Abstract
Backgrounds:
Acinar cell carcinoma of the pancreas is a rare malignancy, and its features remain unclear. We aimed to analyze the clinical characteristics, treatment and prognosis of acinar cell carcinoma with our institutional case series.
Methods:
Patients diagnosed with acinar cell carcinoma in our hospital between 2005 and 2019 were reviewed. Investigations on clinicopathological features, treatment details and long-term survival were performed.
Results:
A total of 45 pathologically confirmed acinar cell carcinomas were identified. The median age at diagnosis was 58 years with a male-to-female ratio of 3.1:1. There were 24 (53.3%) localized, 5 (11.1%) locally advanced and 16 (35.6%) metastatic cases, with a pancreatic head-to-body/tail ratio of 1:1.4 for all the primary lesions. In the localized group, there were 10 pancreatoduodenectomy, 12 distal pancreatectomy, 1 total pancreatectomy, and 1 distal pancreatectomy combined with proximal gastrectomy. Among the locally advanced and metastatic cases, 13 patients received chemotherapy, 1 received concurrent radiochemotherapy, 1 underwent synchronous resection of primary tumor and liver metastasis, 1 underwent palliative operation, 1 underwent exploratory laparotomy, and 4 required no treatment. The median overall survival of this series was 18.9 months with a 5-year survival rate of 19.6%. Moreover, the resected acinar cell carcinoma patients were associated with prolonged survival compared with the unresected cases (36.6 vs. 8.5 months, P < 0.001).
Conclusions:
Surgical resection could improve the long-term survival of acinar cell carcinoma patients, which might also improve the prognosis of selected metastatic cases. Large-scale studies are needed to further clarify the biological behavior and clinical features, and to seek the optimal treatments.
Introduction
Acinar cell carcinoma (ACC) of the pancreas is a rare malignancy, and accounts for approximately 1%-2% of exocrine pancreatic neoplasms. 1 ACC arises from the pancreatic acinus, which secretes digestive enzymes (e.g., trypsin, chymotrypsin, amylase) and makes up more than 80% volume of the pancreas. Histologically, ACC is characterized by the rich tumor cells, forming lobular, solid, glandular or trabecular patterns, and poor fibrous stroma. 2 Radiologically, ACC typically presents as a sizable, well-defined mass with an enhancing capsule, frequently companied with central hypodensity and occasionally with internal calcification in the computed tomography (CT) images. 3 However, definite diagnosis could be rarely achieved through the radiologic examinations. Even for pathological diagnosis, ACC could be difficultly distinguished from pancreatic neuroendocrine tumor (pNET) or solid pseudopapillary tumor due to their similarities. 2 Therefore, immunohistochemical staining for pancreatic enzymes or other markers, such as Bcl-10, is often required for confirmation. 4
Compared with ductal cell adenocarcinoma (DCA), ACC is a distinct entity. Typical DCA-associated mutations in KRAS, SMAD4, and TP53 genes are rarely or less frequently detected in ACC. 5,6 ACC harbors frequent alterations in genes of the adenomatous polyposis coli (APC)/β-catenin pathway, which is similar to colon cancer. 7 For the clinical manifestations, ACC shares many non-specific symptoms with DCA, including abdominal discomfort, weight loss and weakness, nausea and vomiting, melena, and diarrhea. But painless obstructive jaundice, commonly presented in DCA, is infrequently seen in ACC. 8 In addition, a few ACC patients could develop lipase hypersecretion syndrome because of the massive lipase released by the tumor, manifested with subcutaneous fat necrosis, polyarthralgia or eosinophilia. 9,10 As a moderate malignancy, ACC carries a better prognosis in comparison to DCA. The previously reported median overall survival (OS) of ACC patients ranged from 17.6 to 47 months. 9 -16 In the latest ACC series from 3 French centers, Egal et al. 17 showed a favorable median OS of 55.5 months in all staged patients with 106.5 months in patients who underwent curative-intent resections.
Currently, no broad consensus has been reached on the diagnosis or treatment for ACC due to its rarity. The staging system and therapeutic strategy of ACC mainly refer to the guidelines for DCA, though the 2 entities possess quite a few distinctive features. In this study, we aimed to share our experience of ACC with the largest single-center case series to date, which might make contribution to better understand the characteristics of ACC.
