Abstract
Purpose
This retrospective analysis aimed to evaluate the clinical outcomes and cost-effectiveness of long-acting granulocyte-colony stimulating factor as primary prophylaxis of neutropenia caused by chemotherapy for breast cancer.
Methods
Patients with breast cancer who received long- or short-acting granulocyte-colony stimulating factor as primary prophylaxis of neutropenia were enrolled in this study, and incidences of neutropenia were compared between two groups. A decision-analytic and a Markov model were used to compare the health benefits and costs of utilizing long- vs short-acting granulocyte-colony stimulating factor as the primary prophylaxis from the perspective of the Chinese health service system. Subsequently, one-way deterministic and probabilistic sensitivity analyses were conducted. The incremental cost-effectiveness ratios were calculated in baseline and sensitivity analyses.
Results
Patients receiving long-acting granulocyte-colony stimulating factor as the primary prophylaxis of chemotherapy-induced neutropenia experienced a significant lower incidence of this adverse event, compared with the short-acting one for 2 to 7 days. The outcomes of baseline analysis indicated that long-acting granulocyte-colony stimulating factor had a gain of 0.08 quality-adjusted life years and costed $149 more than the short-acting one, yielding an incremental cost-effectiveness ratio of $1792 per quality-adjusted life year. The sensitivity analysis proved the stability of our models and economic efficiency of long-acting granulocyte-colony stimulating factor.
Conclusions
Patients receiving long-acting granulocyte-colony stimulating factor as primary prophylaxis of neutropenia experienced lower risk of this event compared with those underusing short-acting one. The long-acting granulocyte-colony stimulating factor may be a more cost-effective strategy for primary prophylaxis of neutropenia than short-acting one, considering the Chinese willingness-to-pay threshold of $12158.6 per quality-adjusted life year.
Keywords
Introduction
Neutropenia is a major complication in patients receiving chemotherapy. The role of neutrophils is to provide immunity, against bacteria, viruses, and other foreign substances, Thus, a decline in their numbers may result in a weakened immune system. Neutrophils originate from hematopoietic stem cells in the bone marrow and have a half-life of 8 to 12 hours. Therefore, they must be continuously produced to maintain enough amounts in vivo. Chemotherapy drugs prevent the bone marrow from performing its hematopoietic role, resulting in a decrease in the number of circulating neutrophils, as mature neutrophils that have undergone apoptosis are not timely replaced. 1 After chemotherapy, an absolute neutrophil count (ANC) lower than 1.0 × 109/L is considered as severe neutropenia (SN). 1 A value of ANC lower than 0.5 × 109/L or an expected decrease to 0.5 × 109/L support a diagnosis of febrile neutropenia (FN), which is defined as a single oral temperature > 38.3°C (or axillary temperature > 38.0°C) or a sustained oral temperature (sustained for 2 h or 2 successive measurements) >38.0°C (or axillary temperature > 37.8°C). 1 FN is considered as a medical emergency and generally prompts immediate hospitalization for assessment and treatment with empiric broad-spectrum antibiotics. It not only affects patients' quality of life but also causes additional financial burden. 2 Furthermore, FN frequently results in chemotherapy dose reductions and treatment delays that may affect long-term clinical outcomes in responsive and potentially curable cancers. 2
The granulocyte-colony stimulating factor (G-CSF) improves the symptoms of neutropenia through stimulating the production of neutrophil precursors and enhancing the function of mature neutrophils. 3 The G-CSF lessens neutropenia’s duration and severity, as well as the incidence of SN, FN, and infection-related mortality in clinical settings.4-6 Several guidelines recommend it for the prevention and management of chemotherapy-induced neutropenia.7-9
