Abstract
Tobacco use accounts for 30% of all cancer-related deaths worldwide and 20% in the US, despite effective, evidence-based interventions for reducing tobacco use and tobacco-related cancers and deaths. In 2012, to reduce the burden of tobacco-related cancer and associated population-level risks across Texas, The University of Texas MD Anderson Cancer Center initiated the EndTobacco® program to promote statewide cancer control activities. We created evidence-based initiatives, established selection criteria, and implemented actions involving policy, education, and tobacco treatment services. As a result, EndTobacco has supported, educated, and convened local and state coalitions in policymaking; provided tobacco treatment education to health professionals; implemented Texas’ only certified tobacco treatment training program; and led an initiative to enhance the tobacco-free culture of the state’s publicly funded university system. Supported by commitments from MD Anderson, we developed and implemented evidence-based actions for tobacco control tailored to the center’s mission, values, expertise, resources, and partnerships. By 2021, the adult smoking rate in Texas dropped from 19.2% (2014) to 13.2%. Contributors to this drop include state tobacco control policies, programs and services from multiple agencies and associations, and EndTobacco activities that complement the statewide effort to prevent youth smoking initiation and increase quit attempts among youth and adults.
Keywords
Introduction
Over the last half century, tremendous progress has been made in the de-normalization of tobacco use through a range of well-established, evidence-based approaches that include policies, mass media health-communication campaigns, and the availability and promotion of community-based tobacco treatment resources. 1 Historically, tobacco control in the United States has been largely addressed at the state level, but a greater need for federal intervention (ie Tobacco 21 legislation and regulation of tobacco products) has emerged. While the federal government (Centers for Disease Control and Prevention [CDC]) compiled the evidence for tobacco control and recommended a framework for effective tobacco control programs, 2 in most cases, states have been responsible for funding and implementation. Overwhelmingly, evidence shows that larger state investments in comprehensive tobacco control programs lead to declines in the prevalence of tobacco use among adults and youth and in cigarette sales (consumption). 1 This eventually results in substantial healthcare savings.3,4
However, declines in tobacco use have slowed or stalled in some populations, particularly among those with mental health disorders, socioeconomic disadvantages, or low educational attainment.5-7 Despite progress within the last 50 years, tobacco use continues to be a leading cancer risk factor. 1 The introduction of electronic nicotine-delivery systems has compounded that risk by creating an alternative path to nicotine addiction that has marred much of the tobacco control progress among youth and young adult populations.8-10 Continued innovations in tobacco control research will be required to face this growing challenge.
National Cancer Institute (NCI)-designated cancer centers “serve as a major source of discovery of effective approaches to prevention, diagnosis, and therapy” 5 ; create shared research resources; collaborate with other NCI-funded programs and investigators; and disseminate research findings for the benefit of each catchment area community. In 2016, the NCI expanded funding requirements for its designated cancer centers to include community outreach and engagement (COE) 11 that would broaden the academic health-system focus on individual-patient care to include population health management and improvement. 12 While each cancer center addresses the COE requirement based on their center’s commitment, 13 COE criteria also present an opportunity to complement and extend existing state tobacco control efforts to reduce the leading cause of preventable cancers and deaths in many catchment area populations. Cancer centers are still determining how they will optimally address the new COE criterion, including how they can best contribute to effective tobacco control in their catchment area populations.
The University of Texas MD Anderson Cancer Center (MD Anderson) is one of 11 Medicare Prospective Payment System-exempt, NCI-designated cancer centers in the US. In 2012, the institution was charged by its then president to make an unprecedented and sustained commitment to end tobacco use at the institutional, local, state, national, and even international levels. This manuscript summarizes the processes by which MD Anderson developed its comprehensive, multi-component, tobacco control program, EndTobacco®, including implementation, early outcomes, and lessons learned. Our experience may encourage other academic cancer centers or healthcare organizations not historically involved in tobacco control to engage in efforts led by the CDC, World Health Organization (WHO), and other public health agencies. Such concerted actions could help to establish and sustain a tobacco-free culture in the US, which would represent one of the broadest, most compelling, and most impactful advances in population health.
