Abstract
Introduction
Perioperative therapy is standard for patients with borderline-resectable pancreatic ductal adenocarcinoma (BR-PDAC); however, an optimal neoadjuvant regimen is lacking. We assessed the efficacy of FOLFIRINOX chemotherapy followed by gemcitabine-based chemoradiation as preoperative therapy.
Methods
Patients received 4 cycles of FOLFIRINOX, followed by 6-weekly gemcitabine with concomitant intensity-modulated radiation. The primary endpoint was the R0 resection rate. Secondary outcomes included resection rate, overall-response, overall survival (OS), progression-free survival (PFS), and tolerability. The trial was terminated early due to slow accrual. A Simon’s optimal two-stage phase II trial single arm design was used. The primary hypothesis of treatment efficacy was tested using a multistage group sequential inference procedure. The secondary failure time analysis endpoints were assessed using the Kaplan-Meier procedure and the Cox regression model.
Results
A total of 22 patients enrolled in the study, 18 (81.8%) completed neoadjuvant treatment. The bias corrected R0 rate was 55.6% (90% CI: 33.3, 68.3; P value = .16) among patients that received at least 1 cycle of FOLFIRINOX and was 80% among patients that underwent surgery. The median OS was 35.1 months. The median PFS among patients that underwent surgery was 34 months.
Conclusion
An R0 resection rate of 55.6% is favorable. Neoadjuvant FOLFIRINOX followed by concomitant Gemcitabine with radiation was well-tolerated. NCT01897454
Keywords
Introduction
Historically, pancreatic adenocarcinoma (PDAC) is recognized as an aggressive malignancy that is mostly diagnosed at advanced stages and carries a grim prognosis with a 5-year overall survival (OS) of 3% and 13-40% in metastatic and non-metastatic disease, respectively. 1 In the non-metastatic setting, achieving negative resection margins (R0) has been shown to improve overall survival from 10-15 to 23-28 months,2,3 which approaches the survival of patients with resectable disease. 4 In this setting, neoadjuvant therapy serves several purposes, including targeting micrometastatic disease present in most patients with BR-PDAC at diagnosis, downstaging tumors to increase the probability of R0 resection, and selecting patients with favorable tumor biology who would benefit from an extensive surgery. 5 Thus, neoadjuvant chemotherapy has been adopted as the standard of care for patients with BR-PDAC, however, an optimal regimen remains controversial.6,7
The evidence-based selection of an optimal neoadjuvant regimen has been precluded by several challenges that researchers face when studying BR-PDAC. 5 First, only 20% of all pancreatic cancer cases have BR-PDAC at diagnosis, hindering patient accrual. 1 Therefore, several retrospective and prospective trials have allowed the inclusion of locally-advanced PDAC (LA-PDAC) patients, who differ in outcomes, and complicate result interpretation.8,9 Moreover, other clinical trials have closed before completing its accrual target, which has left questions unsolved due to underpowered statistical analysis. Furthermore, criteria to define resectability have not been not uniformly established among societies such as the National Comprehensive Cancer Network (NCCN), MD Anderson Cancer Center (MDACC) and America Hepato-Pancreato-Biliary Association/Society of Surgical Oncology/Society for Surgery of the Alimentary Tract (AHPBA/SSO/SSAT).10,11 Moreover, resectability is still subject to the evaluation of a multidisciplinary team in high-volume cancer centers. As a result, NCCN guidelines currently recommend either FOLFIRINOX or gemcitabine with nab-paclitaxel with consideration of chemoradiation using capecitabine as the preferred chemotherapeutic agent, while gemcitabine is also listed as an alternative. 12
While the National Comprehensive Cancer Network guidelines favor this approach, there is no evidence to support a specific neoadjuvant regimen for this population. 12 Thus, a regimen selection is based on data extrapolated from randomized clinical trials, which have established superiority by increasing the OS with 5-fluorouracil/irinotecan/oxaliplatin (FOLFIRINOX) or gemcitabine/nab-paclitaxel (Gem/NP) in the metastatic setting13,14 and FOLFIRINOX or gemcitabine/capecitabine in the adjuvant setting15,16; compared to gemcitabine monotherapy. Moreover, the addition of neoadjuvant chemoradiation, has been evaluated given the radiosensitizing properties of drugs such as gemcitabine and 5-fluorouracil/capecitabine.