Methods
Patients
Consecutive patients who received treatments for ACC from June 2005 to November 2019 in our hospital were carefully reviewed. Only pathologically proven ACC cases (acinar differentiation and positive staining of trypsin and/or chymotrypsin) were included into analysis. Demographic, clinical and pathological data were extracted from the medical records. The values of serum tumor markers, including CA19-9, AFP, and CEA, were recorded according to the latest test prior to the treatments. Primary ACC diameters were recorded based on the pathological reports for the resected cases or imaging reports for the unresected cases. TNM stage was defined according to the American Joint Committee on Cancer staging system (eighth Edition). OS was calculated as the period from the date of pathological diagnosis to the date of death or last follow-up. Follow-up was preformed either by searching the electronical outpatient system of our hospital or by telephone interviews, which was ended in December 2019.
Statistical Analysis
Categorical and continuous variables were presented as frequency counts and percentages, and medians and interquartile ranges (IQR), respectively. The Kaplan-Meier method was used to performed survival analysis, and survival differences between the groups were examined using the log-rank test. Univariate and multivariate Cox regression analyses were performed to identify the risk factors for OS. All the above statistical analyses were completed using SPSS software (version 25.0, IBM Corp., Armonk, NY, USA). P values < 0.05 with 2-side were considered statistically significant. Survive curves were plotted using R software (version 3.6.3, https://www.r-project.org/).
Results
Patient Characteristics
Forty-five ACC cases were confirmed by the pathology. The median age of these patients was 58.0 years (IQR, 47.5-67.5 years) with a male-to-female ratio of 3.1:1. The major complaints of the patients at admission were abdominal discomfort (n = 24, 53.3%) and positive detection in the periodic physical examination (n = 11, 24.4%). The median diameter of the primary tumors was 4.7 cm (IQR, 3.3-5.9 cm) with a pancreatic head-to-body/tail ratio of 1:1.4. About one-third of the patients were detected with metastasis. According to the TNM staging system, there were 6, 15, 8, and 16 cases classified as stage I, II, III and IV, respectively (Table 1). Pathologically, apart from the 38 pure ACCs, there were 6 mixed acinar-endocrine carcinoma and 1 mixed acinar-ductal carcinoma cases in this retrospective analysis (Table 2).
Baseline Characteristics of Patients With ACC.

ACC, acinar cell carcinoma; CA19-9, carbohydrate antigen 19-9; AFP, alpha fetoprotein; CEA, carcinoembryonic antigen; AJCC, American Joint Committee on Cancer; IQR, interquartile range; a, n = 40.
Treatment and Pathological Characteristics of Patients With ACC.

ACC, acinar cell carcinoma; a, n = 24; b, n = 42; c, n = 40; d, n = 25.
Treatment and Prognosis
Among the localized cases, 10 patients underwent pancreaticoduodenectomy, 12 underwent distal pancreatectomy, 1 underwent distal pancreatectomy combined with proximal gastrectomy, and 1 underwent total pancreatectomy, respectively. Sixteen of the resected patients received adjuvant chemotherapy with either gemcitabine alone (n = 9) or gemcitabine-based regimens (n = 7). Of the locally advanced cases, 4 patients received treatments, including palliative operation (n = 1), exploratory laparotomy (n = 1), chemoradiation (n = 1), and chemotherapy (n = 1), and the remaining 1 did not receive any treatments. Of the metastatic cases, all the patients were detected with liver metastasis, 2 of who were additionally discovered with peritoneal metastasis. One patient with isolated liver metastasis underwent synchronous pancreatectomy and hepatectomy, and 12 patients received chemotherapy, 5 of who underwent transcatheter arterial chemoembolization for liver metastases. The remaining 3 metastatic patients required no treatment. The pathological findings in all the resected cases (24 localized cases and 1 metastatic case) indicated that 10 primary tumors harbored necrosis, 8 harbored microvascular invasion, 8 harbored lymph node metastasis, and 7 harbored nerval invasion, respectively (Table 2).
On the last follow-up day, 31 (68.9%) patients had died, 9 (20.0%) were still alive and the remaining 5 (11.1%) could not be contacted. Of the localized cases, 18 patients were detected with relapses, including local recurrences (n = 5), isolated liver metastases (n = 11), and liver combined with retroperitoneal lymph node metastases (n = 2). The median OS of the whole series was 18.9 months with a 5-year survival rate of 19.6%. Survival analyses showed that the median OS of the localized patients was significantly longer than that of the locally advanced patients (36.6 vs. 10.1 months, P = 0.002) and the metastatic patients (36.6 vs. 7.4 months, P < 0.001) (Figure 1A). No statistical difference in OS was observed between the locally advanced and metastatic cases (10.1 vs. 7.4 months, P = 0.870). In addition, the resected patients correlated with significantly prolonged OS compared with the unresected cases (36.6 vs. 8.5 months, P < 0.001) (Figure 1B).

