Abstract
Background
The purpose of this study was to explore the feasibility, safety and efficacy of iodine-125 seed implantation in the treatment of dysphagia of advanced esophageal cancer.
Methods
We retrospectively analyzed patients with advanced esophageal cancer who underwent EUS-guided iodine-125 seed implantation or conventional chemoradiotherapy in our hospital. The propensity score match was used to reduce the baseline differences.
Results
A total of 127 patients were enrolled, 17 patients received EUS-guided iodine 125 seed implantation (Group A), 31 patients received radiotherapy (Group B), 38 patients received chemotherapy (Group C) and 41 patients received chemotherapy combined with radiotherapy (Group D). At half month postoperatively, the dysphagia remission rate in Group A (100%) was better than that in Groups B (39.3%), C (20%), D (15.8%), respectively, in the original cohort (P < 0.01); At 1 month postoperatively, the dysphagia remission rate in Group A (86.7%) was better than that in Group B (57.1%) (P > 0.05), Group C (25.7%) (P < 0.05) and Group D (34.2%) (P < 0.05), respectively, in the original cohort. There was no statistically significant difference in median overall survival (OS) between Group A (16 months) and Group B (37 months) (P = 0.149), and between Group A (16months) and Group C (16 months) (P = 0.918) in the original cohort. The mean OS of Group D (54 months) was better than that of Group A (20 months) in the original cohort (P = 0.031). The incidences of grade ≥2 myelosuppression in Groups B, C, and D were 12.9%, 28.9%, and 43.9%, respectively; the incidence of grade ≥2 gastrointestinal adverse events in Groups B, C, and D were 12.9%, 15.8%, 12.2%, respectively. No serious adverse events were found in Group A. The radiation dose around the patient was reduced to a safe range after the distance from the implantation site was more than 1 m (4.2 ± 2.6 μSv/h) or with lead clothing (0.1 ± 0.07 μSv/h).
Conclusions
Compared with conventional radiotherapy or chemotherapy alone, iodine-125 seed implantation might improve dysphagia more quickly and safely, further clinical data is needed to verify whether it could effectively prolong the OS of patients.
Introduction
Esophageal cancer was most common in East Asia and East Africa, and the majority of cases concentrated in China with approximately 307,000 cases. 1 According to Global Cancer Statistics 2020, esophageal cancer ranked seventh in incidence (604,000 new cases) and sixth in mortality (544,000 deaths). 2 For resectable esophageal cancer, surgery was the mainstay, while radiotherapy(RT) and chemotherapy were the best adjuvant treatments. 3 For unresectable esophageal cancer, chemotherapy and radiotherapy were still the main methods. Conventional radiochemotherapy, and immunotherapy were constantly improving, while new treatment technologies were constantly being developed to combine them, they were continuously optimizing the treatment of esophageal cancer. 4 However, some patients are in the advanced stage or advanced age at the time of diagnosis, and could not tolerate conventional radiochemotherapy due to their poor basic status. 5 There was an urgent search for an effective alternative treatment for them. As main particles of brachytherapy, iodine-125 seeds were widely applied in the management of malignant tumors, especially in prostate cancer.6-8 In this study, we were the first to attempt to implant iodine-125 seeds into esophageal cancer tissue under the guidance of endoscopic ultrasonography (EUS). In order to explore the feasibility and efficacy of EUS-guided iodine-125 seed implantation in esophageal cancer, we compared it with conventional radiochemotherapy to supply more scientific foundation for the application of this method.
Material and Methods
Population
From January 2017 to March 2022, a total of 127 patients with definite diagnosis of esophageal cancer or gastroesophageal junction cancer who received iodine-125 seed implantation (Group A), radiotherapy alone (Group B), chemotherapy alone (Group C), or radiotherapy combined with chemotherapy (Group D) were collected. Group A was the experimental group, and the other three groups were the control group. This is a retrospective study. The collected information mainly includes age, gender, tumor stage, dysphagia grade, overall survival (OS), adverse events and so on. The reporting of this study conforms to STROBE guidelines. 9 The collected adverse events included the entire process from the beginning of treatment to the end of follow-up. For the same adverse event recurring in case records, only the most severe events were collected. Adverse events that require subjective evaluation should be evaluated by two investigators. Controversial adverse events needed to be discussed and decided with a third investigator. This study has been approved by the Ethics Committee of the Chinese People’s Liberation Army General Hospital (S2021-421-01). The clinical data in this study was retrospective and anonymous. Therefore, the informed consent was waived. All patients or their families have been informed of the general procedures, possible adverse events and signed informed consent.