Program Development
A multi-disciplinary, 18-member working group (Work Group) of MD Anderson scientists, clinicians, and administrative leaders convened in February 2013 in response to the charge from the institution’s president: to develop a program to reduce (and eventually eliminate) tobacco use and tobacco-related cancers through evidence-based actions implemented at multiple organizational and sociocultural levels to end cancer in Texas, the nation and the world. In addressing these aspirations, the Work Group used recommendations from the WHO’s MPOWER framework 14 and the CDC’s Best Practices for Comprehensive Tobacco Control Programs—2014. 2 These frameworks provided solid evidence, a comprehensive scope, global deployment capability, and a critical link to numerous potential partners and collaborators. We first mapped the MPOWER framework, a structure to review and recommend evidenced-based best practices in tobacco control, against expert opinions in the Work Group to provide a strategic plan that aligned with the institution’s mission and values. We completed this work by recruiting experts, allocating resources, and developing partnerships. Subsequently, we mapped these recommendations to the CDC best practices document. 2
Guiding Principles for Program Implementation.

Complementary to the charge to develop a comprehensive tobacco control program, MD Anderson leadership had previously developed a framework for advancing cancer control to distinguish it from the institution’s other three mission domains of research, clinical care delivery, and healthcare provider-oriented education and training. In alignment with broader institutional cancer control actions, cancer control was defined as “the implementation and dissemination of evidence-based actions in public policy (P), public and professional education (E), and delivery of community-based clinical services (S)” to affect a meaningful and lasting reduction in the burden of cancer and its associated risk factors at the population level. 15 Because tobacco control is an important subset of cancer control, the Work Group also adopted these PES domains to organize EndTobacco.
As overarching long-term goals, the Work Group adopted the CDC’s three recommended goals for comprehensive tobacco control programs: (1) prevent initiation among youth and young adults, (2) reduce exposure to secondhand smoke, and (3) promote quitting among adults and youth. 2 A fourth CDC-recommended goal—to identify and eliminate tobacco-related disparities—was not explicitly adopted by the Work Group; rather, the group defined tobacco-related disparities as an overarching, cross-cutting goal and considered it more feasible to tailor specific PES recommendations to special populations with high rates of tobacco susceptibility or use.
Program Implementation
In 2015, after the initial program conceptualization and development phase was complete, a dedicated program director and a program manager, each with experience in the creation, implementation, and management of tobacco control programs, were recruited to support all program operations. While the EndTobacco Work Group members contribute subject-matter expertise to develop and shape the program’s ongoing actions and strategies, the director and program manager provide extensive efforts toward development, education, coalition-building, and evaluation for the initiative.
Implementation Partners.

Outcomes
The primary outcome of the EndTobacco Work Group was an organizational construct comprised of a set of strategic and tactical recommendations codified into an initial plan to reduce the tobacco burden at various levels (Table 1). This framework—a compilation of CDC best practices 2 and WHO MPOWER strategies 14 that adhered to MD Anderson’s definition of cancer control—provided both a comprehensive approach and specific actions that could be taken in targeted settings.
Over the last seven years, significant progress has been made in implementing Work Group recommendations (Figures 1 and 2). Table 3 details strategies and example actions from the most mature EndTobacco projects in policy, professional education, clinical service delivery; and towards a comprehensive effort encompassing all three cancer control domains. EndTobacco timeline. EndTobacco program logistics, Abbreviations: CTTTP, Certified Tobacco Treatment Training Program; ETU, End Tobacco Use Initiative; TCCEP, Tobacco Cessation Clinic Enhancement Program. The EndTobacco® Construct with Representative Examples of Recommended Strategies and Actions Implemented within Various Populations. aInstitutional level: MD Anderson is the focus, consistent with EndTobacco’s guiding principle to lead by example; will also consider working with other institutions in a similar space in the greater Texas Medical Center and beyond. Local/regional/state level: Texas is the initial focus, with potential dissemination in later years to other states. National/international level: institutional and state-level actions are leveraged to expand nationally or to other countries, as resources and collaborations permit. bInvolves actions across the three cancer control domains (policy, education, and clinical service delivery).