The National Comprehensive Cancer Network (NCCN) 7 and Chinese Society of Clinical Oncology (CSCO) 1 guidelines recommend G-CSF for primary prophylaxis when the risk of FN is high (>20%), either owing to chemotherapy alone or a combination of chemotherapy and individual patient factors like advanced age, a history of chemotherapy or radiotherapy, and poor physical fitness. For reducing hospitalization rates during the novel coronavirus disease 2019 (COVID-19) pandemic, the CSCO 1 guidelines also suggests that primary G-CSF prophylaxis is feasible in individuals with an intermediate risk of FN (10% to 20%). Some chemotherapy regimens for the treatment of breast cancer, such as TE (docetaxel + epirubicin), TC (docetaxel + cyclophosphamide), and TAC (docetaxel + doxorubicin + cyclophosphamide), require primary prophylaxis with G-CSF.1,7
Two forms of G-CSFs are in clinical use: the short- and long-acting G-CSFs. The short-acting G-CSF requires daily administration because it is cleared through neutrophil- and renal-mediated processes and has a plasma half-life of 3-4 hours. 9 The long-acting G-CSF is produced by PEGylated of short-acting G-CSF, resulting in a long plasma half-life and thus, needing a single injection per chemotherapy cycle. 10 No significant differences were found in the incidence of G-CSF-related bone, joint and muscle pain, between the two varieties.11,12 A total of 10-11 doses of short-acting G-CSFs following chemotherapy is as efficacious as the long-acting one.11,12 However, in clinical practice, daily short-acting G-CSF is sometimes administered in shorter courses of therapy than recommended (i.e., 3 to 6 doses), which may lead to diminished effectiveness. 13
Results of clinical trials or real-world data were previously used to assess cost-effectiveness of the long-acting G-CSF.14-17 The consensus was that long-acting G-CSF represented a more cost-effective strategy than the short-acting one. The analyses often concentrated on the cost and utility values associated with FN patients, since neutropenia characterized by values of neutrophil count equal or greater than 0.5 × 109/L and lower than 1.8 × 109/L, is frequent in clinical practice. Our study was designed to assess the clinical outcomes and cost-effectiveness of long-acting G-CSF for primary prophylaxis of neutropenia caused by chemotherapy against breast cancer, in patients with values of ANC lower than normal, not limited to only FN. Our study was designed to assess the clinical outcomes and cost-effectiveness of long-acting G-CSF.
Methods
Study Design
We conducted a retrospective study in a large 3A hospital in China. We collected information on patients who consecutively underwent chemotherapy for breast cancer between December 2016 and September 2021 from the electronic medical record. Subjects were eligible if they met the following criteria: (ⅰ) age ≥ 18 years old; (ii) had a histopathology-confirmed diagnosis of breast cancer; (iii) had received the long- or short-acting G-CSF for primary prophylaxis of neutropenia; (ⅳ) had undergone chemotherapy with TE, TC, or TAC. Additionally, they were excluded if they (ⅰ) had insufficient clinical information, such as undergoing less than two chemotherapy cycles; (ii) were male patients; (iii) had another tumor or any other treatment.
The sample size was determined by Power and Sample Size calculator (http://www.powerandsamplesize.com/). We estimated the incidence of FN in short- and long-acting G-CSF-treated groups to be 0.11 and 0.17, 18 with α = 0.05, β = 0.20 and sampling ratio=1. We aimed to include 521 patients in every group.
The collected information of patients, included age, the tumor node metastasis (TNM) stages, chemotherapy regimen, medication history, values of ANC in blood routine examination and records of temperature in every cycle.
The study was approved by the Ethics committee of Henan Provincial People’s Hospital (date of approval: June 20, 2022; approval number: 2022-72) and owing to the retrospective nature of the study, individual consent for this analysis was waived. All patient details were de-identified. This study conforms to CHEERS guidelines. 19
Clinical Outcomes
Based on the values of ANC, the patients were divided into four groups: (ⅰ) normal level of ANC (ANC ≥ 1.8 × 109/L); (ⅱ) non-SN, meaning that ANC was decreased, but patients have not yet reached SN (1.0 × 109/L ≤ ANC < 1.8 × 109/L); (ⅲ) SN, meaning that ANC was reduced to SN (ANC < 1.0 × 109/L) but patients had no fever; (ⅳ) FN, meaning that ANC was decreased (ANC < 1.0 × 109/L) and was accompanied by fever. The blood test was conducted prior to the subsequent chemotherapy round, and patients were included in one of these four categories. Patients with normal ANC level further received chemotherapy. We calculated the incidence of neutropenia, including non-SN, SN and FN.
