Abstract
Background:
There is strong and well-documented evidence that socio-economic inequality in cancer survival exists within and between countries, but the underlying causes of these differences are not well understood.
Methods:
We systematically searched the Ovid Medline, EMBASE, and CINAHL databases up to 31 May 2020. Observational studies exploring pathways by which socio-economic position (SEP) might causally influence cancer survival were included.
Results:
We found 74 eligible articles published between 2005 and 2020. Cancer stage, other tumor characteristics, health-related lifestyle behaviors, co-morbidities and treatment were reported as key contributing factors, although the potential mediating effect of these factors varied across cancer sites. For common cancers such as breast and prostate cancer, stage of disease was generally cited as the primary explanatory factor, while co-morbid conditions and treatment were also reported to contribute to lower survival for more disadvantaged cases. In contrast, for colorectal cancer, most studies found that stage did not explain the observed differences in survival by SEP. For lung cancer, inequalities in survival appear to be partly explained by receipt of treatment and co-morbidities.
Conclusions:
Most studies compared regression models with and without adjusting for potential mediators; this method has several limitations in the presence of multiple mediators that could result in biased estimates of mediating effects and invalid conclusions. It is therefore essential that future studies apply modern methods of causal mediation analysis to accurately estimate the contribution of potential explanatory factors for these inequalities, which may translate into effective interventions to improve survival for disadvantaged cancer patients.
Introduction
Socio-economic position (SEP) is a complex construct of several aspects of a person’s social, financial and occupation position. 1 Cancer patients with lower SEP consistently show worse survival than those with higher SEP, regardless of whether individual-level SEP or area-based measures are used. 2,3 Comprehensive reviews conducted by the International Agency for Research on Cancer (IARC) in 1997 2 and Woods et al, in 2005 3 found solid evidence for socio-economic inequalities in cancer survival for most malignancies and in many countries. The extent of the survival differences by SEP is moderate for most cancer sites, but substantial for cancers of the breast, colon, bladder and corpus uteri, which all have relatively good prognosis. 2 Stage at diagnosis was reported to be the primary explanatory factor, but its estimated mediating effect has differed by cancer site and between countries and studies. 3,4 Few studies have assessed the contribution of treatment to survival differences among socio-economic groups. 3,4 The degree to which patient characteristics such as the presence of co-morbid conditions and health-related behaviors explain socio-economic differences in cancer survival also remains unclear.
In this systematic review, we assessed studies exploring underlying reasons for socio-economic inequalities in cancer survival, with the aim of identifying potential contributing factors and determining the validity of published estimates of their mediating effects.
Methods
This systematic review was planned, conducted and reported in adherence to the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analysis Protocols (PRISMA-P). 5 The review protocol was registered with the International Prospective Register of Systematic Reviews–PROSPERO (registration number CRD42016039227).
Search Strategy
A systematic search of studies published in English from 1 January 2005 to 31 May 2020 was conducted in Ovid Medline, EMBASE and the Cumulative Index to Nursing and Allied Health Literature (CINAHL) databases to identify those that investigated the underlying reasons for socio-economic inequalities in cancer survival (Supplementary Table 1). The bibliographies of selected studies were reviewed to locate eligible articles that might not have been detected through the above process. Finally, we carried out a further manual search using Google Scholar and reviewed the first 3 pages to ensure that potentially relevant studies were not missed.
Eligibility Criteria
Eligible studies met all of the following criteria: (1) observational study of adults (men or women diagnosed with cancer at age ≥15 years); (2) written in English and published in a peer-reviewed journal since 2005; (3) investigated the underlying causes of socio-economic inequalities in cancer survival; (4) assessed death from any cause or death from a specific type of cancer; and (5) reported an estimate of a hazard ratio (HR), odds ratio (OR), or excess mortality rate ratio (EMRR), with a corresponding 95% confidence interval (CI) or standard error. The EMRR is the ratio of the excess mortality rate due to cancer diagnosis in one group of people (e.g., people with low SEP) versus the excess mortality rate in another group (e.g., people with high SEP). We excluded eligible abstracts if full text was not available.
Study Screening and Data Extraction
N.A. performed the literature search and excluded irrelevant or ineligible studies based on the titles and abstracts. Full reports of selected articles were imported to Covidence, a web-based program for conducting systematic reviews, for independent screening by N.A. and R.L.M. Any disagreements were resolved after consulting D.R.E. Data from the selected studies were extracted by N.A. with assistance from R.L.M. For each study, we extracted the following information: the first author’s last name, year of publication, country where the study was conducted, sources of data, diagnosis years, range of age at cancer diagnosis, cancer types studied, measures and categories of socio-economic position, factors considered as potentially contributing to socio-economic inequalities in cancer patient survival, statistical methods and covariates included in the analyses.
Assessment of Risk of Bias
N.A. and R.L.M independently assessed the risk of bias of eligible studies using the domains of bias from the ROBINS-E (Risk of Bias In Non-Randomized Studies-of Exposures) tool [http://www.bristol.ac.uk/population-health-sciences/centres/cresyda/barr/riskofbias/robins-e/]. The following domains were reviewed: confounding, selection of participants into the study, classification of the exposure, adjustment for mediators, level of missing data, measurement of the outcome, and reporting of results.
