Abstract
Pancreatic cancer with synchronous liver metastasis has an extremely poor prognosis, and surgery is not recommended for such patients by the current guidelines. However, an increasing body of studies have shown that concurrent resection of pancreatic cancer and liver metastasis is not only technically feasible but also beneficial to the survival in the selected patients. In this review, we aim to summarize the short- and long-term outcomes following synchronous liver metastasectomy for pancreatic cancer patients, and discuss the potential criteria in selecting appropriate surgical candidates, which might be helpful in clinical decision-making.
Introduction
Pancreatic cancer (PC) is a lethal digestive system malignancy with a dismal 5-year survival rate of 9%. 1 More than half of the PC patients are detected with distant metastases, of which liver is the most common site, accounting for 37%-41.9% of the initially diagnosed cases. 2,3 PC patients with liver metastasis suffer from an extremely poor prognosis with a 5-year survival rate less than 1% and median overall survival (OS) of only 5 months. 4 Currently, systemic chemotherapy is the optimal choice for most metastatic patients, and surgery is not recommended in the regular clinical practice by the guidelines. However, surgical resection of the liver metastasis has shown a significant improvement in the long-term survival for colorectal cancer, neuroendocrine tumor, and gastric cancer, which was also “neglected” in the previous treatments for these malignancies. 5 -7 Here we aim to discuss the role of surgery in PC patients with synchronous liver metastasis by reviewing the recent literature.
The Short- and Long-Term Outcomes Following Synchronous Liver Metastasectomy
The feasibility of synchronous pancreatectomy and hepatectomy for metastatic PC has been verified in the most previous reports with mortality rates < 5% (Table 1). 8 -13 But Gleisner et al. 14 reported that the 30-day postoperative mortality was up to 9.1% in the patients who underwent synchronous liver metastasectomy for periampullary or pancreatic adenocarcinoma. This was significantly increased compared with 4.5% in the non-metastatic patients who underwent surgical resection, as well as 3% in the patients who underwent palliative bypass. Takada et al. 15 observed the same high mortality in an earlier retrospective study, implying the potential risks of the combined surgery should be carefully evaluated even with the advancements in surgical techniques and perioperative managements.
Previous Studies Regarding Synchronous Resection for PC With Liver Metastasis.

ASA, American Society of Anesthesiologists; DFS, disease-free survival; m, months; MR, metachronous resection; met, metastasis; NA, not available; OS, overall survival; PC, pancreatic cancer; SR, synchronous resection; a, median; b, mean; c, calculated in the patients with pancreatic (n = 20) and distal bile duct tumors (n = 5); d, calculated in the synchronously resected patients for liver metastasis (n = 62) and inter-aortocaval lymph node metastasis (n = 43).
The postoperative morbidity rates varied greatly in the published studies. 24 Tachezy et al. 17 reported the highest morbidity rate of 68% for synchronous resection by analyzing liver metastatic PC cases from 6 European pancreas centers, which was significantly elevated compared with that in the non-resected patients but comparable to the published data on patients with pancreatic resection only. In the study including the most synchronous resection cases, Andreou et al. 22 reported the overall morbidity rate of 50% and major morbidity rate of 16%. Many factors, such as the patient conditions, resection extents, and surgeon techniques, could affect the complications following pancreatectomy alone or combined with hepatectomy. The majority of the previous studies on the synchronous resections for liver metastatic PC indicated that the postoperative morbidity rates were acceptable in comparison to the internal control groups and the reported data.
The survival benefit of PC patients with liver metastasis is the major concern for surgeons to make therapeutic decisions. The median OS following synchronous liver metastasectomy and pancreatectomy for PC ranged from 5.9 to 15.7 months in the literature.
14,18
After analyzing 14 cases, Dünschede et al.
10
found that the median survival of the patients who underwent concurrent resection was not superior to that of those receiving chemotherapy (8
Selection of Candidates for Synchronous Liver Metastasectomy
Though the morbidity and mortality following synchronous pancreatectomy and hepatectomy should be well evaluated, survival benefit from this combined surgery is more important be assessed when selecting cases. Quite a few studies reported the potential risk factors impacting the long-term survival of liver metastatic PC patients, including the liver metastatic number, neoadjuvant chemotherapy, tumor biomarker level, and so on. 26 Thus, these factors could be the major concerns when evaluating appropriate surgical candidates.
