Abstract
Purpose
Our specific aim was to develop and assess the consensus-based validity of common measures for understanding health behaviors and ancestry in Florida’s population subgroups and establish the feasibility of wide-scale implementation of the measures and biospecimen collection within three cancer centers’ catchment areas.
Methods
Using the National Cancer Institute’s Grid-Enabled Measures web-based platform and an iterative process, we developed the Florida Health and Ancestry Survey (FHAS). We then used three sampling approaches to implement the FHAS: community-engaged, panel respondent, and random digit dialing (RDD). We asked a subset of participants to provide a saliva sample for future validation of subjective ancestry report with DNA-derived ancestry markers.
Results
This process supported the FHAS content validity. As an indicator of feasibility, the goals for completed surveys by sampling approach were met for two of the three cancer centers, yielding a total of 1438 completed surveys. The RDD approach produced the most representative sample. The panel sampling approach produced inadequate representation of older individuals and males. The community-engaged approach along with social media recruitment produced extreme underrepresentation only for males. Two of the cancer centers mailed biospecimen kits, whereas one did not due to resource constraints. On average, the community engaged approach was more productive in obtaining returned biospecimen samples (80%) than the panel approach (48%).
Conclusions
We successfully developed and implemented the FHAS as a common measure to show its feasibility for understanding cancer health disparities in Florida. We identified sampling approach successes and challenges to obtaining biospecimens for ancestry research.
Keywords
Comprehensive understanding of cancer health disparities is hindered by inadequate understanding of heterogeneity in risk factors, prevention behaviors, clinical trials, and access to care among diverse population subgroups within Florida. Ranked as the state with the second highest number of cancer cases as well as cancer incidence and mortality, Florida has notable cancer health disparities that disproportionately burden historically marginalized racial and ethnic groups as well as underserved urban and rural residents.1-4 Current surveillance data, however, fail to capture and summarize the scope and mechanisms of cancer disparities in unique populations and local communities served by Florida’s three academic cancer centers that comprise the Florida Academic Cancer Center Alliance (FACCA). Therefore, with FACCA support, the Moffitt Cancer Center (MCC), University of Florida Health Cancer Center (UFHCC), and University of Miami Sylvester Comprehensive Cancer Center (UMSCCC) initiated efforts to improve the data related to cancer health disparities throughout Florida. The purpose of this article is to describe our approach to conducting a collaborative, evidence-based project for advancing cancer health equity research in Florida, a large, geographically diverse state with significant multiculturalism and a high cancer burden.
Academic cancer centers in Florida are well positioned to characterize the diversity of catchment area participants and to investigate the varied mechanisms that produce disparities in cancer incidence and outcomes. Florida has a uniquely diverse ancestry population. More than 42% of this population is Black (16.9%) or Hispanic (26%), approximately 20% is foreign-born, 5 and over 2 million (approximately 10%) of Floridians reside in rural areas. 6 Florida’s diverse population is thought to reflect the future demographics of the nation. The areas covered by the three cancer centers include 42 of the 67 counties in Florida. The UFHCC catchment area includes Alachua, Baker, Bradford, Citrus, Clay, Columbia, Dixie, Gadsden, Gilchrist, Hamilton, Jefferson, Lafayette, Lake, Leon, Levy, Madison, Marion, Putnam, Sumter, Suwannee, Taylor, Union, and Wakulla counties in North Central Florida. UMSCC catchment area includes Miami-Dade, Broward, Palm Beach, and Monroe counties in South Florida and MCC catchment area includes Lake, Charlotte, Sumter, Sarasota, Hernando, Pasco, Pinellas, Lee, Citrus, Manatee, Highlands, Hillsborough, Hardee, Polk, and Desoto counties in West Central Florida.
To facilitate better data sharing and comparison, our collaborative efforts built on National Cancer Institute (NCI) initiatives by developing and testing a core set of self-report measures. The use of the core set of self-report measures would produce data that represent the distinct populations within Florida, which is essential to directing research, clinical practice, programmatic, and policy priorities of each academic cancer center. Furthermore, we can use the common measures to build a more comprehensive understanding of the mechanisms at play in cancer health disparities across Florida and leverage our joint resources to strategize for statewide cancer control. Simultaneously, our approach also allowed each cancer center to add items to accommodate the unique needs of each cancer center’s catchment area and/or the focus of each center’s cancer control efforts, making this model of academic cancer center collaboration possible in other diverse states with high cancer burden. The specific aim of the project presented here was to develop and assess the consensus-based validity of common measures for understanding health behaviors and ancestry in Florida’s population subgroups and establish the feasibility of wide-scale implementation of the measures and biospecimen collection within each catchment area relevant to the FACCA.