Kaplan-Meier survival analyses in the whole ACC series divided by the tumor status (A) and resection (B), and in the localized subgroup divided by the TNM stage (C) and adjuvant chemotherapy (D). ACC, acinar cell carcinoma.
In the subgroup of localized cases, survival analyses indicated that the median OS of stage I and stage II patients was significantly longer than that of stage III patients (stage I vs. III, 37.3 vs. 9.2 months, P = 0.004; stage II vs. III, 36.6 vs. 9.2 months, P = 0.003) (Figure 1C). Moreover, patients who received postoperative chemotherapy acquired an improvement in the long-term survival compared with those who had no adjuvant chemotherapy (37.7 vs. 18.9 months, P = 0.010) (Figure 1D). Cox regression analyses demonstrated that TNM stage (stage I vs. III, hazard ratio [HR] 0.025, 95% confidence interval [CI] 0.002-0.347, P = 0.006; stage II vs. III, HR 0.026, 95% CI 0.002-0.400, P = 0.009) and postoperative chemotherapy (HR 0.221, 95% CI 0.070-0.691, P = 0.009) were independent risk factors of OS for the localized ACC (Table 3).
Univariate and Multivariate Cox Regression Analyses of Risk Factors for Overall Survival in the Localized ACC Patients (n = 24).

ACC, acinar cell carcinoma; HR, hazard ratio; CI, confidence interval; ref, reference.
Discussion
ACC is a low incidence rate malignancy, and the most published reports contained relatively limited cases, which might weaken the power of the corresponding conclusions (Table 4). 8 -11,13 -19 Though there have been several database studies with hundreds of ACC cases, they mainly focused on the prognostic analyses, particularly in resected patients. 12,20 -23 Certain characteristic information, such as clinical symptoms, hematological examinations, and pathological details, was unavailable in these studies. In the present research, we investigated the clinical feature, treatment, and prognosis of ACC with a 14-year series in our high-volume center, which performed more than 400 pancreatic operations annually.
Previous ACC Literature Containing Surgical Cases.
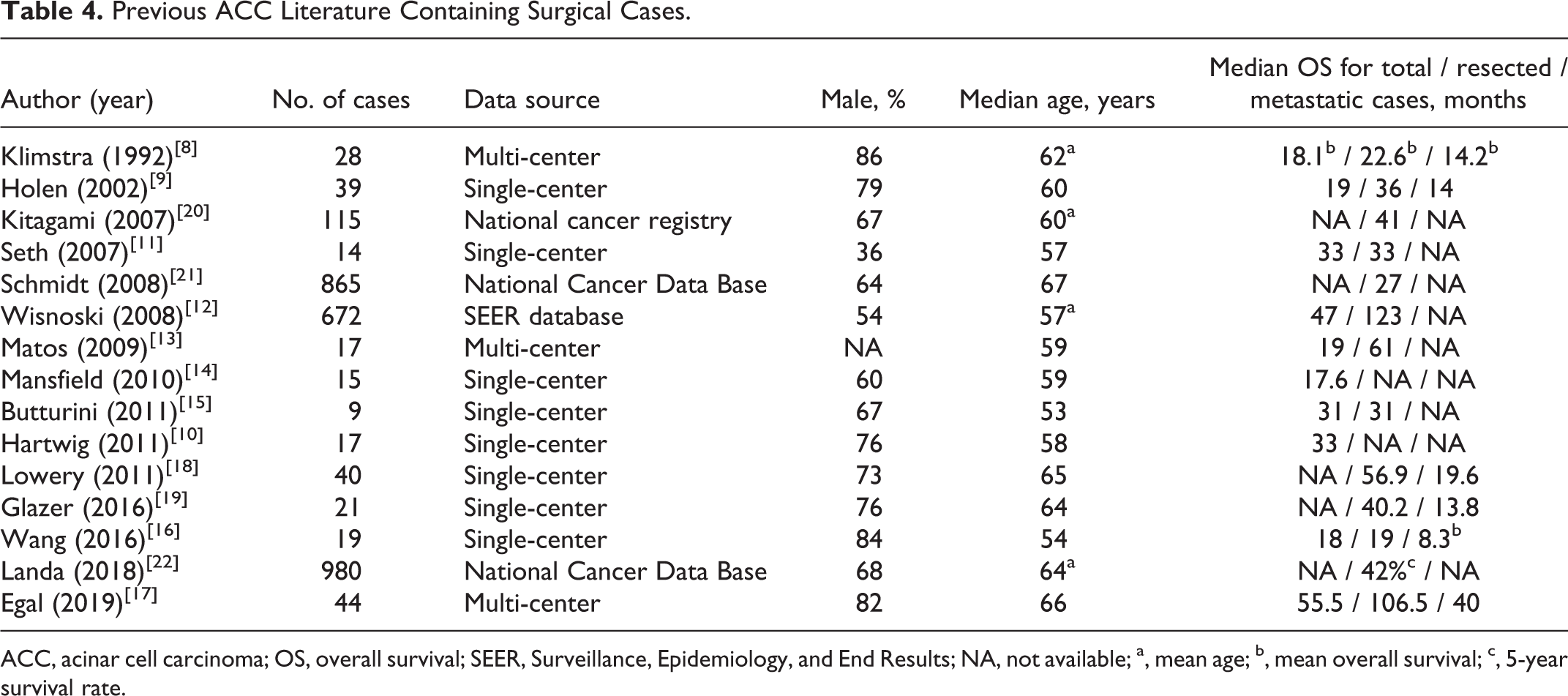
ACC, acinar cell carcinoma; OS, overall survival; SEER, Surveillance, Epidemiology, and End Results; NA, not available; a, mean age; b, mean overall survival; c, 5-year survival rate.
Consistent with many previous reports, ACC patients in our series were relatively young (median age 58 years) with the predominance of males (75.6%). 9,10,14 -16,20 Non-specific abdominal discomfort urged more than half of the patients to seek medical advice initially. Only 3 patients presented with obstructive jaundice here. Except for the cases with primary tumors located at the pancreatic body and tail, ACC still had a much lower rate of jaundice (15.8%) than pancreatic head DCA, which was up to approximately 80% in the published literature. 24,25 This may indicate the less aggressive behavior of ACC as it generally pushes rather than directly invades into the bile duct. 26 No typical symptom caused by the lipase overproduction was observed in this series, though 3 of the 4 patients with documented serum lipase data had elevated levels (415, 447, and 2708 U/L; normal range, 13 to 63 U/L). These lipase levels might not be high enough to generate the clinical signs, or the elevated periods in the patients were relatively short.
