Abstract
Despite significant advances in oncology, cancer care globally continues to face critical challenges, including stark disparities in access, insufficient preventive focus, fragmented primary health care (PHC) integration, unsustainable financing models, workforce shortages, and inadequate community involvement. This paper revisits the Alma Ata Declaration’s principles—health equity, universal access, preventive care, and community participation—as a conceptual framework to address these persistent issues in cancer care. We highlight opportunities to strategically integrate oncology services within strengthened PHC systems, balancing centralized specialist resources with decentralized community-based care. Evidence from diverse settings illustrates how reinforcing PHC infrastructures enhances preventive measures, early detection, and survivorship care, thus mitigating geographic and socioeconomic disparities. Sustainable financing mechanisms and targeted workforce strategies, including task-shifting and multidisciplinary training, are proposed as essential components. Effective community engagement models demonstrate improved care relevance, acceptance, and outcomes. Additionally, we emphasize the critical role of health policy alignment with universal health coverage objectives, robust pharmacoeconomic evaluations, and evidence-based national cancer control plans. Integrating Alma Ata’s principles into contemporary oncology provides a viable, scalable model to advance equitable, accessible, and sustainable cancer care globally, laying the theoretical groundwork for future research initiatives and informed policy development.
Plain Language Summary
Why was this study done? Despite many medical advances, cancer care remains very unequal globally. Many people worldwide still have poor access to cancer services because of high treatment costs, lack of trained medical staff, uneven healthcare resources, and weak healthcare systems. This work explores how the primary healthcare principles set by the Alma Ata Declaration—a major global health agreement from 1978 aimed at ensuring accessible health care for all—could help improve cancer care today. What did the researchers do? We reviewed challenges faced by current cancer care systems, including poor prevention, unequal access to care, high treatment costs, shortages of specialist healthcare workers, and limited community involvement. We then proposed a way to improve these issues by applying the Alma Ata principles—such as universal access, prevention-focused healthcare, and community involvement—to cancer care. We also highlighted successful real-world examples where these ideas have improved cancer services in different countries. What did the researchers find? Strengthening primary healthcare could significantly improve cancer outcomes by helping to detect cancers earlier, preventing many cases, reducing healthcare costs, and making care fairer for everyone, no matter their location or income. Involving local communities directly in healthcare planning and creating a balanced system combining specialized centers and local health services could benefit patients greatly. What do the findings mean? Applying the Alma Ata primary healthcare approach to cancer care can make it more accessible, affordable, and effective worldwide. This provides a clear roadmap for researchers, healthcare leaders, and policymakers seeking fairer and better cancer care for all people.
Keywords
Introduction
