Abstract
Background
As yet, there is no unified method of treatment for the evaluation and management of gastric low-grade intraepithelial neoplasia (LGIN) worldwide.
Methods
Patients with gastric LGIN who had been treated with Helicobacter pylori eradication were gathered retrospectively. Based on several relevant characteristics described and analyzed by LASSO regression analysis and multivariable logistic regression, a prediction nomogram model was established. C-index, the area under the receiver operating characteristic curve (AUC), calibration plot, and decision curve analysis (DCA) were adopted to evaluate the accuracy and reliability of the model.
Results
A total of 309 patients with LGIN were randomly divided into the training groups and the validation groups. LASSO regression analysis and multivariable logistic regression identified that 6 variables including gender, size, location, borderline, number, and erosion were independent risk factors. The nomogram model displayed good discrimination with a C-index of .765 (95% confidence interval: .702-.828). The accuracy and reliability of the model were also verified by an AUC of .764 in the training group and .757 in the validation group. Meanwhile, the calibration curve and the DCA suggested that the predictive nomogram had promising accuracy and clinical utility.
Conclusions
A predictive nomogram model was constructed and proved to be clinically applicable to identify high-risk groups with possible pathologic upgrade in patients with gastric LGIN. Since it is regarded that strengthening follow-up or endoscopic treatment of high-risk patients may contribute to improving the detection rate or reducing the incidence of gastric cancer, the predictive nomogram model provides a reliable basis for the treatment of LGIN.
Introduction
Gastric cancer is one of the most common types of cancer in the world. In 2020, there were 1.09 million new cases of gastric cancer diagnosed around the world, among which, the new cases and deaths from China accounted for 44% and 49% of the total numbers worldwide respectively. 1 Early diagnosis is crucial for the prevention and treatment of gastric cancer. To date, Correa 2 cascade reaction is a widely accepted mode in terms of the pathogenesis of gastric cancer. The carcinogenic effect of gastric cancer is considered to be a continuous progression from atrophic gastritis to intestinal metaplasia (IM), intraepithelial neoplasia (GIN), and finally to adenocarcinoma. 3 During that process, low-grade intraepithelial neoplasia (LGIN) is regarded as a precancerous lesion. According to the tumor classification suggested by the World Health Organization in 2000, the recommended clinical treatment guidelines for gastric LGIN are as follows: (i) conservative treatment: drug treatment and follow-up; (ii) endoscopic therapy: (a) lesion mucosal resection: endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD); (b) Lesion mucosal injury: the main methods include high-frequency electrocoagulation, argon plasma coagulation, radiofrequency ablation, holmium laser treatment, microwave coagulation treatment, etc. Additionally, according to the guidelines of the American Gastroenterology Association (AGA) 4 and the British Gastroenterology Association (BSG), 5 endoscopic resection is recommended regardless of the size of the adenoma and whether it is associated with dysplasia. The American Society of Gastroenterol Endoscopy (ASGE) guideline 4 also suggests endoscopic resection as an effective treatment responding to gastric LGIN lesions that can still be found after 1 year of follow-up. Nevertheless, although clinical guidelines provide various treatment principles, there is still no unified solution for the evaluation and management of gastric LGIN all over the world currently, contrasting to the fact that endoscopic treatment is consistently recommended in many clinical guidelines for patients with high-grade intraepithelial neoplasia (HGIN).