Burden of Liver Metastasis
Tumor burden predicts the prognosis of patients with metastatic malignancies. An early systemic review performed by Michalski et al.
27
indicated that the long-term survival in PC patients who underwent concurrent resection for one or few liver metastases was comparable to the cases without evidence of metastasis. Recently, Tachezy et al.
17
considered that PC patients with a small metastatic tumor burden could benefit from this aggressive surgery even without preoperative chemotherapy, on account of the result that the median OS in the patients with isolated liver metastasis (n = 65) was greatly prolonged in comparison to that in the patients with ≥ 2 metastases (n = 73) (11.6
Neoadjuvant Chemotherapy
Neoadjuvant chemotherapy has been paid much attention to in the borderline resectable and resectable PC for its potential advantages: degrading tumor stage to improve resection rate, eliminating micrometastasis to prevent early recurrence, and guiding postoperative chemotherapy.
29,30
Though neoadjuvant chemotherapy for metastatic PC patients was explored much less, some recent studies have shown promising results. Wright et al.
20
reported 23 metastatic patients (liver metastasis, n = 16) who underwent pancreatectomy with or without metastasectomy (liver metastasectomy, n = 9) after favorable response to chemotherapy. The study indicated that the median OS from surgery and disease-free survival (DFS) and OS from diagnosis were 18.2, 8.6 and 34.1 months, respectively. Crippa et al.
21
also reported 11 liver metastatic PC patients who underwent either primary or combined resection following complete or partial response to chemotherapy, and these patients obtained significantly longer OS than the liver metastatic cases receiving palliative chemotherapy (n = 116) (39
Tumor Markers
Serum carbohydrate antigen 19-9 (CA19-9) has been a useful marker in the evaluation of resectability and prognosis for resectable and borderline resectable PC with different cut-off points, which implies its potential value in selecting suitable metastatic PC candidates for surgical resection. 32 -34 Crippa et al. 21 reported that CA19-9 reduction < 50% of baseline level after chemotherapy was an independent risk factor of OS in the liver metastatic patients following resection. They thought that major biochemical response (CA19-9 decrease > 90%), as a complementary evaluation to the radiologic response, should be a required condition for surgery. Carbohydrate antigen 125 (CA125) was also reported as an important biomarker for synchronous liver metastatic PC by Shi et al. 18 They found that this marker could be used not only in the evaluation of resectability for synchronous liver metastasis but also in the prognostic prediction following this combined resection, indicating its supportive value in Lewis-negative individuals, who secrete very little CA19-9.
Pancreatic Cancer Location
Recently, Yang et al.
23
retrospectively analyzed their single-center experience of 23 liver oligometastatic PC cases with synchronous resection. They showed that the OS of patients with pancreatic head cancer who underwent synchronous resection did not differ from that of the systemic chemotherapy counterparts (6.9
To date, no prospective research with regard to synchronous resection of liver metastatic PC has been published. Several previous studies mentioned their general indications of hepatectomy, but these indications might not be of great referential value in selecting the suitable cases (Table 1). Currently, 2 ongoing studies could be found in the https://ClinicalTrials.gov database, both of which take liver metastases ≤ 3 as a resection criterion (Table 2). In addition, Yu et al focus on the role of chemotherapy in screening the potential candidates in a large cohort of 300 estimated participants, which deserves to look forward to.
Ongoing Clinical Studies on Synchronous Resection for PC With Liver Metastasis.