The Alma Ata Conference Declaration (AACD), issued in 1978 during the International Conference on Primary Health Care co-hosted by WHO and UNICEF, revolutionized global health care by establishing primary health care (PHC) as essential and health as a fundamental human right. 1 Prior to this landmark conference, access to essential health care services was severely limited, especially in rural regions and among impoverished urban populations. Health systems were primarily reactive, disease-oriented, and heavily centralized in urban hospitals, which perpetuated significant health disparities. 2 The AACD introduced a transformative vision emphasizing PHC as a cornerstone of health systems, advocating health equity, universal accessibility, community participation, and a holistic approach that explicitly addresses social determinants of health. 2
Following the declaration, global health strategies increasingly embraced community-oriented approaches, focusing on preventive care and intersectoral collaboration. In Low and Middle-Income Countries (LMICs), these strategies substantially improved healthcare access despite persistent economic and logistical challenges. 3 The AACD’s call for universal health care (UHC) catalyzed national and international efforts to reduce health disparities and enhance access to health care for underserved populations. However, fully realizing the Alma Ata goals has been hindered by economic constraints, emerging health crises, and geopolitical changes. 4 In 2018, the Astana Declaration marked the 40th anniversary of Alma Ata, reaffirming the enduring importance of PHC and renewing the global commitment to UHC, emphasizing AACD’s ongoing relevance to contemporary global health policy. 4
Within oncology, the core principles outlined by the AACD remain relevant. Despite remarkable advancements in cancer diagnosis and therapeutics, contemporary oncology confronts persistent challenges including significant disparities in access to care, high costs of innovative treatments, and an overwhelming focus on advanced therapies often inaccessible to those most in need. These issues frequently overshadow vital preventive strategies.5–7 Additionally, the centralization of oncology services within specialized urban hospitals continues to widen gaps in both access and quality, particularly affecting availability of novel therapies and clinical trial participation.8–10
This manuscript, structured as a narrative review and policy analysis, explores how integrating AACD’s principles into oncology can help reshape cancer care systems, advancing equity, strengthening preventive approaches, and enhancing PHC and community participation. We propose a conceptual framework (Figure 1) that aims to align the foundational AACD’s principles—health equity, universal accessibility, preventive care, and community participation—with contemporary oncology practice. Conceptual Framework Integrating Alma-Ata Principles Into Oncology. The Model Links Primary Healthcare, Specialized Oncology Services, and Community-Based Support, Backed by Enabling Factors, to Deliver Equitable, Sustainable Patient-Centered Care Across the Cancer Continuum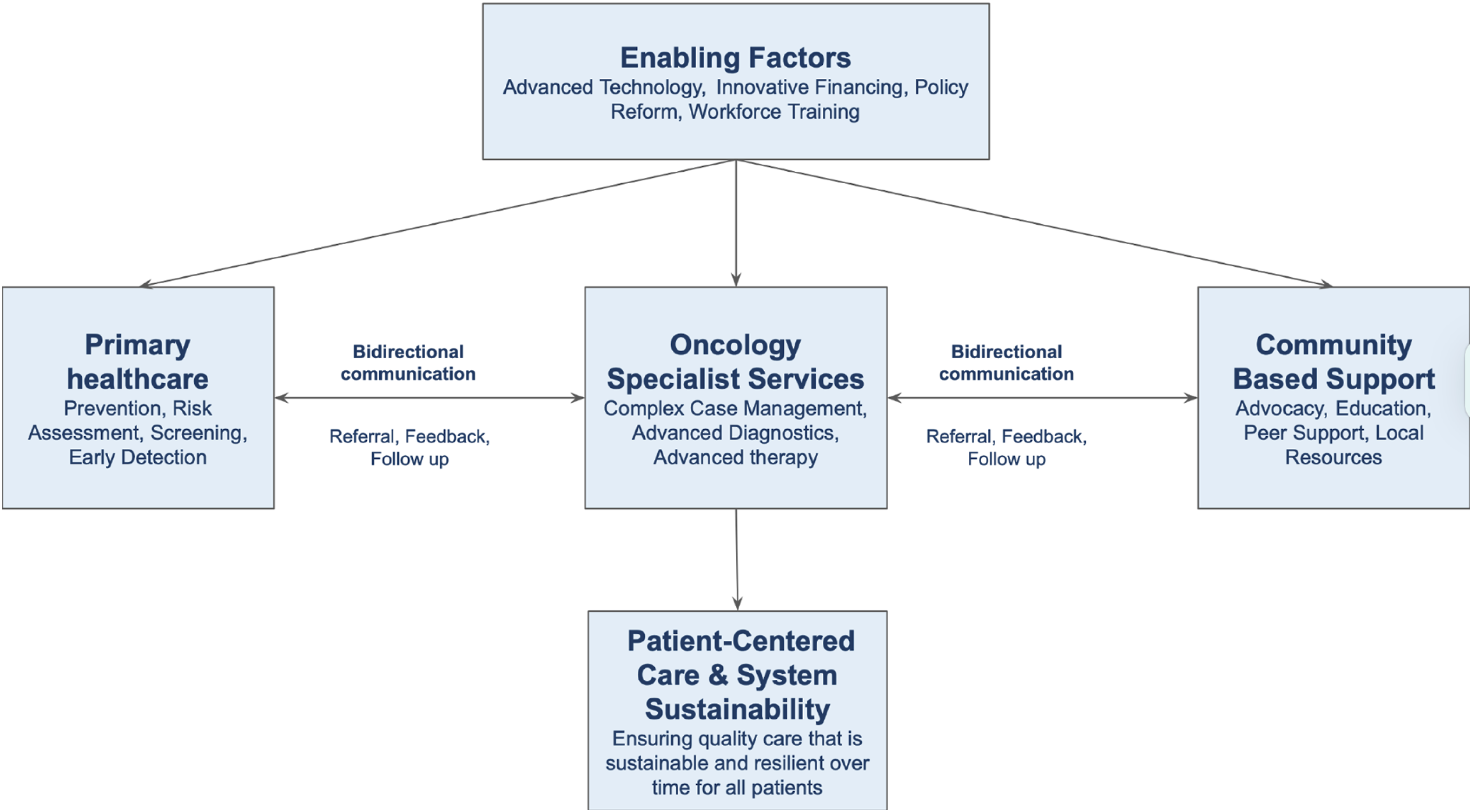
Current Challenges of Oncology Care
Cancer continues to be a significant public health challenge worldwide. As of 2022, the global cancer burden escalated to an estimated 20.3 million new cases and nearly 10.9 million cancer-related deaths, projected to reach 35 million cases by 2050 due to aging populations, lifestyle changes, and increased risk factor prevalence.
