Abstract
Objective
The incidence of papillary thyroid microcarcinoma (PTMC) has increased sharply during the past decades. Yet, whether or not nodal dissection should be performed remains controversial. This article aims to assess the high-risk factors associated with cervical lymph node metastasis (LNM) in patients with PTMC, which may potentially guide clinical management decision-making.
Methods
Medical records of 449 PTMC patients who underwent thyroidectomy in our hospital from August 2016 to July 2017 were retrospectively reviewed. Clinical and pathological factors of the patients were anonymously extracted from the charts and analyzed.
Result
The patients were classified into two subgroups according to maximum tumor size measured through post-surgical pathology: smaller PTMC group (≤5 mm) and larger PTMC group (>5 mm). Larger tumor size was found to be associated with a higher rate of LNM (P = .001), particularly central lymph node metastasis (CLNM) (P = .001). Tumor size was also associated with extrathyroidal tumor extension (ETE) (P < .001), bilateral lesions (P = .015), and BRAFv600e mutation (P = .004). LNM was found to be more common in older patients (>55 y) (P = .030), and those with multifocal cancer (P < .001). In PTMC patients with unilateral lesions without ETE, tumor size was not significantly associated with LNM (P = .121).
Conclusions
For the PTMC population, tumor size was an independent risk factor for LNM, particularly for patients of old age (>55 y), and multifocality. However, in PTMC patients with unilateral lesions without extrathyroidal extension, tumor size was not related to the risk of LNM. These findings may potentially guide clinical decision-making in terms of cervical nodal dissection.
Introduction
Thyroid carcinoma is the most common endocrine malignant tumor worldwide, with papillary thyroid carcinoma (PTC) being the most common subtype. 1 Papillary thyroid microcarcinoma (PTMC) is a special subgroup of PTC, defined by the WHO as PTC with a maximum diameter less than or equal to 10 mm. 2 Given the frequent use of cervical ultrasound and fine-needle aspiration biopsy (FNAB) for thyroid nodules, the incidence of thyroid carcinoma including PTMC has increased worldwide. 3 It is reported that the rate of PTMCs accounts for nearly 30% of PTCs.4,5 Although the prognosis of PTMC is favorable,4,6,7 its treatment remains controversial. The 2015 American Thyroid Association guidelines for thyroid nodules and cancer recommend regular surveillance for thyroid nodules with a diameter less than 10 mm regardless of whether it is benign or malignant.2,8 The Japanese Society of Thyroid Surgeons also recommends active surveillance for low-risk PTMC. 9 Yet, the Chinese guideline for PTMC suggests that PTMC with a diameter larger than 5 mm is an indication for operation. 10 Moreover, it is further debatable whether prophylactic cervical nodal dissection is warranted or not during the surgery.
Even though most PTMC patients have favorable outcome, poor prognosis still exists for a portion of patients. An important factor related to the poor prognosis of PTMC is cervical lymph node metastasis (LNM).11-13 Recent studies have attempted to establish predicting models for LNM development based on the features of PTMCs.14-16 However, a definitive conclusion has not been reached.
17
Therefore, in the current study, we aimed to identify the independent risk factors for LNM in a relatively large patient population with PTMC, which may potentially guide clinical management decision-making in the future.
Methods
Patients
A total of 449 adult patients diagnosed with PTMC on histopathology from August 2016 to July 2017 at Ruijin Hospital were enrolled in this study. We retrospectively reviewed the medical records, including pathology reports of all patients. All of the patients met the inclusion criteria, which were as follows: (1) age ≥18; (2) thyroid nodules were preoperatively assessed by fine-needle aspiration biopsy (FNAB) and proven malignant; (3) underwent total thyroidectomy, near-total thyroidectomy, or unilateral thyroid lobectomy, with prophylactic or therapeutic central neck lymph node dissection (CLND) and ipsilateral lateral neck lymph node dissection (LLND); (4) dissected thyroid nodules and lymph nodes were assessed on postoperative pathology and proven to be PTMC, of which the maximum tumor size was ≤1 cm (the maximum tumor size was defined as the greatest dimension for solitary lesion, or greatest dimension of the maximum lesion for multifocal lesion); (5) absence of distant metastasis evidence. According to the above criteria, TNM staging of all patients was categorized to T1a,3b,4N0∼1M0 according to the American Joint Committee on Cancer/Union for International Cancer Control (AJCC/UICC) staging system.
This study was approved by the Institutional Ethics Committee of our hospital (approval number was 2021-416), and the informed consent was waived given the retrospective nature of the study. Patients’ clinical data (age, sex, post-surgical pathology findings) were anonymously extracted from the patients’ charts without patients’ personal information.
