Abstract
Introduction
Gastric cancer remains an important disease worldwide. It is the fifth most commonly diagnosed malignancy and third most common cause of cancer-related deaths. 1 Up to 65% of gastric cancer cases are diagnosed at locally advanced or advanced stages and have poor prognosis. 2 Age is one of the biggest risk factors for cancer, and the incidence of most solid organ tumors increases with age. Older age is also associated with changes in the pharmacokinetics and pharmacodynamics of cancer therapy, and increased susceptibility to toxic complications. 3
In a study comparing surgery with epirubicin, cisplatin, and infused 5-fluorouracil (ECF) regimens administered perioperatively to patients with potentially operable gastric cancer, progression-free survival and overall survival (OS) were found to be significantly better in patients receiving perioperative ECF, and the ECF regimen was used in perioperative treatment for many years. 4 In another study, ECF was compared with FLOT (5-Fluorouracil, leucovorin, oxaliplatin, and docetaxel). The OS was significantly longer in the FLOT arm. After this study, FLOT regimen became the standard in neoadjuvant treatment of gastric cancer. In these studies, the FLOT regimen was shown to be effective and tolerable in geriatric patients aged ≥70 years, accounting for 24% of all patient groups. 5 National Comprehensive Cancer Network (NCCN) and European Society for Medical Oncology (ESMO) guidelines recommend neoadjuvant chemotherapy for locally advanced non-metastatic eligible patients with adequate organ function.6,7
The immune system is extremely important in the treatment of malignant tumors. Serum albumin and lymphocyte levels are important indicators of immune function. The prognostic nutritional index (PNI) is an index that uses serum albumin and peripheral blood lymphocyte count to predict the likelihood of postoperative complications by assessing the preoperative nutritional status of patients. 8 Recent studies have demonstrated the high accuracy of PNI in predicting treatment outcomes for various cancers, particularly cancers of the gastrointestinal tract.9,10 Park et al investigated the prognostic effect of PNI in stage 2 and 3 gastric cancer patients. They revealed that higher preoperative PNI scores were associated with better prognosis. Patients with a high PNI preoperatively who maintain a high PNI postoperatively have better outcomes compared to those whose PNI decreases. Conversely, patients with a low preoperative PNI who improve their PNI post-surgery have a better prognosis than those who remain with a low PNI. 9 The PNI, which reflects nutritional and immune status, is crucial for predicting the risk of surgery and the outcome of gastric cancer treatment. Liu et al. found that survival rates were significantly better in the high PNI group compared to low PNI. This finding indicating that a higher PNI is associated with better survival outcomes after surgery. 10 To sum up, a higher PNI is linked to better nutritional status and immunity, which are essential for successful recovery and prolonged survival after surgery.
The traditional markers of inflammation, neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR), have also been used to assess the prognosis of patients with gastric cancer. 11 In a study by Zhao et al. in patients with metastatic gastric cancer, patients with lower NLR and PLR values showed significantly better survival rates. They suggested that high NLR and PLR levels may contribute to adverse anti-tumor functions, thereby worsening prognosis. 11
There are still no reliable indicators to predict tumor response and prognosis of patients before chemotherapy, and ultimately to optimize the treatment strategy. Several biomarkers have been derived from complete blood counts and routine biochemistry. However, the prognostic value of these biomarkers in elderly patients remains unclear, as most studies on these biomarkers have been performed on young individuals, and very few studies have included only the elderly individuals.
The aim of our study was to investigate the value of pretreatment PNI, serum albumin, total lymphocyte count, NLR, and PLR in predicting response to treatment in elderly gastric cancer patients treated with FLOT regimen as neoadjuvant chemotherapy.