Results
Study Selection
The electronic database search identified 9,245 articles; 2,069 duplicate citations were removed, and an additional 7,026 articles were excluded based on their title and abstract, leaving 150 articles for further assessment. We excluded 76 studies after full-text screening; therefore, 74 articles met the eligibility criteria for inclusion in the review (Figure 1).
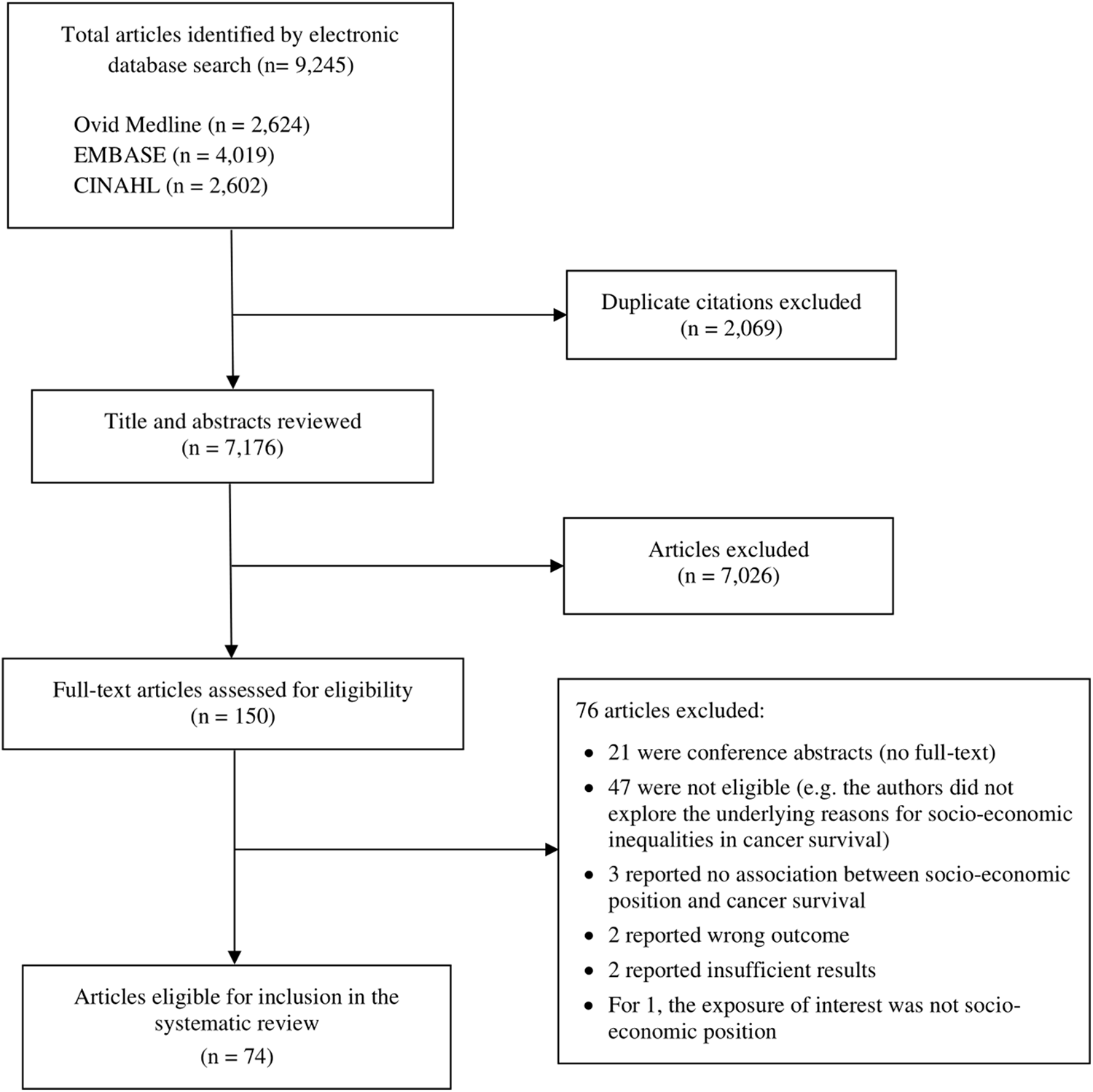
Flow diagram describing selection of studies for inclusion in the systematic review of factors explaining socio-economic inequalities in cancer survival. CINAHL, Cumulative Index to Nursing and Allied Health Literature.
