechanisms for MiNENs are rare.
Methods
We report an unusual colonic MSI-MiNEN uncovered in an 89-year-old woman and the review of the literature.
Results
Pathologic inspection revealed a high-grade carcinoma composed of tumor cells with neuroendocrine histologic traits and immunophenotype intermixed with mucin-containing signet ring–like cells arranged in nested and micronodular patterns. Loss of MLH1 and PMS2 mismatch repair proteins was detected in tumor cells. INSM1 immunostaining highlighted about 50% of the tumour, further reinforcing the MiNEN diagnosis. Next-generation sequencing identified multiple carcinogenic mutations. Because of the advanced stage of the tumor and its adhesion to the adjacent organs, surgical resection was aborted; immunotherapy was initiated. The tumor is in remission 30 months following initiation of treatment, and the patient remains asymptomatic.
Conclusion
This unique MSI MiNEN was characterized by its immunohistochemical and molecular signatures and illustrated how correctly diagnosing MSI can strongly improve a patient’s outcomes.
Introduction
Gastrointestinal tract mixed neuroendocrine and non-endocrine neoplasms (MiNENs) are a conceptual category of epithelial neoplasms in which both neuroendocrine and non-neuroendocrine components are present, with each component being histologically and immunophenotypically recognizable and constituting at least 30% of the neoplasm. 1 The molecular mechanisms affecting the initiation and progression of MiNENs have not been fully understood, but it has been discovered that the disease outcome is predominantly determined by the proliferative potential of the neuroendocrine component. 2
MiNENs constitute a great pathologic and clinical challenge because of their rarity and heterogeneity. Pathologically, the neuroendocrine component can range from a low-grade well-differentiated neuroendocrine tumor to a high-grade neuroendocrine carcinoma (NEC), such as a large-cell or small-cell NEC, whereas the non-neuroendocrine component can be a precursor lesion or a carcinoma (not necessarily adenocarcinoma). 1 Hence, MiNEN used to be classified as mixed adenoneuroendocrine carcinoma (MANEC), in which both components were described as clonally related. 1 Clinically, MiNENs with a NEC component carry worse prognoses than conventional non-neuroendocrine adenocarcinomas and are associated with dismal patient outcomes similar to those for patients with NECs; therefore, when treating patients with MiNENs, it’s preferable to target a high-grade neuroendocrine component if one is identified.2-4 Despite this, MiNEN staging follows the criteria for adenocarcinoma. 1
Although diagnostic criteria have been established for MiNEN, they are primarily used for diagnosing surgically resected tumors if the distinct neuroendocrine and non-neuroendocrine components could be seen colliding into each other. 4 However, other lesser-known MiNEN patterns have also been documented in literature: cell types can be intimately intermingled within the tumor mass (ie, a composite MiNEN), or the tumor can be composed of amphicrine cells with dual neuroendocrine and non-neuroendocrine differentiations (ie, amphicrine MiNENs). 5 MiNENs are associated with aggressive behavior and poor patient outcomes, especially if they are metastatic at the time of diagnosis.1,2,6 Because of the poor prognosis associated with MiNENs, a correct and timely diagnosis is crucial for effective clinical action and positive patient outcomes; however, accurate diagnosis is extremely rare, particularly if the diagnosis is based on biopsy material.
In practice, rendering an MiNEN diagnosis remains challenging due to various reasons, including low incidence rates and unconventional morphologic and immunophenotypic profiles. MiNEN diagnosis also could be hindered by lack of clinical suspicion, small tissue sample size, unfamiliarity with the double differentiation of MiNENs, and incomplete or improper ancillary testing. 1 Available data suggest that MiNENs may be frequently undiagnosed 5 ; our institutional review indicated that, among consult and review cases sent from community institutions, ∼50% of MiNENs were erroneously diagnosed as poorly differentiated carcinomas (unpublished investigation). The main contributing factors to missing or misdiagnosing MiNENs include low awareness of MiNENs and suboptimal or improper ancillary testing (unpublished investigation).