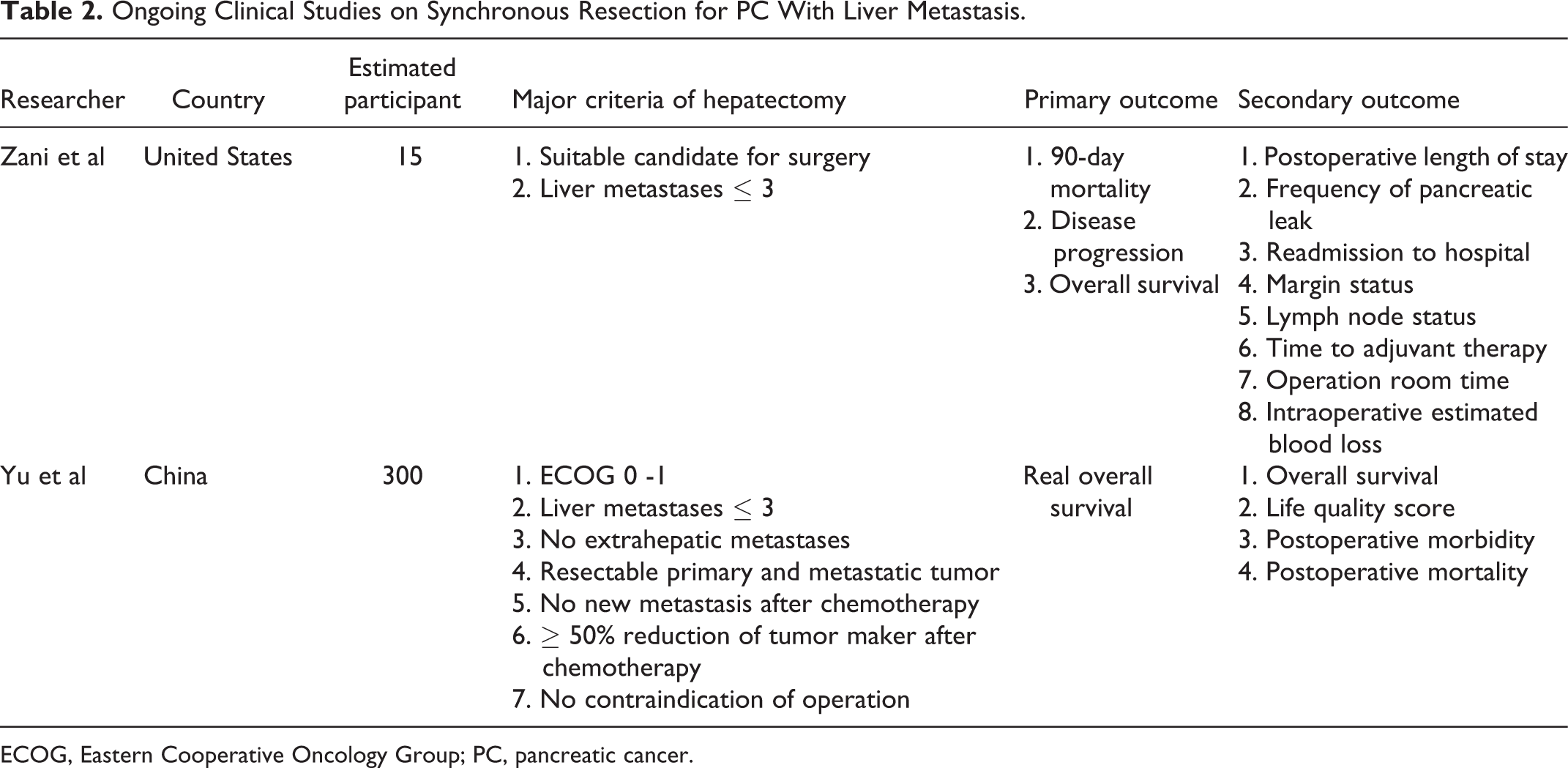
ECOG, Eastern Cooperative Oncology Group; PC, pancreatic cancer.
Conclusion
A growing body of evidence supports the role of synchronous resection in the selective PC patients with liver metastasis. But no consensus has been successfully reached for selecting appropriate surgical candidates. The current major concerns on patient selection include the metastatic tumor burden, chemotherapeutic efficacy, tumor marker level, as well as candidates’ health condition. We think liver oligometastatic PC patients, achieving major radiological and biochemical responses to neoadjuvant chemotherapy, could be suitable for further resections, particularly for whose primary tumor located at the pancreatic body/tail. All the previous results regarding surgery in the liver metastatic PC came from retrospective studies, where the included cases were quite limited and highly heterogeneous. Registry studies, especially with nationwide cooperation, should be able to provide a mass of patients for more efficient analysis before starting randomized controlled trials.
Footnotes
Abbreviations
CA125, Carbohydrate Antigen 125; CA19-9, Carbohydrate Antigen 19-9; DFS, Disease-free Survival; OS, Overall Survival; PC, Pancreatic Cancer.
Acknowledgments
The authors thank Dr. Ashely Shin for her helpful assistance in the language polishing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Shanghai Municipal Health Commission (No.201840190).