Study Characteristics
Table 1 summarizes the characteristics of the included studies and factors considered as potentially contributing to socio-economic inequalities in cancer-specific and overall survival. Forty-four studies were conducted in Europe 6 -48 (1 study used data from England and Australia), 49 19 in the United States of America (US), 50 -68 4 in Canada, 69 -72 3 in Australia, 73 -75 2 in New Zealand, 76,77 and 2 in Asia. 78,79 These studies assessed the following cancers: female breast, 8,9,13,17,18,26,29,30,32,36,44,49,53,59 -61,65,67,77 male breast, 58 cervix, 24,50 ovary, 23,42,45 endometrium, 33 prostate, 7,34,43,64 penis, 47 colorectum, 12,15,22,28,40,48,55,57,66,73 lung, 10,11,16,20,25,41,56 head and neck, 31,54,70,71 brain and central nervous system (glioma), 62,63 esophagus, 27 pancreas, 39,79 liver, 72 kidney, 52 melanoma, 19,35 acute myeloid leukemia 37 and non-Hodgkin lymphoma, 21,38 as well as selected groups of malignancies. 6,14,46,51,68,69,74 -76,78 The majority of the studies used population-based cancer registry data, 7 -11,13,16 -20,25,27 -30,34 -36,38 -41,44,49,50,52,54 -56,58,60 -63,66,68,69,71 -79 , some linking these with healthcare administration, public and private hospital, screening and treatment datasets. 8,20,38,50,54,71,73 The remaining studies used data from a variety of sources including cohort and case-control studies, 6,12,14,26,43,59,64 hospitals, 31,37,46,70 cancer surveillance programs, 57 national cancer audit 48 or other cancer databases. 15,21 -24,32,33,42,45,47,51,53,65,67 Twenty-three studies reported cancer-specific survival, or relative or net survival, where the cancer under study was considered as the cause of death, 10,13,18,19,27,28,30,32,34 -36,44,49,52,60,61,66,71,73 -77 while 36 studies presented overall survival (i.e., death from any cause). 6 -8,11,14,16,17,20 -26,33,37 -39,41,42,45,46,48,50,51,53,55 -58,62,63,70,72,78,79 Other studies reported both overall and cancer-specific or relative/net survival. 9,12,15,29,31,40,43,47,54,59,64,65,67 -69
Characteristics of Included Observational Studies on Potential Explanations for Socio-Economic Inequalities and Cancer Survival, 2005-2020.

The measurement of SEP of cancer patients at diagnosis varied across studies. Several studies used composite measures or indices of SEP or deprivation such as census tracts socio-economic status, 38,51 -56,58,61 -63,65,66,68,70,71 Townsend index, 17,27,28 the index of multiple deprivation, 12,20,25,29,32,34,41,44,45,48 the index of relative socio-economic advantage and disadvantage or the index of relative socio-economic disadvantage, 73 -75 the New Zealand deprivation index, 76,77 the Scottish Index of multiple deprivation, deprivation index, 30,37,42 socio-economic index 11 or the Small Area Health Research Unit index of social deprivation. 36 Other studies defined SEP using measures such as educational level, income, unemployment rate, poverty-level, median house-hold income or median property value across aggregated or geographical areas based on address, postal code or neighborhood. 7 -9,31,49,50,59,60,64,67,69,72,78 The remaining studies used individual measures including education, gross household or disposable income, last occupation and housing status (rental or owner occupied). 6,10,13 -16,18,19,21 -24,26,33,35,39,40,43,46,47,57,59,64,67,79
Different approaches were used to attempt to explain the underlying reasons for socio-economic inequalities in cancer survival. Most studies applied the “difference method,” which compares regression coefficients for exposure on outcome from models with and without adjusting for potential mediators. 6 -8,10 -15,17 -28,30,31,33,35 -37,39 -44,46,47,50 -53,55 -57,59,60,62 -67,69 -79 Other studies examined the distribution of cases across socio-economic categories by the mediator(s) of interest or stratified the relative risk or survival rate estimates by potential mediators. 9,16,32,34,45,48,49,58,61 Four studies applied causal mediation analysis. 29,38,54,68
Factors Explaining Socio-Economic Inequalities in Survival
Cancer-specific survival
Sixteen studies examined the underlying causes of educational and socio-economic inequalities in breast cancer survival: 8 from Europe, 9,13,18,30,32,36,44,49 5 from the US, 59 -61,65,67 2 from New Zealand 76,77 and 1 from Australia. 74 Studies from Switzerland, Italy, Ireland, New Zealand and Australia applying the difference method reported consistent findings that stage at diagnosis, other tumor characteristics, method of detection or presentation (symptomatic, screening, incidental, unknown), and receiving suboptimal treatment and sector of care (private or public) only partly explained the observed socio-economic inequalities in breast cancer survival. 13,30,36,76,77,74 In contrast, studies from the Netherlands and Sweden found that the observed lower survival among disadvantaged women, or those with low level of education, was not explained by variation in stage of breast cancer at diagnosis, other tumor characteristics, number of nodes examined or the treatment received. 9,18