Another underrecognized factor that may affect MiNEN diagnosis and treatment is the tumor’s microsatellite instability (MSI) status. Microsatellites are composed of multiple repeats of short segments of DNA base pairs distributed throughout the genome and are maintained by the mismatch repair (MMR) system, which predominantly includes MLH1, MSH2, MSH6, and PMS2 proteins.7,8 Defective mismatch repair (dMMR) function leads to MSI and affects many cellular events implicated in carcinogenesis. MSI or dMMR has been identified in ∼15% of colorectal carcinomas (CRC), especially those found at the right colon; around 12% of all CRC are sporadic MSI or dMMR tumors, and the remaining 3% MSI or dMMR cases are due to a germline mutation in the MMR system that is recognized as Lynch syndrome.7,9,10 An understanding of MSI and dMMR has led to new therapeutic strategies targeting CRC that have improved patient outcomes, so when treating patients with CRC, an analysis of MSI or MMR status should be performed regardless of clinical suspicion, the patient’s history, or microscopic histomorphology of the tumor.
Unlike CRC, there are few reports about the molecular pathology of MiNENs in the literature. Some of these investigations identified similar mutational profiles between the neuroendocrine and adenocarcinoma components within a MiNEN, including KRAS, APC, TP53, RB1, and SMARCA4 mutations.11,12 Other investigations have not successfully identified the same alterations between the 2 components within a singular MiNEN.13-15
Due to the low prevalence of MiNENs, our understanding of its molecular etiology and mutational mechanisms is very poor, especially when dMMR function is considered. La Rosa et al 4 examined the CpG methylator phenotype (CIMP) and MSI tumors and identified the MSI/CIMP + phenotype with loss of MLH1 and PMS2 proteins in 5 of 27 NECs but 0 of 12 MiNENs. Sahnane et al 6 studied MSI and dMMR among 89 gastroenteropancreatic NECs and MiNEN tumors and identified 11 NEC and MiNEN tumors (12.4%) with MSI. Among these tumors, 9 showed MLH1 promoter hypermethylation and MLH1 protein absence; another 2 cases showed loss of MSH2 or PMS2 proteins. In addition, BRAF mutations were identified as linked to MSI in 6 cases. They concluded that a subset of dMMR-enteric NEC/MiNEN tumors display distinct behaviour and confer a better prognosis than non-dMMR tumors. Based on the conclusions drawn from Sahnane et al and La Rosa et al, MSI and/or MMR status should be checked when determining a treatment plan for a newly diagnosed NEC or MiNEN.
Regarding treatment strategies, it is important for practicing pathologists to note that MSI and dMMR are involved in a wide spectrum of malignancies in addition to CRC; MSI/dMMR status may have implications for clinical management strategies regardless of cancer type. MiNENs are usually diagnosed at advanced stages, rendering the tumors nonresectable. A limited clinical arsenal is available for chemoradiation management, and satisfactory therapeutic strategies have yet to be established for MiNENs. Immunotherapies and targeted treatments against signaling pathways and gene mutations have shown promising results for treating other cancers, including those that are MSI, so it is conceivable that these therapies could also be beneficial for managing MiNENs.
Here, we report the first diagnosed right colon MSI-MiNEN, which was diagnosed from a biopsy sample. Following an aborted surgical procedure that originally aimed to resect this advanced malignancy, a modified therapeutic regimen based on the MSI diagnosis was prescribed. This regimen led to a markedly positive patient outcome.
Data
Written informed consent was obtained from the patient for the publication of this case report, and the Informed Consent Form can be made available upon reasonable request. This publication is considered exempt from Institutional Review Board review at Moffitt Cancer Center because the retrospective review of records for publication of a single-patient case report is not considered to be research involving human subjects.
An 89-year-old female originally presented at our tertiary cancer center’s Genitourinary Clinic in March 2020 because of a history of urothelial carcinomas. She underwent a right ureterectomy in 2005 for a pT1 grade 3 urothelial carcinoma and a cystectomy, lymphadenectomy, and an ileal conduit urinary diversion in 2010 for another pT1N0 urothelial carcinoma. In 2017, the patient underwent a right nephroureterectomy and reconstruction of the ileal conduit, during which a pT3NxMx urothelial carcinoma from the renal pelvis and a pT1NxMx urothelial carcinoma involving the ureter were removed. At this time, the patient deferred systemic chemotherapy and opted for close observation.