We conducted a single arm Simon two-stage phase II trial to evaluate the efficacy, defined as the proportion of R0 resections, of FOLFIRINOX chemotherapy followed by gemcitabine based chemoradiotherapy as preoperative therapy in patients with BR-PDAC.
Methods
Eligibility
Adult patients with histologically confirmed, treatment-naïve PDAC diagnosed at Montefiore Medical Center, deemed to have BR-PDAC by a multidisciplinary team according to the Expert Consensus Statement definition by Callery et al 17 were eligible for the study (see Appendix). The inclusion criteria were as follows: performance status ≤ 1 per the Eastern Cooperative Oncology Group (ECOG) criteria, 18 life expectancy greater than 6 months, adequate organ function, defined as leukocyte count ≥3000/μL, absolute neutrophil count (ANC) ≥1500/μL, platelet count ≥100,000/μL, total bilirubin (TB) ≤ 2 mg/dl, aspartate transaminase (AST) and alanine transaminase (ALT) ≤ 2.5 times the upper limit of normal and creatinine within normal limits. Subjects were enrolled from November 2012 to April 2019. Participants with uncontrolled intercurrent illness, human immunodeficiency virus, pregnant women, or those with any concurrent active malignancy other than non-melanoma skin cancer or carcinoma in-situ of the cervix were excluded.
Study Design
This was a single-center, phase 2 trial using a Simon’s Optimal two-stage design 19 to evaluate the efficacy of neoadjuvant FOLFIRINOX followed by gemcitabine-based chemoradiotherapy in patients diagnosed with BR-PDAC. The primary endpoint was the proportion of R0 resection, defined as the absence of gross and microscopic tumor involvement in the resection margins, among participants receiving at least 1 cycle of FOLFIRINOX chemotherapy.
The secondary endpoints included the proportion of patients that underwent surgery, the R0 resection rate among patients that underwent surgery, overall response rate (ORR), defined as the proportion of patients that achieved PR or CR as per the Response evaluation criteria in solid tumors criteria (RECIST v.1.1) 20 ; OS defined as the duration of time from diagnosis to death; progression-free survival (PFS), defined as the duration of time from diagnosis to the time of clinical or radiographic progression or death among patients who underwent surgery, and toxicity, evaluated according to the National Cancer Institute Common Terminology Criteria for Adverse Events, version 4.3 (CCTAE). 21
The study protocol was approved by the Institutional Review Board and conducted in accordance with ethical principles per the Declaration of Helsinki. The trial was registered on ClinicalTrials.gov and all participants provided written informed consent.
Procedures and Treatments
The induction phase consisted of FOLFIRINOX (oxaliplatin 85 mg/m2, leucovorin 400 mg/m2, irinotecan 180 mg/m2, 5-fluorouracil 400 mg/m2 IV bolus; followed by 5-fluorouracil, given as a continuous infusion of 2400 mg/m2 over a 46-hour period). This regimen was given every 2 weeks for 4 cycles, and the response was assessed by computed tomography (CT) at the end of 4 cycles. Patients who did not have progression of disease (POD) proceeded to the chemoradiation phase, while those with POD continued management per their oncologist.
Chemoradiation, starting within 6 weeks of completing the induction phase, was given by external beam radiation therapy (EBRT) using intensity-modulated radiation therapy (IMRT) and was delivered once daily for 5 days a week to a total dose of 50.4 Gy in 28 fractions. Patients underwent a 4D CT simulation for treatment planning and if there was significant motion, patients were either treated with deep-inspirational breath-hold or gating using 3 to 4 gating phases with the least amount of target motion. The treatment planning (simulation) CT scan was used to define gross tumor volume (GTV) defined as all known gross disease and clinical tumor volumes (CTV), defined as the areas of subclinical risk around the GTV. Elective nodal irradiation for microscopic disease was not performed. The Planning Target Volume (PTV) to compensate for treatment set-up uncertainty was determined by putting a margin around the CTV or iCTV which accounted for internal organ motion. Surrounding critical normal structures (small bowel, stomach, duodenum, liver, kidneys, lung and heart) were delineated and were constrained as shown in Supplementary Table 1. Concomitantly, patients received weekly gemcitabine dosed at 400 mg/m2. After 4-6 weeks of treatment completion, treatment response was evaluated by imaging (CT with pancreatic protocol) and assessed by a multidisciplinary board. Patients deemed to have resectable disease underwent surgery. All treatment was given at Montefiore Medical Center.