With regard to drug treatment, follow-up, and endoscopic treatment, in most cases, clinicians usually decide on the diagnosis and treatment plan based on their personal clinical experience and the requirements of patients. Although the methods of treatment of gastric LGIN from various clinical guidelines differ, long-term follow-up is consistently recommended by almost all guidelines. However, this suggestion leads to increases in economic burden, psychological burden, and potential medical risk for patients. 6 Furthermore, current research shows that about 16.1% to 48.9% of biopsies diagnosed with gastric LGIN tend to undergo pathologic upgrade.7,8 Clinical strategies that are merely based on follow-up or drug treatment may miss the great opportunity of optimal treatment, and there is also a risk of missed diagnosis or misdiagnosis. It is widely recognized that early endoscopic intervention for patients with high-risk gastric LGIN brings considerable clinical and economic benefits.6,9,10 Therefore, it is of significant clinical necessity and value to find a new method to predict and evaluate the possibility of gastric LGIN progression. All in all, this study aims to develop a simple but effective predictive tool to evaluate the risk factors of gastric LGIN patients by the endoscopic diagnosis as well as to efficiently predict the pathologic upgrade risk of those patients. Based on that, the model would help to identify high-risk groups who may develop gastric cancer. Thus, the incidence rate of gastric cancer can be reduced by intensive monitoring and active treatment of the identified patients, and excessive physical examination and waste of medical resources can be avoided, as it provides valuable guidance for the clinical intervention of gastric LGIN patients.
Methods
Patients
The overall flow diagram is shown in Figure 1. The clinical data of 431 patients who had been diagnosed with gastric LGIN after the first gastroscopy in the Second Affiliated Hospital and Yuying Children’s Hospital of Wenzhou Medical University from December 2015 to December 2020 were retrospectively analyzed. The inclusion criteria of this study were: (i) diagnosis as gastric LGIN for the first time; (ii) reviewed gastroscopy at least 1 year after diagnosis; (iii) the result of examination on H. pylori (HP) by 13C-Urea Breath Test (UBT) was positive, and it was re-examined 1 month after the completion of quadruple therapy to ensure the eradication of HP; (iv) all patients did fine endoscopic examination: Mucosal lesions were observed and biopsied by 2 experienced endoscopists using Narrow Band Imaging (NBI) and Magnifying Endoscopy (ME) by means of painless endoscopy. At least 2 biopsies were grabbed and at least 3 clear pictures of the biopsy site were kept for the follow-up. The exclusion criteria are shown in Figure 1. Flow diagram of the study.
Ethics Approval Declaration
The study was conducted according to the World Medical Association Declaration of Helsinki and approved by the Ethics Committee of the Second Affiliated Hospital and Yuying Children’s Hospital of Wenzhou Medical University (LYCK2022-288). The informed consent form was signed by each patient. And all methods were carried out under relevant guidelines and regulations.
Demographic and Clinicopathological Variables
All information was gathered from the medical record systems and telephone follow-up inquiries. Demographic data of patients were retrospectively collected and recorded, which include age, gender, family history of gastric cancer, and concomitant diseases. Endoscopic characteristics of lesions were recorded, including lesion diameter, lesion location, boundary, multiple, rough mucosa, mucosal swelling, morphological characteristics (heave, flat, depression), erosion ulcer, and lesion color (redness, whiteness, yellowing). The patients who progressed to HGIN or cancer were incorporated into the upgrade group and those without upgrade pathology were included in the stable group.
Statistical Analysis
Random numbers were applied to classify 70% (n = 217) of all enrolled patients into the training group and 30% (n = 92) into the validation group. The least absolute shrinkage and selection operator (LASSO) method was used to select the optimal predictors of risk factors from the patients with gastric LGIN. Multivariable logistic regression analysis was adopted to build a predicting model, incorporating the predictive factors selected from the LASSO regression model. Based on the factors above, a nomogram model of pathologic upgrade prediction of gastric LGIN patients was constructed. Sociodemographic variables with a P-value of .1 and all variables associated with clinical characteristics were included in the model. To quantify the discrimination performance of the nomogram, Harrell’s C-index and the area under the receiver operating characteristic curve (AUC) were measured in both the training group and the validation group. In addition, calibration accuracy was evaluated by a calibration plot and the clinical effectiveness was assessed by a decision curve analysis (DCA).
SPSS (version 26.0) and the R software (Version 3.4.1; https://www.R-project.org) were applied for statistical analysis.
Results
Baseline Patient Characteristics in Training Cohort
Different Demographic and Clinical Characteristics Between the Stable and the Upgrade Groups.