Materials and Methods
Study Population
The present study was a single-center, retrospective study and includes geriatric patients with gastric cancer who received neoadjuvant chemotherapy between 2016 and 2024 at Van Yüzüncü Yıl University Medical Faculty Hospital. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). Ethical approval was obtained from the Ethics Committee of Van Yüzüncü Yıl University Medical Faculty (No. 2024/02-08). The requirement for informed consent was waived due to the retrospective nature of the study. The inclusion criteria for our study are as follows: (I) Age ≥65-year-old, (II) cytological or histological proven gastric carcinoma, (III) clinical stage two and three disease, (IV) received at least one cycle of chemotherapy, and (V) received FLOT regimen as chemotherapy regimen. Those who did not fulfil these criteria and whose file data could not be accessed were excluded from the study. The participant inclusion process is illustrated in Figure 1. The International Union for the Control of Cancer (UICC) tumor-node-metastasis (TNM) classification was used for the classification of clinicopathological factors.
12
After the diagnosis of gastric cancer in all patients, the decision to administer neoadjuvant treatment was made by a multidisciplinary council that included related departments. FLOT neoadjuvant therapy; Docetaxel (60 mg/m2), oxaliplatin (85 mg/m2), leucovorin (200 mg/m2), and 5-fluorouracil (2600 mg/m2, via a port catheter, continuous infusion over 24 hours), all administered on day one, once every two weeks. All patients were treated surgically two to four weeks after the end of chemotherapy. The reporting of this study conforms to REMARK guidelines.
13
Flow Chart of Included Patients.
Evaluation
Thoraco-abdominal contrast-enhanced computed tomography (CT) or fluorodeoxyglucose positron emission tomography/enhanced tomography (FDG PET-CT) was performed before the start of neoadjuvant treatment and every eight weeks during treatment, and radiological response was evaluated according to the Response Evaluation Criteria in Solid Tumors (RECIST) criteria. 14 The pathological response evaluation of patients who underwent surgery after neoadjuvant treatment was performed according to the Tumor Regression Grade Becker criteria as follows; Grade-1, complete (0% residual tumor; Grade-1a) or subtotal tumor regression (<10% residual tumor per tumor bed; Grade-1b); Grade-2, partial tumor regression (10-50% residual tumor per tumor bed), and Grade-3, minimal or no tumor regression (50% residual tumor per tumor bed). 15 Laparoscopy for staging was not performed on patients. Disease staging was performed using imaging modalities (CT or FDG PET-CT). Patients were divided into two groups as treatment-responsive group (grade-1 and grade-2) and treatment-nonresponsive group (grade-3).
Definitions and Follow-Up
Data including demographic characteristics, tumor localization, complete blood count, serum albumin level (g/dL), other biochemical tests, serum tumor markers (CEA and CA19-9), comorbidities (diabetes mellitus, hypertension, ischemic heart disease, cerebrovascular disease, chronic renal failure, congestive heart failure, chronic obstructive lung disease, and others), and other clinicopathological parameters, were recorded from all patients’ medical files before chemotherapy. The blood samples of the patients were analyzed in the laboratory at the time the blood was taken. These blood samples are routinely analyzed before treatment in all patients. PNI = 10 × albumin g/dl + 0.005 × total lymphocyte mm3, neutrophil/lymphocyte and platelet/lymphocyte ratios were calculated. The variables used to calculate the PNI, NLR, and PLR were obtained from blood analyses before treatment and before receiving any medical support (e.g. nutritional support). Median follow-up time was 28.7 months (12.3-48.1). The OS was calculated as the time to death or last follow-up. Disease-free survival (DFS) was defined as the period from the initiation of neoadjuvant treatment to disease local recurrence or distant metastasis. In our study, there were 2 patients with stage 1 disease. As these patients were clinically T2 N0, these patients received neoadjuvant treatment.
Statistical Analysis
The primary outcome measure was the difference in relapse rate between the treatment-responsive and treatment-nonresponsive groups. The standard effect size was set at 0.88 with a 5% margin of error (95% confidence interval) and 80% power. A minimum of 20 cases was planned to be included in the groups. The mean, standard deviation, median minimum, maximum, frequency, and ratio values were used for the descriptive statistics of the data. The distribution of variables was measured using the Kolmogorov-Smirnov test. The effect level was analyzed by univariate and multivariate logistic regression. Kaplan-Meier survival analysis was used. Independent sample t-tests and Mann-Whitney U tests were used to analyze quantitative independent data. In the analysis of qualitative independent data, the chi-squared test was used, and Fischer’s exact test was used when the chi-squared test conditions were not met. The effect level and cut-off values were investigated using ROC curve. Statistical analyses were performed using IBM SPSS version 28.0 (IBM Corp., Armonk, NY, USA). A P-value < .05 was considered statically significant level.