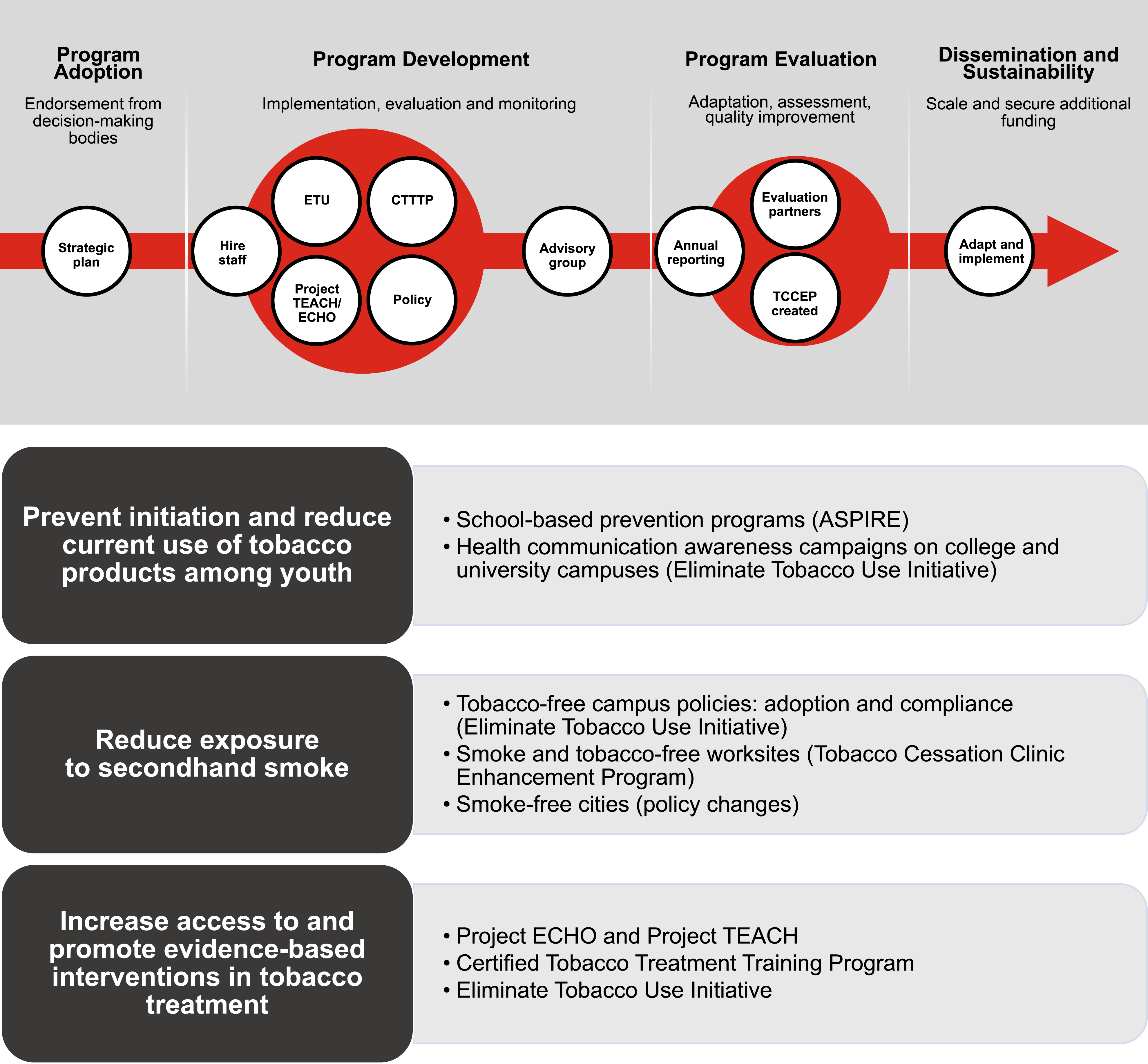

Building on its long-standing tobacco-free campus policy that began in 1989, MD Anderson adopted a tobacco-free hiring policy that became effective in January 201516,17: a two-step pre-employment procedure consisting of a cotinine-based, evidence-of-smoking screener coupled with an invitation to receive our comprehensive tobacco treatment services (counseling assisted by pharmacotherapy at no cost through MD Anderson’s Tobacco Research and Treatment Program [TRTP]). 18 Upon successful tobacco cessation, applicants can re-apply for open employment positions following a waiting period of 180 days and completion of a new two-step, tobacco-use screening test. Annually, fewer than 1% of applicants have tested positive, suggesting the policy’s minimal adverse impact on the applicant hiring process. EndTobacco implemented this new policy in accordance with its guiding principles to 1) lead by example to establish a more comprehensive and complete tobacco-free culture on campus, and 2) make the end of tobacco an institutional aspiration. The policy’s implementation also reinforced the institution’s efforts to intentionally and impactfully balance promotion of a tobacco-free culture with empathy and compassion for tobacco users. It also addressed institutional-level recommendations that advanced all three goals of the EndTobacco program (Table 3).
For engagement with partners on local- and state tobacco policies, MD Anderson, a state institution, defined its role as a convener, coalition-builder, and provider of educational resources for any government-oriented policymaking process. As an educational resource and community engagement partner, the EndTobacco team joined the Smoke-Free Fort Worth-, 19 San Antonio Tobacco 21-, 20 and Texas Tobacco 2117,21 coalitions to support their successful efforts to reduce tobacco use among youth in local communities and across the state. These local- and state policy initiatives, while not due to the sole efforts of the EndTobacco team, significantly advanced EndTobacco’s goals 1 (prevent tobacco initiation and reduce current use of tobacco among adults and youth) and 2 (prevent/reduce exposure to second-hand smoke).