Surgical Procedures and Definitions
All patients underwent ultrasonography to evaluate primary thyroid lesions and LNM. FNAB was performed for sonographically suspicious thyroid nodules (TI-RADS 4A or above) and suspicious lymph nodes. Unilateral thyroid lobectomy was performed for preoperatively proved (or suspicious) unilateral malignant thyroid nodules by FNAB diagnosis (Bethesda V and VI). Total thyroidectomy or near-total thyroidectomy was performed for preoperatively proved (or suspicious) bilateral malignant thyroid nodules. CLND and therapeutic ipsilateral LLND were performed for clinically proved lateral lymph node metastasis (LLNM) by FNAB diagnosis or suspected LLNM during operation. Prophylactic CLND was performed for patients without any preoperative or intraoperative evidence of LNM. Total thyroidectomy was defined as removal of all grossly visible thyroid tissue, and near-total thyroidectomy was defined as removal of all grossly visible thyroid tissue while leaving only a small amount (<1 g) of tissue adjacent to the recurrent laryngeal nerve. LLND was performed from level II to level V. CLND was bilaterally performed from level VI to level VII. All acquired pathology specimens were microscopically diagnosed and cross-checked by two or more experienced pathologists at our hospital. Pathological features analyzed in this study included surrounding thyroid tissues type (normal or Hashimoto thyroiditis), maximum tumor size, extrathyroidal extension (ETE, defined as the tumor invading strap muscles or extending into major neck structures), multifocal lesions (two or more malignant thyroid nodules), bilateral lesions, and LNM. The PTMC patients were classified into two subgroups according to maximum tumor size measured on pathology: smaller PTMC group (≤5 mm) and larger PTMC group (>5 mm).
Statistical Analysis
All data were entered in EpiData (version 3.1; The EpiData Association, Odense, Denmark) and statistical analysis was carried out with SPSS (version 24.0; SPSS Inc., Chicago, Illinois, USA). Qualitative data were expressed as a percentage or frequency. Quantitative data were shown as mean ± standard deviation (SD) when accorded with normal distribution. Descriptive statistics were used to summarize the data. Differences in clinical parameters between two groups or three groups were compared using χ 2 -test or Fisher exact test for qualitative data and independent t-test for quantitative data. Logistic regression analysis was used in multivariate analysis. P < .05 was considered statistically significant.
Result
Patient Characteristics in PTMC
Patients’ characteristics.

LNM: lymph node metastasis; CLNM: central lymph node metastasis; LLNM: lateral lymph node metastasis; ETE: extrathyroidal extension.
Clinicopathological Differences Between the Small and Large Tumor Groups
Univariate analysis of patients’ clinicopathological factors of tumor size.

LNM: lymph node metastasis; CLNM: central lymph node metastasis; LLNM: lateral lymph node metastasis; ETE: extrathyroidal extension.
Univariate analysis of clinicopathological factors of LNM.