Statistical Analysis
SPSS 23.0 software and GraphPad Prism 9.0 softwares were used for statistical analysis and mapping. Measurement data was analyzed by independent samples t-test, and count data was analyzed using the chi-square test. Two-way analysis of variance was used to assess statistical significance, and p values < 0.05 were considered statistically significant.
Model Structure
Two models (Figure 1), created in Microsoft Excel 2010, were used to compare the health benefits and costs of utilizing the long- or short-acting G-CSF as the primary prophylaxis therapy, in two fictitious cohorts of women, aged 49 years old, with breast cancer stage II, III, and IV, receiving chemotherapy. We build the models using real world data and published literature.17,20 The first decision-analytic model monitored the effects of chemotherapy on neutropenia, while the second Markov model assessed how dosage reduction affected long-term survival. Model structures including a decision-analytic model (A) and a Markov model (B). G-CSF, the granulocyte-colony stimulating factor; SN, severe neutropenia; FN, febrile neutropenia; RDI, relative dose intensity.
In model 1 (Figure 1A), each patient received the long- or short-acting G-CSF as primary prophylaxis for neutropenia following chemotherapy and was described as belonging to the “chemotherapy + G-CSF” state. Based on transition probabilities, patients could alternate among three health states: “non-SN,” “SN,” and “FN”, or remain in the previous state. When a patient had SN or FN, infection or even death could occurred. If ANC was normal, the patients returned to the “chemotherapy + G-CSF” state until they finished four cycles of chemotherapy or died from an infection. The cycle length was 3 weeks, and the time horizon was 12 weeks. No discounting was applied in this model.
As shown in Figure 1B, a post-chemotherapy model was created to calculate the impact of decreasing relative dose intensity (RDI) on long-term survival of breast cancer. The health benefits of the long and short acting G-CSFs were examined in terms of quality-adjusted life years (QALYs). A 30-year time horizon was used while adopting cycles of one year. The chosen discount rate was 5%.
Analysis was conducted from the perspective of the Chinese health service system. In this study, all costs were recorded in US dollars ($). A willingness-to-pay (WTP) threshold of $12158.6 per QALY (one fold of the Chinese percapita gross domestic product in 2021) was applied.
Inputs of the Chemotherapy Model (Model 1)
The parameters in the decision-analytic model.

aSame as chemotherapy.
bSame as FN inpatient.
cCalculated by: 1-risk of hospitalization if FN.
G-CSF, the granulocyte-colony stimulating factor; ANC, absolute neutrophil count; SN, severe neutropenia; FN, febrile neutropenia; DSA, deterministic sensitivity analyses; PSA, probabilistic sensitivity analysis; Ⅰ, the retrospective study; Ⅱ, expert opinion.
The average cost of chemotherapy was computed using data from one hundred patients. The prices of long- and short-acting G-CSFs were obtained from local winning drug prices and the average prices was calculated. In patients receiving short-acting G-CSF as primary prophylaxis for neutropenia, its injection times were the most often occurring counts of short-acting G-CSF consumption. Experts calculated the costs for the management of ANC following chemotherapy, which mainly consisted in ANC monitoring and prescriptions to improve neutropenia. Seven oncologists and seven pharmacists were invited to estimate these costs after chemotherapy in light of the following aspects: (ⅰ) laboratory testing of ANC was required for all patients following chemotherapy, and additional ones were added when ANC was lower than 1.8 × 109/L. (ii) when ANC was lower than 1.0 × 109/L, especially if concurrent fever was present, oral formulations for improving neutropenia, such as Diyushengbai tablet, and injectable short-acting G-CSF were potentially used, despite being against the recommendations.1,7 Patients who underwent chemotherapy and had normal levels of ANC required regular monitoring of ANC. When estimating the cost of non-SN, further laboratory ANC testing should be considered. When calculating the costs of SN and FN outpatient, it was important to take into account additional ANC laboratory tests, use of oral medication and short-acting G-CSF into account. The costs of FN inpatient, hospitalization and infection were obtained from published literature. 16
The QALYs were adjusted by utility scores. The utility scores for chemotherapy, SN, and FN inpatient and outpatient were captured from published literature,15,17,20 as shown in Table 1. Utility scores for non-SN and infection were considered to be 0.70 and 0.33, respectively, which were equivalent to the utilities for chemotherapy and FN inpatient.