For various reasons, clinical providers often lack the time, or necessary skills to provide evidence-based treatment for tobacco addiction. Recent data suggest that just 60% of smokers receive advice to quit smoking from their health care provider, only 30% actually seek help, and fewer than 5% receive the most effective evidence-based treatment (a combination of medication and counseling). 22 To address these issues, in 2017 the EndTobacco team piloted MD Anderson’s Certified Tobacco Treatment Training Program (CTTTP), which provides education to providers from all healthcare disciplines using a multi-day curriculum led by addiction specialists and counselors within our well-established TRTP. 18 The curriculum is based on the most up-to-date tobacco treatment research and evidence-based treatment strategies. It addresses goal 3 of EndTobacco (increase access to tobacco treatment and counseling in ambulatory settings) at the local/regional/state and national/international level and is accredited by the National Council for Tobacco Treatment Training Programs. MD Anderson’s CTTTP is the only program of its kind within Texas and its bordering states and one of only two such programs in the southwestern US. 23 Tobacco use in surrounding states ranges from 16.1% (New Mexico) to 20.5% (Arkansas) and (22%) (Louisiana, Mississippi, Georgia, and Florida).
Since its inception, the CTTTP has trained 979 participants from 33 US states and the countries Egypt, Spain, Nepal, and Guam. At the request of Arkansas Department of Health, we have trained 297 healthcare professionals from all 75 counties in that state. MD Anderson’s CTTTP data show that mean knowledge increased by 24% between pre- and post-test assessments, while 95% of participants have passed the credentialing exam to earn the Tobacco Treatment Specialist certification. Providers completing the CTTTP certification are subsequently encouraged to participate in MD Anderson’s Project TEACH (Tobacco Education and Cessation in the Health System) to sustain their training through case consultations about challenging cases, emerging treatment innovations, guideline updates, and other areas of interest.