6
The responsibility for the treatment of these patients falls in the hands of modern oncology, a field that has made remarkable strides with the advent of precision medicine and novel therapies. The current landscape, though marked by these advancements, now confronts a complex array of critical challenges that threaten equitable access, sustainable financing, and truly patient-centered outcomes (Figure 2). • Centralization of Oncology Care: Centralizing oncology services within academic centers significantly hinders equitable access to advanced treatments, technologies, research, and clinical trials, particularly affecting populations in remote or socioeconomically disadvantaged areas.11–13 Conversely, decentralizing care could lead to suboptimal treatments, inefficient resource use, and a lack of specialized expertise.8,14 There is a critical need for research on effectively integrating both centralization and decentralization models. Existing studies on this are primarily conducted in high-income countries (HICs) and often fail to address population-specific needs at the country or health system level.8,10,14 • Focus on treatment rather than prevention: While a treatment-centric approach may cure non-metastatic disease, it is usually only marginally effective in advanced stages. This focus often overlooks the substantial benefits of early intervention and preventive measures, which include primary prevention through health promotion and secondary prevention via targeted screening for specific tumors.15–17 It is noteworthy that more than 50% of cancer cases could potentially be prevented.
14
Investing in prevention requires reallocating resources not only within oncology but across the entire healthcare system.14,18 Some estimates suggest that investing in public health for prevention could be significantly more cost-effective, potentially generating up to four times the quality-adjusted life years per unit of expenditure compared to traditional healthcare spending.
19
• Global Cancer Care Disparities: The global landscape of cancer care is marked by significant disparities, particularly in LMICs, where less than 25% of patients have access to safe, cost-effective, or timely surgical interventions.
5
Geographic and socioeconomic factors drastically impact outcomes, with over two million women annually facing survival risks from breast or cervical cancer due to limited access.5,20–22 In many regions, the absence of UHC further restricts access to cancer care, creating a global scenario where both location and financial means determine the level and quality of care.23–26 Even within high-income regions like Europe and countries themselves, coverage can vary between different geographic areas due to uneven resource allocation.
27
• Community Participation: Community engagement in cancer care remains inadequate in many settings, limiting the effectiveness and cultural relevance of cancer programs. This engagement operates in two directions, shaping health strategies by highlighting critical needs and perspectives, and assessing patient satisfaction and outcomes. Both facets are increasingly vital for responsive, effective care. Engaging communities and enhancing public awareness are essential steps towards a more inclusive and effective approach to cancer care.28,29 • Challenges in Sustainable Financing: Sustainable financing faces significant problems, particularly regarding inequity in resource allocation and the costs of cancer treatments.
7
In LMICs, underfunded and overstretched health systems hamper adequate care, while in HICs, rising therapy expenses lead to financial toxicity for patients, families, and the broader health system.30,31 Advanced therapies often come at prohibitive prices, and the pharmaceutical industry’s influence on escalating costs underscores the urgency for a pharmacoeconomic framework that ensures evidence-based, consistent decisions about which interventions to fund.7,30–32 Governments must also reflect on how heavily cancer therapy development relies on the pharmaceutical industry, as inadequate support for large-scale academic projects can limit the rational use of resources.33–35 • Shortage of Specialist Workforce: Oncology faces a critical global shortage of specialists, notably in LMICs, severely restricting effective cancer management. In regions from Africa and Asia, one oncologist may be responsible for over 1000 new cancer patients, reflecting significant disparities in workforce availability.
