Abstract
Programmed cell death (PD-1) and programmed cell death ligand 1 (PD-L1) inhibitors have been increasingly used in cancer therapy. The aim of this study was conducted a meta-analysis to assess the efficacy and safety of PD-1/PD-L1 inhibitors in patients with unresectable hepatocellular carcinoma (uHCC). A total of 1657 patients were included. The completed phase III trials with details data, such as overall survival (OS), progression-free survival (PFS), objective response rate (ORR), disease control rate (DCR), and adverse effects (AEs) were included. The pooled hazard ratio (HR) of OS and PFS were .75 (95% CI: .61–.92) and .74 (95% CI: .56–.97) with heterogeneity between PD-1/PD-L1 inhibitors groups and control groups. Sensitivity analysis revealed IMbrave-150 could be the most important factor of heterogeneity for OS, while CheckMate-459 was the main fact of heterogeneity for PFS. In addition, the relative risk (RR) of ORR and DCR were 2.43 (95% CI: 1.80–3.26) and 1.26 (95% CI: 1.11–1.43) with low heterogeneity in PD-1/PD-L1 inhibitors groups. The therapeutic effect of PD-1/PD-L1 inhibitors was better in females, Asia without Japan, BCLC status C and infected hepatitis groups. The RR of AEs from any cause and serious adverse events (SAEs) for patients receiving PD-1/PD-L1 inhibitors were 1.03 (95% CI: .93–1.13) and 1.13 (95% CI: .89–1.44), respectively. Pruritus was the most common AEs reported in 10% of patients or more (RR = 1.69, 95% CI: 1.33–2.15). In conclusion, PD-L1 inhibitor combined with anti-VEGF antibody could improve the prognosis of patients with uHCC. However, caution should be taken for AEs during patients receiving PD-1/PD-L1 inhibitors.
Keywords
Introduction
Liver cancer is the sixth most common cancer and the fourth most common cause of cancer-related death worldwide. 1 Hepatocellular carcinoma (HCC) accounts for 80%–90% of liver cancer with global variations in the incidence and mortality. 2 Despite improved anti-viral treatments decreased the risk of de novo HCC development, the other risk factors such as fatty liver and alcohol remain significant burden of HCC. 3
To date, the curative or non-curative treatments of HCC included liver surgery, interventional radiology, liver transplantation, targeted therapy, and immunotherapy. 3 Palliative treatments for unresectable HCC (uHCC) remain complicated due to the lack of series of evidence-based medicine. Multi-kinase inhibitors, such as sorafenib and lenvatinib, have been approved for first-line systemic treatments of patients with uHCC. 4 However, kinase inhibitors have been shown to fail to improve overall survival (OS) or recurrence-free survival (RFS). 5 Over the past 2 years, application of immune-checkpoint inhibitors (ICIs) provided a new systemic therapy for patients with uHCC, especially for programmed cell death-1 (PD-1)/programmed cell death ligand 1 (PD-L1) inhibitors. 6
PD-1/PD-L1 inhibitors have been demonstrated to improve the prognosis of patients with advanced cancers in multiple phase II/III trials, such as melanoma, renal cell carcinoma, non-small cell lung cancer, and urothelial bladder cancer. 7 However, The efficacy of PD-1/PD-L1 inhibitors monotherapy or combination is variable for uHCC in phase II trials.8,9 For patients with uHCC, the improvements of OS in phase II and III trials were not complete coincident.10,11 In addition, only a minority of patients have durable response. 12 Furthermore, toxicities including immune-related adverse events (irAEs) are increasingly being reported, posing therapeutic challenges for clinicians. 7 To derive more comprehensive estimation of PD-1/PD-L1 inhibitors in uHCC, a systematic review and meta-analysis of phase III trials were performed.
Methods
Search Strategy
The current meta-analysis was conducted by reference to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). 13 The protocol was registered in International Platform of Registered Systematic Review and Meta-analysis Protocols (INPLASY) (NO. INPLASY202210067). Six databases, including PubMed, Embase, Cochrane Library, Web of Science, Scopus, and ClinicalTrials.gov, were searched from inception to May 11, 2020 for relevant publications using the following retrieval terms to define the therapy: “programmed cell death 1 receptor,” “PD1 receptor,” “programmed cell death 1 ligand 1,” “B7-H1 antigen,” “nivolumab,” “pembrolizumab,” “atezolizumab,” “durvalumab,” “cemiplimab,” and “avelumab.” The terms used to define the disease included “liver neoplasm,” “hepatocellular carcinoma,” “liver cancer,” and “HCC.”