Project TEACH, an institutionally sanctioned continuing education and telementoring program, was developed and facilitated by TRTP faculty and staff. 24 Its goal is to sustain specialized tobacco-treatment practices by bolstering healthcare provider self-efficacy in delivering tobacco treatment services using the Project ECHO framework—created by The University of New Mexico 16 and adopted by MD Anderson for several cancer care projects. 25 Now fully administered and operated by the EndTobacco team, Project Teach addresses tobacco treatment strategies through two 30-minute practices (didactic- and case-based learning) per session to increase ambulatory access to tobacco treatment (goal 3, Table 1). Learning from the program is bi-directional as MD Anderson leadership teams gain greater knowledge about healthcare in diverse patient groups.
During its first five years of operation, Project TEACH has steadily increased the number of providers it serves, from an average of 11-15 participants per session in the first year to 50 per session in 2022. Approximately 40 clinics are held annually and in 2021 the program reached over 600 unique providers from 36 states and five countries. Since the project’s inception, a total of 924 American Medical Association continuing medical education hours and 2902 continuing-education credits have been awarded to participants. Results from a 2021 survey of all Project TEACH participating providers suggest that sessions improve their ability across different organizational settings to provide high-quality, clinical, tobacco treatment services to a diverse group of patients.
An estimated one million, currently enrolled, US college students are projected to die prematurely from smoking, 26 illustrating the staggering statistic that 99% of adult smokers start smoking before age 26. 27 As tobacco and nicotine products evolve, institutions of higher education can become agents of change for young adults ages 18 – 24. Colleges provide an ideal environment to nurture lifetime behaviors and can influence the social norms around smoking and tobacco use during a time of growth and experimentation for many young people. In response to these and other data, in 2015 the EndTobacco team collaborated with the UT System, which comprises 14 health and academic institutions, and with state and national tobacco control partners to form the End Tobacco Use (ETU) Initiative. The ETU fostered a tobacco-free culture across all UT institutions and developed a model for effective tobacco control in college settings. The ETU Initiative implements several EndTobacco actions at an institutional level to advance its three programmatic goals that ensure that each campus has (1) a comprehensive tobacco-free policy that features consistent and compassionate enforcement, (2) access to evidence-based educational and preventive programming, and (3) access to tobacco treatment services for all students and staff across the UT campuses.
Key elements of the ETU Initiative model include university system-level leadership endorsement, campus-specific task forces, annual summits, a dashboard to track and monitor progress, health communication tools, and a team of dedicated individuals with expertise in public health program implementation and coalition-building (the EndTobacco team). Use of this model resulted in significant achievements in tobacco control across the UT System. Most notably, 18 months into the initiative’s work (May 2017), the entire UT System became tobacco free, making it the first university system in the state, and the largest Texas employer, to achieve that status. This effort included nearly 240 000 students; 104 000 faculty and staff; and over 199 000 people covered under the UT System’s insurance plan.
In 2021, a new initiative, the Tobacco Cessation Clinic Enhancement Program (TCCEP), was developed and launched to increase tobacco-free activities, to enhance provider knowledge and practice, and most importantly, to deliver treatment services for populations who have notably high use of tobacco. Frequently these populations seek services from Federally Qualified Health Centers and non-profits that provide comprehensive population-health programs to medically underserved communities. Delivery of these services is ongoing as we implement a comprehensive, evidence-based, systems-change approach that addresses policy, education, and tobacco treatment service on each campus. Currently, the EndTobacco team and a faculty member are piloting the program in two clinics: OmniPoint Health outside of Baytown, Texas, and The Montrose Center, which serves LGBTQ+ residents in the Greater Houston area. These clinics were chosen for the TCCEP pilot program for multiple reasons, such as clinic’s interest, need, and readiness; high rates of tobacco use in clients/patients; and proximity to MD Anderson.
Through a variety of interventions, the adult smoking rate in Texas dropped from 19.2% in 2014 to 13.2% in 2021. Contributing factors to this meaningful decline include state and local tobacco control policies; and numerous programs and services from multiple agencies, associations, and EndTobacco activities that align with and complement the statewide effort to prevent youth smoking initiation and increase quit attempts among youth and adults.