The incidences of non-SN, SN and FN inpatient in the retrospective study.
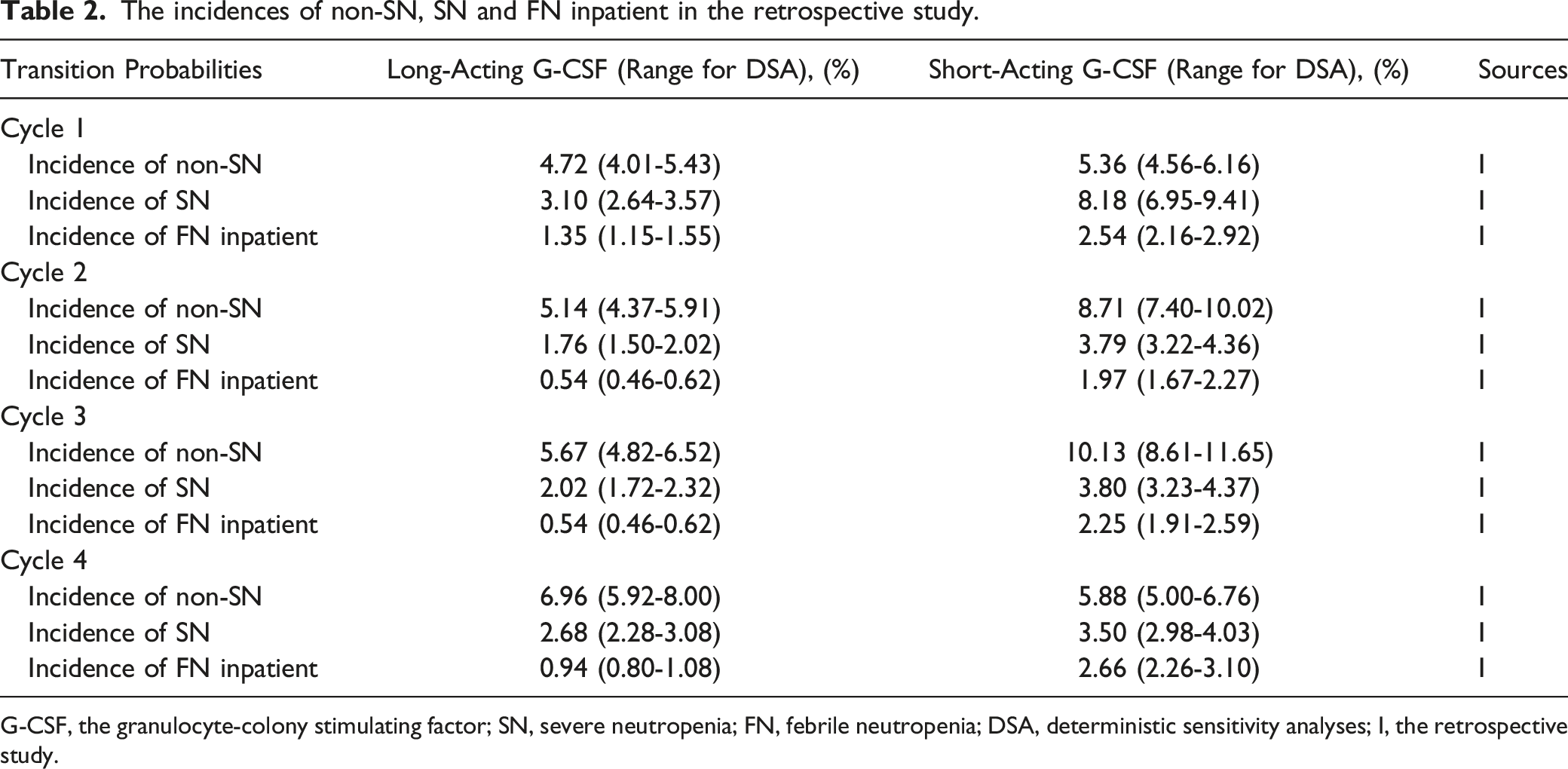
G-CSF, the granulocyte-colony stimulating factor; SN, severe neutropenia; FN, febrile neutropenia; DSA, deterministic sensitivity analyses; Ⅰ, the retrospective study.