Selection Criteria
Literatures met the following predetermined criteria were eligible for inclusion: (1) patients with uHCC received PD-1/PD-L1 inhibitors; (2) phase III randomized controlled studies (RCTs); (3) sufficient data of efficacy outcomes including OS, progression-free survival (PFS), objective response rate (ORR), disease control rate (DCR); and (4) data available on rates of adverse effects (AEs) from any cause, grade 3–4 AEs, or serious adverse events (SAEs).
Data Collection and Risk of Bias Assessment
The potentially relevant articles were screened independently by 2 investigators (WY and LYZ). Disagreements were resolved through discussion with a third reviewer (WQQ). Data from included studies were extracted by 2 co-authors (JWS and XHJ) independently. The data included first author, year of publication, trial name, study registration number, study size, study drugs, demographic and clinical information of the participants, outcomes of interest, and rates of any AEs.
Risk of bias in the included studies was evaluated with Jadad score by independent authors (LYZ and WQQ). 14
Statistical Analysis
The data of hazard ratio (HR) with 95% confidence intervals (CI) from each study was extracted. After logarithmic transformation, effect/CI was used to calculate pooled effect of HR by random effect models. The heterogeneity was evaluated by I-squared statistic. I-squared value greater than 50% suggested high heterogeneity. Subgroup analyses were further explored according to varied factors. Sensitivity analysis was used to estimate the influence of a single study on the overall results of the meta-analysis. ORR, DCR, and the rate of AEs were estimated by relative risk (RR). P value <.05 was considered statistically significant. All of these were performed by STATA version 14 (STATA Corp, College Station, TX, USA). Publication bias was evaluated by Deeks’ funnel plot.
Results
Search Results and Quality Assessment
Based on the search terminology and evaluation of other references, a total of 4479 records were retrieved. After reviewing the abstracts and full texts, 3 RCTs involving 1657 patients were included in the final analysis.10,11,15 The study selection process was shown in Figure 1. Flow diagram of studies selection.
Characteristics of Included 3 Phase III Trials.

HR, hazard ratio; 95% CI, 95% confidence intervals; OS, overall survival; PFS, progression-free survival; NR, no report; Ref., references.
The included studies reported complete outcome data. According to the Jadad score, the RCTs studies had good quality with scores in the range of 5 to 7.
Efficacy Analysis
For OS analysis, the pooled HR was .75 (95% CI: .61–.92; I-squared =52.8%, P=.006) (Figure 2A), indicating the test for overall effect had significant statistical difference with moderate heterogeneity between PD-1/PD-L1 inhibitors groups and control groups. The pooled HR for PFS was .74 (95% CI: .56–.97; I-squared = 80.0%, P = .007), implying significant heterogeneity existed in these data (Figure 2B). Forest plots of pooled overall survival (OS), progression-free survival (PFS), objective response rate (ORR), and disease control rate (DCR). (a) OS; (b) PFS; (c) ORR; (d) DCR.
The data related to ORR were conducted from 3 RCTs and DCR were reported in 2 RCTs. The RR of ORR and DCR were 2.43 (95% CI: 1.80–3.26; I-squared =.0%, P=.000) (Figure 2C) and 1.26 (95% CI: 1.11–1.43; I-squared =13.7%, P = .000) (Figure 2D). Statistical significance improvement of ORR and DCR was noticed in PD-1/PD-L1 inhibitors groups with low heterogeneity.
Sensitivity Analysis
For OS, the data of I-squared were significantly changed after deleting IMbrave150, while for PFS, I-squared changed significantly after deleting CheckMate-459 (Supplementary Figure 1). This result signified IMbrave-150 might be the most important cause of heterogeneity for OS and CheckMate-459 might be the most important cause of heterogeneity for PFS.