Results
All patients’ personal details were de-identified. A total of 216 eligible participants were assessed for eligibility. Participants were selected based on age, disease diagnosis, clinical stage of the disease, and chemotherapy regimens. Of these 216 patients, 125 were excluded (88 patients were under 65 years, 26 patients received non-FLOT regimens, and data for 11 patients were inaccessible). Consequently, 91 patients were enrolled in this study. A total of 91 patients, 25 females (27.5%) and 66 males (72.5%), were included in our study. There were 22 patients (24.1%) in the treatment-responsive group and 69 patients (75.9%) in the treatment-nonresponsive group. 86.8% of the patients (n = 79) were in clinical stage III disease. The most common tumor locations were the gastroesophageal junction and cardia, with a rate of 51.6% (n = 47). 56% of the patients (n = 51) had moderately differentiated tumors, and 31.9% (n = 29) had pathological stage III tumors whereas 35.2% had pathological stage II tumors. In 80.2% of patients, HER-2 was negative (n = 73). 67% of patients (n = 61) had no comorbidities. Finally, the ECOG performance score was 0 in 83.5% of patients. 12 patients had missing data regarding clinical T stage and 13 patients had missing data on HER-2status. The patients received a mean of four cycles of neoadjuvant chemotherapy. The median platelet count was 249x103, median neutrophil count was 4300, median lymphocyte count was 1.700, CEA was 4, CA19.9 was 11, NLR was 3, PLR was 148.3, and PNI was 45.1. Grade 3-4 toxicity was observed in 38.5% of patients (n = 35). Poor or no response was observed in 24.1% of the patients (n = 22), and 22% of the patients (n = 20) relapsed. 71.4% of patients (n = 65) are alive.
Demographic, Clinical and Histopathological Features of the Groups.

Abbreviations: ECOG, Eastern Cooperative Oncology Group. *Bold and italics values are statistically significant.
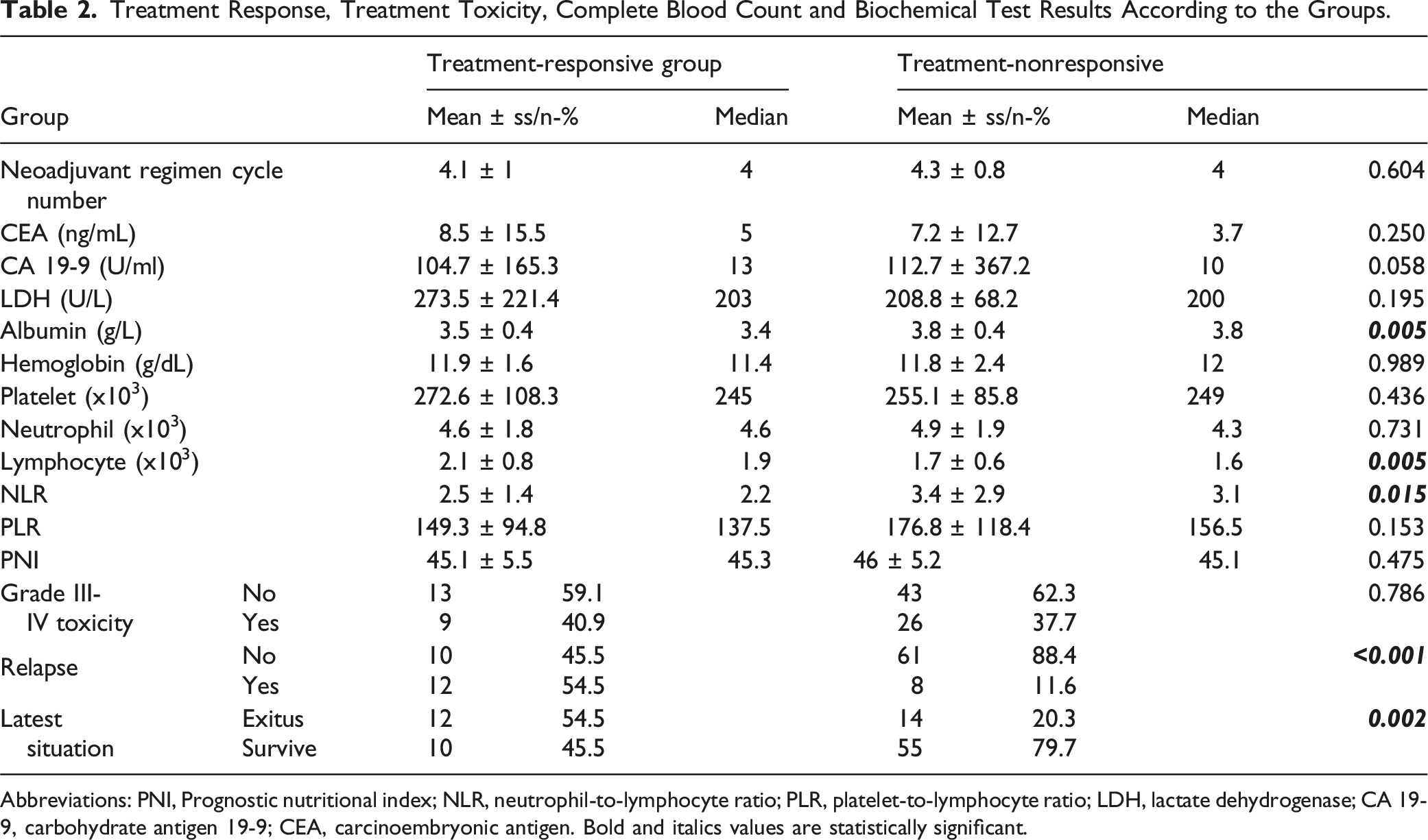