Methods
The project team achieved the aim through an iterative process based on cancer literature review, stakeholder engagement, consensus building, and implementation of a variety of community outreach approaches as described in the following sections. The desired outcomes were: (1) a content-valid common set of measures to characterize the cancer risk behaviors of individuals living in the academic cancer centers’ catchment areas, (2) defined sampling frameworks for reaching individuals, (3) feasibility indicators of the measure using the sampling frameworks; (4) feasibility indicators of saliva sample collection from a subsample for genetic ancestry characterization, and (5) feasibility of validating subjective ancestry report with DNA-derived ancestry markers. The work reported here focuses on the outcomes 1-4. The findings for outcome 5 and survey results for outcome 3 will be reported elsewhere.
Consensus Building Through GEM
FACCA work group and investigators.

The GEM platform is a dynamic, web-based collaborative tool that can be used to gain consensus on the use of common measures for prospective research. 8 Stakeholders are enabled to evaluate measures using collaborative workspaces. GEM is available publicly and can be used by invitation (see: https://www.gem-measures.org/Public/Home.aspx). The goal is to achieve harmonized data that can be shared and analyzed. To identify commonalities in survey items and item format, the Workgroup created a construct table from the individual survey tools provided by each cancer center and GEM. Comparing the survey questions and GEM items, many questions were similar but had different format (ie, age in years vs date of birth). Workgroup members set up a private GEM Workspace and uploaded the survey items from the construct table. We invited workgroup members, members of the three cancer centers’ Cancer Population Science (CPS) research programs, and recruited community members to vote on the inclusion and preferred format for each item. Faculty, team members and community advocates recruited through word of mouth participated in the voting. Each site was responsible for selecting 10 participants to complete the survey.
FACCA Common Survey Instrument
Constructs and description of measures with source.

Each Florida academic cancer center planned to recruit a sample of 300 respondents using either random digit dialing (RDD) (probability-based sampling) or a respondent panel purchased from a panel provider (population-based sampling). Each cancer also planned to recruit an additional 100 participants using a community-engaged approach (convenience sample). Each cancer center proceeded with the sampling approach feasible locally.
Sampling Framework UF, MCC, UM catchment area population and actual achieved by sampling approach.

Abbrevuation: UFHCC, University of Florida Health Cancer Center; MCC, Moffitt Cancer Center; UMSCCC, University of Miami Sylvester Comprehensive Cancer Center.
aNo data.
University of Miami Sylvester Comprehensive Cancer Center conducted a hybrid phone sampling frame in its 4 county catchment area (Table 3). Its approach combined random digit dialing (RDD), targeted landline, and out-of-area consumer cell sampling to construct a representative sample, while oversampling certain hard to reach populations, such as Native American and Black residents, as well as those residing in rural areas. As of November 2019, a sample was obtained from Marketing Systems Group (M-S-G) that was 80% RDD cell, 15% targeted landline, and 5% out-of-area cell. The UMSCCC data were weighted in order to produce reliable population estimates. While reflecting the selection probabilities of sampled respondents, weighting also seeks to compensate for practical limitations of sample surveys, such as differential nonresponse and under-coverage. To account for this, an iterative proportional fitting method (ie, raking) conducted utilizing the WgtAdjust procedure of SUDAAN was used to simultaneously adjust the weighted distributions along several dimensions, including gender, race, ethnicity, education, income, and county.
MCC conducted both community-based and survey panel approaches targeting counties in West Central Florida. The UF Survey Research Center at the BEBR identified residents that were: (1) ages 18+; (2) resided in Moffitt’s catchment area counties; (3) were able to speak and read English or Spanish; and (4) identified as Black or Hispanic in the survey panel database. BEBR collected the MCC data between December 17th, 2020 and March 2nd, 2021. Data collection for the community-based sample in the MCC’s 15-county catchment area began on January 25th, 2021 and was completed on June 1st, 2021. MCC utilized its network of outreach teams and community partners to share initiative information (eg, flyers, social media postings, email listservs) with community members and refer interested individuals to the study team.