LASSO Regression Analysis and Multivariate Logistic Regression Analysis
The LASSO regression model was used to select 6 predictors that present relatively high correlation from the 11 features in the training set. As result, gender, lesion size, location, borderline, number, and erosion proved to be the more important predictors with non-zero coefficients, as shown in Figure 2. After multivariate logistic regression analysis, gender (male), lesion size(≥1 cm), location (cardia or pylorus), borderline (yes), number(single), and erosion(no) were identified as independent predictors of the occurrence of pathologic upgrade in patients with gastric LGIN (Table 2). Figure 3 illustrated the association between these indicators and the probability of pathological upgrading using a forest map. Demographic and clinical feature selection using the LASSO binary logistic regression model. Multivariable Logistic Regression of Predictors for Pathologic Upgrade in Gastric LGIN Patients. Note: β is the regression coefficient. Associations between predictors and pathologic upgrade. Odds ratios with 95% confidence intervals were given.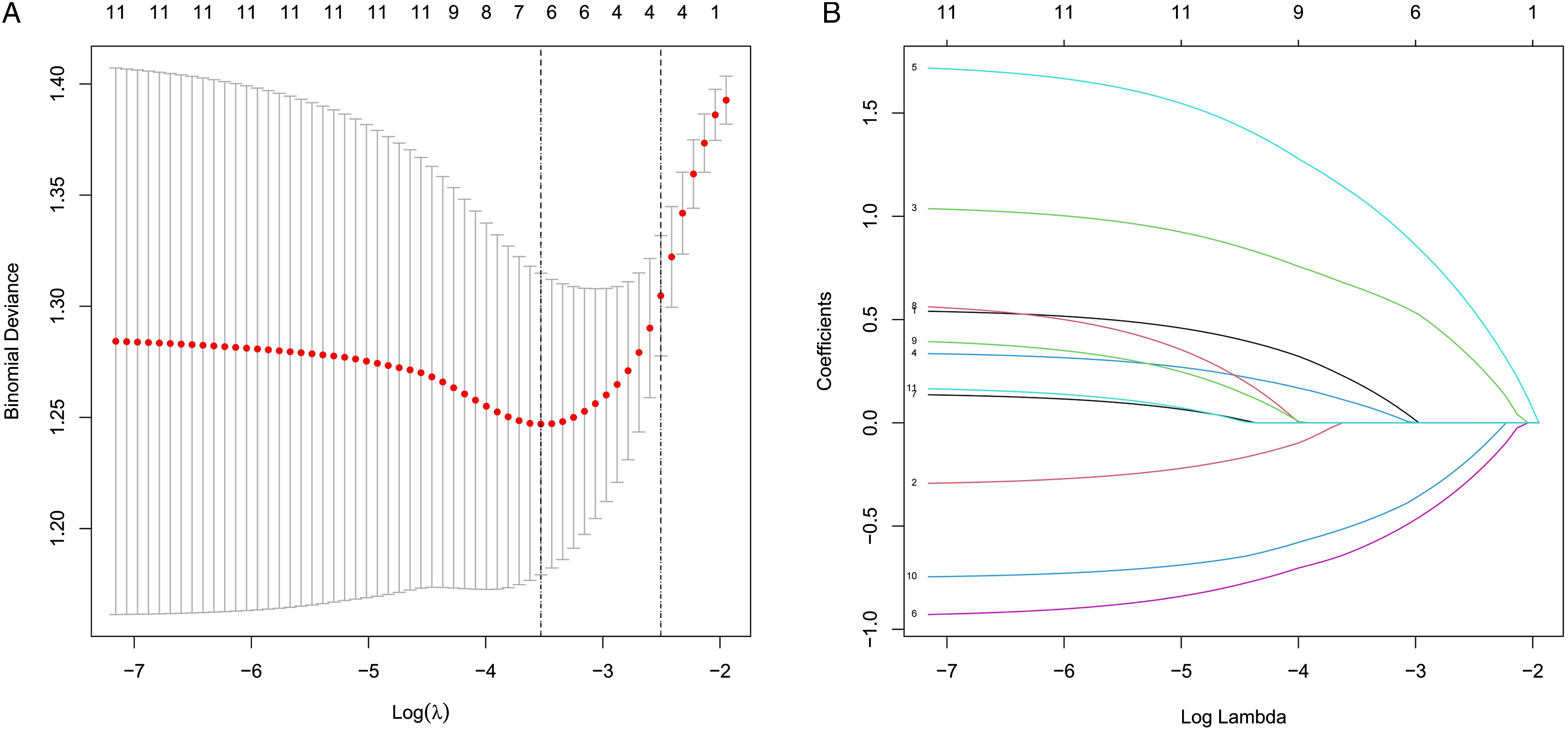


Development of a Nomogram Model of Pathologic Upgrade Prediction
Based on the Lasso analysis, a nomogram model for predicting pathologic upgrade in gastric LGIN patients was designed using 6 variables including gender, size, location, borderline, number, and erosion. As shown in Figure 4A, each of the independent predictors was projected upward to the value of the “points” at the top level of the nomogram to obtain a score within the range of 0 to 100. The total score of these points was recorded and then the corresponding probability of pathological progression was obtained through the total score line at the bottom of the nomogram. The higher the total score, the higher the risk of progression. The risk score distribution for each patient in the training and validation cohorts was shown