Inclusion criteria: (1) Patients were diagnosed with esophageal cancer or gastro-esophageal cancer by imaging, pathology or digestive endoscopy; (2) Patients lost the opportunity for R0 resection or had poor response to radiotherapy and chemotherapy; (3) No severe cardiorespiratory dysfunction or infection; (4) Expected OS greater than 3 months; Exclusion criteria: (1) The patient has previously undergone radical surgery for esophageal cancer; (2) The patient with other primary malignancies; (3) The patient with non-primary esophageal cancer; (4) Postoperative dysphagia score could not be accurately assessed in patients.
Iodine-125 seed (China Isotope Radiation Co, LTD, Beijing, China) consists of a titanium capsule containing iodine-125 adsorbed on a passivated palladium rod, which could generate 27.4 keV X-rays with a half-life of 59.4 days. The activity of single seed was 0.4 mCi.
Patient and Public Involvement
Patients or members of the public were not involved in the study since it was a deidentified data and a retrospective study.
Dysphagia Assessment
Dysphagia was scored according to the following criteria 10 (Grade 0: normal diet; Grade 1: able to eat some solid foods; Grade 2: able to eat only semi-solid foods; Grade 3: able to swallow liquids only; Grade 4: complete dysphagia).
Implantation of Iodine-125 Seeds Under the Guidance of EUS
Before the procedure, the patients and their families were informed of the general process and possible adverse events, then signed relevant informed consent. The main procedure was performed by an experienced endoscopist. Preoperative upper gastrointestinal angiography was performed to assess the location, length and extent of esophageal strictures, chest CT were performed to measure the tumor size to determine the number of implanted seeds. Iodine-125 seeds were transported in lead tanks by professional personnel before being used. All personnel involved in the operation were trained in radiation safety before operation. Similar to our previous research, 11 based on our clinical experience, we adopted a straightforward method for seed implantation. Under EUS (Olympus Corporation, Tokyo, Japan) guided, the19 G puncture needle was inserted into the distal edge of the tumor, avoiding blood vessels and vital organs. Then the needle core was removed and the seed was inserted into the needle. When the needle core was withdrawn, the seeds were pushed into the tumor tissue, and the distal end of the seed was guaranteed to be 1-2 mm apart. The seeds were evenly implanted in a straight line with a density of 5-10 seeds/cm. In order to prevent the seed from flowing out of the esophageal lumen during needle extraction, the implantation was suspended when the needle was pulled to the outer edge of the esophageal lumen at 2-3 mm. There was no overlap between puncture tracks, which meant the last seed implanted could not be seen during the next puncture. The seeds were guaranteed as evenly as possible within the tumor. Finally, the number of implanted seeds was accurately recorded and X-ray was used to evaluate the position and distribution of iodine-125 seeds. The operation was completed.
Treatment Policy
After obtaining the consent of the patients and their primary family member, the treatment regimens were selected according to the patient’s condition and physical condition. In our hospital, for patients with an opportunity for R0 resection, surgery was recommended. If the disease progressed and the opportunity for surgery had been lost, chemotherapy was recommended. Whether to combine radiation therapy required to be judged by the radiologist based on the patient’s physical condition and the size of the irradiation range. If the patient required it or if it was considered a better option than chemotherapy after evaluation by the attending physician, radiotherapy was recommended. Especially for upper esophageal cancer, radiotherapy was usually recommended due to the difficulty of surgery and poor postoperative quality of life. Iodine-125 seed implantation was recommended for patients who could not tolerate or refuse radiotherapy or chemotherapy and lose the opportunity for surgery.
Chemotherapy and Radiotherapy
The initial treatment plan of Group C and Group D was mainly to use first-line double-drug combined chemotherapy regimen, at least 4 cycles. The chemotherapy regimens were mainly the Nab-paclitaxel (260 mg/m2) plus platinum-based chemotherapy ((Nedaplatin (25 mg/m2), or Lobaplatin (50 mg/m2), or Carboplatin (300 mg/m2), or Cisplatin (75 mg/m2)). A small number of patients were given docetaxel (l75 mg/m2) plus platinum or etoposide (100 mg/m2) plus platinum, and some patients were given immunotherapy (Pembrolizumab (200 mg, q3w), Nimotuzumab (100 mg, q1w), Toripalimab (3 mg/kg, q2w)) during the treatment. Chemotherapy combined with the radiotherapy group meant that the first cycle of chemotherapy was not less than 1 cycle, and radiotherapy was added to the subsequent treatment. After first-line chemotherapy, if the patient’s disease progressed or intolerable adverse events occurred, second-line chemotherapy would be selected.