Subgroup Analysis
Subgroup analysis of OS and PFS were performed according to sex, geographic region, Barcelona Clinic Liver Cancer (BCLC) status, and viral status. As shown in Figure 3, the OS and PFS of uHCC patients receiving treatment with PD-1/PD-L1 inhibitors were substantially higher than placebo or sorafenib. Females (OS: HR: .56, 95% CI: .25–1.25, P = .156; PFS: HR: .60, 95% CI: .40–.89, P = .012) derived better survival benefit than males (OS: HR: .72, 95% CI: .58–.89, P = .002; PFS: HR: .66, 95% CI: .53–.83, P = .000). In addition, patients came from Asia without Japan (OS: HR: .56, 95% CI: .40–.79, P = .001; PFS: HR: .49, 95% CI: .36–.66, P = .000) gained the best survival benefit in all subgroups. It is of note that uHCC patients received PD-1/PD-L1 inhibitors in BCLC status C had better survival outcomes than BCLC status B. Although there was no difference between patients with hepatitis B virus (HBV) (OS: HR: .54, 95% CI: .38–.75; PFS: HR: .57, 95% CI: .38–.82) and hepatitis C virus (HCV) infected liver cancer (OS: HR: .64, 95% CI: .29–1.41; PFS: HR: .58, 95% CI: .38–.89), the OS and PFS in virus subgroup were better than patients with non-viral liver cancer (OS: HR: .89, 95% CI: .67–1.17; PFS: HR: .74, 95% CI: .58–.94). Subgroup analysis of overall survival (OS) and progression-free survival (PFS). (a) OS; (b) PFS.
Safety
The therapy with PD-1/PD-L1 inhibitors resulted in a slightly higher risk for all-grade AEs. RR of AEs from any cause, AEs in Grade 3-4 severity, and SAEs were 1.03 (95% CI: .93–1.13), 1.06 (95% CI: .93–1.21), and 1.13 (95% CI: .89–1.44), respectively.
Adverse Events in as-Treated Patients.
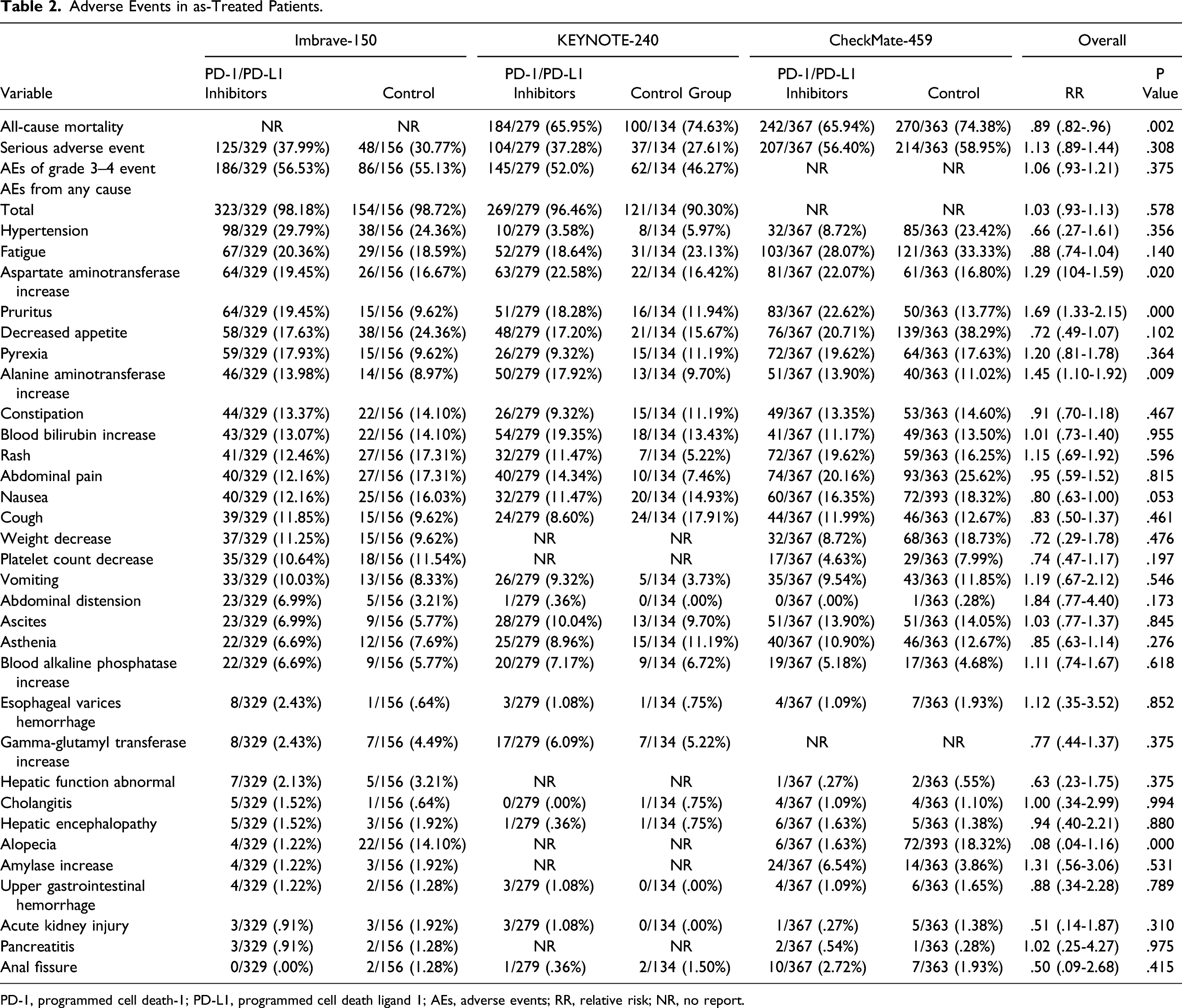
PD-1, programmed cell death-1; PD-L1, programmed cell death ligand 1; AEs, adverse events; RR, relative risk; NR, no report.
Publication Bias
Due to the small number of studies in our meta-analysis, the publication bias was only assessed by funnel plot asymmetry (Supplementary Figure 2). There was no potential publication bias in this study.
Discussion