in Figure 4B. The example given below served as an illustration. An adult male patient underwent a gastroscopy, and it discovered a borderless, erosive polyp with a diameter of .5 cm in the stomach antrum. According to the nomogram, the diameter of .5 cm, the location of the stomach antrum, the borderless score, and the erosion all received 0 points. The male score is 35 points, and the number a single score is 58 points. The final score is 93, and as a result, the probability of pathological upgrading is .20. Developed gastric LGIN pathologic upgrade nomogram. 
Validation of the Nomogram
The self-verification of the nomogram model was conducted. The C-index for the prediction nomogram was .765 (95% CI: .702-.828) for the training cohort. Then we drew the receiver operating characteristic curve (ROC) of predicted probability and the AUC value for the prediction of pathologic upgrade risk was .764, which indicated that the nomogram prediction model performed with great discrimination (Figure 5A). The calibration curve of the nomogram in gastric LGIN patients also demonstrated good agreement in this cohort (Figure 5B). The evaluation of the performance of the nomogram predicting the risk of pathologic upgrade in patients with gastric LGIN in the training cohort. 
Furthermore, in the validation cohort, there were 51 progressed cases (55.4%). The C-index was .768(95% CI: .672-.864), indicating that the prediction model is of considerable discriminability. The AUC value in the validation cohort was .757, although slightly lower than that of the training cohort, which still indicated that the prediction model showed high accuracy, as presented in Figure 6A. In addition, the Calibration curve manifested that the nomogram prediction model was of high consistency and fitting degree, as shown in Figure 6B. The evaluation of the performance of the nomogram predicting the risk of pathologic upgrade in patients with gastric LGIN in the validation cohort. 