Radiotherapy was performed using a 6 MV linear accelerator. Preoperative localization was performed with enhanced CT and given rotational Intensity Modulated Arc Therapy (IMAT) or Volumetric Modulated Arc Therapy (VMAT). The tumor target area and the corresponding lymphatic drainage area were treated with radiotherapy with a total dose of 40-70 Gy, 25-35 times of radiotherapy, 5 times a week, 1.8-2.2 Gy for each time.
Post-Treatment Care and Follow-Up
Iodine-125 seeds Implantation
The patient’s vital signs were monitored for at least 6 hours, and the patient was instructed to fast for 12 hours. Close attention was paid to if any suspicious adverse events had happened, while patients without them could be discharged. Patients were followed up regularly after discharge. They were required to return to the hospital for re-examination, and seed replantation was performed if necessary 2-3 months later. All patients and their primary family members received relevant radiation protection training, including lead clothing, safe distance, and monitoring feces (if shedding seeds were found, they were required to send back in lead boxes).
Radiotherapy and Chemotherapy
Close attention was paid to whether the patient had gastrointestinal reactions, bone marrow suppression, allergic reactions, and damage to important organs (such as the heart and liver, etc.). Medication was administered to prevent or reduce gastrointestinal reactions and liver damage. Lesion size, blood routine and tumor markers were routinely monitored after operation.
Dysphagia improvement was defined as a ≥1 point decrease in dysphagia score. Technical success was defined as the successful completion of the procedure. Clinical success was defined as improvement and disappearance of dysphagia. OS was defined as the time from the first treatment (experimental group: the first iodine-125 seed implantation; control group: the first radiotherapy or chemotherapy) to the death or loss of follow-up. All patients were followed up by telephone until March 2022.
Statistical Analysis
SPSS25.0 was used in this study. Continuous variables were compared between the two groups using the independent sample t-test, and categorical variables were compared using the chi-square test or Fisher’s test. Paired sample t-test was used for the statistics of radiation dose rate. Normally distributed quantitative data were expressed as
To minimize differences in baseline clinical characteristics between the two groups, propensity-matched scores (PSM) were used when comparing iodine-125 seed implantation (Group A) with chemotherapy (Group C) and chemotherapy combined with radiation (Group D). Age, gender, tumor location, pathological type, and clinical stage were used as covariates, and propensity score values were estimated by logistic regression. The 1:3 nearest neighbor matching method was used for the matching between Group A and Group C, as well as between Group A and Group D, but the caliper value of the former was 0.2, and the caliper value of the latter was 0.3. Statistically significant differences were indicated by two-tailed P < 0.05.
Results
Baseline Clinical Characteristics
Baseline characteristics of iodine125 seeds and the radiotherapy.

Characteristics of the original cohort and matched cohort.