The number of chemotherapy cycles, CEA, CA 19-9, LDH, hemoglobin, platelet, neutrophil, PLR, PNI, and grade III-IV toxicity rate were not significantly different between the groups with and without neoadjuvant treatment response (P > .05). Additionally, the ROC curve showed an AUC of 0.627 (0.478-0.775) (P = .085). The cut-off value for PNI was 44.6 with a sensitivity of 62% and a specificity of 60% (Figure 2). In the treatment-responsive group, albumin, and NLR values were significantly higher than those in the treatment-nonresponsive group (P = .005, P = .015, respectively). The lymphocyte count in the treatment-responsive group was significantly lower than that in the treatment-nonresponsive group (P = .005). The recurrence rate in the treatment-responsive group was significantly lower than that in the treatment-nonresponsive group (P = .00). The exitus rate in the treatment-responsive group was significantly lower than the treatment-nonresponsive group (P = .002) (Table 2). ROC Curve Analysis of PNI. Treatment Response, Treatment Toxicity, Complete Blood Count and Biochemical Test Results According to the Groups. Abbreviations: PNI, Prognostic nutritional index; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; LDH, lactate dehydrogenase; CA 19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen. Bold and italics values are statistically significant.
Significant [AUC 0.700 (0.580-0.820)] efficacy of albumin value was observed in differentiating patients with and without neoadjuvant treatment response. A significant [AUC 0.651 (0.515-0.786)] efficiency of albumin 3.62 cut-off value was observed in differentiating patients with and without neoadjuvant treatment response. The sensitivity, positive predictive value, specificity, and negative predictive value of albumin 3.62 cut-off value were 71%, 84.5%, 59.1%, 59.1%, and 39.4%, respectively. Significant [AUC 0.700 (0.579-0.822)] efficacy of the lymphocyte value was observed in differentiating patients with and without neoadjuvant treatment response. A significant [AUC 0.707 (0.591-0.823)] efficacy of lymphocyte 1.62 cut-off value was observed in differentiating patients with and without neoadjuvant treatment response. The sensitivity, positive predictive value, specificity, negative predictive value, and specificity were 55.1%, 55.1%, 92.7%, 86.4%, 86.4%, and 38%, respectively, at the cut-off value of lymphocyte 1.62 (Figure 3). Significant Efficacy of Albumin Levels and Lymphocyte Count.