Lessons Learned
The EndTobacco program represents a focused and sustained commitment from MD Anderson to provide leadership, investment, and collaboration that advances comprehensive, evidence-based tobacco control as a core element of the institution’s mission in cancer prevention and control (Figure 3). Our outcomes affirm what is possible when an NCI-designated cancer center commits to addressing a leading cancer risk factor. The EndTobacco program: (1) supported, educated, and convened local and state coalitions and collaborated with government officials in the health policymaking process to achieve three significant policy successes (ie, Smoke-Free Fort Worth, San Antonio Tobacco 21, and Texas Tobacco21); (2) credentialed 979 providers by implementing the only CTTTP in the state; (3) provided tobacco treatment education to over 600 health providers across the US and in five countries through an evidence-based telementoring model (Project TEACH); and (4) led a system-wide initiative (Eliminate Tobacco Use Initiative) to enhance the tobacco-free culture of the state’s largest publicly funded university system. The expert providers of MD Anderson’s TRTP,
22
established in 2006 to encompass both patient care and research activities, contributed to the creation and maintenance successes within both the CTTTP and Project TEACH. This model may be useful to other cancer centers as they seek to establish or enhance existing tobacco treatment programs through the NCI Cancer Center Cessation Initiative.
28
EndTobacco planning conceptual framework. Abbreviations: CDC, Centers for Disease Control and prevention; CTTTP, Certified Tobacco Treatment Training Program; ETU, Eliminate Tobacco Use™; TCCEP, Tobacco Cessation Clinical Enhancement Program; WHO, World Health Organization; TEACH, Tobacco Education and Cessation in the Health System.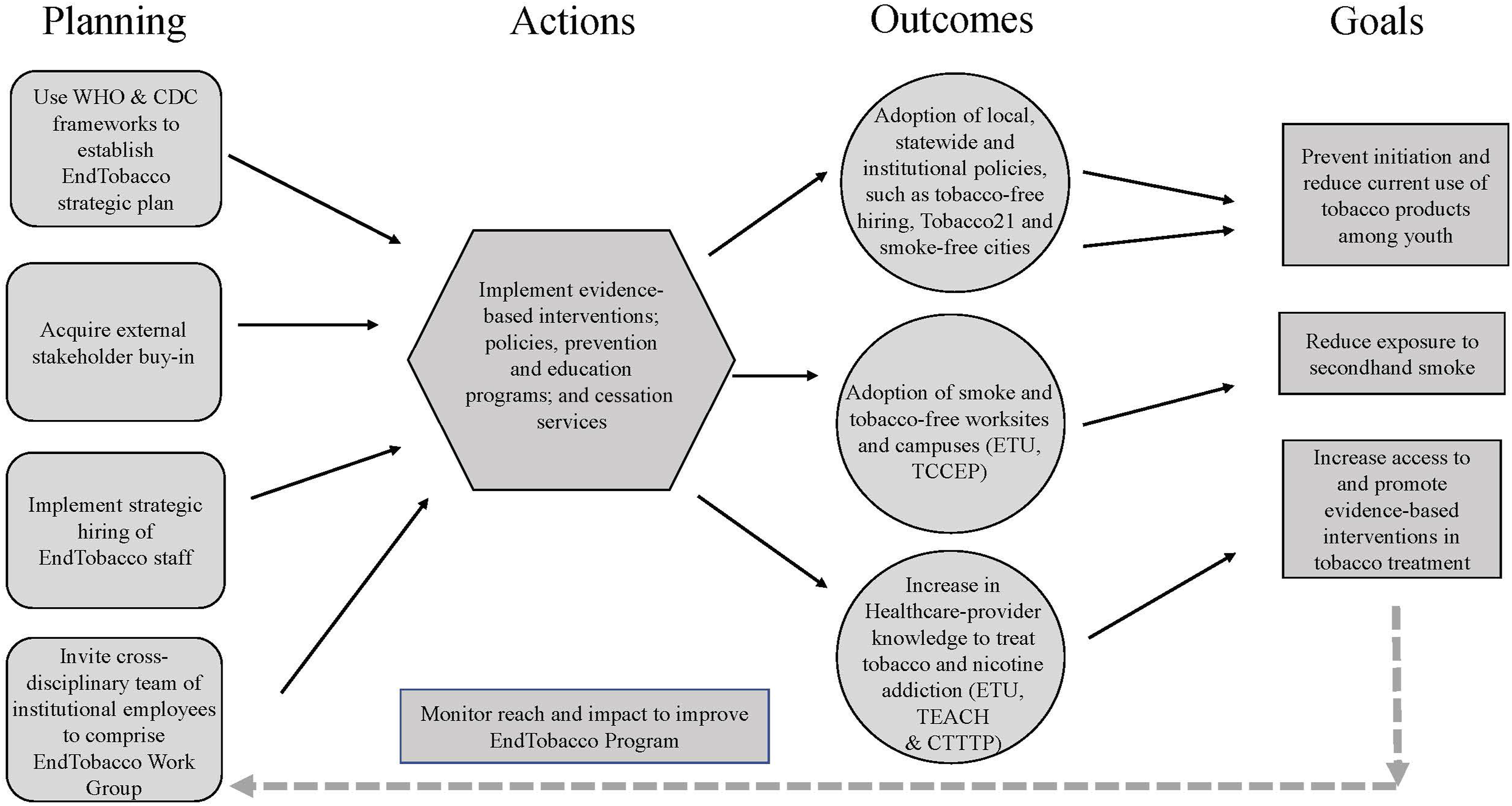
Near-term next steps for EndTobacco will be to disseminate the CTTTP to high tobacco-use prevalence areas within Texas and the nation, and to assist in replicating the ETU Initiative within other college and university campuses around the country. These activities have already begun with collaborative efforts in Arkansas, Missouri, New Jersey, and Virginia.
To best realize the potential of communities, new NCI-issued COE criteria require that NCI-designated cancer centers first engage with those communities to discuss and understand their priorities prior to developing, implementing, and disseminating evidence-based interventions, public educational programs, and public health policy recommendations within those catchment areas. A national network of cancer centers working to implement the well-established, tobacco control evidence base within their respective catchment areas could meaningfully reduce population-level tobacco use and associated diseases, including cancers. Below, we highlight aspects of the EndTobacco program relevant to pursuing comprehensive tobacco control in alignment with the new COE requirements.