Improvement in Dysphagia
It could be seen from Figure 1A that among all available dysphagia data in original cohort, two weeks after the operation, the proportion of patients with dysphagia relieved in Group A was better than the other three groups (A vs B, 100% vs 39.3%, P = 0.000; A vs C, 100% vs 20%, P = 0.000; A vs D, 100% vs 15.8%, P = 0.000). One month after the operation, the percentage of patients with dysphagia relieved in Group A was 86.7%, which was significantly better than that in Group C (25.7%) (P = 0.000) and Group D (34.2%) (P = 0.002), and also better than Group B (57.1%) (P = 0.104), although there was no significant difference. Two months after the operation, there was no significant difference in the percentage of patients who achieved dysphagia improvement in the comparison of Group A with the other three groups (A vs B, 76.9% vs 75.0%, P = 0.065; A vs C, 76.9% vs 48.6%, P = 0.152; A vs D, 76.9% vs 42.1%, P = 1.000). In the original cohort, There was no significant difference in the proportion of dysphagia relief between Group A and the other three groups at 3 (A vs B, 72.7% vs 79.1%, P = 0.322; A vs C, 72.7% vs 56.3%, P = 0.544; A vs D, 72.7% vs 50.0%, P = 0.685), 6 (A vs B, 57.1% vs 66.7%, P = 0.846; A vs C, 57.1% vs 51.9%, P = 1.000; A vs D, 57.1% vs 69.4%, P = 0.678), and 9 months (A vs B, 66.7% vs 62.5%, P = 1.000; A vs C, 66.7% vs 60.9%, P = 1.000; A vs D, 66.7% vs 72.7%, P = 1.000) postoperatively (Figure 1A). Percentage of patients with improvement in dysphagia after treatment. A. Original cohort, 0.5 month: 100%, 39.3%, 20%, 15.8%; 1 month:86.7%, 57.1%, 25.7%, 34.2%; 2 months:76.9%, 75.0%, 48.6%, 42.1%; 3 months: 72.7%, 79.1%, 56.3%, 50.0%; 6 months: 57.1%, 66.7%, 51.9%, 69.4%; 9 months: 66.7%, 62.5%, 60.9%, 72.7%; B. Matched cohort, 0.5 month: A vs C,100% vs 23.5%; A vs D, 100% vs 23.5%; 1 month: A vs C, 85.7 vs 35.7%; A vs D, 85.7% vs 47.1%; 2 months: A vs C, 66.7% vs 52.9%; A vs D, 66.7% vs 52.9%; 3 months: A vs C, 40.0% vs 40.0%; A vs D, 60.0% vs 58.8%.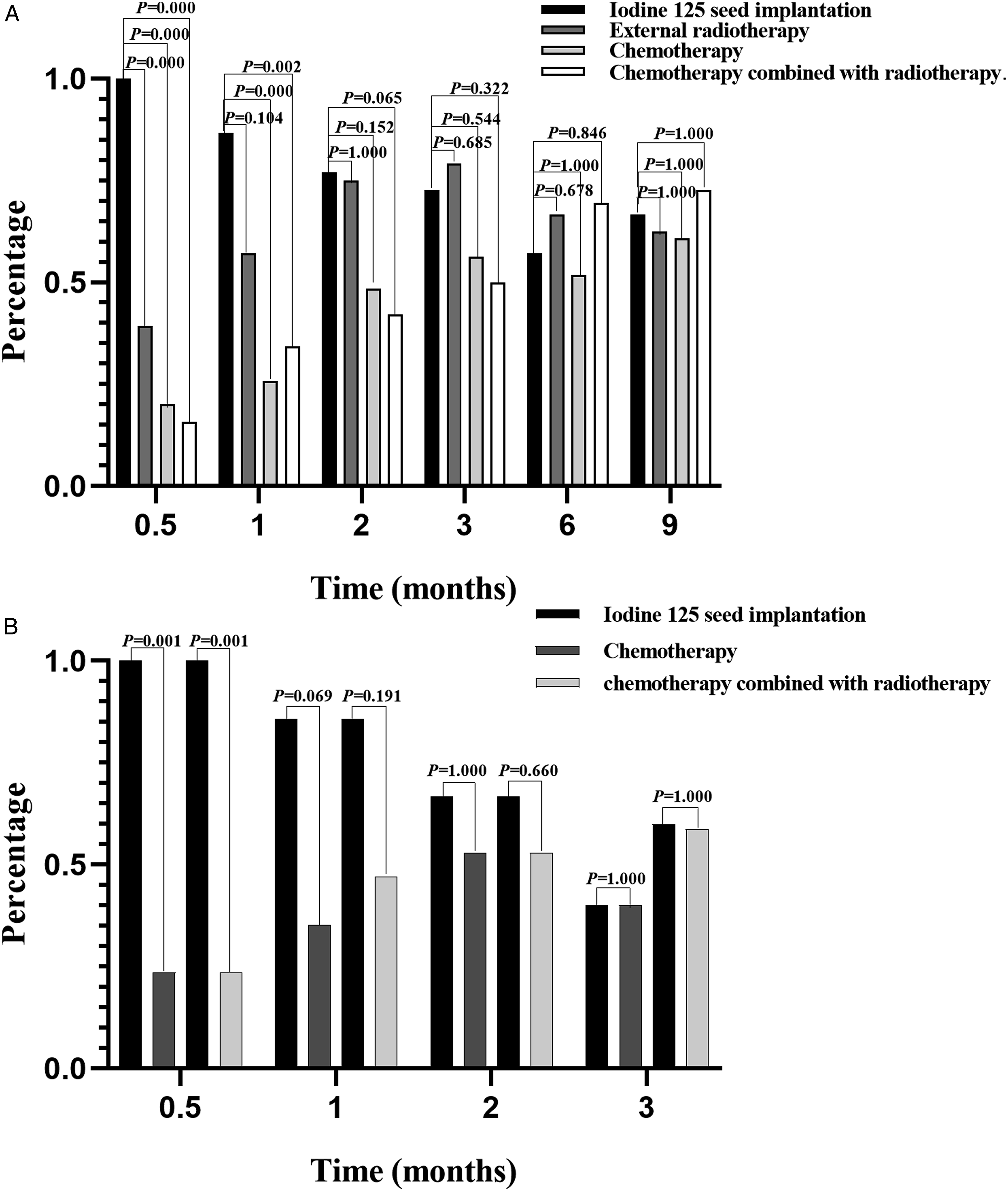