Univariate and Multivariate Analysis of Parameters According to Neoadjuvant Treatment Response Status.

Abbreviations: NLR, neutrophil-to-lymphocyte ratio; CI, Confidence interval; OR, Odds Ratio; CEA, carcinoembryonic antigen; CA 19-9, carbohydrate antigen 19-9. *Bold and italics values are statistically significant.
Predicted DFS in the treatment-nonresponsive group (18.7 months) was significantly (P = .00) lower than in the treatment-responsive group (43.4 months). Predicted OS was significantly (P = .00) lower in the treatment-nonresponsive group (19.9 months) compared to the treatment-responsive group (37.4 months) (Figure 4). Overall Survival and Disease-free Survival of the Patients.
Discussion
The prognosis of malignancies in geriatric patients is influenced by a variety of factors, including the type and stage of cancer, the patient’s overall health, and the treatment approach. Geriatric patients often present with unique challenges such as comorbidities and age-related physiological changes that can affect cancer management and outcomes. 16 Consequently, the most effective therapeutic approach for geriatric patients constitutes a compelling clinical dilemma. The majority of randomized clinical trials have historically excluded older individuals due to age restrictions and the presence of specific comorbidities as exclusionary factors. 17
In this comparative study, we demonstrated that pretreatment serum albumin levels, total lymphocyte count, and NLR values can predict treatment response in geriatric gastric cancer patients treated with a neoadjuvant FLOT regimen. PNI and PLR, which are other nutritional or inflammatory markers, were not found to be associated with treatment response. The group that responded to neoadjuvant treatment had lower T and N stages and the probability of treatment response decreased as the clinical stage of the patient increased. In a study comparing the FLOT and ECF regimens, 38% of patients showed little or no response. 18 Similarly, 24.2% of patients had poor or no response in our study.
Elderly patients frequently demonstrate hypoalbuminemia, attributable to factors including malnutrition, inflammation, and chronic diseases. 19 This condition is linked to heightened morbidity and mortality, rendering it a major issue in geriatric healthcare. 20 There is a close relationship between serum albumin level and cancer prognosis. Crumley et al. found that low serum albumin levels were an independent risk factor affecting the survival of gastric cancer patients. 21 In addition, low serum albumin levels may lead to poor prognosis in cancer patients by impairing cellular immune function. 22 In our study, serum albumin levels were higher in the treatment-responsive group than in the treatment-nonresponsive group, in accordance with the literature. In addition, significant efficacy of albumin value in differentiating patients with and without treatment response was observed. At a cut-off value of 3.62, albumin level predicted neoadjuvant treatment response. In addition, albumin levels were associated with treatment response in both univariate and multivariate analyses. Based on the data obtained, we concluded that serum albumin level can be used as a biomarker to predict the response to neoadjuvant treatment in geriatric patients with gastric cancer.
Lymphocytes play an important role in the immune response against cancer. The immune system that comes with age exhibits a decline in the numbers of both B and T lymphocytes, which leads to a heightened vulnerability to infections and cancer. 23 Lymphopenia among the elderly population serves not only as an indicator of frailty but also as a prognostic factor for deleterious health consequences, encompassing heightened morbidity and mortality rates. 24 Low peripheral lymphocyte count has been associated with poor outcome in various cancers. Fumagalli et al. reported that among metastatic renal cell carcinoma patients receiving IL-2, those with low lymphocyte counts at baseline had worse survival rates. 25 Fogar et al. also reported that survival in patients with pancreatic cancer was mainly determined by tumor stage, but a low lymphocyte count was also associated with worse survival. 26 In our study, lymphocyte counts were significantly lower in patients who responded to chemotherapy. In general, a low lymphocyte count is associated with poor prognosis, but there are no studies on its relationship with response to neoadjuvant treatment in geriatric patients. A significant efficacy of lymphocyte count in differentiating patients with and without response to neoadjuvant treatment was also observed in our study. At a cut-off value of 1.62, lymphocyte count predicted the neoadjuvant treatment response. In addition, lymphocyte counts were associated with treatment response in both univariate and multivariate analyses Based on the findings of our study, we concluded that lymphocyte count can be used as a biomarker to predict the response to neoadjuvant treatment in geriatric patients with gastric cancer.