Decision Curve Analysis of the Prediction Model
In addition, DCA was adopted to calculate the net benefit so as to evaluate the clinical utility of the nomogram, which is presented in Figure 7. The decision curves indicated that the nomogram was capable of acquiring great net benefits across a large range of high-risk thresholds (range from .06 to .91). Decision curve analysis for the validation set. A horizontal line indicates that all samples are negative and not treated, with a net benefit of zero. An oblique line indicates that all samples are positive, when the net benefit shows a negative slope.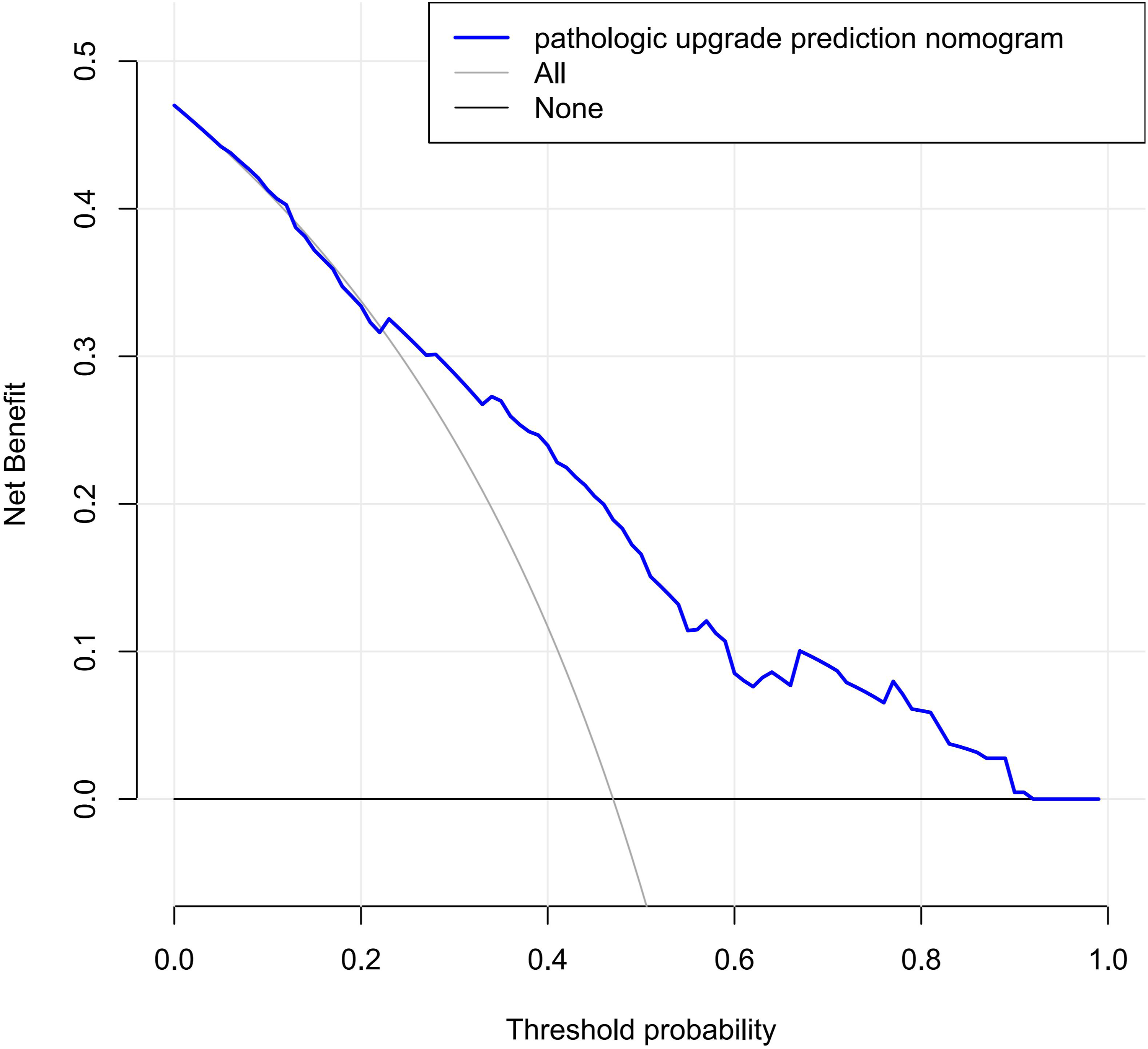
Discussion
Generally, according to the degree of cell atypia and structural disorder, GIN is divided into low-grade intraepithelial neoplasia (LGIN) and high-grade intraepithelial neoplasia (HGIN). Among that, LGIN is sub-classified into mild dysplasia and moderate dysplasia, and HGIN has severe dysplasia and carcinoma in situ. For patients with pathological diagnoses of gastric HGIN, endoscopic treatment is undisputedly recommended by most guidelines, but there is still controversy over the treatment of gastric LGIN.