A study from England using stage-specific analysis found that lower survival from breast cancer among women with mid-level SEP, relative to the most advantaged, was entirely explained by stage at diagnosis, while stage had no mediating effect on lower survival among the most disadvantaged patients. 32 Another study using data from England and Australia observed no differences in survival across socio-economic groups in New South Wales, Australia after stratifying on mode of detection (screen-detected or non-screen-detected) 49 ; however, in the West Midlands, England, there were inequalities in survival between affluent and disadvantaged women with both screen-detected and non-screen-detected breast cancer, which were not explained by extent of disease at diagnosis. 49 In contrast, another study from England reported that inequalities in survival, irrespective of screening status (screen-detected or non-screen-detected), were only partly explained by differences in stage at diagnosis and co-morbidities; differences in other tumor characteristics and treatment did not contribute to lower survival in disadvantaged women. 44
US studies measuring SEP at the individual and community level found that higher risk of death among the most disadvantaged women was mostly influenced by variation in annual screening mammography participation and disease stage, and less by lifestyle factors and first course treatment. 59,60 Other US studies reported that differences in stage at diagnosis, breast cancer subtype (triple negative, HER2 or luminal), co-morbid conditions and the treatment received did not explain lower survival for residents of disadvantaged areas, 61,65 while another study reported health-related lifestyle behaviors and co-morbidity, but not treatment, as contributing factors to the observed gap in breast cancer survival. 67
Of the 7 studies that investigated cancer-specific survival after diagnosis with colon or rectal cancer, studies from Sweden, Australia and the US found that lower survival in disadvantaged or low educated patients was not explained by variations in stage at diagnosis, patient factors or other tumor characteristics, nor by differences in co-morbidities, hospital type/characteristics, and treatment e.g. elective or emergency surgery or preoperative radiotherapy for rectal cancer. 15,66,73,74 In contrast, studies from England, Switzerland and New Zealand concluded that rural-urban residence, extent of the disease and provision of treatment within the first month of diagnosis contributed to the observed lower survival in disadvantaged patients and those with low-level education. 28,40,76
Three studies assessed the association of socio-economic disadvantage with survival from head and neck cancer. A study from Canada found that the anatomic extent of the tumor accounted for 3-23% of differences in survival from glottic cancer 71 ; the authors also reported waiting time to receive treatment as an explanatory factor for lower survival among disadvantaged patients. 71 A US study of oral and pharyngeal cancer reported cigarette smoking as a contributing factor to socio-economic inequalities in survival; the authors reported that the indirect effect of smoking was larger for patients in the middle socio-economic group (21%) than for those in the lowest category (13%). 54 A Scottish study comparing HRs with and without adjusting for potential mediators proposed that socio-economic differences in survival from head and neck cancers (deprived compared with affluent cases, unadjusted HR 1.33; 95%CI 1.06-1.68) were explained by variations in stage at diagnosis, tumor differentiation, alcohol drinking, smoking status and patient performance status (fully adjusted HR 0.93; 95%CI 0.64-1.35). 31
Three studies investigated socio-economic differences in survival from prostate cancer. 34,43,64 A study from Scotland stratifying the relative risk estimates by Gleason score found that differential stage of prostate cancer contributed to some of the observed deprivation-based gap in survival. 34 A US study concluded that differences in co-morbidities, health-related behaviors, hospital characteristics and environmental factors did not explain the observed lower survival for men with low level of education, although the gap in survival by socio-economic position (disadvantaged men compared with advantaged cases, base model HR 1.56; 95% CI 1.11-2.19) decreased after adjusting for these variables (fully adjusted HR 1.41; 95% CI 0.98-2.03). 64 A study from Denmark reported treatment and metabolic indicators such as body mass index and diabetes as contributing factors to lower survival among men with lowest education, whereas co-morbidities did not explain the observed gap in survival. 43
Two studies from Sweden 19,35 and 1 from New Zealand 76 assessed cancer-specific survival from melanoma by education level or an area-based measure of SEP. A Swedish study, comparing models with and without adjusting for mediators of interest, found that stage at diagnosis largely explained worse survival among low educated relative to highly educated individuals (age and sex adjusted HR 1.58; 95%CI 1.40-1.77 decreased to HR 1.19; 95%CI 1.06-1.34 after adjusting for clinical stage). 19 Another study from Sweden reported consistent findings. 35 A New Zealand study showed that 50% of the observed deprivation gap in survival from melanoma was explained by extent of the disease. 76
Of the remaining studies, a study of esophageal cancer from France concluded that lower survival among patients living in deprived areas was not explained by differences in stage, morphology, surgery, radiotherapy or chemotherapy. 27 A US study conducted tumor characteristics-specific analysis and suggested that lower survival from renal cancer among individuals from lower socio-economic background may be partly explained by advanced tumor at diagnosis, or tumor size or grade. 52 Another study of gastrointestinal cancers from the US, applying causal mediation analysis, showed that differences in surgery explained one third of socio-economic inequalities in survival for patients with esophageal adenocarcinoma, extrahepatic cholangiocarcinoma, and pancreatic adenocarcinoma. 68