36
This deficit extends beyond physicians, including shortages in skilled nurses, technicians, anesthesiologists, palliative care specialists, and PHC professionals integral to multidisciplinary cancer care.5,37,38 • Gaps in PHC-Driven Cancer Care: A limited PHC infrastructure, hinders timely cancer detection, referral, and follow-up.39,40 PHC is recognized as a fundamental element of a health system, yet insufficient funding, personnel and organizational support have led to significant gaps in care delivery. This shortfall, along with insufficient communication channels among PHC, oncology, and supportive care teams, delays diagnosis and treatment initiation, ultimately affecting patient satisfaction and outcomes.29,41–43 This under-resourced and disjointed approach leads to suboptimal patient outcomes and increased burden on healthcare systems. • Government Strategies and Policy Challenges: Health policies frequently fail to match actual fiscal capacities, resulting in unrealistic objectives and ineffective resource allocation for cancer care.44,45 Decision-making often lacks transparent, evidence-based pharmacoeconomic frameworks.
27
Collaboration with academia and clinicians in policy development is usually insufficient, leading to health plans that do not fully mirror population needs. Unresolved tensions between privatization and public investment further complicate system resilience, equity, and viability.27,46,47 Current Challenges of Oncology Care

Integrating Alma Ata Principles into Oncology
Integrating the AACD principles into oncology requires a clear shift toward equity, accessibility, and community-oriented care. Effective community engagement, robust referral networks, and coordinated care pathways further support this integrated approach. This section discusses how such integration can be practically realized, aligning with the conceptual framework presented earlier (Figure 1), which explicitly links primary healthcare, specialized oncology services, and community-based support to deliver equitable, sustainable, and patient-centered cancer care.
Balancing Centralization and Decentralization in Cancer Care
Cancer care systems must strategically balance centralizing specialized services in high-volume centers with decentralizing care closer to communities. HICs countries have often pursued centralization for complex cancer surgeries and treatments due to evidence that higher case volumes may improve outcomes. For example, the Netherlands introduced minimum volume standards for pancreatic and esophageal cancer surgeries, significantly enhancing survival rates. 48 However, centralization increases travel distances, creating access barriers that disproportionately affect disadvantaged populations, as observed in the USA, potentially exacerbating geographic and socioeconomic inequities.10,49,50
Some LMICs, traditionally reliant on centralized services in major cities, are evaluating decentralization approaches such as the “hub-and-spoke” model. Kenya, for instance, decentralized oncology services by establishing regional cancer centers within provincial hospitals, significantly enhancing patient access in underserved rural communities and facilitating earlier diagnosis and treatment. 51 Similar initiatives in the Democratic Republic of Congo demonstrated that decentralization combined with local capacity-building effectively reduces morbidity and mortality by overcoming geographic barriers. 52 In Norway, illustrating successful decentralization in an HIC, the Orkdal model integrated oncology and palliative care at local hospitals, improving timely access and patient outcomes through decentralized, community-level service delivery. 53 Similarly, Italy’s Territorial Oncology Care program provided cancer treatment at community hospitals, significantly reducing patient travel burden and achieving high patient satisfaction by delivering care closer to home. 54
An optimal approach integrates both models through a tiered system: high-complexity treatments remain centralized to maintain quality, while preventive care, early detection, basic chemotherapy, and survivorship care are decentralized into PHC or community oncology, connected by robust referral mechanisms.48,51,52
Emphasis on Prevention and Early Detection
Preventive care is crucial in oncology to reduce cancer incidence and mortality. PHC teams are uniquely positioned to lead cancer prevention through lifestyle interventions, vaccination, and screening programs.55,56
In HICs, preventive initiatives integrated into PHC have demonstrated effectiveness. For example, structured lifestyle interventions in the USA and the UK have reduced risk factors like obesity and tobacco use.57,58 In LMICs, where resources for advanced care are limited, prevention is even more critical and often delivered through PHC and community programs. Tobacco control interventions integrated within PHC in India, using community health workers for education and cessation support, could reduce smoking rates, potentially lowering tobacco-related cancer incidence. 59 In South Africa, brief interventions for alcohol reduction embedded within PHC visits have effectively reduced excessive alcohol consumption. 60 In Rwanda, nationwide HPV vaccination implemented through PHC and school-based platforms achieved over 90% coverage rate. 61 Thailand’s integration of hepatitis B vaccination into routine PHC immunization programs decreased the prevalence of hepatitis B infection among young adults from 8%-10% to less than 1%, which may reduce liver cancer incidence over two decades. 62
Equity in Cancer Care
Achieving equity in cancer care involves ensuring universal access irrespective of geographic or socioeconomic status. The WHO emphasizes that strong PHC is key for health systems and equitable cancer outcomes. 63 For example, Brazil’s PHC reforms under the Family Health Strategy expanded access to underserved populations, costing only about $50 per person annually, reducing hospitalizations and mortality.64,65