It could be seen from Figure 1B that among all available dysphagia data in matched cohort, two weeks after the operation, the proportion of patients with dysphagia relieved in matched Group A was better than matched Group C (matched A vs matched C, 100% vs 23.5%, P = 0.001) and matched Group D (matched A vs matched D, 100% vs 23.5%, P = 0.001). One month after the operation, the percentage of patients with dysphagia relieved in matched Group A was better than matched Group C (matched A vs matched C, 85.7% vs 35.3%, P = 0.069) and matched Group D (matched A vs matched D, 85.7% vs 47.1%, P = 0.191). At 2 and 3 months postoperatively, there was no significant difference between matched Group A and the other two groups (2 months, matched A vs matched C, 66.7% vs 52.9%, P = 1.000; matched A vs matched D, 66.7% vs 52.9%, P = 0.660) (3 months, matched A vs matched C, 40.0% vs 40.0%, P = 1.000; matched A vs matched D, 60% vs 58.8%, P = 1.000) (Figure 1B).
Overall Survival
The median OS was 16 months in Group A (95%CI, 9.734-22.266) and 37 months (95% CI, 23.637-50.363) in Group B (P = 0.149) (Figure 2A). Before matching, median OS was both 16 months in Group A (95%CI, 9.734-22.266) and Group C (95%CI, 9.594-22.406) (P = 0.918) (Figure 2B); after matching, median OS was 12 months (95%CI, 0.000-27.416) in Group A and 19 months (95%CI, 5.253-32.747) in Group C, but the difference was still not statistically significant (Figure 2C) (P = 0.453). Before matching, the mean OS was 20 months in Group A (95%CI, 12.685-27.824) and 54 months (95%CI, 42.469-65.331) in Group D (P = 0.031) (Figure 2D); after matching, the mean OS was 18 months (95%CI, 8.102-28.648) in Group A and 59 months (95%CI, 42.435-75.222) in Group D (P = 0.045) (Figure 2E). Survival curves for different groups. A. Survival curve of iodine 125 seeds implantation (Group A) and external radiotherapy (Group B); B. Survival curves of the original cohort of iodine-125 seeds implantation (Group A) and chemotherapy (Group C); C. Survival curves of the matched cohort of iodine-125 seeds implantation (matched Group A) and chemotherapy (matched Group C); D. Survival curves of the original cohort of iodine-125 seeds implantation (Group A) and chemotherapy combined with radiotherapy(Group D); E. Survival curves of the matched cohort of iodine-125 seeds implantation (matched Group A) and chemotherapy combined with radiotherapy (matched Group D).
Adverse Event Rate and Radiation Dose Monitoring
Adverse event (Original cohort).

Radiation dose was measured at different monitoring distances after iodine-125 seed implantation (X ± S, μSv/h).