First, a multi-disciplinary campus advisory work group is essential when developing a comprehensive tobacco control program. Planning is enhanced by the multi-disciplinary perspectives of various experts who provide valuable guidance and enable staff to see a broader and more comprehensive perspective.
Second, much of our work relied on creating and activating partnerships not always typical of cancer centers. Building partnerships and collaborations with groups that already work in tobacco control or have large target populations—non-profit advocacy groups, medical societies, major employers, schools, and public health groups at the local, state, and federal levels—is essential for implementing and sustaining impactful initiatives. EndTobacco activated more than 100 national, state, and local partnerships over a seven-year period to support shared, formally constructed goals. The team conducted weekly interactions with at least 15 organizations to develop accountable partnerships in advancing tobacco control initiatives. Further, foundational elements positioned EndTobacco to deliver program outcomes including direct and sustained involvement of institutional executive leaders; coordination of dedicated leadership and staff with deep expertise in tobacco control; philanthropic support incentivizing high-risk, high-reward programmatic investments; sustained institutional support via tobacco settlement funds ranging from $300,000 to $500,000 annually; and institutional activation of a dissemination team within an innovative, milestone-driven, public-health-promoting incubation environment.
Third, the strength, reputation, and reach of MD Anderson as a national and international leader in cancer care delivery translational research, prevention, and education enabled elevation and prioritization of tobacco control and prevention efforts through the EndTobacco program. MD Anderson’s size and status as a state agency was leveraged to gain comprehensive UT System Administration buy-in to attract and prioritize investments in tobacco control. Many steps were required for institutional approval of EndTobacco as a cancer prevention program. Several stakeholders reviewed and approved the EndTobacco plan, including the EndTobacco Work Group and the MD Anderson Board of Visitors (a non-fiduciary development board). As a component of The UT System, MD Anderson gained support from the UT System Board of Regents for the proposed EndTobacco strategic plan, allowing for the hiring of dedicated personnel. These elements ultimately served to strengthen the broad-based support for MD Anderson’s proactive role towards addressing the burden of tobacco for patients, employees, and their families and communities.