An Australian study concluded that stage at diagnosis was the underlying reason for lower survival from ovarian cancer in women living in the most disadvantaged areas. 74 Another study from New Zealand found that the extent of disease fully explained the observed lower survival from cervical cancer among more deprived women, while it only contributed to 5% of the deprivation gap in survival from uterine cancer. 76 A Swedish study of lung cancer showed that better survival from early-stage disease in women with high educational level, relative to low educated, persisted after adjusting for treatment (HR 0.33; 95%CI 0.14-0.77), while for stage III, the observed lower survival comparing high versus low educated men also remained (fully adjusted HR 1.41; 95%CI 1.04-1.90). 10 Another study from Sweden found that stage at diagnosis and co-morbidities did not contribute to socio-economic inequalities in survival from penile cancer. 47 Lastly, an Australian study of all malignancies found that stage at diagnosis did not explain the observed lower survival among cancer patients living in more disadvantaged areas. 75
Overall survival
We identified 8 studies that assessed overall survival following breast cancer diagnosis. A Dutch study observed socio-economic differences in overall survival for women with screen-detected, non-screen-detected and interval breast cancers. 8 Lower survival among more disadvantaged women diagnosed via screening was partly explained by the higher prevalence of co-morbidities. For women with non-screen-detected and interval breast cancers, inequalities in survival appeared to be largely explained by differences in stage at diagnosis, while treatment explained very little. 8 A Canadian and a British study found that socio-economic inequalities in overall survival after breast cancer were partly mediated by differential stage and surgical treatment. 29,69 Similarly, studies from England, the US and Canada concluded that the higher risk of death for disadvantaged women was largely explained by their generally more advanced stage and co-morbid conditions. 17,51 Another study from the US reported insurance status, disease stage and treatment as major explanatory factors, although co-morbidities explained very little of the observed gap in survival. 53 A study conducted in Denmark showed that metabolic indicators, smoking status and alcohol intake accounted for part of the higher risk of death among women with lower educational attainment, while disease-related prognostic factors and co-morbidities played only a minor role. 26 The only study of male breast cancer, conducted in the US, suggested that later stage due to poor access to diagnostic and health care services may partly explain worse overall survival among men from lower socio-economic neighborhoods. 58
Seven studies investigated the association of SEP and education with overall survival following diagnosis with colon or rectal cancer. Two studies from England reported that variations in co-morbid conditions, urgency of surgery and curative resection status explained part of lower overall survival among the most deprived patients with colorectal cancer. 12,48 A study of colon cancer, conducted in Canada, found no evidence that stage at diagnosis contributed to socio-economic differences in overall survival, 69 while a study from Norway found that educational inequality in overall survival from colon and rectal cancer is explained partly by stage at diagnosis and although less so, by smoking and alcohol drinking. 14 One study from Denmark noted that observed socio-economic differences in overall survival from colorectal cancer were partly mediated by co-morbidities and to a lesser extent by health-related behaviors, while stage at diagnosis, mode of admittance (admitted for surgery electively or acutely), type of surgery and specialization of surgeon did not contribute to these differences. 22 Two US studies found that lower overall survival among disadvantaged individuals with colon and rectal cancer was explained in part by disease stage, tumor grade and differential access to treatment. 55,57
Six studies examined socio-economic and educational inequalities in overall survival from lung cancer. Of the 3 studies conducted in England, 1 found that the higher overall survival in the most affluent group, especially those with early-stage disease, was partly explained by variations in co-morbidities and treatment. 11 Two other studies also reported that differences in receipt of treatment explained part of the worse overall survival among patients residing in the most deprived areas. 20,25 A study from US found differential disease stage, tumor grade and receipt of the treatment explained some of the observed overall survival disadvantage among lung cancer patients from areas with higher concentration of deprivation and lower levels of education. 56 Similarly, a study from Denmark noted that educational inequalities in overall survival were partially explained by differences in stage of lung cancer at diagnosis, delivery of first-line treatment, co-morbidity and performance status. 16 In contrast, a German study found that lower survival observed for residents of more disadvantaged regions was not explained by differential stage at diagnosis or tumor grade. 41