Previous studies have researched on the pathological progression in patients with gastric LGIN, Park etc. Found that during a median follow-up of 58 months, 26.9% (7/26) of gastric LGIN patients had progressed to HGIN or cancer. 11 After excluding misdiagnosis, Zou etc. Detected that 12.2% (12/98) of patients with gastric LGIN had undergone pathological progression, with a median progression duration of 39.5 months. 9 Another recent study also showed that about 23% gastric LGIN cases tend to progress to cancer, while the incidence rate of HGIN was even higher (60% - 85%). 12 These data suggested that the risk of gastric LGIN deterioration to malignancy should not be ignored. For patients with high-risk of pathologic upgrade, it is necessary for us to recommend early endoscopic treatment, including endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD). For this purpose, our nomogram model aims to effectively identify gastric LGIN patients of different risk levels, so that accordant degrees of clinical intervention can be conducted to achieve more effective clinical utility.
6 risk factors were identified and included based on statistical analysis in this study: patient gender, lesion size, location, border line, number, and erosion. A national multicenter study13,14 showed that gender was an independent risk factor for high-risk gastric cancer, with OR 2.52 (1.92 to 3.30), P < .001, which was consistent with our results. However, our results indicated that there was no significant correlation between gastric LGIN progression and age. It contradicted the study of CAI et al, 13 but was consistent with previous studies,6,15 which was probably due to the influence of factors such as age structure and population size. Additionally, the lesion diameter was distinguished as ≥ 1 cm and < 1 cm in this study, implying that it was an independent risk factor for the risk of pathologic upgrade in patients with gastric LGIN. This is consistent with the guideline of endoscopic mucosal dissection 16 by the European Society of gastrointestinal endoscopy, the latter recommending endoscopic mucosal dissection for lesions >1.5 cm. Noticeably, lesion size is consistently considered a closely related feature to malignant tumor progression, but the threshold of risk classification remains to be further confirmed among a larger sample size. What’s more, this study detected that factors including the location of lesions (in the gastric fundus or cardia), the boundary, single onset, and whether there is erosion can impact the risk of pathological progression of patients with gastric LGIN. Kang et al retrospectively analyzed the data of 1006 cases of gastric LGIN resected by endoscopic submucosal dissection. 17 They observed that the lesion size ≥1 cm and various surface changes (erythema, nodule, depression, and erosion) were significantly related to the diagnosis of gastric LGIN. Yuqian C et al suggested that multiple location was an independent risk factor for prolonged or advanced progression in patients with LGIN via univariate and multivariate analysis. 6 Our research also found that the location of the lesion was to impact the pathologic progression, and we further pointed out that the fundus/cardia lesions might be more likely to increase the risk of progression into HGIN or cancer. The characteristics that describe the polyp surface shape, including roughness, swelling, shape, and color, were not demonstrated as independent risk predictors in this study. These 4 factors were subjective in judgment, which might be 1 of the reasons affecting the results. It suggested that we need more unified standards and a larger sample size to confirm the reliability of this model in the future. Although few studies have focused on gastric LGIN to date, based on the limited existing research results, we can find that the model constructed in this study has strong reliability for the risk prediction of pathological progression in patients with gastric LGIN.
However, referring to the published studies, the erosion on the lesion surface was also considered as a risk factor, which was contradictory with the results in this study. It may be due to the fact that among the selected cases reported above those with negative HP infection or that had turned negative after treatment were not strictly screened, given that the surface erosive ulcer was reported to be closely correlated with HP infection.18–21 Therefore, it is still doubtful whether the erosive ulcer at the lesion site in the above report above can be used as an independent risk factor if not excluding HP factors. In this study, all patients selected showed positive results in HP infection after the first examination and underwent quadruple bactericidal therapy. HP eradication was confirmed 1 month after completion of the treatment course, as we aimed to control the variables of HP infection. It implied that the lesion surface erosion and ulcer might be the protective factors for the pathological upgrading of gastric LGIN, although it still needs further confirmation by extended studies in the future.