Statistical Analysis
Sample size determination: Assuming a 40% R0 resection rate among patients with BR-PDAC treated in the preoperative setting, 12 the Simon optimal two-stage design tested the hypothesis of H0: resection rate (R0) ≤40% vs the alternative hypothesis of Ha: R0 ≥ 60%. A total of 46 patients (16 and 30 patients for stages 1 and 2, respectively) were required to evaluate the primary endpoint at 5%, one-sided, type I error, and 80% power. At the first stage, if 7 or fewer patients of 16 patients had R0 resection, the intervention would be considered futile, and the study would be terminated. In the second stage, an additional 30 patients would be accrued. If at least 24 patients had achieved R0 resection, the null hypothesis would have been rejected, and intervention would be considered efficacious. The study design had an expected sample size of 24.52 and a probability of early termination of 71.6%.
Due to slow accrual, the trial was terminated early when 22 participants were enrolled. The study successfully completed phase I of study and continued to phase 2, assuming a single-stage design under a two-stage scenario to report the R0 resection rate will be biased and acknowledging the need for proper adjustment when the actual sample size is different from the planned sample size, 22 we used a multistage group sequential inference procedure 22 to account for both two-stage and sample size adjustment. The bias-adjusted response rate was estimated using the Whitehead method23,24 to be consistent with the hypothesis testing, a 90% confidence interval along with the P-value.