Fourth, while the EndTobacco program leaders have more than 30 years of combined experience in public health, tobacco control, and program planning, it is a lean operation, growing from one to three full-time employees over a five-year period. The team collaborates across departments within the institution and with other like-minded public health organizations to implement programs that achieve results.
Finally, the multi-year institutional investment to fund the EndTobacco program was derived from philanthropic donations to MD Anderson’s Moon Shots Program®. After the EndTobacco program’s fourth year, revenue generating activities were initiated and institutional funding was received, contributing to its current financial diversification that enables sustainability and continued growth.
Also of note: several studies on smoking behaviors during the COVID-19 pandemic have been published including a report that noted a decline in US annual smoking prevalence from 16.3% to 15.3% in 2016 to 2019 and a second decline to 14.2% in 2020. 29 A meta-analysis found mixed results for smoking prevalence worldwide during the pandemic: 21% smoked less, 27% smoked more, and 50% had unchanged smoking. 30 Altered smoking behaviors during the pandemic may have contributed to the overall reduction in smoking rates we described.
The outcomes and lessons learned from MD Anderson’s creation and implementation of EndTobacco suggest that focused investments and intentional actions related to core mission-based institutional priorities can lead to significant and efficient results. The construct we describe may be relevant to other organizations interested in tobacco control or in implementing frameworks to address other chronic diseases.
Footnotes
Acknowledgments
We thank Karen Colbert Maresso, MPH, and Stephanie Martch, MS, ELS, for editorial assistance with the manuscript. We also thank the following individuals for their contributions to the EndTobacco program: David Lakey, MD, Nagla Elerian, MPH, and Daniel Oppenheimer, MFA, with the University of Texas System Office of Population Health for co-developing the Eliminate Tobacco Use Initiative and co-hosting the annual summits; Michael Mackert, PhD, Jessica Wagner, MPH, CHES, and Dasha Rakasovic, with The University of Texas Austin Moody College of Communication for developing the Health Communication Campaigns and website for the Eliminate Tobacco Use Initiative; Maher Karam-Hage, MD, Diane Beneventi, PhD, Jan Blalock, PhD, Mark Evans, MSW, LCSW, and Rosario Wippold, RN, department of Behavioral Science at MD Anderson, for their contributions to the development and implementation of the Certified Tobacco Treatment Training Program and Project TEACH; Elizabeth Frenzel, MD, MPH, FACOEM and Georgia Thomas, MD, MPH, department of Employee Health and Well-Being, for their contribution to the tobacco-free hiring process and gathering of the data for this manuscript; and the institution’s EndTobacco Work Group members for developing the first edition of the EndTobacco plan and/or advising on the EndTobacco program annual activities. Thank you to Ronald DePinho, MD, past president of MD Anderson and current faculty member in the institution’s Cancer Biology Department, for his leadership and actions that led to the development of the EndTobacco plan and the EndTobacco program.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by philanthropic support to MD Anderson’s Moon Shots Program. The EndTobacco Program and the Tobacco Research and Treatment Program is supported by State of Texas Tobacco Settlement funds provided by MD Anderson Cancer Center. E. Hawk is supported in part by the T. Boone Pickens Distinguished Chair for the Early Prevention of Cancer and by the MD Anderson Support Grant 5P30CA016672-37 from the National Institutes of Health. J. Tektiridis is supported in part from the Duncan Family Institute for Cancer Prevention and Risk Assessment. P. Cinciripini is supported in part by the Margaret and Ben Love Chair in Clinical Cancer Care in honor of Dr Charles A. LeMaistre.
Ethical Approval
This article does not report on any studies with human or animal participants; as such, the inclusion of information about informed consent is not applicable.