Inputs of the Post-chemotherapy Model (Model 2)
RDI is defined as the ratio of the actual chemotherapy dose patients received to the intended dose of the standard chemotherapy regimen within a given time period. The complete and partial response rates, as well as the progression-free survival rate, are consistently correlated with the chemotherapy dose intensity. 21 Age ≥ 65 years and a personal history of FN are risk factors for receiving RDI less than 85%, although these factors are not independent of one another. 22
The parameters of post-chemotherapy model.

aCalculated based on OR; RDI, relative dose intensity; FN, febrile neutropenia; OR, odds ratio; RR, relative risk; HR, hazard ratio; PSA, probabilistic sensitivity analysis.
The total costs and QALYs of the long- and short-acting G-CSFs were calculated in the baseline analysis. The incremental cost-effectiveness ratios (ICERs) were calculated in baseline and sensitivity analyses.
Sensitivity Analysis
To determine the main variables affecting the ICER, we performed a one-way deterministic sensitivity analysis (DSA). A variance of 20% was used for costs, since the prices of drugs and medical services were changed greatly owing to local or national medical reforms. 30 In this study, the minimum and maximum numbers of administrations of short-acting G-CSF were established as 2 and 7, respectively, which were the least and most injection times of short-acting G-CSF in retrospective study. The variances in utilities and transition probabilities were fixed at 15%. Tables 1 and 2 display the ranges of each parameter.
To examine the uncertainty of the parameters when they are simultaneously altered within a particular pattern of distribution, a probabilistic sensitivity analysis (PSA) using 5000 Monte Carlo simulations was performed. Tables 1 and 3 displayed the distributions of these parameters. For each set of parameter values, the ICERs were calculated as in the baseline analysis. To determine if the long-acting G-CSF was incrementally cost effective as a main prophylactic strategy over a range of WTP thresholds, the PSA results were presented in the forms of a scatterplot and cost-effectiveness acceptability curve.
Results
Clinical Outcomes
Demographic and baseline characteristics.

G-CSF, the granulocyte-colony stimulating factor; TNM stages, the tumor node metastasis stages; TAC: docetaxel + doxorubicin + cyclophosphamide; TC: docetaxel + cyclophosphamide; TE: docetaxel + epirubicin.

The incidences of neutropenia in patients receiving long or short G-CSF as primary prophylaxis following chemotherapy. G-CSF, the granulocyte-colony stimulating factor; SN, severe neutropenia; FN, febrile neutropenia.
Baseline Analysis
The results of baseline analysis.

G-CSF, the granulocyte-colony stimulating factor; QALYs, the quality adjusted life years; ICER, the incremental cost-effectiveness ratio.
To ascertain which parameter was the most sensitive for ICER, the one-way DSA was performed. The cost of long-acting G-CSF has the greatest impact on ICERs, followed by injection times of short-acting G-CSF (Figure 3A). When the cost of long-acting G-CSF per cycle rose to $595.9 or the injection times of short-acting G-CSF dropped to 2, the ICERs were lower than the WTP threshold of $12158.6 per QALY. The ICERs remain under the WTP threshold despite the variations of the incidence of non-SN, SN, and FN inpatients (Figure 3B). Tornado diagram of the one-way deterministic sensitivity analysis on costs, utilities and partial parameters on transition probabilities (A), and incidences of non-SN, SN and FN (B). SN, severe neutropenia; FN, febrile neutropenia; G-CSF, the granulocyte-colony stimulating factor; ANC, absolute neutrophil count; Max, maximum value of each parameter; Min, minimum value of each parameter.