Of the 3 studies that investigated prostate cancer, 2 were conducted in Netherlands 6,7 and 1 in the US. 51 One study found that co-morbid conditions, physical activity level and smoking status did not contribute to lower overall survival in patients with lower levels of education. 6 Another study reported that socio-economic-based inequalities in overall survival were partly mediated by differences in treatment selection and by co-morbidities. 7 The US study found that disease stage explained at least part of lower survival among prostate cancer patients either living in poverty or having low educational level. 51
With respect to head and neck and brain cancers, a study from Canada found that lower overall survival among disadvantaged patients was explained by differences in cigarette smoking, alcohol consumption, and stage at diagnosis. 70 In contrast, a second Canadian study of laryngeal cancer specifically, found that survival differences among socio-economic groups were not explained by stage at diagnosis. 71 Of the 2 studies that investigated socio-economic inequalities in overall survival from glioma, a study from US concluded that variations in receiving chemotherapy and radiotherapy did not contribute to the observed gap in survival. 62 In contrast, a second US study showed that lower survival in cases living in more disadvantaged areas was partly explained by differences in the receiving surgery and radiation therapy. 63
We identified 8 studies that explored educational or socio-economic inequalities in relation to overall survival from cancers of the cervix, ovary, corpus or endometrium. A US study of cervical cancer showed that tumor characteristics and treatment explained some of the inequalities in overall survival. 50 A study from Japan reported similar findings. 78 A study from Denmark found that overall survival disadvantage in low educated women with cervical cancer was partially explained by stage at diagnosis and, to a lesser extent, by co-morbidities and smoking status. 24 Both studies, from Japan 78 and Denmark, 33 investigating uterine and endometrial cancer found that the lower survival among disadvantaged or less educated women was partly mediated by disease stage and histology, while co-morbid conditions and treatment had no mediating effect. Of the 4 studies that assessed ovarian cancer, studies from France and the US found that the observed gap in overall survival was partly explained by variations in stage at diagnosis and the treatment received. 42,45 A Danish study reported differential stage at diagnosis, tumor histological type, co-morbidities and health-related lifestyle behaviors as some of the explanatory factors. 23 In contrast, a study from Norway found that stage of ovarian cancer or smoking status prior to diagnosis did not contribute to overall survival gap between education groups. 14
Of the remaining studies, studies from Denmark and Ireland found that late stage at diagnosis and emergency presentation contributed in part to educational inequalities in overall survival form non-Hodgkin’s lymphoma. 21,38 A French study of acute myeloid leukemia showed that variations in initial treatment did not explain the observed gap in overall survival by socio-economic position. 37 Of the 2 studies that assessed pancreatic cancer, a study from Denmark concluded that differences in surgical resection and chemotherapy explained very little of the gap in overall survival across household incomes. 39 Another study conducted in Japan found no evidence that stage at diagnosis, smoking habits, surgery and chemotherapy contribute to lower survival observed among unemployed patients and those with lower levels of occupation. 79 A Canadian study reported variations in the provision of curative treatment and co-morbidity prevalence as major contributing factors to lower overall survival in disadvantaged patients with hepatocellular carcinoma. 72 Lastly, a study from Germany that assessed all cancer types found that cases with blue-collar jobs, vocational training and lower level of income have lower cancer survival which was not explained by differences in health-related lifestyle behaviors. 46
Risk of Bias
Results from the assessment of the risk of bias of the eligible studies are summarized in Supplementary Table 2. All included studies had low risk of bias with respect to measurement and classification of the exposure and the outcome.
Age at cancer diagnosis, sex (for non-sex-specific cancers), ethnicity/race (for studies conducted in the US and New Zealand), and year of diagnosis (where applicable i.e. the period of diagnosis was ≥10 years) were considered to be important confounding domains that should be adjusted for in the analyses. Studies that applied a relative survival framework to estimate EMRRs should have used socio-economic- or deprivation-specific population life tables. With respect to confounding, several studies had moderate risk of bias; 7 studies did not use socio-economic-specific population life tables, 9,12,15,18,27,30,49 A US study did not adjust for ethnicity/race, 66 5 did not control for sex, 28,31,62,69,71 and 6 studies did not adjust for year or period of diagnosis. 26,43,47,71,72,78 Based on existing literature, 2,3,80 we considered health-related lifestyle factors, screening participation, stage of cancer at diagnosis, other tumor characteristics, co-morbid conditions, emergency presentation and treatment modalities as potential mediators on the causal pathway that should not be adjusted for in the analyses (Figure 2). Consequently, all studies that used the difference method were classified as at high risk of bias. We also assumed that patient’s rural-urban residence is a potential mediator as socio-economic position generally determines where a person lives, while acknowledging that it might be also a confounder (i.e. area of residence affects SEP as well).