At a follow-up evaluation in March 2020, physical examination and laboratory testing were unremarkable. The patient reported mild abdominal discomfort but denied any weight loss, melena, or hematochezia. Corresponding CT imaging showed stable surgical changes from the cystectomy and right nephrectomy with a right abdominal ileal conduit; unexpectedly, abnormal right colon wall thickening was noted, with loss of normal haustral folds involving the ascending colon. Despite an absence of gastrointestinal symptoms or patient complaints, a malignant neoplasm (most likely an adenocarcinoma) was clinically suspected.
A subsequent colonoscopy in March 2020 identified a 5-cm right colonic mass occluding more than 50% of the lumen, with a poorly defined margin (Figure 1). Microscopically, the right colon mass biopsy was composed of sheets of epithelioid oval-to-polygonal neoplastic cells with high-grade cytologic atypia, arranged in nests, trabecula, and possible tubules, forming a closely packed sheet under low power magnification (Figure 2A). Higher magnification illustrated intriguing cytology of these closely packed and intermixed neoplastic cells: though some cells showed intracellular vacuoles or mucin, more cells were devoid of vacuoles or mucin but showed slightly eosinophilic and finely granular cytoplasm; mitoses and apoptosis were easily identified in this limited biopsy (Figure 2B-2D). Presumably, these findings could be seen with a poorly differentiated carcinoma, likely adenocarcinoma because of the foci of intracellular mucinous material (Figure 2D). Colonoscopy images of the large mass lesion in the right colon of an 89-year-old female. A 5-cm tumor occupied more than 50% of the circumference; clear demarcation from adjacent mucosa was not seen. (A-D) (magnification of 100×, 400×, 400×, and 600× for panels (A, B, C, and D), respectively): The tumor was composed of sheets of oval-to-polygonal malignant cells with eosinophilic and finely granular cytoplasm containing focal intracellular mucinous vacuoles; nodules, nests, and subtle strands were present. (E) (600×): Mucicarmine and (F) (600×): PAS-diastase special stains illustrated multifocal intracellular mucin production by tumor cells.
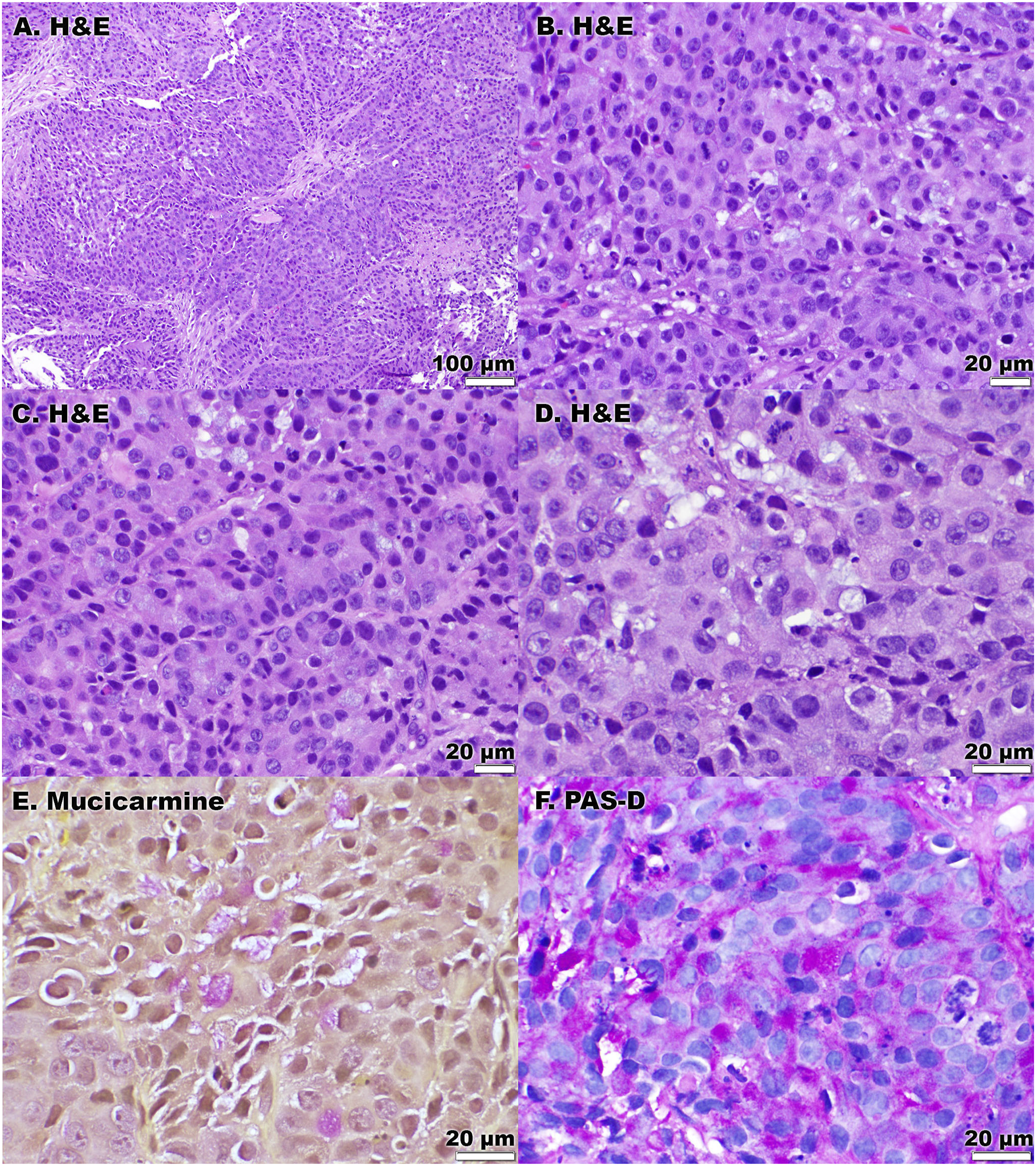