Biospecimen Collection
In addition to each site collecting self-reported information, we also assessed the feasibility of collecting biospecimens to support genetic ancestry estimation (via genotyping) at 2 sites. The analysis will provide further insight into the biological diversity within our catchment areas based on resultant molecular profiles. A subset of participants in both the survey panel and community-based sample, was asked to indicate their willingness to provide saliva sample for this purpose. The participants from the panel and community-based sampling approaches were selected consecutively until the accrual goal, as measured by returned collection kits, was reached (UF) or the study recruitment period was completed (MCC). For participants declining this option, reasons for refusal were documented at this stage and responses were collected in an open-ended question, included within the questionnaire.
The study received IRB approval for a waiver of signed informed consent. Participants who opted in to contribute a biospecimen, were provided with an Informed Consent form that provided information about the research that included elements of consent but the participant was not required to sign or return the form. Participants also received detailed instructions and materials for conducting the saliva collection at home. Briefly, the instructions to participants provided step-by-step guidance on successful collection of a 2 mL volume saliva sample using a DNA Genotek Oragene (Ottawa, Canada) OGR-600 saliva collection kit apparatus to be returned to the study team via pre-addressed mailing materials. Upon receipt by the study team, information about the saliva specimens were logged into an Excel file or a Research Electronic Data Capture (REDCap) database for the study to include: (a) date, time, and volume of specimen collection; (b) linkage of the specimen to the participant’s de-identified subject identifier (SID) by recording the DNA Genotek barcode (unique to each collection kit); and (c) generating the gift card incentive for the participant. Germline DNA from saliva samples was extracted at each institution using compatible manual DNA extraction protocols or automated platforms (eg, Qiagen Autopure LS) according to the manufacturer’s protocol; resultant DNA aliquots were stored at −80°C and batched for sequencing (ie, genotyping). Germline genotyping will be completed soon to facilitate an estimation of the proportion of the genetic ancestry derived from major global populations (eg, European, Indigenous American, East Asian, and African) represented within each participant and to molecularly characterize individuals in Florida and within the catchment area. Ancestry proportions will be described using descriptive statistics and visualized using principal components analysis and t-Distributed Stochastic Neighbor Embedding (t-SNE). 9
Results
Validity
Based on the stakeholder votes on the GEM platform and a consensus conference with the multidisciplinary group of representatives from each cancer center, the final common measure included 29 main items. Several of the items included multipart questions. Every question had the response option of “Prefer not to answer.” Since the items came from previously validated measures, and the assessment focused on validity for the populations within our catchment areas, this consensus process supported the content validity of the common measure. 10
To further support content validity, each cancer center modified the survey to add previously validated questions specific to the populations in their catchment area. The modifications included the addition of questions on rural identity, questions specific to Latinx populations, and questions integral to local CPS priorities. The additional questions were drawn from existing valid and reliable instruments. For example, the UFHCC survey tool contained 48 items, of which 37 had multiple parts including, the Brief Pain Inventory - Short Form (BPI-sf), 11 eHealth Literacy Scale (eHEALS), 12 National Cancer Institute’s (NCI) Health Information National Trends Survey (HINTS), 13 BRFSS, 14 NHANES, 15 and COVID-19 (investigator derived, format adapted from Palliative Care & Supportive Oncology Workgroup Survey and eHEALS). 12 The MCC survey tool included additional questions about COVID-19 and community engagement with MCC programs. University of Miami Sylvester Comprehensive Cancer Center removed questions about rurality, since its catchment area is almost entirely urban, and sequenced questions about US nativity with questions about country of origin and length of residence in the United States to be sensitive to rampant fears in immigrant populations about risk of deportation during the data collection period. University of Miami Sylvester Comprehensive Cancer Center did not include any questions about COVID-19, as data collection in its catchment area began in November 2019 and additional questions would have been disruptive to overall data collection.