The distribution of patient and clinical characteristics was numerically summarized using descriptive statistics. The secondary endpoints were assessed at the end of the study. The overall response rate (ORR) was presented as a proportion along with 95% Clopper-Pearson exact confidence interval. The failure-time events, OS and PFS, were assessed using Kaplan – Meier product limit estimator along with 95% confidence intervals. The difference in survival distribution was examined using the log-rank test. Adverse events were tabulated using frequency count and percentages by category, severity, and their relations to the intervention. All analyses were performed using R 4.0.5 software 25 and Stata software version 17.0. 26
Results
Patient Characteristics
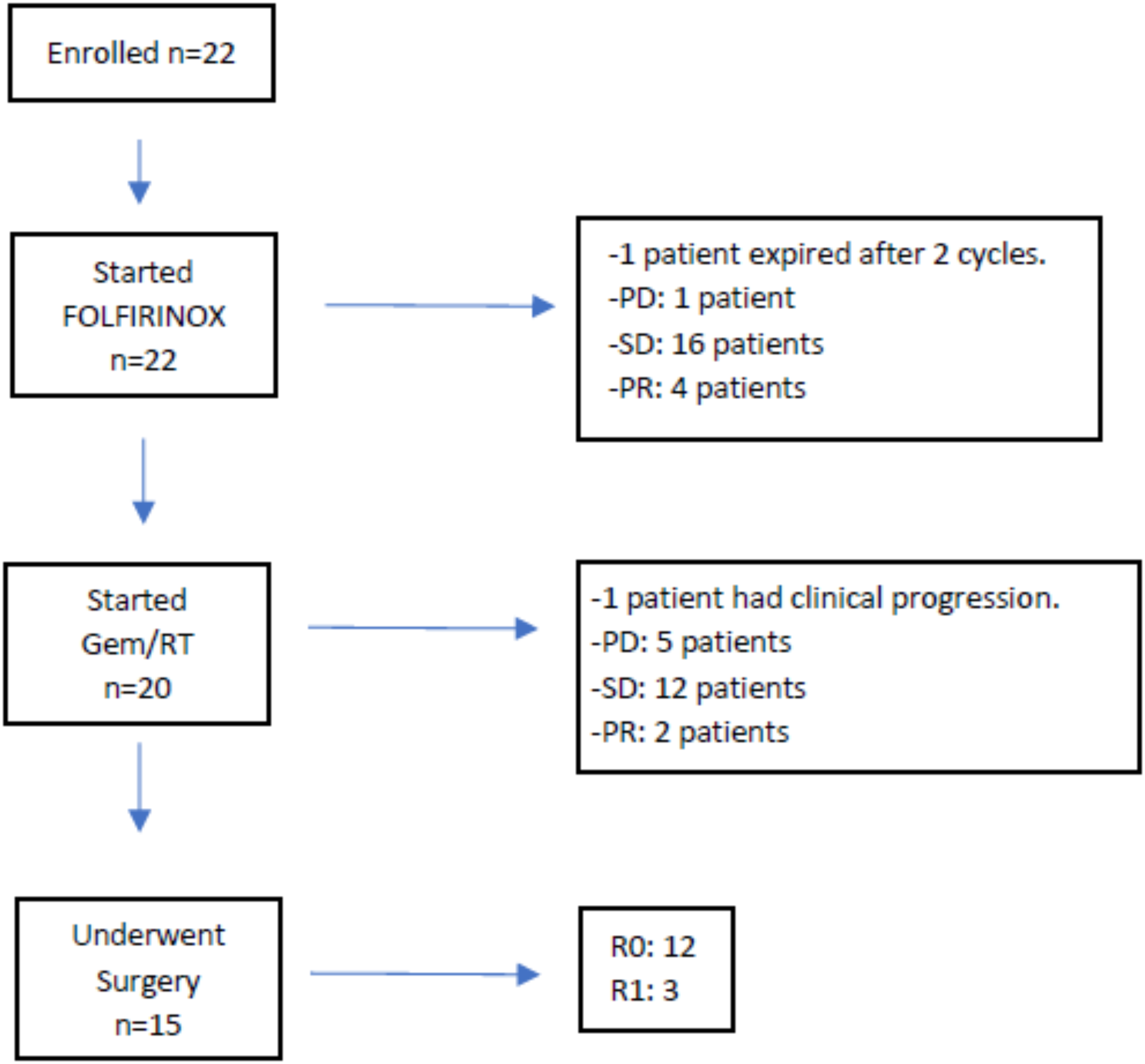
Between November 2012 and April 2019, 22 patients were enrolled in the study. Figure 1 is a flowchart that summarizes the course of all enrolled participants. At diagnosis, the median age was 63.5 years (SD: 7.8), and 45.5% (n = 10) were male. Nearly 82% were from minority racial/ethnic groups (Hispanics: 10 [45.5%] and Non-Hispanic Blacks: 8 [36.4%]). Tumors were more commonly localized in the pancreatic head and neck (95.5%), measured between 2 and 4 cm in maximum dimension (T2: 40.9%), had metastases in 4 or more regional lymph nodes (N2: 40.9%), and had venous vessel involvement (63.6%). Clinicopathological characteristics are presented in Table 1. Flowchart of participants with borderline-resectable pancreatic ductal adenocarcinoma enrolled in the trial. Baseline characteristics of patients with borderline-resectable pancreatic cancer (n = 22).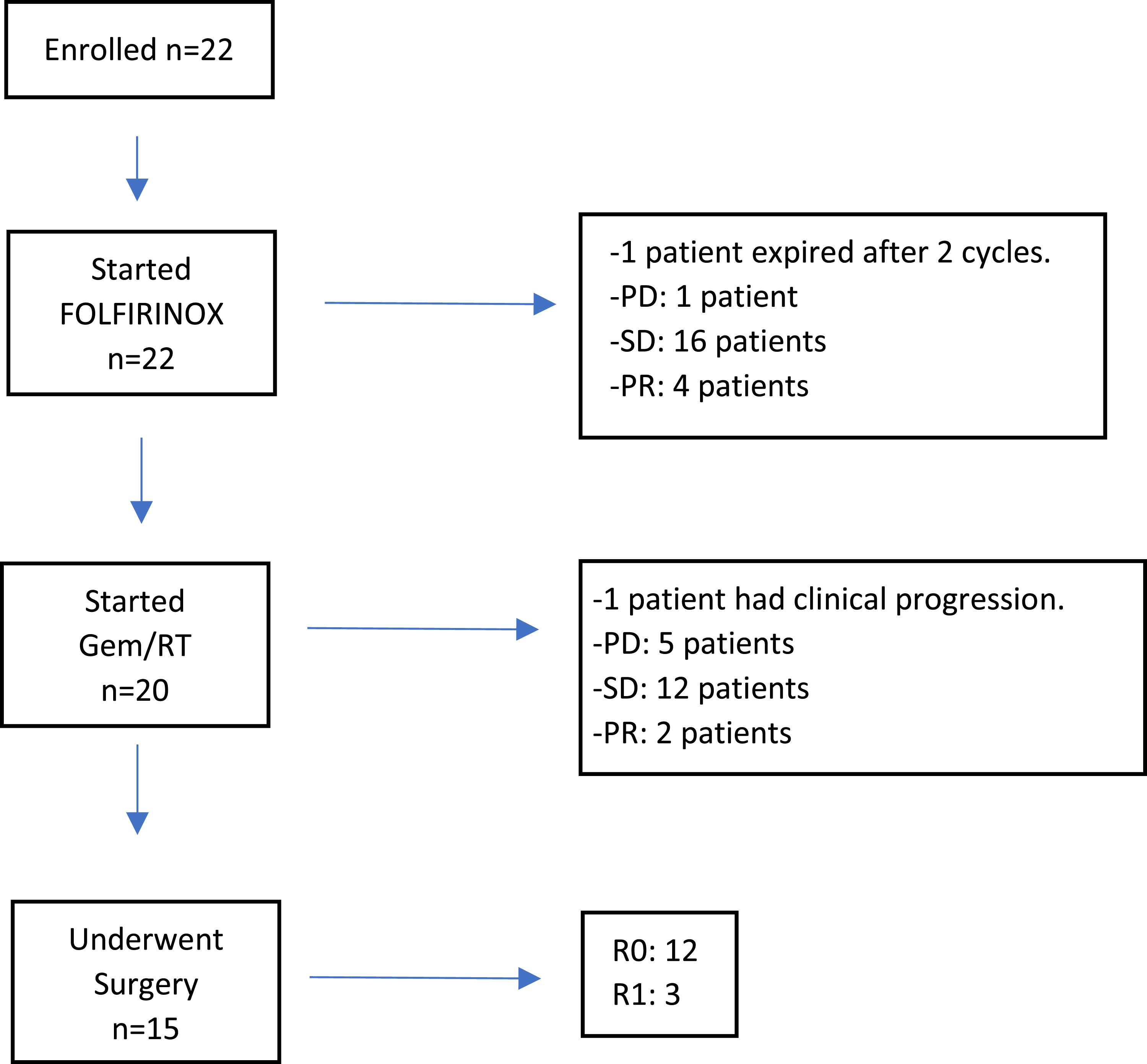