Sensitivity analysis
The fact that all 5000 replications are in the northeast and southeast quadrants shows that in most scenarios, long-acting G-CSF is more effective than short-acting G-CSF (Figure 4). When WTP threshold is larger than $3000, the strategy of using long-acting G-CSF as primary prophylaxis is favored, according to the cost-effectiveness acceptance curve (Figure 5). With a WTP threshold of $12158.6 per QALY, long-acting G-CSF had a 97% chance of being cost-effective, while short-acting G-CSF had a 3% chance of the same. Scatterplot of probabilistic sensitivity analysis for 5000 simulations. QALYs, quality-adjusted life years; WTP: willingness-to-pay. Cost-effectiveness acceptability curves for long or short G-CSF as primary prophylaxis of neutropenia. WTP, the willingness-to-pay.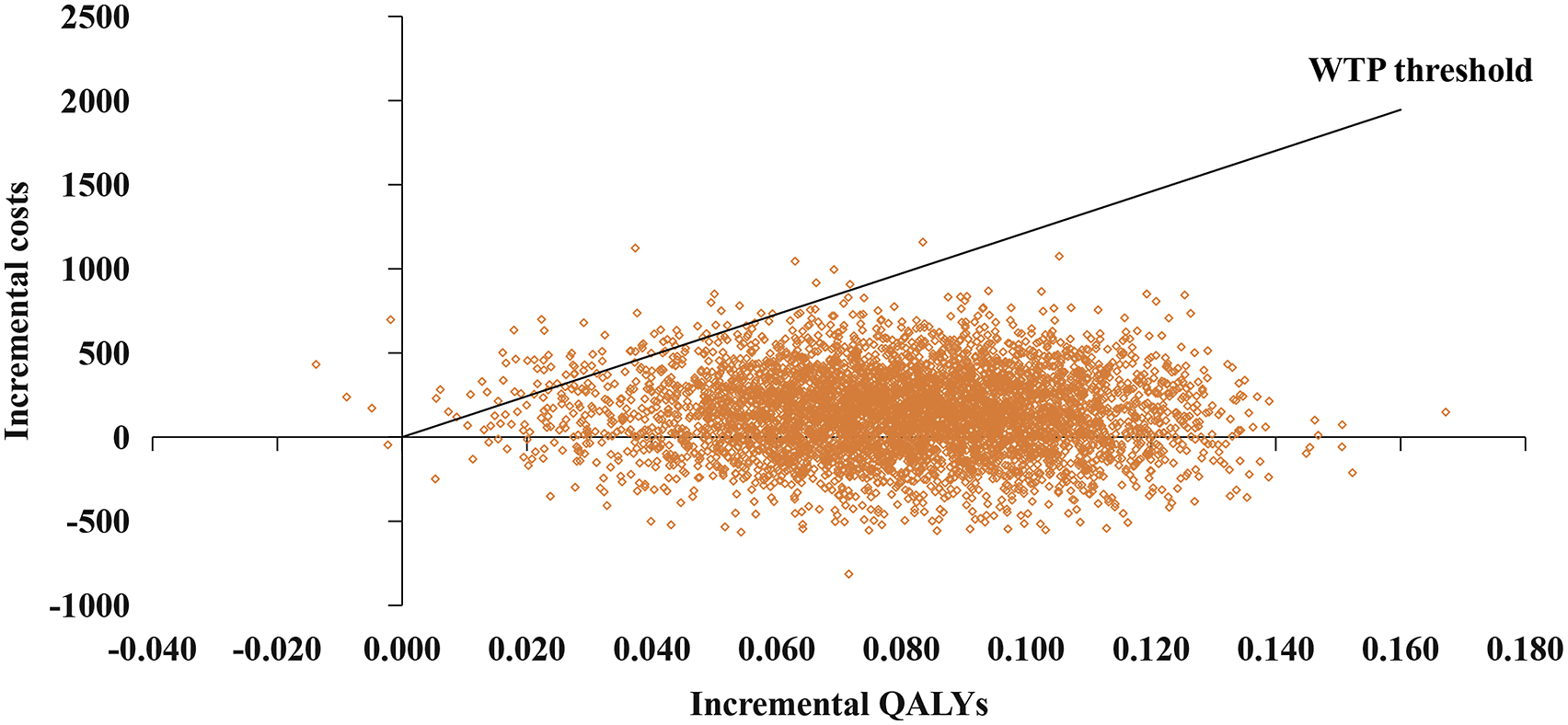

Discussion
According to the results of the current study, patients receiving long-acting G-CSF had significantly lower rates of neutropenia than those receiving short-acting one with 2 to 7 days. The use of long-acting G-CSF yielded an ICER of $1792 per QALY, which was lower than the Chinese WTP threshold of $12158.6 per QALY.