Feasibility of Survey Measure
The survey measure was completed in approximately 30 to 40 minutes using community-engaged approach (phone interview) or panel approach as a self-administered online survey. As noted previously, the original goal per cancer center was 300 participants from RDD or panel approaches and 100 participants from community-engaged approaches. As shown in Table 3, the UFHCC community-engaged sampling approach yielded 100 completed surveys whereas the Bureau of Economic and Business Research (BEBR) conducted panel approach yielded 330 completed surveys. The UMSCCC did not complete a community-engaged sampling approach due to the COVID-19 pandemic, but using the RDD approach the BEBR yielded 716 completed surveys. The UMSCCC over sampled to achieve another institutional goal related to characterization of their catchment area, something that the UFHCC completed previously. The MCC community-engaged sampling approach yielded 39 completed surveys whereas the BEBR panel approach yielded 250 completed surveys.
Based on the proportions obtained by the various sampling approaches in Table 3, it is apparent that the RDD approach yielded sample proportions similar to the catchment area population in terms of age, gender, race, and ethnicity. The UFHCC convenience sampling and panel approaches yielded sample proportions within 5% of the population for 6 of the 14 categories for age, gender, race, ethnicity, and rural status. The UF panel approach yielded underrepresentation of individuals 55-79 years of age, men and Hispanics whereas the convenience sampling approach yielded overrepresentation of the 55-79 ages and non-Hispanic Whites and underrepresentation of males and Hispanics. The MCC convenience sampling approach yielded sample proportions within 5% of the population proportions for 3 of the 14 categories for age, gender, race, ethnicity, and rural status; the panel approach for 2 of the 14 categories. The MCC eligibility called for oversampling of Black and Hispanic populations since that was part of the eligibility criteria. Both approaches yielded overrepresentation of individuals 18-54 years of age and females, and underrepresentation of males. The MCC convenience sample yielded underrepresentation of those with rural identity and individuals 65 years of age or older. The MCC panel yielded underrepresentation in individuals 55-79 years of age.
Feasibility of Biospecimen Collection
Biospecimen sample kits mailed and actual received.
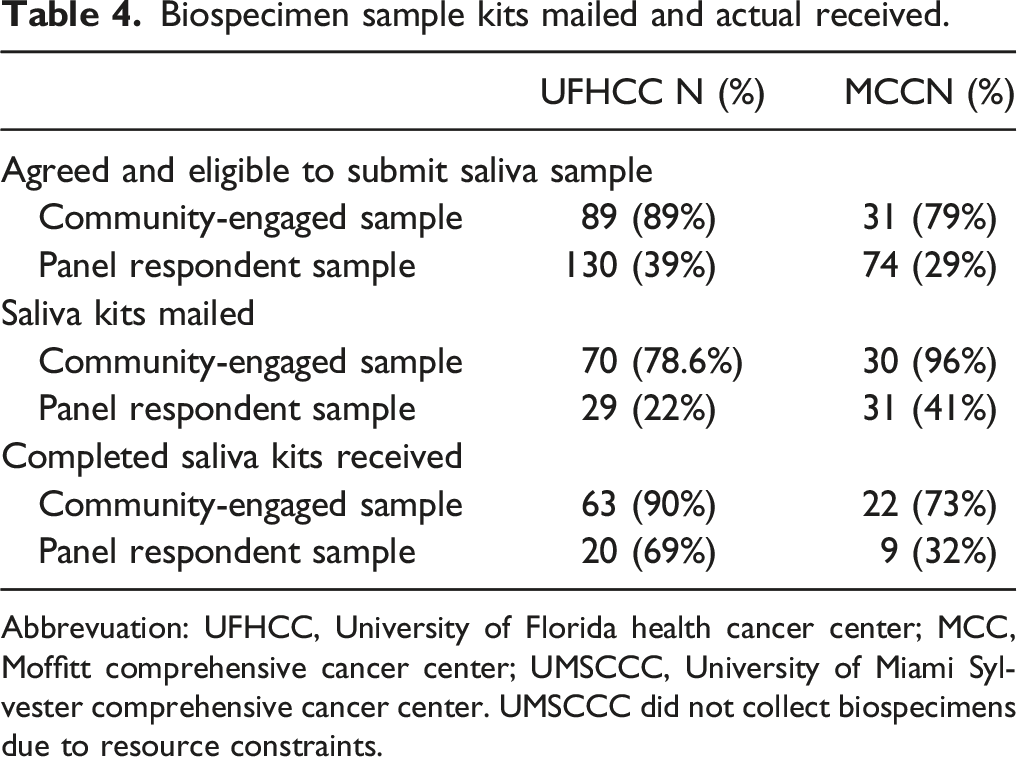
Abbrevuation: UFHCC, University of Florida health cancer center; MCC, Moffitt comprehensive cancer center; UMSCCC, University of Miami Sylvester comprehensive cancer center.