In HICs, patient navigation programs have helped underserved groups overcome obstacles and receive timely cancer diagnoses and treatment, reducing outcome gaps. 66 The Boston Patient Navigation Research Program, effectively helped underserved groups overcome barriers to care. In this program, navigation was associated with a significantly higher likelihood of achieving diagnostic resolution. 67 A historical case from an underserved U.S. community (Harlem, New York) also showed that combining free screening with patient navigation, as a community-based support intervention, dramatically closed the survival gap for poor patients. Breast cancer 5-year survival in Harlem improved from 39% to 70% after introducing these measures. 68 In LMICs, equity can be promoted by bringing closer services to the community. Rwanda’s rural Butaro Cancer Center, for instance, brings chemotherapy, surgery, and palliative care to remote areas and even provides transport and lodging for patients who otherwise would lack access to care. 69
Community Involvement and Participation
AACD affirmed that people have both the right and duty to participate in planning and implementing health care. Community health workers (CHWs) and volunteers play crucial roles in LMICs by raising awareness, facilitating screenings, and providing follow-up care. A systematic review found CHWs in LMICs play an important role in community education, outreach for screening, and follow-up. 70 For example, a community-integrated cervical screening initiative in rural Uganda (the ASPIRE Mayuge trial) increased screening participation by deploying CHWs for door-to-door education and self-sampling outreach. 71 Another example comes from Kerala, India, where a community-driven palliative care network mobilized local volunteers alongside health professionals to care for cancer patients at home. Over 15 000 trained volunteers, with support from nurses and doctors, now help provide home-based care in all districts of Kerala, covering more than 60% of patients in need.72,73
HICs are also embracing community involvement. For instance, community-based participatory research (CBPR) can engage local stakeholders in designing and evaluating cancer prevention and intervention programs, enhancing their cultural relevance and effectiveness. A recent study utilized CBPR approaches to adapt educational materials for cancer screening among Native American communities, resulting in culturally tailored resources. 74 Additionally, partnerships between healthcare systems and community organizations can successfully tailor cancer awareness campaigns for different populations. 75 Church–academic partnerships with African American congregations have successfully delivered tailored cancer screening programs to help reduce disparities. 76
Global Cancer Care Disparities
Global cancer care disparities remain profound, with vastly different survival outcomes between HICs and LMICs, illustrated by breast cancer survival rates exceeding 89% in the United States but below 66% in India and other LMICs.21,77,78 Similar disparities exist across other malignancies, highlighting a persistent gap in cancer care between high-resource settings and LMICs or disadvantaged populations within wealthier nations.79–81
Central to these disparities is uneven access to essential cancer prevention, screening, and diagnostic services. For instance, over 40% of HICs report moderate-to-high cervical cancer screening coverage, while fewer than 4% of low-income countries achieve even 50% coverage. 82 Limited infrastructure, including pathology, radiology, and specialist surgical facilities, further exacerbates late-stage presentations, significantly worsening outcomes.36,83
Strengthening PHC is essential to mitigating these disparities. Regular engagement with PHC prior to cancer diagnosis significantly reduces the likelihood of metastatic disease and lowers cancer-specific mortality, as demonstrated in the U.S. Veterans Affairs system. 84 Complementing PHC, UHC ensures equitable access to essential services without financial hardship, making it crucial for sustainable cancer control, particularly in resource-constrained settings. Integrating cancer services into PHC under UHC frameworks facilitates proactive management throughout the cancer continuum, from prevention and early detection to survivorship and palliative care.82,85
Challenges in Sustainable Financing
Cancer care faces financial challenges driven by rapidly escalating treatment costs and rising global cancer incidence. Between 2020 and 2050, global cancer care expenses are projected to reach approximately $25.2 trillion. 86 This economic burden creates severe financial hardship for both patients and healthcare systems. Over half of cancer patients globally (56.1%) experience catastrophic health expenditures, often resulting in severe debt, asset loss, or sacrificing basic needs. 87
The situation is especially dire in LMICs, where the absence of UHC and reliance on out-of-pocket payments place immense strain on patients, often causing delays or abandonment of treatments.87–89 Even in HICs that have no UHC, the rising costs of modern targeted therapies and immunotherapies significantly increase financial distress among patients. A U.S. study reported nearly 20% of patients prescribed oral anticancer agents abandoning or delaying treatment due to high out-of-pocket expenses. 90 These financial pressures broadly impact healthcare systems, limiting their capacity for sustainable cancer care.91–93 Health systems often manage competing health priorities, making high-cost cancer treatments particularly challenging.