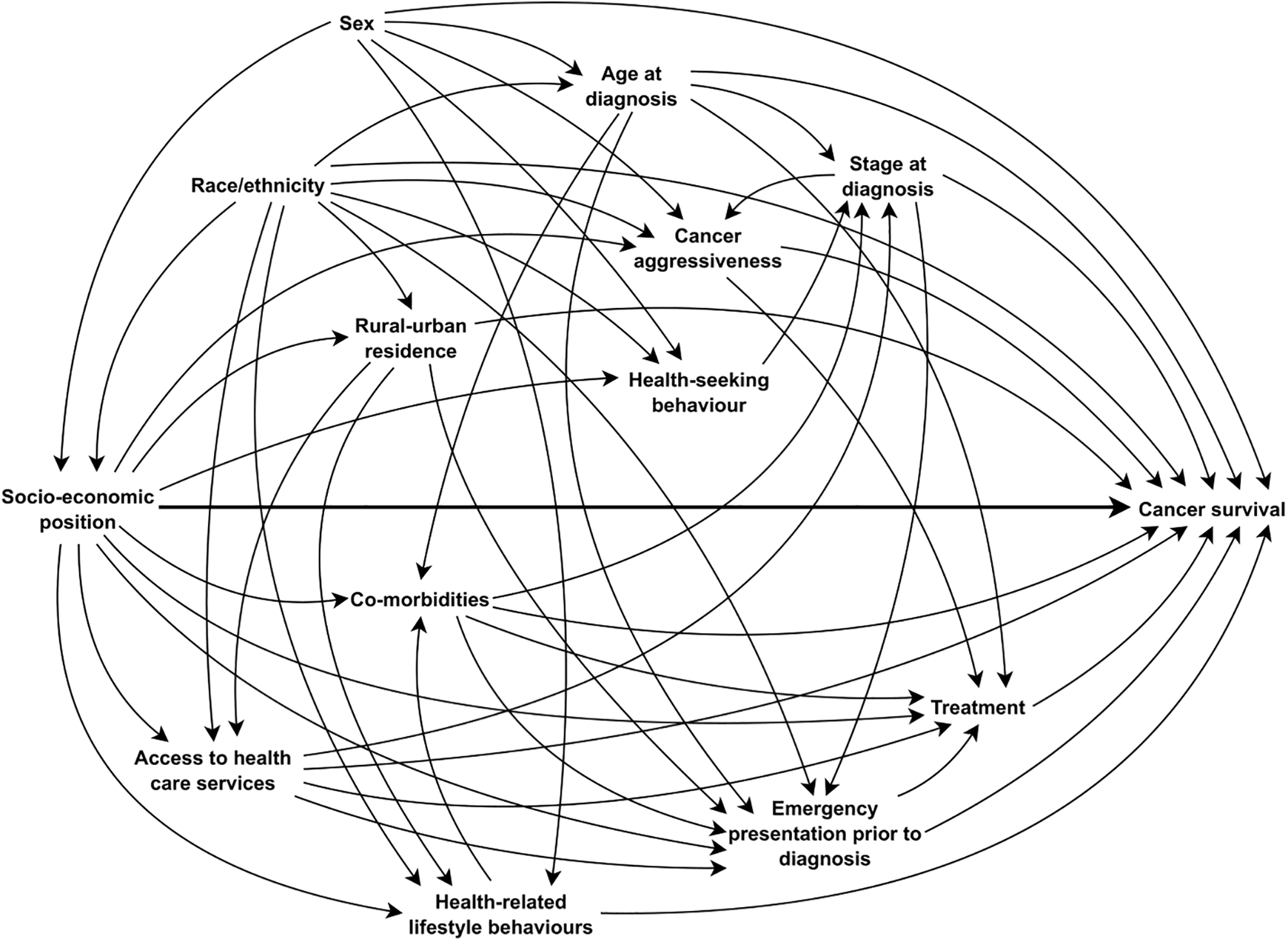
Directed acyclic graph (DAG) showing assumed causal associations between socio-economic position and cancer survival.
Bias in the selection of participants into the study was low for all studies except 4 that identified cancer patients from a hospital, 31,37,46,70 which is problematic when estimating the mediating effects of treatment. All studies, except 3, had a low risk for the domain of bias due to selective reporting of results. 41,43,47 With respect to missing data, we assigned moderate risk of bias to twenty-three studies that had 10-20% of their data missing, 7,13,20,25 -27,30,34,37,38,40,42,44,45,53,54,57,58,61,64,67,69,78 and high risk to eleven studies with more than 20% missing data, 33,39,41,43,46,48,65,66,72,75,79 regardless of whether multiple imputation was applied.
Discussion
In this systematic review, studies relatively consistently reported differences in disease stage, other tumor characteristics such as size, grade or morphology, lifestyle behaviors including smoking and excessive alcohol intake, co-morbid conditions and the treatment received as contributing causes of socio-economic inequalities in cancer-specific survival, but the estimated mediating effects of these factors varied across countries and cancer sites. Of the studies investigating survival from breast cancer, the majority reported stage at diagnosis as a contributing factor to lower survival among disadvantaged women, while a few acknowledged the potential role of co-morbidities and treatment. Most research on survival following colon or rectal cancer showed that stage of disease did not explain inequalities in survival, whereas treatment and co-morbidities partly contributed to the observed differences. Research on survival from head and neck cancer found stage at diagnosis, smoking and alcohol intake as the primary reasons for these inequalities. Similarly, for melanoma and cancers of the ovary, cervix, kidney and prostate, disease stage and treatment were reported as contributing factors to the observed survival gap across socio-economic groups. We found no obvious differences in explanatory factors by year of publication or by country.
Findings of studies investigating underlying reasons for socio-economic inequalities in overall survival following a cancer diagnosis were generally in line with studies that assessed cancer-specific survival, although the estimated effects of potential mediators varied relative to studies considering death from a particular cancer as the outcome.
The most recent review published by IARC highlighted several factors, which may contribute to observed inequalities in cancer outcomes. There is compelling evidence from existing literature that individuals with lower levels of income and education have limited awareness about adverse effects of health-related behaviors; therefore, unhealthy diet, physical inactivity, smoking, heavy alcohol intake and co-morbid conditions are more prevalent among disadvantaged people. 80 Stage at diagnosis is cited as the primary reason for inequalities in cancer survival by socio-economic position, possibly due to higher rates of screening participation and better access to diagnostic services among advantaged people. 80 Differential access to treatment facilities and the quality of care received have also been reported as potential contributing factors to lower cancer survival. 80 We should consider the fact that the mediating effect of these factors may be country-specific, generally due to variation in healthcare systems.