Due to the specificity, serum lipase level may serve as a potential marker for differential diagnoses in patients with radiologically suspected pancreatic malignancies. However, as it abnormally increased in less than half of the ACC patients in the previous research, its real utility needs to be verified. 13,18 AFP is another potential serum marker proposed in some studies for facilitating the identification of ACC from DCA, but it only abnormally elevated in 3 (7.5%) documented patients in this series. 20 Currently, no reliable laboratory biomarker has been revealed to detect or differentiate ACC. Given the distinct gene mutation patterns between ACC and DCA, sequencing of the circulating tumor cells or DNA could be promising directions.
Pathological features of the resected cases showed that the rates of lymph node metastasis, nerval invasion, and microvascular invasion were lower in ACC compared with the corresponding data regarding DCA from the previous reports, which again reflected the less aggressive behavior of ACC. 27,28 In addition, 40% of the resected cases presented with necrosis, which was quite rare in DCA. 29 This might ascribe to the high proportion of tumor cell component in ACC, requiring sufficient blood supply to sustain metabolism, whereas DCA was predominated by the fibrous stroma and less likely affected by the deficient blood flow.
Surgical resection is the first-line treatment for resectable ACC. The OS of the resected patients in our series was 36.6 months, which was consistent with the data in the previous literature, and significantly prolonged in comparison to that of the unresected counterparts. 9,11,15,19,20 Noticeably, one patient with solitary liver metastasis were misdiagnosed as pNET preoperatively, and underwent synchronous distal pancreatectomy and hepatic wedge resection. No obvious evidence for recurrence has been observed since the patient underwent the operation 89 months ago. This favorable outcome raises an issue whether surgical resection should be an acceptable choice for solitary liver metastatic ACC. Technically, the feasibility and safety of combined pancreatectomy and hepatectomy have been demonstrated in many published studies. 30 -32 In terms of oncology, radical resection has been suggested in well-differentiated pNET patients with single liver metastasis by the European Neuroendocrine Tumor Society. 33 Even for more aggressive DCA, quite a few studies have shown the survival benefits in liver metastatic patients who underwent synchronous resections, particularly in oligometastatic cases. 34,35 Given the rarity of ACC, the value of surgery in liver metastatic situations was seldom explored. Seth et al. 11 reported one patient who underwent concurrent hepatectomy for metastasis acquired the longest survival period of 95 months among all the 14 resected ACC patients. Another analysis performed by Hartwig et al. 10 showed that the 2-year survival rate of ACC patients with limited metastatic disease underdoing synchronous resection (3 additional hepatectomy and 1 omentectomy) was comparable to that of nonmetastatic patients treated with resection (75% vs. 66%, P = 0.85). A more recent study containing 340 metastatic ACC cases from National Cancer Data Base (NCDB) indicated that patients required resection correlated with an increased 5-year survival rate compared with those who did not undergo resection (18.7% vs. 3.8%, P < 0.001). 22 As liver is the most common metastatic site of ACC patients, this result might also provide evidence to support the role of surgical resection in liver metastatic cases. However, it needs to be well evaluated in large-scale studies with good designs.