Addressing this financial toxicity requires multi-level initiatives. 93 At the individual level, measures such as financial counseling, patient navigation, and medication assistance programs are critical. 94 Community-level interventions include advocacy and fundraising initiatives supporting vulnerable patients. 95 Nationally, governments should integrate cancer treatments into UHC frameworks, regulate drug prices, and promote cost-effective generics and biosimilars. 93 India’s Ayushman Bharat scheme, for instance, expanded cancer care coverage to reduce patient expenditures. 96 Globally, multilateral agencies and donors should champion pooled procurement frameworks and solidarity funds to alleviate financial burdens in low-resource settings. The Access to Oncology Medicines (ATOM) Coalition, led by the Union for International Cancer Control (UICC), exemplifies this approach by pooling resources to negotiate better procurement terms for essential cancer medicines in LMICs.97,98
Shortage of Specialist Workforce
There is a widespread shortage of oncology workforce, with stark contrasts between HICs and LMICs. 36 The number of cancer doctors in African nations ranges from zero in some countries to only a few hundred in others, meaning many patients never see an oncologist. 36 By contrast, HICs have more specialists but still face significant shortfalls as cancer cases rise. The USA anticipates a shortage of over 2300 oncologists by 2025, 99 while the UK similarly projects a severe shortage of clinical oncologists. 100 Rural and underserved areas within well-resourced nations also struggle significantly to attract and retain oncology professionals. 101 In South Africa, the shortage of specialists is particularly severe in rural public hospitals. 102
Addressing this challenge requires innovative strategies such as task-sharing and task-shifting, where non-specialist healthcare providers undertake oncology roles. Models include training PHC doctors, clinical officers, and nurses in essential oncology skills, enabling them to manage aspects of cancer care. 36 For instance, Rwanda successfully trained general practitioners to deliver chemotherapy and manage common cancers. 69 Nurse-led clinics have expanded oncology access, enabling nurses to independently manage follow-up care, symptoms, and palliative care. 103 In Bangladesh, oncology nursing training was crucial for establishing a bone marrow transplant unit. 104
Gaps in PHC–Driven Cancer Care
Although PHC plays a critical role across the cancer care continuum—from prevention and early detection to survivorship and palliative support—it is often underutilized.55,56,105,106 Common challenges include inadequate oncology training among PHC providers, limited cancer education in medical and nursing curricula, and insufficient referral systems linking PHC with specialized oncology services. 107 These shortcomings result in delayed diagnosis, suboptimal management, and poorer patient outcomes.55,56 In Botswana, for instance, patients experienced median delays of 160 days from initial PHC consultation to specialist cancer treatment. 108
This underuse creates gaps in continuity of care: once patients are referred to specialist services, PHC is frequently sidelined, causing fragmented follow-up and lost opportunities for ongoing support. 107 Key underdeveloped areas in PHC include palliative care and survivorship. In Saudi Arabia, a pilot initiative trained family physicians in basic palliative care, establishing dedicated clinic days at PHC centers. This approach resulted in earlier interventions, improved symptom control, and 90% patient satisfaction. 109 In Canada, the INTEGRATE Project demonstrated that integrating palliative care into primary care practices increased providers’ confidence and facilitated earlier advance care planning for cancer patients, addressing gaps in continuity of care and coordination between primary and specialized oncology services. 110 Survivorship care remains rarely integrated into PHC; even advanced PHC practices in the U.S. often lack formal survivorship programs due to unclear roles, limited information exchange, and inadequate health IT support. 111
Bridging these gaps requires strategies aligned with AACD’s principles of integrated, people-centered care, including strengthened bidirectional referral systems, clear post-treatment transition plans, improved oncology training for PHC teams, and engaged community support networks.112,113 Health systems with robust PHC foundations demonstrate better outcomes and lower costs, even for complex diseases like cancer.56,114,115