UMSCCC did not collect biospecimens due to resource constraints.
MCC intended to provide 100 participants with a saliva collection kit, 50 each for the panel respondents and community-engaged samples respectively. From the panel respondent sample, 74 responded “yes” to “willing to donate sample.” Of the 74 eligible panel respondents, 31 disclosed a valid mailing address. Of the 31 saliva collection kits mailed to panel respondents, 9 completed kits were returned. Of the community-engaged sample, 31 participants agreed to donate a saliva sample, 30 participants were mailed a kit and 22 returned the completed kit.
University of Miami Sylvester Comprehensive Cancer Center did not collect biospecimens due to the timing of when this part of the work was intended to start and the onset of the COVID pandemic. University research, particularly that which was community-facing, was halted starting in mid-March 2020 through early Fall 2020. Given community concerns about COVID and exacerbated tensions around research, a strategic decision was made not to pursue this focus of inquiry.
Discussion
The three academic cancer centers in Florida worked together successfully to develop the FACCA Common Measure, the FHAS, from which aggregate data can be obtained to characterize residents’ cancer risk behaviors within the centers’ catchment areas which, combined, covers much of the population of Florida. The FHAS requires 30 to 40 minutes for completion as a phone interview or self-administered online survey, and it is valid and feasible to survey participants in each center’s catchment area. The hybrid RDD approach produced the most representative sample within UMSCC’s 4 county catchment area. UFHCC’s and MCC’ panel sampling approach, which was expected to represent their respective catchment areas, had inadequate representation of older individuals and males. With UFHCC’s larger sample and catchment area, the convenience sampling approach along with social media, email, and extension agent-facilitated recruitment produced extreme underrepresentation only for males.
Since our primary intent was to examine the yield of the survey data relative to the age, gender, and race of our catchment area, the study provides important insights to plan future studies. The RDD approach yielded the sample most representative of the catchment area, however, this is likely due to the number of participants completing the survey rather than being reflective of the sampling approach or reflective of site performance. Overall, the community-engaged sampling approach was negatively impacted by the COVID-19 pandemic. Each cancer center planned to recruit 100 participants using the community-engaged sampling approach, however, due to the COVID-19 pandemic changes were necessary. Although UFHCC did meet the targeted number of participants, changes to the community-engaged approach for UFHCC meant no face-to-face meetings and recruitment occurred online and surveys were completed over the telephone. UMSCC did not to conduct the community-engaged sampling approach, and the MCC did not meet the targeted number of participants through the community-engaged sampling approach.
Our next steps are to complete data analysis within and across the cancer centers, complete the genotyping for genetic ancestry component estimations, and conduct an analysis of the congruence of the self-reported ancestry and DNA-derived ancestry markers. The findings from these analyses are expected to lead to joint publications and funding awards to support research focused on improving cancer health equity in Florida. We have already shared the FHAS with other research groups in Florida. This collaborative spirit has potential to unite researchers in Florida to discover solutions toward cancer health equity. This work is sorely needed to improve cancer outcomes for Floridians.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by the State of Florida, the Florida Academic Cancer Center Alliance (FACCA), and the University of Florida Health Cancer Center (UFHCC), Cancer Population Sciences research program, Biostatistics & Quantitative Sciences Shared Resource (UFHCC BQS-SR) and the Biostatistics and Bioinformatics Shared Resource (BBSR) at Moffitt Cancer Center (MCC). The Institutional Review Boards of the University of Florida (IRB #201901264, the University of Miami Sylvester Comprehensive Cancer Center (UM IRB Protocol # 20180390), and the Moffitt Cancer Center (Advarra IRB #00000971) approved the study. This publication was made possible by Grant Numbers U54CA233444 and 5P30CA076292 from the National Institutes of Health (NIH), National Cancer Institute (NCI). The content is solely the responsibility of the authors and does not necessarily represent the official views of the State of Florida, FACCA, the UFHCC, MCC, or the NCI. The final peer-reviewed manuscript is subject to the National Institutes of Health Public Access Policy.