Current recommendations for the optimal decisions of HCC were based on multidisciplinary expertise. The quick advancement of systemic therapy and immunotherapy gave the hope to patients with uHCC. 6 Previous study reported systemic treatment with sorafenib could improve OS compared to no treatment. 16 Recently, ICIs have demonstrated to be the important second-line treatment options after the intolerance to first-line agents (sorafenib and lenvatinib) or disease progression. 6 Although the results of PD-1/PD-L1 inhibitors were promising, there are patients that are not benefitting from these treatments due to specific characteristics of individual patients. 17 Therefore, the present meta-analysis was to evaluate the efficacy and safety of PD-1/PD-L1 inhibitors in adjuvant setting for uHCC based on current phase III trials data.
The high-quality data indicated the 25% lower hazard of death and a corresponding 24% decrease in the hazard of disease progression or death with heterogeneity were observed in PD-1/PD-L1 inhibitors groups. IMbrave-150 could be the most important factor of heterogeneity for OS, while CheckMate-459 was the main fact of heterogeneity for PFS. The statistical significance improvements of ORR and DCR were observed in PD-1/PD-L1 inhibitors groups with low heterogeneity. The RR of AEs from any cause and SAEs were 1.03 (95% CI: .93–1.13) and 1.13 (95% CI: .89–1.44), respectively, with a significant difference.
Overall survival and PFS, as the primary endpoint, remained the important indicators for evaluating therapeutic effect and prognosis in most phase III trials for HCC. It is of note that KEYNOTE-240 and CheckMate-459 did not meet the statistical dual end points for improvements of OS and PFS with PD-1 inhibitors.10,11 In addition, the HR of PFS was .93 (95% CI: .79–1.10) in CheckMate-459 and .72 (95% CI: .47–.76) in KEYNOTE-240, which are higher than the threshold of .6. 18 Possible reasons for these results included significant heterogeneity, complex pathogeny, and the effect of other drugs on OS after disease progression. Therefore, these results for supporting PD-1 inhibitors as sufficient treatment are pending. Surprisingly, Imbrave-150 revealed PD-L1 inhibitor (atezolizumab) combined with anti-VEGF antibody (bevacizumab) contributed to better benefit of OS and PFS than sorafenib in patients with uHCC. 15 This may be the main reason for the heterogeneity of OS.