Ideally, this means patients receive timely, competent evaluations locally, rapid referral for specialized treatment, and effective follow-up and palliative support at home—a continuum of care anchored in PHC. Such a model aligns with the AACD vision, placing revitalized PHC at the core of equitable, integrated, and people-centered cancer care systems.2,116
Government Strategies and Policy Challenges
The integration of AACD principles into oncology ultimately depends on effective health policies. Governments play a decisive role in shaping equitable cancer care through national planning, resource allocation, health insurance design, and regulatory frameworks. Recently, there has been growing policy recognition of the need to prioritize cancer alongside other non-communicable diseases (NCDs). By 2017, the World Health Assembly urged member states to develop or update National Cancer Control Plans (NCCPs) as part of the NCD agenda,117,118 resulting in more than 120 countries implementing NCCPs. 118 These plans typically address prevention, early detection, treatment, palliative care, and research.
However, having a plan does not always translate into effective implementation. Analyses reveal critical gaps in many NCCPs. In LMICs, inadequate financial planning is common, with fewer than 30% specifying costs or resource projections, despite frequent references to health workforce strategies. In HICs, common shortcomings include insufficient use of evidence-based strategies, limited alignment with global health initiatives, and low engagement with patient stakeholders, as only 18% of NCCPs involve cancer survivors in planning. 118
Policy implementation also varies widely. Thailand provides a notable success story: it achieved UHC in 2002 and integrated cancer care into this system in 2005. By investing in preventive measures, such as tobacco and alcohol control via taxation, and expanding treatment progressively, Thailand aligned its cancer plan with available resources, significantly improving access. 119 Similarly, South Korea’s 10-Year Cancer Control Plan increased cancer screening rates and improved 5-year survival rates through UHC integration. 119 Japan introduced systematic cost-effectiveness evaluations to manage oncology costs, illustrating adaptable sustainability strategies. 119
Nevertheless, significant challenges remain in many regions. In Nigeria, oncology services are severely under-resourced, with shortages of specialized cancer services and trained professionals, causing patients to often receive care in general wards under suboptimal conditions. 120 Policy experts in Nigeria emphasize expanding UHC to cover cancer care as crucial. 121 India, despite long standing NCCPs, experiences substantial disparities between states and healthcare sectors. Public hospitals face overwhelming demand, resulting in treatment delays averaging two months post-diagnosis, while private facilities offer high-quality care primarily to affluent populations, highlighting inequitable access. 96 Some governments fund these initiatives through taxes or cross-subsidies (eg, tobacco/alcohol levies). 122 Kenya, for example, established public-private partnerships to develop radiotherapy centers and diagnostic services, complementing public resources. 123
Robust regulatory and pharmacoeconomic frameworks are essential for global cancer control policies. Health technology assessment (HTA) guides decisions on the adoption, coverage, and pricing of interventions. 124 In HICs, agencies like the Food and Drug Administration (FDA) and European Medicines Agency (EMA) approve new cancer drugs, while entities like the UK’s National Institute for Health and Care Excellence (NICE) conduct HTAs to determine therapy coverage, influencing the availability and pricing of innovations.124,125 Often, novel drugs are initially not cost-effective at list price, prompting payers to demand price reductions or conditional reimbursement until sufficient data emerge.125,126 China applied HTA before adding immunotherapy drugs to its national formulary, achieving price cuts of nearly 80%. 127 Trastuzumab’s high cost delayed its uptake in LMICs, with fewer than 10% including it in their formularies until lower-cost biosimilars became available later. 128
Practical measures and examples.