Efficacy
Of 22 patients that received at least 1 cycle of FOLFIRINOX, the bias-corrected R0 resection rate was 55.6% (90% CI: 33.3 - 68.3%; P = .16). The treatment showed promise, but this was not statistically significant.
Secondary Endpoints
Of 22 enrolled patients, 15 (68.2%) underwent pancreaticoduodenectomy, and vascular reconstruction was required in 5 (33.3%). Radiographic responses are shown in Figure 2. R0 margins were achieved in 12 of 15 (80%) patients that underwent surgery (Figure 2). Patients were followed for a median of 27.6 months (range: 2.3 - 86.3 months). The probability of survival at 12, 24, and 36 months were 85.1%, 74.5%, and 49.7%, respectively. The median OS was 35.1 months (95% CI: 18.4, 78.5 months). The median OS was higher in participants that achieved R0 compared to all others (78.5 vs 18.4 months, log-rank P-value = .02). The probability of recurrence-free survival among patients that underwent surgery at 12, 24, and 36 months were 100%, 52.4%, and 43.6% respectively. The median recurrence-free survival was 34 months (95% CI: 18.8, - ). The Kaplan-Meier plots for recurrence-free and overall survival are presented in Figure 3. (A) Tumor response (n = 19) and (B) Clinical Outcomes after neoadjuvant treatment with FOLFIRINOX followed by Gemcitabine/RT (n = 22) in patients with borderline-resectable pancreatic cancer who underwent surgery. Recurrence Free-Survival and Overall Survival of patients with BR-PDAC treated with neoadjuvant FOLFIRINOX followed by Gemcitabine/RT (n = 22)

Adverse Events
Toxicity assessment.