To our knowledge, multiples of immune tolerance pathways were involved in HCC microenvironment. 19 Previous studies have demonstrated the combination therapy could result in a greater benefit than monotherapy.15,20 Preclinical studies suggested that the combination of PD-1 inhibitors and anti-VEGF antibody improved tumor T-cell infiltration. 21 Results from phase III trials of atezolizumab combined with bevacizumab for renal cell carcinoma, non-squamous non-small cell lung cancer, and uHCC supported the basic researches.10,11,15,22,23 However, there was no published phase III trials data related to PD-1/PD-L1 inhibitors combined with locoregional therapies, such as transarterial chemoembolization or radiofrequency ablation. In addition, data on comparisons among different PD-1/PD-L1 inhibitors combinations remain limited. Together, more prospective, randomized studies on the best managements of HCC are needed to explore in the future.
In the subgroup analysis, PD-1/PD-L1 inhibitors brought benefit to survival for patients came from Asia without Japan, with 44% lowest hazard of death and a corresponding 51% decrease in the hazard of disease progression or death. Moreover, patients with uHCC in females, BCLC status C and infected hepatitis groups showed better OS and PFS than males, BCLC status B and non-viral liver cancer groups, respectively. Notably, upregulation of PD-1 in HBV/HCV specific T cells and PD-L1 in HBV/HCV infected hepatocytes were observed during virus infection, supporting the inclusion of PD-1/PD-L1 inhibitors towards functional cure of HCC with chronic hepatitis virus infections. 24
A meta-analysis revealed the incidence of pneumonitis was significantly higher among patients receiving PD-1 inhibitors compared with PD-L1 inhibitors (3.6% vs 1.3%). 25 Grade 3 or higher AEs occurred in approximately 10% of patients receiving PD-1 inhibitors. 7 In the present study, the most common AEs reported in 10% of patients or more were pruritus. Remarkably, compared with anti-cytotoxic T lymphocyte antigen 4 (CTLA-4) antibodies, toxicities owing to PD-1/PD-L1 inhibitors could take a long time to resolve.7,26 Thus, the choice of interventions should strike a balance between efficacy improvement and toxicity.
Our meta-analysis has several limitations as well. First, the number of phase III trials for uHCC treatments with PD-1/PD-L1 inhibitors were limited, especially for PD-L1 inhibitors. Second, the including 3 phase III trials were supported by companies, which could be a possible source of bias. Third, the studies of CheckMate-459 published with abstract and results only on ClinicalTrials.gov, resulting in insufficient relevant information, such as lack of P value of PFS. These may affect further interpretation of the results. Moving forward, ongoing studies are needed to explore the optimal treatment strategy for uHCC patients who might benefit from monotherapy or combination PD-1/PD-L1 inhibitors treatments.
Conclusions
In conclusion, treatment with PD-1/PD-L1 inhibitors for uHCC patients was associated with better OS, PFS, ORR, and DCR than sorafenib and placebo, especially for PD-L1 inhibitor combined with anti-VEGF antibody with statistical significance. However, the incidence of AEs in uHCC patients receiving PD-1/PD-L1 inhibitors were slightly higher. Therefore, the safety of PD-1/PD-L1 inhibitors in uHCC patients remains necessary to estimate in future clinical studies.
Supplemental Material
Supplemental material - Survival Outcomes and Safety of Programmed Cell Death/Programmed Cell Death Ligand 1 Inhibitors for Unresectable Hepatocellular Carcinoma: Result From Phase III Trials
Supplemental material for Survival Outcomes and Safety of Programmed Cell Death/Programmed Cell Death Ligand 1 Inhibitors for Unresectable Hepatocellular Carcinoma: Result From Phase III Trials by Linyan Zeng, Junwei Su, Wenqi Qiu, Xuehang Jin, Yunqing Qiu and Wei Yu in Cancer Control.
Footnotes
Author Contributions
The work presented here was carried out in collaboration between all authors. WY and YQQ developed the concept and designed the study. LYZ and WY carried out the literatures research and studies selection. JWS and XHJ co-worked on associated data collection. The qualities of included studies were carried out by LYZ and YQQ. Disagreements were resolved by WQQ. Data synthesis and analysis were carried out by LYZ and WY. The manuscript was written by LYZ and corrected by YQQ and WY. All authors discussed the results and implications and commented on the manuscript at all stages.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Public Welfare Technology Research Project of Zhejiang Province in China (Project No: LGF18H310002).
Consent for Publication
All authors have seen and approved the content and fulfill the journal’s requirements for authorship.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