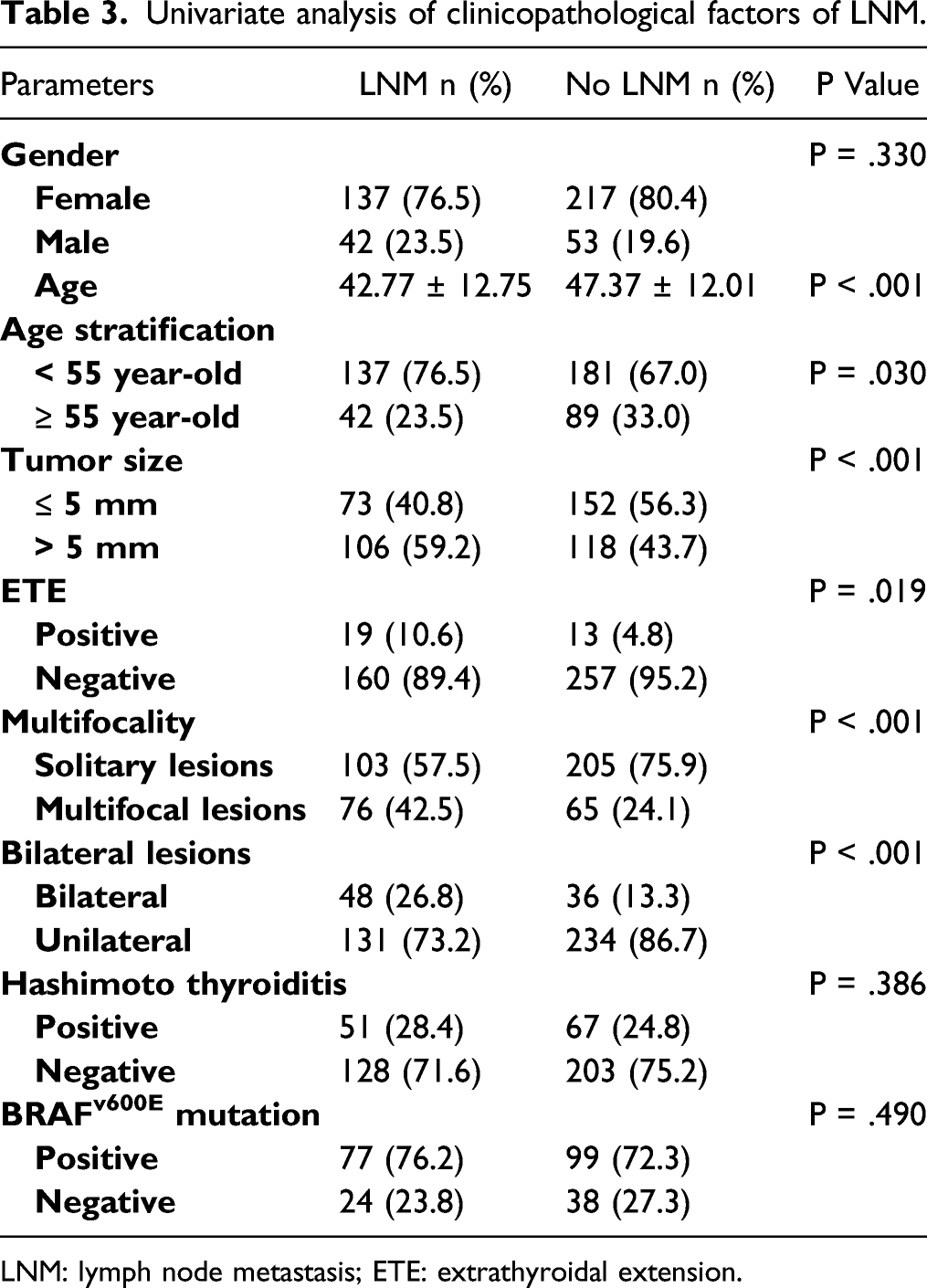
LNM: lymph node metastasis; ETE: extrathyroidal extension.
The hazard ratios of clinical and pathological features were analyzed by multivariate analysis (Figure 1). As shown in Figure 1, multiple lesions (HR = 1.96, P =.026) and larger tumor size (HR = 1.75, P = .009) remained as independent risk factors. Older age (≥55 year) (HR = .60, P = .023) was also an independent protection factor for LNM. Bilateral lesions and ETE were not independent risk factors for LNM in PTMC. Multivariate analysis of parameters of LNM. Hazard Ratio was shown as mean (95%CI).
Correlation Between Tumor Size and LNM in Unilateral PTMC Patients With Staging T1a
Analysis of tumor size and LNM in different situation.

LNM: lymph node metastasis; PTMC: papillary thyroid microcarcinoma.
Discussion
Increasing incidence of PTC has been reported over recent years, which could be attributed to the significantly increased incidence of PTMC. 18 Surgical resection is generally accepted to be the most effective treatment for PTC, yet whether or not PTMC should be treated surgically remains controversial, nodal dissection in particular.19,20 Advocates suggest that a large proportion of patients with PTMC are associated with LNM, and without timely surgical intervention, the risk of subsequent surgery being required increases as tumors progress. 21 Conservatives on the other hand believe that only dynamic medical surveillance is needed for PTMC, and surgery is only required when significant tumor progression or LNM occurs. 22 In China given the higher rate of LNM in Asian PTMC patients and limitations of preoperative examinations in detecting CLNM (especially in level VII), most institutions prefer surgical interventions with nodal dissection rather than surveillance alone.21,23 In our cohort, the percentage of PTMC patients with LNM was up to 40% (179/449).
In our cohort, there was a statistical association between tumor size and LNM, in which larger PTMC had a higher incidence of LNM and CLNM, but not LLNM. Li et al 24 analyzed 273 PTMC patients with clinically negative central lymph nodes, which showed a large tumor diameter had a strong correlation with a larger number of CLNM. Feng et al 11 did a study including 785 PTMC patients who underwent thyroidectomy and CLND and showed the incidence of CLNM was 18.9% in smaller PTMC and 42.7% in larger PTMC, similar to our results.
The number of CLNM plays an important role in evaluating the aggressiveness of PTC. Bohec et al 25 reported that patients with more than 5 positive CLNM had significantly higher aggressiveness of PTC and had a high risk of LLN involvement. In our research, the larger PTMC group had a greater number of CLNM than the smaller PTMC. Other studies also showed that tumor size had a positive correlation with the number of metastatic lymph nodes, and these results were concluded by a meta-analysis by Liu et al. 26 The meta-analysis showed that regardless of PTMC or non-PTMC, larger tumor size was significantly associated with a greater count of CLNM.