However, under low to high magnification, the subtle eosinophilic and granular cytoplasm was reminiscent of that of neuroendocrine tumors of the gastrointestinal tract (Figure 2C, 2D and Figure 3A). Hence, confirmatory ancillary testing was conducted to further classify this malignancy. Interestingly, the tumor cells were positive for synaptophysin, with stronger signal observed in the top and right portion (Figure 3B); chromogranin (not shown) and INSM1 (Figure 3C, right portion) were also positive. INSM1 highlighted ∼50% of tumor cells’ nuclei, illustrating their neuroendocrine nature. Notably, INSM1 has been identified as having a unique diagnostic utility for MiNENs, as its specificity for neuroendocrine differentiation can help distinguish between neuroendocrine and non-neuroendocrine components.16,17 In our case, INSM1 staining reinforced the MiNEN diagnosis. (A) (magnification of 100×): Microscopically, tumor cells formed a relatively blunt frontier pushing into adjacent benign colonic mucosa. (B) (100×): By immunohistochemistry, the tumor cells were positive for synaptophysin. (C) (100×): By immunohistochemistry, the tumor cells were focally positive for INSM1. (D) (400×): Tumor cells were also labelled by CDX2, confirming their intestinal lineage. (E) (200×): GATA3 was negative in tumor cells. (F) (200×): P40 was negative in tumor cells. (G) (100×): Tumor cells lost expression of PMS2 and MLH1 (not shown). (H) (100×): Tumor retained MSH6 and MSH2 (not shown). An arrow indicates benign mucosa in (A, B, G, and H).
Tumor cells were labeled for CDX2 (Figure 3D) and pankeratin OSCAR (not shown) diffusely, confirming their epithelial nature with intestinal lineage. Because of the patient’s history of multiple urothelial carcinomas and the nested tumor growth pattern in this biopsy, GATA3 (Figure 3E), P40 (Figure 3F), TTF1 (not shown) staining were performed, and all were negative in tumor cells. The mucin production by the neoplastic cells was confirmed by the positive intracellular signal of mucicarmine and Periodic acid–Schiff– (PAS-) diastase in over 30% of the tumor cells (Figure 2E, 2F). Ki-67 immunostaining showed a nuclear labeling index of ∼84% (not shown). The combination of these immunohistochemical results and ancillary findings (Figures 2 and 3) illustrated a malignant mixed neuroendocrine and non-neuroendocrine neoplasm—a MiNEN. Because of the finding of intimately intermixed neuroendocrine and non-neuroendocrine components each occupying over 30% of the tissue, the specific possibility of a composite MiNEN was entertained, 5 though the identified pattern carried no additional clinical weight for this patient’s management planning beyond the diagnosis of MiNEN.