Discussion
Dysphagia was a common symptom of esophageal cancer. 12 Both in the original cohort and the matched cohort, we found that the percentage of patients with dysphagia remission two weeks, one month and two months after EUS-guided iodine-125 seed implantation were all better than the other three groups (Figure 1). It was worth mentioning that, two weeks after the operation, the percentage of dysphagia remission was significantly better than the other three groups, both in the original cohort and the matched cohort (Figure 1A,1B). Although previous studies have reported that the improvement of dysphagia after external beam radiotherapy could reach 67%, 13 it was still worse than iodine-125 seed implantation (100%). We also found that, one month after the operation, the percentage of dysphagia relief in Group A was also significantly better than that in Group C and Group D in the original cohort, respectively, and although it was also better than Group B, the difference was not statistically significant. In the matched cohort, although the percentage of dysphagia relief in Group A from 1 to 3 months after the operation was not significantly different from the other three groups, the overall trend was similar to the original cohort. The reason for the lack of significant difference may be the small number of patients available for study after matching. For the same reason, it was regrettable that we were unable to obtain valid data on dysphagia relief at 6 and 9 months postoperatively in the matched cohort. Therefore, iodine-125 seeds implantation may be a more effective way to improve dysphagia in patients with esophageal cancer in the short term. It is undeniable that radiotherapy is also an effective treatment for improving dysphagia in patients with esophageal cancer. It should be pointed out that most of therapeutic regimen for Group D were chemotherapy first, followed by radiotherapy, resulting in that the improvement of dysphagia in Group D was not better than Group C in the short term. During follow-up, we found that in the original cohort, the percentage of dysphagia remission was 55.1% and 66.7% in the iodine-125 seed implantation group at 6 and 9 months, respectively, which was not statistically different from the other three groups. Therefore, the long-term effect of iodine-125 seed implantation is also encouraging.
Iodine-125 seed implantation might improve dysphagia better and faster than the other three groups. Although external radiation therapy is also an effective modality for improving malignant dysphagia, it was always accompanied by intolerable adverse events. 14 These serious adverse events, such as gastrointestinal reactions and radiation esophagitis, also interfered with the patient’s normal diet. As a systemic drug regimen, chemotherapy had limited improvement in dysphagia and there were few relevant studies; conversely, dysphagia was often caused during chemotherapy. 15 Due to the severe digestive tract reaction of radiochemotherapy, patients had a poor appetite in the early stage of treatment, and some patients needed intravenous nutrition, which required a recovery time. However, the implantation of iodine-125 seeds had less damage to the patient’s gastrointestinal tract, and hardly affected the patient’s appetite. With the guidance of EUS, iodine-125 seeds could be uniformly implanted into the target lesions, and the esophageal cancer tissue could be irradiated more accurately. Furthermore, its half-life is close to two months, so tumor tissue could be accepted irradiation continuously. Through regular follow-up, new esophageal lesions could be replanted in time. Therefore, its long-term effect of relieving dysphagia could also be guaranteed. In summary, compared with conventional chemotherapy or radiotherapy, EUS-guided iodine-125 seed implantation might more quickly improve dysphagia.
Due to the small number of patients available for study, we had to summarize all patients who received iodine-125 seed implantation. Currently, Group A could be divided into the following three types: 1. The patient had received radiotherapy or chemotherapy in the early stage, but the effect was not significant, so he/she chose iodine-125 seed implantation (8 cases); 2. The patient was found to have no indication for radiotherapy or chemotherapy by preoperative evaluation, and then received iodine-125 seed implantation (6 cases); 3. The patient had indication of radiotherapy and chemotherapy but the patient refused, and then chose iodine-125 seed implantation (3 cases). Strictly speaking, patients with the third condition were more suitable for comparison with the other three groups. Of these 3 patients, 2 died and 1 survived, and the OS were 34, 27, and 40 months, respectively. In a study comparing it with 3-Dimensional Conformal Radiotherapy, the median OS of Intensity-Modulated Radiotherapy was 43.2 months, 16 which was close to the median OS (37 months) in our radiotherapy group. Only from the survival curve in Figure 2A, the OS of Group A was worse than Group B, but the difference was not statistically significant. Moreover, the OS of two of the last three cases in Group A was very close to the median OS of Group B. In a study of carboplatin and paclitaxel as first-line chemotherapy for esophageal cancer, the median OS was 15.5 months. 17 And the median OS was 16 months in the original cohort of our chemotherapy group. The differences in OS between Group A and Group C were not statistically significant, either before or after matching (P > 0.05). However, the OS of the last 3 patients in group A was better than that of median OS in Group C. In the original cohort and the matched cohort, the median OS was not available in Group D due to too few deaths. However, it could be seen from Figures 2(D) and 2(E), the mean OS of Group A was significantly shorter than that of Group D (P < 0.05). Therefore, compared with radiotherapy or chemotherapy alone, although no significant survival advantage has been found in iodine-125 seeds implantation group, the median OS beyond 12 months was also encouraging. Iodine-125 seeds implantation might have application prospects. Whether iodine-125 seed implantation has a survival advantage compared to conventional radiochemotherapy requires more data to verify in the future.