The number of lymphocytes decreases with age, while the number of neutrophils remains the same. 27 In a study involving 268 patients with metastatic gastric cancer, survival was lower in patients with a low NLR. 28 In 1028 patients with gastric cancer undergoing gastrectomy, a high pretreatment NLR was shown to be an independent risk factor for OS. 29 Jung et al evaluated 293 patients who underwent curative gastrectomy. They revealed that a high preoperative NLR was significantly associated with poor OS in patients with stage III or IV gastric cancer. 30 In another study, NLR change (NLRc) was evaluated in 734 patients who underwent surgical or endoscopic resection of gastric cancer. NLRc was shown that NLRc was more valuable in predicting survival than baseline NLR. 31 In a meta-analysis of 3264 patients with gastric cancer who underwent curative resection and articles including preoperative NLR values, it was found that a higher NLR was associated with a lower five-year OS. 32 In our study, NLR was significantly higher in the treatment-responsive group. The percentage of total lymphocytes and the absolute numbers of T and B cells gradually decrease with age. 33 However, studies show that absolute neutrophil counts remain relatively constant in the absence of infection in different age groups. For example, a study comparing young and older people found no significant difference in neutrophil counts in the absence of infection between these groups. 34 Consistent with the literature, in our study, although not statistically significant, the mean age of responders was higher than that of non-responders and the lymphocyte count was significantly lower in this group. In addition, although not statistically significant, the neutrophil count was also higher, but in normal range, in responders than in non-responders. Therefore, the NLR may have been higher. Although a high NLR has been associated with poor prognosis in previous studies, no studies showing its relationship with response in neoadjuvant treatment.
Geriatric patients often present with a complex interplay of comorbidities, altered immune responses, and age-related physiological changes that can influence inflammatory markers like PLR. In a meta-analysis of eight studies including a total of 4513 patients with gastric cancer, a high PLR was found to be associated with a higher risk of lymph node metastasis, serosal invasion, and advanced stage (III + IV) gastric cancer compared with normal PLR, but no association with OS was found. 35 Recent studies have shown that the prognostic significance of inflammatory indices can vary widely across different cancer types and patient populations, highlighting the need for tailored approaches in geriatric oncology. Mohammadi et al. demonstrated that NLR is a significant prognostic factor for survival in gastric cancer patients, whereas PLR is not. 36 The lack of significant differences in PLR may reflect these complexities, suggesting that a one-size-fits-all approach to using PLR as a prognostic marker may be inadequate. Similarly to this study, PLR were not found to be associated with treatment response in our study.