A systematic review and meta-analysis revealed that, no statistically significant difference was found in clinical outcomes of long- and short-acting G-CSFs when administered ≥ 7 days, if their usage and dosage complied with the recommended guidelines. 31 Another meta-analysis showed that long-acting G-CSF reduced neutropenia just as effectively and safely as short-acting G-CSF, with the premise of using a sufficient dose of the latter. 32 Contrary to the findings of the above studies, in our study, neutropenia episodes were much less frequent in patients receiving long-acting G-CSF than in those receiving short-acting G-CSF. The clinical outcomes of short-acting G-CSF were less favorable than those of the long-acting type. The likely cause could be 4-day short-acting G-CSFs were widely employed. According to a retrospective analysis, prophylaxis with G-CSF for fewer than 7 days was linked to a higher incidence of FN hospitalization. 33 When the mean duration of prophylaxis with filgrastim was 4.8 days, patients who received pegfilgrastim as prophylaxis had a lower risk of neutropenia-related or all-cause hospitalization than those who received filgrastim. 34 The insufficient usage of short-acting G-CSFs is caused by the patients' insufficient understanding on G-CSF, decrease in the dose intensity of the chemotherapy, adverse response, costs, or protocol of hospital or country/region. 35
Numerous studies have examined the cost-effectiveness of prophylactic therapy with long-acting G-CSF for neutropenia. A WTP survey 35 examined the willingness of patients with small-cell lung cancer to receive either pegfilgrastim or short-acting G-CSF. Pegfilgrastim was more expensive than short-acting G-CSF, but most patients were willing to pay the extra costs, because of its method of administration, consisting in a single injection per cycle of chemotherapy. Compared with 6-day short-acting filgrastim, pegfilgrastim had higher costs, more QALYs, and was more cost-effective in primary prophylaxis of FN,14,15 which was consistent with other economic analysis of long-acting G-CSF based on Chinese national conditions.17,36 The cost-effectiveness analysis of long-acting G-CSF based on real-world data was also provided; however, the findings were conflicting. This might be due to the use of a small sample size.37,38 Based on real-world data, Zhao J, et al 16 evaluated the cost-effectiveness of pegylated recombinant human G-CSF (PEG-rhG-CSF) as the primary prophylaxis of neutropenia in patients with breast cancer. The results revealed that PEG-rhG-CSF produced an ICER of ¥347 per QALY compared with rhG-CSF, for which the outcomes are similar to ours. Some differences exist between the two studies: we included patients with non-SN and SN, not just with FN, since individuals with values of ANC ranging between 0.5 × 109/L and 1.8 × 109/L but no fever, need to be monitored more frequently and even need further injections of short-acting G-CSF. They also contribute more QALYs than FN patients.
This study has some limitations. First, the costs of non-SN, SN and FN were estimated by experts based on clinical practice. However, this may not reflect real-world expenditures very well. Thus, we carried out a sensitivity analysis and discovered the status had a weak impact on the model. Second, the study reflects the perspective of the health service system, only direct medical costs being taken into account. Societal or multiple perspectives might be more advisable. Finally, most parameters were extracted from published literature or expert opinion, except for incidences of non-SN, SN and FN inpatient, and there may be a discrepancy with real world. Therefore, a cost-effectiveness study should be carried out including the perspective of society or multiple perspectives, as well as direct non-medical, indirect and intangible costs.
In conclusions, patients receiving long-acting G-CSF as primary prophylaxis against neutropenia experienced lower risk of neutropenia compared with underused short-acting G-CSF. The long-acting G-CSF may be a more cost-effective strategy for primary prophylaxis of neutropenia than short-acting G-CSF, considering the Chinese WTP threshold of $12158.6 per QALY.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by Ethics Committee of Henan Provincial People’s Hospital (date of approval: June 20, 2022; approval number: 2022-72) and individual consent for this retrospective analysis was waived.