Furthermore, we analyzed the association of LNM and patients’ characteristics. The results showed that larger tumor size and multifocality were both independent risk factors of LNM, and older age was an independent protective factor of LNM. Contrary to some previous studies,12,13,27 our results showed that there was no statistical difference between LNM and gender as well as BRAFv600e mutation.
Multifocality has a certain role in PTMC. Our results showed that PTMC with multiple lesions had a higher incidence of LNM than those with a solitary lesion. Zheng et al 23 analyzed 1587 patients with PTMC who underwent thyroidectomy and lymph node dissection, and results showed that PTMC with multiple lesions had a higher incidence of CLNM and LLNM.
Age is an important characteristic of PTC. Our study found that age has a significant correlation with LNM, that is, patients with LNM had a younger age than those without LNM. This mirrors reports from Ito et al 28 that analyzed 1235 patients with low-risk PTMC who underwent active surveillance. Results showed that younger PTMC patients (<40 years) had a higher incidence of LNM than older patients (≥60 years).
Gender is a controversial factor in studies of PTMC. In our cohort, LNM had no significant correlation with gender in PTMC. Zhao et al 29 analyzed 521 PTMC patients who were diagnosed by FNAB concluding that the male gender is an independent risk factor for LNM, which was different from what we found in the current study. The difference between our studies was that they used ultrasound to detect LNM. However, occult LNM may escape ultrasound detection, which might explain our differences in findings. Lee et al 30 analyzed 2930 PTC patients and concluded that the male gender was an independent risk factor in PTC >1 cm, but was not an independent risk factor in PTMC.
BRAFv600e mutation is known as a biomarker of aggressiveness of PTMC. Our study showed that larger PTMC had a higher incidence of BRAFv600e mutation than smaller PTMC (P = .004). Kim et al 31 conducted a large multicenter retrospective research and reported that BRAFv600e mutation-positive group had a larger tumor size than BRAFv600e mutation-negative group.
Several studies showed that LNM was associated with BRAFv600e mutation.32,33 Our results showed that patients with LNM had a higher incidence of BRAFv600e mutation (76.2% in LNM patients and 72.3% in no LNM patients), whereas there was no clear association between LNM and BRAFv600e mutation (P = .490). Zheng et al 23 who completed BRAFv600e mutation tests of 299 PTMC patients, also revealed that LNM was not associated with BRAFv600e mutation. However, given that whether BRAFv600e mutation tests are conducted is subject to the judgment of surgeons, there might be selection bias in the study. A multi-center study with a larger sample size may be needed.
Based on previous findings, we investigated the effects of tumor size on the risk of LNM of PTMC. After screening out patients with bilateral lesions, we surprisingly found that there was no correlation between tumor size and LNM in unilateral PTMC patients. Considering that T stage had an important effect on primary tumors, we screened out patients with ETE, and analyzed PTMC patients with stage T1a17. We found that tumor size was still not correlated with LNM, and P value was increased. While other studies conducted extensive research on the risk factors for LNM in PTMC patients, focusing on the entire population of PTMC cohort, no further analysis of individual risk factors was performed.34,35 Our study conducted a detailed analysis on the risk factor of tumor size and found that the risk of LNM in unilateral T1a PTMC was independent of tumor size. This finding confirmed that ETE and bilateral lesions, which represent biological characteristics of the primary tumor, played a more important role in TNM staging. 36 More importantly, tumor size itself had little significance as an independent risk factor in PTMC patients. Therefore, when evaluating high-risk PTMC, attention should instead be paid to the effects of ETE, bilateral lesions, multifocality, younger age, and other factors on LNM. These findings will allow for a more comprehensive and accurate risk assessment of PTMC to guide treatment strategies.
Our research had several potential limitations. First, it was a retrospective study and nonrandomized features were inevitably produced. Second, the information of metastatic lymph node size and follow-up post-operation was not collected. Finally, our study only included PTMC patients from a similar environment. Thus, more multi-center studies containing a larger sample of patients are needed to reaffirm our findings.
Conclusion
For the PTMC population, younger age, multifocality, ETE, and larger tumor size were independent risk factors of LNM. However, for T1a PTMC patients with unilateral lesions, tumor size was not related to the risk of LNM. These findings may play important roles in guiding a more complete and detailed assessment of the risk of LNM of PTMC, thus influencing the treatment decisions on PTMC patients.
Footnotes
Acknowledgments
The authors thank Dr. Wengen Chen and Jui-Ming Lin for their helpful assistance in the language polishing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Natural Science Foundation of China (NSFC grant numbers 81471688, 81 671 720, 81 801 726, and 81 971 644) and the Shanghai Sailing Program (18YF1414300).
Ethical Approval
The study was approved by the institutional Ethics Committee of our hospital (approval number was 2021-416), and the consent was waived given the retrospective nature of the study.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