A review of access to cancer treatment trials across socio-economic groups found that disadvantaged cancer patients were underrepresented due to several barriers such as presence of co-morbidities, travel distance to and from clinics and financial concerns. 81 Other research reported an increasing gap between advantaged and disadvantaged patients regarding access to novel targeted cancer treatments such as immunotherapy. 82 Research on the effect of psychological factors on socio-economic inequalities in cancer screening participation and attitudes toward cancer reported that people from lower socio-economic background have more negative beliefs about cancer screening, early detection and treatment such as worrying and not willing to know if they have cancer, as they believe cancer is a death sentence or cancer treatment is worse than cancer itself. 83,84
The contribution of the above factors and social or psychological stresses to socio-economic inequalities in cancer patient survival is not clear due to methodological issues and data limitations. Also, it is unclear whether the interaction between the socio-demographic and clinical characteristics of cancer patients and the health care system partly explain these inequalities. In the absence of more in-depth knowledge, it is challenging to identify and prioritize actionable factors to address socio-economic inequalities in cancer survival and improve outcomes for disadvantaged patients.
Limitations
The findings of this review should be interpreted with caution, mainly due to between-study heterogeneity in measures of SEP (measured at individual or neighborhood level) and the methods used to identify the underlying causes of socio-economic inequalities in cancer survival. A limitation of using single individual-level indicators of SEP in investigating socio-economic inequalities in health outcomes is that each does not address the multi-dimensionality of SEP; for instance, a person can be classified as advantaged by one indicator (high level of education), but not another (low level of income). 85,86 Another issue is that in health inequalities research, it is common practice to adjust for socio-economic indicators other than the one of interest, which ignores the complexity of the pathways that connect SEP indicators to health. 86 Using composite measures of SEP defined by weighting and aggregating several socio-economic dimensions to measure material and social deprivation or social and economic standing, can potentially overcome the issues mentioned above, but these measures might not be suitable to answer particular policy questions. 87 Using area-based SEP measures may lead to misclassification and underestimation of contribution of individual-level SEP to health outcomes, although the characteristics of an area, such as public resources and infrastructure, can also independently affect people’s health, thereby over-estimating the association of individual-level SEP with the outcome of interest. 88
The majority of the studies applied the difference method, by which regression models were compared with and without adjusting for potential mediators. This standard approach has some major limitations. The first problem arises when there are unmeasured mediator-outcome confounders. Adjusting for the mediator in the presence of mediator-outcome confounding is inappropriate as it creates a non-causal association between the exposure and the mediator-outcome confounder, which can induce substantial bias, known as collider bias. 89 Another limitation is the assumption of no interaction between the effects of the exposure and the mediator on the outcome, which may result in invalid inferences. 89,90 Moreover, this approach fails when multiple mediators are of interest and the mediators affect or interact with each other (for example, interactions between stage and treatment or co-morbidity and treatment); in this case, adding mediators one-by-one to the model could give biased estimates. 89,90
About half of the included studies assessed overall survival, which is problematic when exploring the reasons for socio-economic inequalities in cancer survival as there is solid evidence that socio-economic position is associated with death due to causes other than cancer. 91 In addition, for screen-detectable cancers such as breast, colorectal and prostate cancer, the effect of overdiagnosis, lead-time and length-time bias should not be neglected as screen-detected cancers show higher survival, even in the presence of co-morbidities and ineffective treatment.
Conclusion
Socio-economic inequalities in cancer survival appear to be partly explained by differences in disease stage, health-related behaviors, chronic conditions and treatment modalities. Discrepancies in findings across studies could be due to variation in the covariates included in the analyses. It is essential that future studies apply novel methods of mediation analysis to population-based linked health data to generate more reliable evidence about the medical and psychological mechanisms underlying these inequalities, which may lead to better resource allocation and change in cancer control policies to improve cancer survival for all patients.
Supplemental Material
Supplemental Material, sj-pdf-1-ccx-10.1177_10732748211011956 - Factors Explaining Socio-Economic Inequalities in Cancer Survival: A Systematic Review
Supplemental Material, sj-pdf-1-ccx-10.1177_10732748211011956 for Factors Explaining Socio-Economic Inequalities in Cancer Survival: A Systematic Review by Nina Afshar, Dallas R. English and Roger L. Milne in Cancer Control
Supplemental Material
Supplemental Material, sj-pdf-2-ccx-10.1177_10732748211011956 - Factors Explaining Socio-Economic Inequalities in Cancer Survival: A Systematic Review
Supplemental Material, sj-pdf-2-ccx-10.1177_10732748211011956 for Factors Explaining Socio-Economic Inequalities in Cancer Survival: A Systematic Review by Nina Afshar, Dallas R. English and Roger L. Milne in Cancer Control
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: N.A. was the recipient of an Australian Government Research Training Program Scholarship.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