No serious adverse events were found in Group A. On the one hand, it benefited from the operator's superb endoscopic ultrasound technology. On the other hand, due to its unique radiation characteristics, the effective radiation radius of iodine-125 seeds is only 1.7-2.0 cm, so the damage to the surrounding normal organs is small. However, adverse events such as gastrointestinal reactions (nausea, vomiting, diarrhea), bone marrow suppression, radiation esophagitis, and radiation pneumonitis caused by conventional radiochemotherapy still plague the majority of patients (Table 3). Therefore, some elderly patients with poor basic status usually could not tolerate conventional radiochemotherapy. A study suggested that radiotherapy and chemotherapy should be carefully selected for the elderly aged 80 years and above. 18 For these patients, especially those with obvious dysphagia that seriously affected their quality of life, EUS-guided iodine-125 seed implantation with less side effects and significant effect might bring them hope. In particular, we found that the combination of radiotherapy and chemotherapy aggravated the occurrence of adverse event. These adverse events also limited the application of radiochemotherapy. Currently, iodine- 125 seeds were loaded on esophageal stents, which was the main application in esophageal cancer.19-23 However, the seeds loaded into esophageal stents were limited at one time and the therapeutic effect on the primary tumor was also limited. The detachment and displacement of the stent was still an unavoidable problem. In addition, after the patient received iodine-125 seeds implantation, we would monitor the radiation dose rate at different distances in front of the patient’s chest, and found that the radiation dose decreased significantly with the increase of distance. According to the “Basic Standards for Ionizing Radiation Protection and Radiation Source Safety” promulgated in 2002, the radiation dose should be lower than 2.5 μSv/h after iodine-125 seed implantation. We found that the national standard could be reached when the distance was more than 3 feet in front of the chest (4.2 ± 2.6 μSv/h). After the patient wore a 0.25 mmpb lead coat, the radiation dose to the chest dropped to 0.1 ± 0.07 μSv/h. Therefore, iodine-125 seed implantation might be a safe and controllable treatment method.
Based on the current study, it is undeniable that chemoradiotherapy remains the first choice for the treatment of unresectable esophageal cancer. But iodine-125 seed implantation offers a new option for patients who have lost the indication for radiochemotherapy, or who refuse radiochemotherapy for personal reasons. Although we tried our best to perfect this study, there were still some limitations. First, due to the single-center study and the early clinical application of the technology, the number of patients available for study was small. Therefore, in order to ensure a sufficient number of patients to meet the requirements of the study, we had to increase the caliper value of PSM; Second, since very few data on dysphagia improvement were available after matching at 6 and 9 months postoperatively, Figure 1B only showed the proportion of dysphagia improvement within three months after operation. Large-scale randomized controlled and multicenter clinical studies are needed to verify the generalizability of the findings in the future. In summary, compared with conventional chemotherapy or radiotherapy, EUS-guided iodine-125 seed implantation might improve dysphagia more quickly and safely, especially for some patients with poor radiotherapy and chemotherapy effects. Whether it could effectively prolong the OS of patients, further clinical data support is needed.
Strengths and Limitations of This Study
We were the first attempt in the world to directly implant iodine-125 seeds into cancer tissue to relieve dysphagia in patients with unresectable esophageal cancer;
Propensity score match was used to reduce baseline differences between experimental and control groups, minimizing selection bias;
Our research has proven that this is an effective and safe method to relieve dysphagia; The main disadvantage of this study was that the scale of the study was too small, and large-scale randomized controlled trials were needed in the future.
Footnotes
Acknowledgments
Special thanks to the staff of Gastroenterology Endoscopy Medical Center of the Chinese People’s Liberation Army General Hospital for their support and cooperation during the collection of clinical data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors disclosed no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Science and Technology Commission of the Central Military Commission (Grant No. 17-163-12-ZT-002-048-01).
Ethics Approval
This study has been approved by the Ethics Committee of the Chinese People’s Liberation Army General Hospital, Ethics No. S2021-421-01.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