As is shown in previous studies, a risk prediction nomogram presents favorable potential in clinical trial design and evaluation and has been widely used in prognostic models. In this study, a nomogram was used for the first time to construct the prediction model forecasting whether gastric LGIN patients would experience pathological progression. ROC curve analysis and calibration line were used to verify the prediction accuracy of the nomogram. Compared with the risk prediction model constructed by univariate and multivariate regression analysis, 6 the application of LASSO regression and nomogram made the analysis more intuitive, and the specific risk score also strengthened the accuracy of the prediction.
During the follow-up of this study, pathological reversal was found in some patients, although at least 2 biopsies were conducted on each lesion to exclude the interference of operating errors. It was even previously reported that 49.4% of cases were reversed during an average follow-up duration of 15 months. 23 Some studies pointed out that the occurrence of these cases with pathologic upgrade may be due to the difference of gastroscopic biopsy materials or pathological misdiagnosis when gastric LGIN was first diagnosed.22,24 In a study of 138 cases concerning gastric LGIN, the difference between preoperative endoscopic biopsy (PEB) and postoperative pathological examination (PPE) was analyzed. It was found that 47.8% of cases showed pathological upgrading due to misdiagnosis. 10 However, at present, most patients are diagnosed with gastric LGIN through the first gastroscopic biopsy, which inevitably takes on the risk of missed diagnosis and misdiagnosis. Therefore, for gastric LGIN patients of different pathological levels, the prediction of their risk is particularly important for the management of the disease, which can effectively reduce the clinical risk more or less.
Referring to the clinical baseline of the cases included in the training cohort, pathologic upgrade was discovered in 47.0% of the patients with gastric LGIN (102/217). Therefore, it is reasonable to recommend patients diagnosed with gastric LGIN by the initial endoscopy to conduct annual review. Meanwhile, according to the nomogram risk prediction model established in this study, if patients are identified being at high risk, endoscopic treatment (EMR or ESD) should also be performed during the initial gastroscopy, or endoscopic treatment should be performed once pathological escalation is found in more frequent gastroscopy visits.
Our research also has some limitations. Firstly, this nomogram model is based on retrospective single-center data set, which weakens the credibility of the model and limits the scope of its application. Secondly, although self-verification and internal validation were carried out in this study, external validation has not been included. But the latter is also important to prove the accuracy of the model and the lack of. It may lead to statistical analysis deviation after elimination. To establish a more improved prediction model of pathologic upgrade in patients with gastric LGIN that is able to provide more evidence, further multi-center and large sample clinical research is still needed in the future.
Conclusions
In summary, this study developed a nomogram model of pathologic upgrade prediction in patients with gastric LGIN. This model proves to be efficient in providing a valuable reference for clinical decision-making in the treatment of patients with gastric LGIN.
Footnotes
Acknowledgments
We thank the Second Affiliated Hospital of Wenzhou Medical University and all the authors for their joint efforts.
Authors’ Contributions
Yejiao Ruan and Xuanping Xia conceptualized and designed the study. Yejiao Ruan, Zhenzhai Cai, and Xuanping Xia were responsible for the conception, design, quality control of this study, reviewed, and edited the manuscript. Guangrong Lu performed data extraction and statistical analyses. Yejiao Ruan and Yuesheng Zhu were major contributors to writing the manuscript. Xianhui Ma, Yuning Shi, Xuchao Zhang, and Zheng Zhu participated in data extraction and statistical analyses. All authors have read and approved the final version of the manuscript.
Availability of Data and Material
All data generated or analyzed during this study are included in this published article.
Declaration of Conflicting Interests
The authors declare that there are no conflicts of interest.
Ethical Approval
All experimental protocols were approved by the Ethics Committee of the Second Affiliated Hospital and Yuying Children’s Hospital of Wenzhou Medical University (LYCK2022-288). Informed consent was obtained from all subjects. And all methods were carried out under relevant guidelines and regulations.
Informed Consent
All authors have read and approved the final version of the manuscript and consented to publication.