Upon the MiNEN diagnosis, an abdomen and pelvis PET/CT scan detected abnormal thickening of the right colon centered around a 5 cm lesion, with a standard uptake value of 27.6 and associated medial pericolonic stranding and prominent right mesenteric lymph nodes. To further characterize this advanced-stage malignancy, immunostains for mismatch repair proteins were performed. Surprisingly, loss of MLH1 (not shown) and PMS2 (Figure 3G) and intact expression of MSH2 (not shown) and MSH6 proteins (Figure 3H) were observed. These ancillary tests established that this right colon MiNEN was a dMMR tumor.
The dMMR MiNEN diagnosis was conveyed to and discussed with the clinical team, leading to adjustment of management planning. Next-generation sequencing was performed and multiple oncogenic mutations, including a BRAF p.V600E mutation, were detected, which ruled out Lynch syndrome as the cause of dMMR.
Because the diagnosis of a dMMR-MiNEN is unusual, a right hemicolectomy was suggested to the patient in early April 2020 as the main line of therapy. However, during the surgery, the tumor was found to be invading through the colon wall and extending and adhering to the right retroperitoneum, the ileal conduit, and the nephrectomy surgical bed. Thus, it was deemed unresectable. Postsurgical imaging in late April 2020 showed mediastinal/retroperitoneal adenopathy, indicative of metastatic diseases. Because of these findings, the goals of therapy became palliative. A traditional chemotherapy regimen targeting colonic cancers was selected, but it was quickly aborted because of clinical concern for the patient’s borderline kidney function related to her prior nephrectomy. Subsequently, because of the tumor’s dMMR status, pembrolizumab immunotherapy was initiated, and this remains her only treatment regimen.
Since the initiation of immunotherapy in June 2020, the patient has been feeling well, remains asymptomatic, and enjoys a better quality of life than she had before the right colon biopsy. A PET/CT scan performed 3 months after the start of the treatment detected a subtle decrease of the tumor size and lowered standard uptake value (from 27.6 to 9.9); later scans detected continued decrease in size and extent of the F-fluorodeoxyglucose– (FDG-) avid mass involving the ascending colon and lowered signal from right mesenteric lympho nodes. Furthermore, regular clinical follow-ups and PET/CT procedures were carried out with an average interval of 3.5 months, which have detected significant tumor shrinkage and diminished tumor involvement with the adjacent organs/sites. The 2 most recent (March and June of 2022) PET/CT scans detected only normal radiotracer distribution within the gastrointestinal system, with no colonic masses or lesions identified; no abnormal activity was identified within the liver, spleen, pancreas, or other abdominal organs.
Discussion
MiNENs are heterogeneous tumors, and the literature has described mixed tumor histomorphologies, such as sheets of confluent tumor cells intermixed with glands, acini, tubules, and mucin, frequent mitoses, and geographic necrosis. 4 In practice, these typical histomorphological features could be observed when there is ample diagnostic tissue, such as with resected MiNENs. However, these morphological features may not all be present in small or superficial biopsies. When dealing with superficial or limited biopsies, astute attention to the subtle growth pattern of the cell nests, intracellular mucinous vacuoles, brisk mitoses, inconspicuous signet ring cells, and eosinophilic and granular cytoplasm is imperative for reaching an accurate diagnosis. Considering the different patterns of neuroendocrine and non-neuroendocrine components 5 as well as the essential ancillary testing needed for uncovering a MiNEN, diagnosing MiNEN is more challenging and easier to miss if the diagnostic material comes from a small biopsy rather than a surgically resected tumor.