Political and Institutional Resistance
Some institutions, such as national cancer hospitals or specialty networks, may resist reforms perceived to dilute their resources or authority. Limited budgets and infrastructure at the PHC level further impede its integration into cancer care. 130 In practice, these dynamics mean that shifting cancer care to the community requires overcoming vested interests and reallocating resources against the status quo. High-level political commitment is often lacking, as policymakers juggle competing health priorities and may be influenced by entrenched hospital-based lobbies. The result is a slower, uneven uptake of programs in community settings, despite evidence that closer-to-home care could improve access and equity. 130
Institutional inertia and professional silos similarly pose challenges to oncology–PHC integration and further advancing AACD principles. Cancer care has traditionally been the domain of specialists, sometimes leading to a “capture and no release” model where patients stay under oncology follow-up in hospitals long after acute treatment. This culture can foster resistance to shared-care models. Oncologists may doubt primary care providers’ oncology expertise or be reluctant to relinquish control of survivorship care, while general practitioners often lack clear role definitions, training, and resources for cancer management.131,132 Systemic factors reinforce the divide: fee-for-service payment models and hospital budgets reward intensive, specialist-led interventions, creating little incentive to shift aspects of care to community clinics. Fragmented information systems exacerbate the problem – without seamless communication channels, hospital and primary teams struggle to coordinate. 131 Even well-resourced systems face “power dynamics” issues: tertiary centers and specialty providers hold financial and decision-making power, and reforms that empower PHC may encounter pushback within institutional hierarchies.
Conclusion
Achieving meaningful progress in global cancer care requires fully embracing the AACD principles of equity, accessibility, prevention, and community participation. By aligning cancer control strategies with these principles, policymakers and health leaders can create a more just and effective framework for cancer prevention and treatment. This approach calls for ensuring that all populations – regardless of socioeconomic status or geography – benefit equally from advances in cancer care.
133
The Astana Conference reaffirmed the enduring importance of the AACD,
4
yet further efforts are essential to fully integrate its valuable legacy into contemporary cancer control. To operationalize this vision in global oncology, the following health policy principles should be followed: • Ensure equity in cancer care: Prioritize implementing policies that allocate resources and services in a manner that prioritizes underserved populations. This includes investing in cancer facilities and workforce in low-resource areas and reducing financial barriers to care, so that the benefits of new treatments and early detection are equitably realized across all segments of society. • Improve accessibility through PHC integration: Integrate basic oncology services into PHC networks to bring cancer prevention, diagnosis, and treatment closer to where people live. This means strengthening health infrastructure and referral systems at the community and primary-care level, training frontline providers in essential oncology skills, and ensuring availability of vital medicines. • Prioritize prevention and early detection: Make prevention the cornerstone of global cancer control by expanding programs for vaccination, tobacco control, healthy lifestyle promotion, and environmental hazard reduction. Equally important is establishing widespread early detection and screening initiatives through primary care. • Foster community participation in cancer control: Actively involve communities in the planning, implementation, and evaluation of cancer care initiatives. This entails partnering with local leaders, community health workers, patient advocacy groups, and civil society organizations to ensure programs are widely accepted.
Adopting an approach rooted in these principles will guide our communities and nations toward stronger health systems and better cancer outcomes, ultimately bringing the world closer to the longstanding goal of “health for all” in the realm of cancer care.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Asia Ferrández Arias reports receiving travel, accommodations, and expenses from Pfizer. Álvaro Rodríguez-Lescure reports a consulting or advisory role for Roche, Pfizer, Novartis, MSD, AstraZeneca Spain, Daiichi-Sankyo, Seagen, Pierre Fabre, and Lilly; speakers’ bureau participation with Roche, Novartis, Lilly, Pfizer, AstraZeneca Spain, Daiichi-Sankyo, and Seagen; institutional research funding from Roche, Pfizer, Novartis, Lilly, Zymeworks, Bristol Myers Squibb, AstraZeneca Spain, and Radius Health; and travel, accommodations, and expenses from Roche and Pfizer. No other potential conflicts of interest were reported by the remaining authors.