In the present study, PNI was not associated with response to the neoadjuvant treatment. Several studies have previously highlighted the complexity of the relationship between nutritional status and treatment outcomes in gastric cancer. For instance, Hirahara et al. found that while PNI was significantly correlated with prognosis in gastric cancer patients, its predictive ability diminished in certain populations, particularly those with advanced disease or specific treatment regimens. 37 This is consistent with the conclusions of our study, which suggest that the PNI may not be a reliable indicator for assessing treatment response in the geriatric population, which often has complex health problems that may complicate the relationship between nutritional status and treatment response. Zhao’s research emphasizes that preoperative nutritional status, as indicated by PNI, plays a critical role in the surgical outcomes of elderly gastric cancer patients, yet the predictive value of PNI can be confounded by factors such as age-related sarcopenia and chronic inflammation.38,39 Thus, the current findings may suggest that while PNI is a useful tool for assessing nutritional status, its utility in predicting neoadjuvant treatment response in older patients may be limited. Furthermore, the findings necessitate a reconsideration of the clinical relevance of PNI within the framework of neoadjuvant therapeutic interventions. The lack of significant differences in PNI between two groups may reflect the broader context of geriatric oncology, where age-related physiological changes and comorbidities can influence both nutritional status and treatment outcomes. In our study, the mean age of treatment responders was higher than that of non-responders, although this difference was not statistically significant. While a higher PNI was expected in treatment responders, the higher mean age of this group may have caused the PNI to be lower than expected, and a significant difference in PNI between the two groups could not be achieved. Although the PNI was not statistically significant, the individual components of the PNI (lymphocyte and albumin levels) were statically significant predictors of treatment response. Therefore, it is plausible that these parameters alone could be utilized to predict treatment response. Furthermore, it may be predicted that alternative neoadjuvant therapies can be applied to improve treatment response in this patient cohort. To sum up, the current study’s results indicate that PNI does not significantly differ between geriatric gastric cancer patients with and without neoadjuvant treatment response, suggesting limitations in its predictive value in this population. This finding emphasizes the need for further research to explore the multifactorial influences on treatment outcomes in elderly patients and to develop more effective prognostic tools that can guide clinical decision-making in this vulnerable group. In light of these findings, it is essential to consider alternative approaches to assessing treatment response in geriatric gastric cancer patients. Integrating PNI with other biomarkers or clinical assessments may enhance predictive accuracy.
In our study, the recurrence rate was lower in the treatment-responsive group. This suggests that DFS is better and is likely to be reflected in OS. Disease recurrence may predict long-term survival. In addition, the exitus rate was significantly lower in the treatment-responsive group than in the treatment-nonresponsive group. A study by Derieux et al. of 325 patients with gastric adenocarcinoma showed that both OS and DFS were better in those who responded to neoadjuvant chemotherapy than in those who did not.
40
The benefit of neoadjuvant therapies in the geriatric age group is limited owing to the physiological and metabolic consequences of ageing. Owing to the low efficacy expectancy and high toxicity of neoadjuvant treatment in elderly patients, patients who could potentially benefit may be overlooked and deprived of the expected benefit. In this study, we observed that pretreatment serum albumin levels and lymphocyte count serve as predictors of response in geriatric gastric cancer patients treated with neoadjuvant FLOT. Furthermore, both DFS and OS were significantly prolonged in patients who demonstrated a response to treatment. In this way, we conclude that by predicting the group that may benefit from neoadjuvant treatment in geriatric patients with gastric cancer, we can break the prejudice of lower response to treatment in this group and be more alert in patient selection.
Study Limitations
Our study has limitations such as its single-center and retrospective design, lack of mature survival data, and inability to completely exclude the possibility of complications such as subclinical infection that may affect the prognostic values of biomarkers. Despite this, our study is valuable because it included a homogeneous patient group.
Conclusions
While the prognosis of malignancies in geriatric patients is generally poorer compared to younger populations, this is not solely due to age. Factors such as undertreatment, bias, and lack of comprehensive assessments contribute significantly to outcomes. By addressing these issues and utilizing geriatric assessments and predictive models, healthcare providers can improve the prognosis for elderly cancer patients. In conclusion, we can predict response to treatment in gastric cancer patients receiving neoadjuvant chemotherapy using routine and easily accessible blood parameters before treatment. Although the choice of chemotherapy regimen in geriatric patients is difficult owing to the frailty and comorbidities, the serum albumin levels, lymphocyte count, and NLR values, which we found in geriatric patients in our study, may guide us. However, larger prospective multicenter studies are required to confirm this relationship.
Footnotes
Statements and Declarations
Author Contributions
Conception and interpretation of this work: YYÜ, MÜ; Drafting of manuscript: YYÜ; Critical revisions of the manuscript: MÜ; Approved the final version of the manuscript: YYÜ, MÜ; Accountable for all aspects of the work: YYÜ, MÜ
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