Discussion
The results of this phase 2 study of a sequential multimodality regimen using induction FOLFIRINOX followed by concomitant gemcitabine with radiation in patients with BR-PDAC showed good efficacy with a R0 resection rate of 55.6% and was well-tolerated. This resection rate did not reach threshold for statistical significance which might have been impacted by suboptimal sample size as the trial did not reach its accrual goal.
Importantly, this regimen showed a good balance between efficacy and tolerance, as 68.2% of enrolled patients were able to undergo surgery, whereas the remaining could not tolerate treatment or progressed to metastatic disease. Additionally, another measure of efficacy includes the high rate of 80% of patients undergoing surgery that achieved R0 margins, with all of them remaining free of recurrence in the first year. Regardless of the R0 resection rate, a median OS that approaches 3 years with a 1-year OS rate of 85.1% among all patients that were enrolled in the trial makes this a robust regimen.
Thus far, 3 prospective trials have evaluated similar regimens.27-29 In all, induction chemotherapy was given using FOLFIRINOX, while subsequent treatment differed by chemotherapy (gemcitabine or capecitabine) and radiation backbones (EBRT/IMRT or proton therapy). While chemoradiation with concurrent gemcitabine vs capecitabine has not been compared in a clinical trial among patients with BR-PDAC, both seemed effective and had an acceptable toxicity profile with differences that will be highlighted in this discussion.
The surgical resection frequency of 68.2% reported in this trial is comparable to findings of 2 trials that used capecitabine as a radiosensitizer, which reported resection frequencies of 67% and 68%,27,28 whereas this frequency was 63% in a trial using gemcitabine with radiation. 29 In the case of the R0 resection rate, higher frequencies (63.8% and 64.6%) were achieved in capecitabine than gemcitabine trials (65% and 64% compared to 52%),27-29 which is similar to the frequency we report in our cohort (54.5%). As all these trials used FOLFIRINOX as the induction regimen, it seems that using capecitabine instead of gemcitabine in combination with radiation could offer higher R0 resection rates. This finding, favoring the use of capecitabine over gemcitabine as a radiosensitizer agent after a course of induction chemotherapy, has also been observed in patients with locally advanced PDAC, as reported in the open-label, randomized Scalop trial. 30
Although gemcitabine is a strong radiosensitizer, the main limitation for its use is the lack of evidence to support an adequate, tolerable dose. In this study, despite starting gemcitabine at 400 mg/m2, dose delays and dose reductions were required in 31.6%, and 26.3% of patients, respectively, and 16.7% suffered grade 3 or higher adverse events. Likewise, Tran et al. required dose modifications due to frequent grade 3 or higher toxicities in up to the third part of a cohort using concomitant radiation with gemcitabine initially dosed at 1000 mg/m2. 29 It is plausible that suboptimal dosing of gemcitabine due to inability to tolerate the combination with radiation could explain lower R0 rates, despite somewhat similar resection rates, compared to capecitabine.