In our case, some of the abovementioned microscopic features were present, but well-formed glands were absent (Figure 2A-2C). Solid growth patterns, signet ring cells, absence of glands, and intracellular mucinous vacuoles can frequently be seen with poorly differentiated carcinomas. When the histomorphological findings were combined with the facts that tumor cells were labeled for pankeratin OSCAR (not shown), CDX2 (Figure 3D), mucicarmine and PAS-diastase (Figure 2E, 2F), and that diagnoses of urothelial (Figure 3E) and squamous cell carcinoma (Figure 3F) have been excluded, a poorly differentiated adenocarcinoma seemed to be the mostly likely diagnosis. However, upon appreciating the delicate cytoplasm, prominent nuclei, and high-grade cytology, we suspected a neuroendocrine nature and conducted ancillary testing, which detected expression of synaptophysin (Figure 3B), chromogranin (not shown), and more importantly INSM1 (Figure 3C) in tumor cells. As a recently developed immunophenotyping tool, INSM1 has shown superior specificity and sensitivity than the traditional markers in confirming neuroendocrine neoplasms. 18 These subtle histologic features can be easily missed, which may explain the documented tendency of underdiagnosing MiNENs. 5
Uncovering the neuroendocrine and non-neuroendocrine components only partially solved the puzzle for this case. Because of the patient’s personal history of multiple urothelial carcinomas and the various malignancies diagnosed among multiple family members, the possibility of a cancer-prone condition needed to be addressed. Lynch syndrome has been associated with accelerated carcinogenesis in the gastrointestinal tract, gynecologic tract, bladder, ureter, renal pelvis, and other organs and sites.7,19 Immunostains demonstrated MSI, and follow-up genetic testing excluded the possibility of Lynch syndrome in this MiNEN.
Despite Lynch syndrome being excluded, the MSI finding led to modified clinical scheduling and management planning, as studies have shown that MSI tumors tend not to respond as well to 5-fluorouracil–based management as microsatellite-stable tumors and are sensitive to immune checkpoint inhibitors such as PD-1 blockade.8,20-23 Notably, in 2017, the FDA approved the use of immune checkpoint inhibitors for any MSI tumors, regardless of the primary site.22-24 In order to combat this aggressive tumor following the aborted surgery, conventional chemotherapy was initially considered but quickly deemed unfeasible due to the patient’s baseline kidney function; subsequently, pembrolizumab immunotherapy was identified as her only treatment option.
From the perspective of clinical practice, once a diagnosis of MiNEN is established, regardless of clinical suspicion, the pathologist should evaluate the possibility of MSI and dMMR status and consider ordering relevant test(s), such as immunohistochemistry and/or molecular or mutational analysis. Additionally, pathological investigation should not end following the detection of MSI and tumor classification, especially if an advanced disease or a derailed clinical course is encountered; molecular and mutational analyses, such as next-generation sequencing, should be considered, if clinically feasible, to optimize disease management and personalize prognostication for a patient.
There were a few challenges and pitfalls encountered during this investigation that should be noted. First, thorough review of a patient’s personal and family history is pivotal when initiating patient care—a systemic review likely would have raised suspicion about a tumor-prone condition for this patient and led to ancillary testing including (but are not limited to) MSI evaluation, and a closer clinical follow-up schedule with shorter intervals might have detected this tumor at an earlier, less challenging stage. Second, the determination of MSI/dMMR status was an important consideration for disease management, even though this patient did not have Lynch syndrome. Additionally, diagnosing a MiNEN on a biopsy is challenging; whenever available, a resection specimen is preferred to ensure the correct diagnosis.
This is the first reported MSI-MiNEN involving the right colon, and it demonstrates the pivotal role of systemic examination and ancillary analysis during a patient’s clinical course. For this MSI-MiNEN case specifically, 30 months after initiation of treatment, the patient is enjoying improved quality of life while receiving low maintaining doses of pembrolizumab alone, and the tumor remains in remission. This outcome wouldn’t have been possible if the MSI status of the MiNEN was not uncovered. This case is a perfect example of pathologic evaluation for MSI making a major difference in patients’ care and outcomes.
Footnotes
Acknowledgments
We thank Dr. Janis de la Iglesia for all of her support, suggestions, and help in the review and submission process of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Written informed consent was obtained from the patient for the publication of this case report, and the Informed Consent Form can be made available upon reasonable request. This publication is considered exempt from Institutional Review Board review at Moffitt Cancer Center as the retrospective review of records for publication of a single-patient case report is not considered to be research involving human subjects.