Adjuvant chemotherapy is a routine option for all resectable DCA following surgical resection in order to eliminate residual tumor cells to postpone recurrence. But the effect of adjuvant chemotherapy in ACC was still controversial in the literature. A single-center case series of 21 ACC patients from Glazer et al. 19 suggested that adjuvant chemotherapy was not associated with improved survival on multivariate analysis (HR 0.030, P = 0.2). Contrarily, Wang et al. 16 reported their institutional experience with 19 ACC cases and indicated that resected patients followed by adjuvant chemotherapy had increased long-term survival in comparison to those who underwent resection alone (P = 0.006). Our present study also showed the survival improvement in ACC patients receiving postoperative chemotherapy (37.7 vs. 18.9 months, P = 0.010), which was further demonstrated as an independent protective factor on multivariate analysis (HR 0.221, 95% CI 0.070-0.691, P = 0.009). Owing to the limited sample sizes, powerful conclusions were difficultly drawn from these institutional reports. Interestingly, 2 database analyses from the NCBD came to the different results. In the year of 2008, Schmidt et al. 21 found that the resected patients with or without adjuvant chemotherapy shared the similar OS after analyzing 865 ACC cases (P = 0.3). However, more recently, Patel et al. 36 reviewed 298 ACC cases from the same database and discovered that adjuvant chemotherapy following surgical resection was associated with a significant improvement in OS compared with resection alone (HR 0.54, 95% CI 0.33-0.89, P = 0.015). This might be explained by the advancements in chemotherapeutic regimens over time, as the patients included in the 2 studies were from 1985 to 2005 and from 2004 to 2015, respectively. According to our own experience, we take an active attitude toward the use of adjuvant chemotherapy in all suitable ACC patients undergoing surgical resection. The regimens of adjuvant chemotherapy were infrequently mentioned in the previous literature and varied among different institutions. Consistent with the major reports, our center adopted gemcitabine or gemcitabine-based chemotherapy for the postoperative treatment. 10,17,18 Glazer et al. 19 proposed the regimen of folinic acid, fluorouracil and oxaliplatin (FOLFOX) for the adjuvant therapy of ACC considering its higher rate of disease control compared with gemcitabine. But no existing evidence has proven the advantage of this fluorouracil-based regimen in improving the survival of resected ACC patients. These issues regarding postoperative chemotherapy, such as agent type, therapy cycle, and response evaluation, need to be further clarified with accumulated cases.
There were some limitations should be mentioned. The retrospective nature and relatively small size of our study could limit the persuasiveness of the statistical results. However, given the rarity of ACC, we still provide useful information to help reveal the clinical features, some of which are unavailable in the database studies. In addition, the exact periods of recurrence and progression for resected and unresected patients, respectively, could not be evaluated here. This should be performed in prospective research with uniform follow-up strategies.
Conclusion
ACC is a moderately aggressive pancreatic malignancy with a low incidence rate. Surgical resection provides a chance to prolong survival for all resected patients, particularly for those receiving postoperative chemotherapy. Moreover, selected liver metastatic patients may also obtain a survival improvement following synchronous resection. Further well-designed, large-scale, prospective studies are needed to powerfully verify the optimal diagnostic, therapeutic and follow-up strategies for ACC patients.
Footnotes
Abbreviations
Authors Note
Wentao Zhou, Xu Han and Yuan Fang contributed equally to this study.
Acknowledgment
The authors thank Dr. Jing Wu for her helpful assistance in the follow-up data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (No.81972218) and Science and Technology Commission of Shanghai Municipality (No.19140901700).
Research Ethics and Patient Consent
This study was approved by the Ethics Committee of Zhongshan Hospital, Fudan University (No. B2019-297). All participants provided written consent forms.