Among retrospective studies, resection rates broadly ranged from 15.2% to 44.1%. This is likely explained by population heterogeneity as all these studies included patients with LA-PDAC,8,9,31,32 for whom resection and R0 rates are expectedly much lower. Of note, a retrospective study limited to 32 patients with BR-PDAC evaluated a different regimen using gemcitabine and nab-paclitaxel for induction, followed by concomitant capecitabine and radiation with preliminary results that demonstrated a high R0 resection rate (80%) among those who proceeded to surgery. 33 If this is confirmed in a subsequent prospective trial, gemcitabine with nab-paclitaxel could be considered an alternative regimen for induction treatment in patients with BR-PDAC. Optimal outcomes with this regimen could be explained by better tolerability of full-recommended doses in both induction and chemoradiation phases. Although there is no data directly comparing the efficacy of FOLFIRINOX and gemcitabine with nab-paclitaxel in this setting, the SWOG 1505 trial, which compared neoadjuvant FOLFIRINOX and gem/nab-paclitaxel in patients with resectable PDAC, showed similar outcomes including R0 resection rates and overall survival. 34
This study has limitations that deserve consideration. First, this trial was initially planned to enroll 46 patients with BR-PDAC; the study was halted due to slow accrual. Second, the criteria for BR-PDAC was based on a consensus statement issued by the AHPBA/SSAT/SSO, 17 which has not been universally adopted. Finally, parent doses of FOLFIRINOX doses were used and may have contributed to some toxicities observed.
The strengths of the study include prospective trial design, long term follow up of patients, and enrollment of an ethnically diverse patient population. Currently, considerations to optimize outcomes include extending the duration of neoadjuvant therapy, omitting the 5-bolus to minimize toxicity, and incorporating stereotactic body radiotherapy in select patients. The role of neoadjuvant radiation in BR-PDAC requires further investigation. The PREOPANC trial showed that neoadjuvant chemoradiation was associated with superior overall survival compared to upfront surgery followed by adjuvant gemcitabine. 35 However, the addition of SBRT to chemotherapy alone in the preoperative setting was not shown to improve outcomes in the ALLIANCE A021501 study. 36 Nevertheless, the ALLIANCE study did establish FOLFIRINOX as a new reference regimen in this setting. Additional randomized prospective studies including PREOPANC-2 and PANDAS/PRODIGE44 (NCT02676349) would help shed light on the question regarding the utility of radiation in BR-PDAC. 37
Conclusion
In summary, a multimodality regimen using FOLFIRINOX followed by gemcitabine with radiation has a tolerable safety profile and leads to high R0 resection rates among those who undergo surgery. We eagerly await the results of ongoing prospective studies evaluating multimodality approaches for BR-PDAC. Given the difficulty of recruiting an adequate sample size, multicenter studies should be encouraged.
Supplemental Material
Supplemental Material - Induction Chemotherapy With FOLFIRINOX Followed by Chemoradiation With Gemcitabine in Patients With Borderline-Resectable Pancreatic Ductal Adenocarcinoma
Supplemental Material for Induction Chemotherapy With FOLFIRINOX Followed by Chemoradiation With Gemcitabine in Patients With Borderline-Resectable Pancreatic Ductal Adenocarcinoma by Ana Acuna-Villaorduna, Viswanathan Shankar, Michael Wysota, Amanda Jirgal, Rafi Kabarriti, Sarah Bellemare, Inessa Goldman, Andreas Kaubisch, Santiago Aparo, Sanjay Goel and Jennifer Chuy in Cancer Control
Footnotes
Acknowledgments
We wish to acknowledge all our patients and their families who participated in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series. All procedures in this study were conducted in accordance with the Einstein IRB. Approval # 2021-570. Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Supplemental Material
Supplement material for this article is available in online.
Pretreatment Assessment of Resectable and Borderline Resectable Pancreatic Cancer: Expert Consensus Statement
Callery, M.P., Chang, K.J., Fishman, E.K. et al. Pretreatment Assessment of Resectable and Borderline Resectable Pancreatic Cancer: Expert Consensus Statement. Ann Surg Oncol 16, 1727-1733 (2009). https://doi.org/10.1245/s10434-009-0408-6
Tumors considered borderline resectable include the following: a. No distant metastases. b. Venous involvement of the superior mesenteric vein (SMV)/portal vein demonstrating tumor abutment with or without impingement and narrowing of the lumen, encasement of the SMV/portal vein but without encasement of the nearby arteries, or short segment venous occlusion resulting from either tumor thrombus or encasement but with suitable vessel proximal and distal to the area of vessel involvement, allowing for safe resection and reconstruction. c. Gastroduodenal artery encasement up to the hepatic artery with either short segment encasement or direct abutment of the hepatic artery, without extension to the celiac axis. d. Tumor abutment of the SMA not to exceed >180° of the circumference of the vessel wall.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
